Abstract
Background
Parkinson's disease (PD) is a neurodegenerative disorder with heterogenous symptoms and progression rates. Some studies have classifying PD into tremor-dominant and non-tremor-dominant PD (TD-PD and Non-TD-PD), finding that TD-PD tend to have a better prognosis, slower disease progression, and lower pathological burden compared to Non-TD-PD. However, this classification does not consider the specific types of tremors, even though recent studies have shown that different types of tremors in PD might have distinct underlying mechanism.
Objective
Data from 517 de novo drug-naïve PD patients was analyzed.
Methods
Survival analysis was carried out including motor subtypes, rest tremor presence, kinetic tremor presence, postural tremor presence, as well as the instability of tremor presence or motor subtypes as predictors. Occurrence of 6 outcomes, including motor and non-motor milestones at follow-up, were used as the time-to-event.
Results
Both TD-PD subtype and rest tremor presence was associated with slower PD progression, while kinetic tremor presence and postural tremor presence, especially kinetic tremor presence, was associated with a faster one.
Conclusions
Our study suggests different types of tremors are associated with distinct PD prognoses, indicating potential differences in underlying mechanisms. Further investigation is warranted to elucidate the specific mechanisms underlying different types of tremors in PD and to explain their relationship to disease prognosis.
Plain Language Summary
Parkinson's disease (PD) is a heterogenous disease with diverse symptoms and rates of progression. Our study aimed to investigate the impact of motor subtype, different types of tremors, and their consistency on the progression of PD. We analyzed data from 537 newly diagnosed, untreated PD patients and monitored them for several important progression milestones. We found that patients with tremor-dominant PD and those experiencing resting tremors tended to have slower disease progression. Conversely, the presence of kinetic tremors and postural tremor, especially kinetic tremor, was associated with a faster progression of PD. These findings suggest that the specific types of tremors in PD may have potentially different mechanisms. Understanding these differences could help in developing targeted treatments that address specific tremor types, potentially improving outcomes for patients. Further research is necessary to explore the underlying mechanisms behind these tremor types and their relationship to PD prognosis.
Keywords
Introduction
Parkinson's disease (PD) is a neurodegenerative disorder with heterogeneous symptoms and progression rates. 1 Based on the observation that certain motor features tend to cluster together, previous studies have suggested that classifying PD into motor subtypes may help to provide a better understanding of the pathology, prognosis, and progression of the disease. 2 Consistently, research has found that tremor-dominant PD (TD-PD) has a better prognosis, slower disease progression rate, and potentially lower pathological burden compared to non-tremor-dominant PD (Non-TD-PD).3–10 However, some studies have reported no difference in prognosis between TD-PD and Non-TD-PD. 2 This discrepancy may stem from the fact that motor subtype classifications based on motor symptom clusters do not account for the distinct mechanisms behind individual symptoms, even within the same cluster. 11 Indeed, motor subtype classification typically relies on the ratio of tremor scores, which includes rest tremor (RT), kinetic tremor (KT), and postural tremor (PT), to scores for other motor symptoms like postural instability, gait disorder (PIGD), or akinetic-rigid features.12,13 While all tremors in PD are associated with the cerebello-thalamo-cortical circuit, their underlying mechanisms might vary. 14 The cerebellum may play a more dominant and causal role in action tremors (KT and PT) than in RT. KT is related to cerebellar lobule VIIB, whereas RT is related to lobule IV-V.14–16 Additionally, KT shows a poorer response to dopaminergic treatment compared to RT. RT has been associated with serotonergic dysfunction, a distinction not observed with KT or PT.17,18 These structural and neurotransmitter differences suggest that tremors might have different associations with PD pathology and prognosis.14,19 Considering the high prevalence of all three tremor types in PD and their potential co-occurrence within the same patient, grouping these tremors into a single category when subtyping may obscure important underlying differences. This may lead to varying underlying mechanisms for the motor subtypes (especially TD-PD), making it difficult to predict the prognosis. For example, patients exhibiting only RT, KT, or PT, as well as those patients with multiple types of tremors, are often classified as TD-PD despite variations in their dominant tremor type.18,20 Therefore, investigating the role of each tremor type in PD prognosis and their association with TD-PD and Non-TD-PD may help understand the heterogeneity of PD and the potential mechanism for the prognosis of motor subtypes of PD better. However, relevant studies are limited.
Another explanation for the inconsistent results on the prognostic value of the motor subtype might be its instability. As PD progresses, motor subtype can change within the same patient.8,21–24 This instability is associated with increased disease burden and more rapid symptom progression.22,24 Thus, the instability of the motor subtype may have an association with the prognosis of PD, suggesting that the fluctuating nature of motor subtypes should be carefully considered or statistically adjusted for in studies examining the prognostic value of the motor subtype. Additionally, the variability of tremor symptoms, which can appear or disappear as PD progresses, should also be considered in relevant studies.3,18
This study investigated the association between motor subtypes and the progression of PD in de novo, drug-naïve patients and the relationship between different types of tremors and PD progression. We considered all PD patients and those within specific motor subtype groups, taking into account the instability of subtypes and tremors throughout the disease course.
Methods
Participants
We included de novo drug-naïve PD patients from the Parkinson's Progression Markers Initiative (PPMI) cohort, an ongoing prospective, observational, international, multicenter study aimed at identifying clinical biomarkers of PD. 25 Detailed inclusion and exclusion criteria are available at www.ppmi-info.org. The study was approved by the institutional review board at all sites, and participants provided written informed consent, in accordance with the ethical standards outlined in the Helsinki Declaration of 1964.
Clinical evaluation
Demographic data, clinical assessment, and derivative data, such as cognitive state and indicator for Progression Milestones (PM) were obtained from the PPMI database in compliance with the Use Agreement.
The assignment of the motor subtype and presence of tremor was performed by reviewing the clinical data at baseline. Limb tremor presence was assessed by the corresponding part of the Movement Disorder Society-Unified Parkinson's Disease Rating Scale (MDS-UPDRS) (3.17a-3.17d for RT, 3.16a-3.16b for KT and 3.15a-3.15b for PT).26,27 The TD and non-TD subtype was defined by the ratio between the Tremor score / PIGD score according to previous study.11,25 A patient can thus be assigned into 4 subgroups, PD patients with or without RT, with or without KT, with or without PT, and TD/non-TD-PD regarding different grouping method.
Instability of Motor subtype or tremor subgroup were determined by reviewing longitudinal data. Patients were assessed for subtype or tremor presence during each follow-up, and instability was defined by changes in subtype or tremor presence over time. To exclude potential influence of daily tremor variability or residual effect of medications, assignment of instability requires two consecutive follow-up visits where the subgroup differs from that at baseline. 18
For the assessment of PD progression, we defined following outcomes: progression to Hoehn and Yahr staging 3 (HY3), progression to mild cognitive impairment (MCI), and the presence of several PM, including activities of daily living (ADL-PM), autonomic dysfunction (AD-PM), functional dependence (FD-PM), and walking & balance (WB-PM). 28 HY3 represents postural instability. 29 The MCI was defined as impairment (>1.5 standard deviations (SD) below the standardized mean scores (MD)), in the range recommended to establish MCI diagnosis on any two cognitive test scores and cognitive complaint. PM was selected as outcomes because they were found to be more stable and are independent of symptomatic therapy. 28 PM requires a test ≥1.5 SD below the standardized MD in the total scores of the corresponding items, which were listed in previous paper. 28 (Figure 1) For WB-PM, items include MDS-UPDRS 2.12 (walking and balance) and 2.13 (freezing), MDS-UPDRS 3.10-3.12 (gait, freezing of gait, and postural instability), and H-Y stage. For AD-PM, items include urinary incontinence part (MDS-UPDRS 1.10, SCOPA-AUT 8-9), orthostatic hypotension part (SCOPA-AUT item 15 and orthostatic blood pressure change), and syncope (MDS-UPDRS 1.12 and SCOPA-AUT item 16). FD-PM is assessed using the Schwab & England scale. ADL-PM is evaluated by MDS-UPDRS 2.3-2.6 (swallowing, eating, dressing, hygiene) and MDS-UPDRS 3.1 (speech).
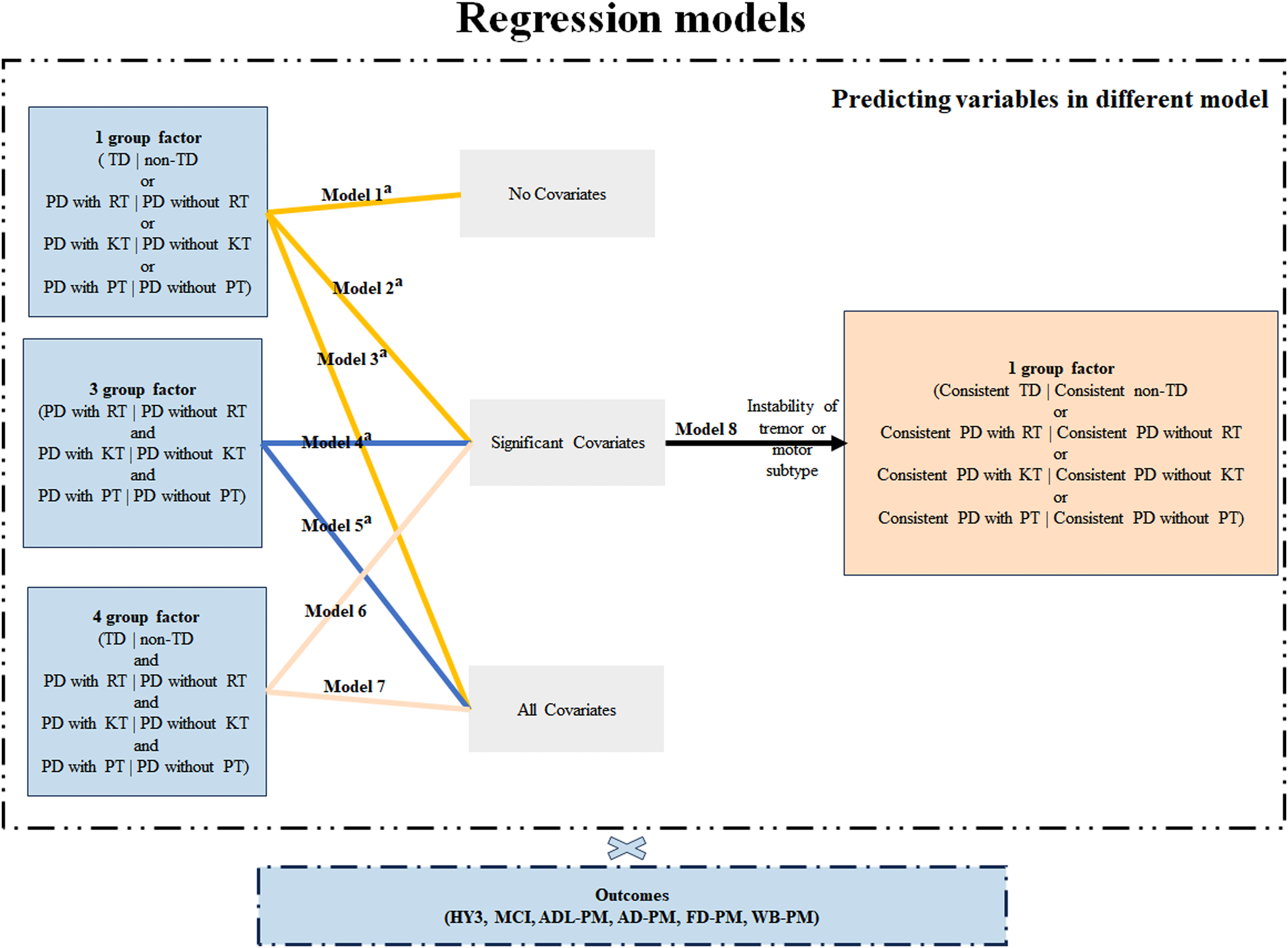
Illustration of the grouping method and analysis procedure. Consistency of tremor or motor subtype was determined by reviewing the adjacent visit. AD-PM: autonomic dysfunction progression milestone; ADL-PM: activities of daily living progression milestone; FD-PM: functional dependence progression milestone; HY3: Hoehn and Yahr staging 3; KT: kinetic tremor; MCI: mild cognitive impairment; Non-TD: non-tremor-dominant subtype; PD: Parkinson's disease; PT: postural tremor; RT: rest tremor; TD: tremor-dominant subtype; WB-PM: walking and balance progression milestones; a models were also tested in TD and Non-TD subtype.
Statistical analysis
Quantitative data were presented as mean ± SD, graded data as median (25th percentile, 75th percentile), and categorical data as ratios or percentages. The Chi-square test was used to compare the counting data, and two-sample independent t-test or Mann-Whitney U was used for measurement data. Data normality was tested using Kolmogorov–Smirnov test.
Survival analysis was conducted using Python 3.11.5 with lifelines v 0.27.7, with statistical significance set at p < 0.05. 30 The time of events was defined as the first recorded occurrence of the outcomes. Participants who did not experience the event during follow-up was coded as not having the event (right-censorship). Longitudinal analysis was carried using Kaplan-Meier survival estimates for each outcome based on different grouping methods, and differences in survival curves were assessed using the log-rank test.
At univariate level, Cox regression was performed with each subgroup variable (i.e. PD with RT vs. PD without RT, PD with KT vs. PD without KT, PD with PT vs. PD without PT and TD vs. non-TD, respectively) as independent variable (model-1). Additionally, univariate regression was conducted with confounding factors identified in previous studies (including age-onset, disease duration, sex, education years, MDS-UPDRS part III score, Montreal Cognitive Assessment, University of Pennsylvania smell identification test and H-Y staging) as independent variable.2,23,28,31 Multivariate analysis was then performed with each subgroup variable as the independent variable and significant factors from the univariate analysis as covariates (model-2).
To further assess the differential impact of tremor types on PD progression, a regression model with all three tremor subgroups (i.e. PD with RT and PD without RT, PD with KT and PD without KT, PD with PT and PD without PT, together) as independent variable was preformed, followed by a model including all four subgroups (i.e. with the TD/non-TD subgroup additionally) as independent variable was performed (model-4 and model-6), while accounting for significant confounding factors. Additionally, all confounding factors, not just those significant at the univariate level, were included in the multivariate models (model-3, model-5, and model-7). In both TD and non-TD subgroups, model-2 to model-5 were repeated to better illustrate the association between different tremor types and progression of PD. Finally, after adjusting for tremor or motor subtype instability and all potential biases, Cox regression was performed to validate priori results (Model-8) (Figure 1). For each outcome, patients who already reached the outcome at baseline were discarded. Cox proportional hazards assumptions tests based on graphical methods and Schoenfeld residuals were used to verify that proportional hazards assumptions were met 30 (Figure 1).
Results
Demographic and clinical character at baseline
At baseline, 537 patients were included, and the demographic and clinical characteristics for different grouping methods are presented in Table 1, TD-PD showed higher education years, lower Geriatric depression scale (GDS), State-trait anxiety inventory (STAI), and had lower MDS-UPDRS scores compared to Non-TD-PD. Patients with RT were older age at onset, had more years of education, and lower MDS-UPRDS part III scores (with and without tremor scores) than those without RT. Patients with KT had a lower percentage of the female sex, higher SCOPA and STAI scores, and higher MDS-UPDRS scores as well as H-Y staging than those without. Patients with PT had higher MDS-UPRDS part III scores (with tremor scores) than those without.
Demographic and clinical features at baseline regarding motor subtype or tremor presence.
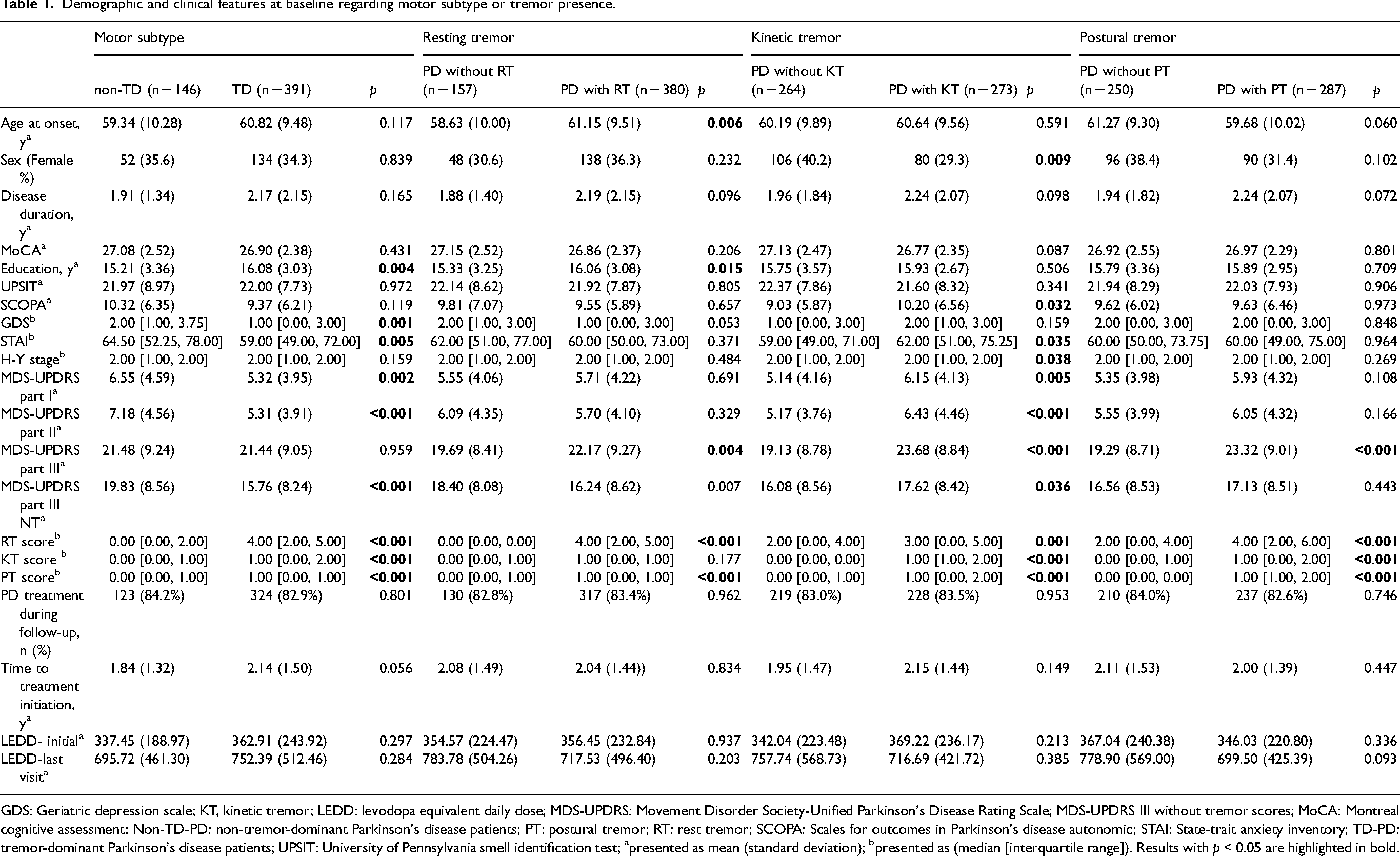
GDS: Geriatric depression scale; KT, kinetic tremor; LEDD: levodopa equivalent daily dose; MDS-UPDRS: Movement Disorder Society-Unified Parkinson's Disease Rating Scale; MDS-UPDRS III without tremor scores; MoCA: Montreal cognitive assessment; Non-TD-PD: non-tremor-dominant Parkinson's disease patients; PT: postural tremor; RT: rest tremor; SCOPA: Scales for outcomes in Parkinson's disease autonomic; STAI: State-trait anxiety inventory; TD-PD: tremor-dominant Parkinson's disease patients; UPSIT: University of Pennsylvania smell identification test; apresented as mean (standard deviation); bpresented as (median [interquartile range]). Results with p < 0.05 are highlighted in bold.
In accordance with previous reports, with the exception of PD with RT and PD without RT having similar scores in KT, patients with one type of tremor had higher tremor scores in all types of tremors18,20,32 (Table 1).
Progression to HY3
Five hundred thirty patients who had not reached the HY3 stage at baseline were included in the survival analysis, among whom 92 patients progressed to HY3 during the follow-up. After adjusting for covariates, TD-PD had a longer survival time compared to Non-TD-PD. Similarly, PD with RT had a longer survival time than PD without RT. When all three tremor types were considered together, PD with RT progressed to HY3 slower than PD without RT. When the motor subtype was additionally added, TD-PD patients progressed to HY3 slower than Non-TD-PD. The association between the presence of RT, the TD subtype, and slower progression to HY3 was confirmed after adjusting for the effects of tremor or motor subtype instability (Figures 2 and 3).
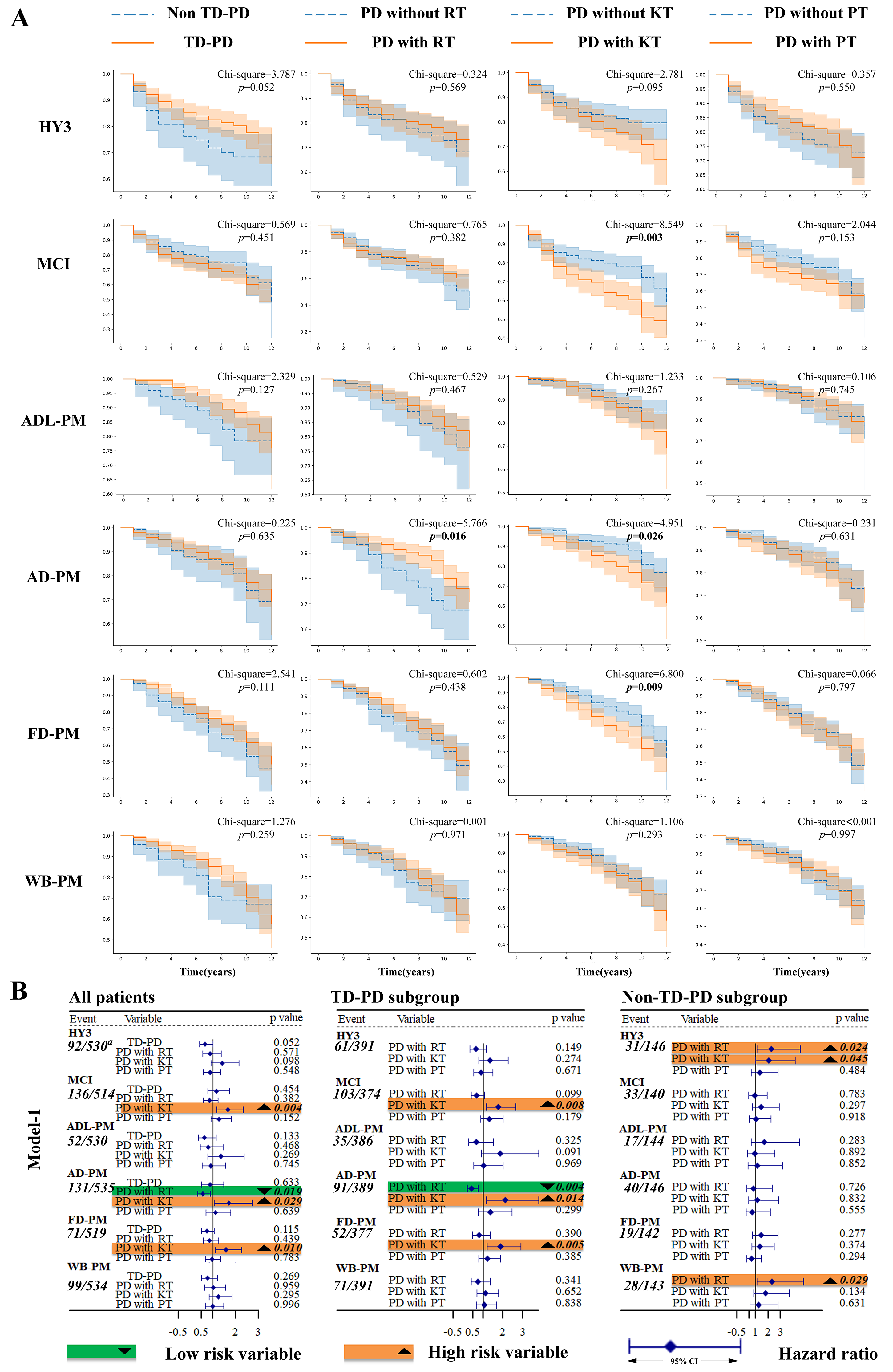
Kaplan-Meier curves and results for log rank test and univariate Cox regression (model-1). (a) Kaplan-Meier curves and results for log rank test (b) model-1. AD-PM: autonomic dysfunction progression milestone; ADL-PM: activities of daily living progression milestone; CI: confidence interval; FD-PM: functional dependence progression milestone; HY3: Hoehn and Yahr staging 3; KT: kinetic tremor; MCI: mild cognitive impairment; Non-TD-PD: non-tremor-dominant Parkinson's disease patients; PD: Parkinson's disease; PT: postural tremor; RT: resting tremor; TD-PD: tremor-dominant Parkinson's disease patients; WB-PM: walking and balance progression milestones. a Represents the ratio of subjects who reached the outcome at follow-up to all subjects who did not reach the outcome at baseline.

Results of multiple variate Cox regression (model-2 to model-8). AD-PM: autonomic dysfunction progression milestone; ADL-PM: activities of daily living progression milestone; CI: confidence interval; FD-PM: functional dependence progression milestone; HY3: Hoehn and Yahr staging 3; KT: kinetic tremor; MCI: mild cognitive impairment; Non-TD-PD: non-tremor-dominant Parkinson's disease patients; PD: Parkinson's disease; PT: postural tremor; RT: resting tremor; TD-PD: tremor-dominant Parkinson's disease patients; WB-PM: walking and balance progression milestones.
Progression to MCI
A total of 514 patients without MCI at baseline were included in the survival analysis, and 136 of them developed MCI during follow-up. The log-rank test and Cox regression revealed that PD with KT had a shorter survival time compared to PD without KT. Cox regression also found that PD with PT had a shorter survival time than PD without PT. Taking all types of tremors into one model, PD with KT still showed a shorter survival time than PD without KT, while PD with RT showed a longer one than PD without RT. When TD/non-TD was additionally added, the association between PD with KT or PD with RT and cognitive survival time persists. A similar result for KT was observed after adjusting for the effects of KT instability (Figures 2 and 3).
Progression to ADL-PM
At baseline, 530 patients who were not at the ADL-PM outcome were involved in subsequent analysis, with 52 patients reaching the outcome during follow-up. After adjusting for the effect of instability, log rank test and subsequent Cox regression found that consistent non-TD had a shorter survival time than consistent TD-PD (Figure 3).
Progression to AD-PM
At baseline, 519 patients who were not at AD-PM outcome were involved in subsequent analysis, with 71 patients reaching the outcome during follow-up. In all patients, the log-rank test found that PD with KT had a shorter survival time compared to those without KT, while PD with RT had a longer survival time compared to those without RT. Univariate and multiple variate Cox regression consistently showed that PD with RT had a longer survival time than PD without RT. Additionally, univariate Cox regression indicated that PD with KT had a shorter survival time than PD without KT. When all types of tremors were included in the model together, PD with RT had a shorter survival time than PD without RT. Similar results were observed when the motor subtype was considered. After adjusting for the effects of instability, PD with RT still had a longer survival time compared to PD without RT. Furthermore, Cox regression also found that Consistent TD-PD had a longer survival time compared to Consistent non-TD (Figures 2 and 3).
Progression to FD-PM
At baseline, 535 patients who were not at the FD-PM outcome were involved in subsequent analysis, with 131 patients reaching the outcome during follow-up. Both log-rank test and univariate Cox regression showed that PD with KT had a shorter survival time than PD without KT. Multivariate regression, whether examining individual tremor types or all tremor types together (model-2 to model-5), demonstrated the association. When the motor subtype was added, PD with KT continued to show a shorter survival time than PD without KT. After adjusting for the effects of tremor or motor subtype instability, similar results were found for KT and RT, while TD-PD was also found to have a longer survival time than non-TD (Figures 2 and 3).
Progression to WB-PM
At baseline, 534 patients who were not at the WB-PM outcome were involved in subsequent analysis, with 99 patients reaching the outcome during follow-up. Cox regression found that TD-PD had a longer survival time than Non-TD-PD (model-3). Additionally, when all types of tremors were considered, PD with RT had a longer survival time than PD without RT (model-5). These results remained consistent after adjusting for the effects of RT or motor subtype instability (Figures 2 and 3).
Results for all subgroup analyses are detailed in Figures 2 and 3. The definite (adjusted) hazard ratio and confidence interval (CI) for all analyses concerning each outcome are provided in the Supplemental Material.
Discussion
In this study, we found that TD-PD was associated with a slower progression of PD. Moreover, RT was associated with slower progression of PD, while KT and PT were associated with rapid progression. These associations were demonstrated after adjusting for the effect of motor or tremor instability.
This study found that TD-PD progressed to HY3 and WB-PM more slowly compared to Non-TD-PD. Furthermore, consistent TD-PD progressed to HY3, ADL-PM, AD-PM, FD-PM and WB-PM more slowly compared with consistent non-TD-PD. These findings are in line with previous studies that found TD-PD presented slower progression, lower burden of motor/non-motor symptoms, and better quality of life compared to PIGD or AR-PD subtypes.4,7,8–10,33–36 These results may be attributed to the relatively lower pathological burden in TD-PD and support the notion that TD-PD is a “benign marker” of the disease.2,3,5,26,36–39
Previous studies often classified motor subtypes of PD using combined tremor scores. 3 However, few have specifically examined the prognosis of PD based on individual tremor types. In this study, we found that PD with RT progressed more slowly to HY3, MCI, AD-PM, FD-PM, and WB-PM. These associations remained significant even after adjusting for motor subtypes, when analyzing in the TD-PD subgroup, or after adjusting for tremor instability during the disease course (Figure 3). Interestingly, both PD with RT and TD-PD were associated with slower progression to HY3 and WB-PM, whereas the other tremor types (KT and PT) were linked to faster disease progression. It seems that RT, rather than KT or PT, best predicts the clinical advantages observed in TD-PD. Accordingly, previous studies have found clinical and pathophysiological similarities between PD patients with RT and TD-PD. Clinically, PD patients with tremor (primarily RT) have been found to perform better cognitively than those without tremor. 34 Pathologically, PD patients with tremor (mainly RT) have less severe dopaminergic defects in the caudate nucleus compared to patients without tremor, and PD with RT have less severe dopaminergic defects in putamen and pallidum than PD without RT.5,40 These findings mirror the milder defects reported in TD-PD.2,26,37 Accordingly, in a recent study, we specifically examined PD patients with RT, excluding those with kinetic or postural tremors, and found that PD with RT has better white matter integrity compared with healthy controls or PD without RT, consistent with findings in TP-PD.6,37,41–43 Further study is needed to explore the clinical and pathophysiological associations between RT and TD-PD over a longer follow-up period.
Our study also found that PD with KT is associated with a greater burden of motor and non-motor symptoms at baseline and a faster progression to MCI-PM and FD-PM. Furthermore, consistent PD patients with KT progress more rapidly to MCI and FD-PM compared with consistent PD without KT. These findings are in line with previous reports that PD patients with KT exhibit more severe motor symptoms than those with only RT, both at baseline and during follow-up, suggesting that PD with KT might have a faster disease course than PD without KT.
18
Moreover, considering that RT is related to a slower progression in PD, KT may underlie different mechanisms in relation to RT. In fact, KT has a higher tremor frequency than RT and is supposed to be a PD-related enhancement of the physiological tremor, while RT is associated with pathological activity originating from the basal ganglia
In our study, PT was associated with faster progression to MCI. Unlike KT or RT, limited outcomes were associated with PT. A possible explanation is that PD patients with PT in our study consist of individuals with tremors related to opposing outcomes. Recent research suggests that PT in PD may consist of two distinct components: re-emergent tremor, which occurs in about 80% of patients and resembles RT in terms of amplitude suppression, frequency, and response to dopaminergic treatment; and pure PT, which differs from RT in these aspects and occurs in about 20% of patients.47,48 Moreover, similar to RT, re-emergent tremor has been causally linked to motor cortex activity. 49 However, because the MDS-UPRDS scale does not differentiate between re-emergent tremor and pure PT, our study, like many previous studies relying on clinical examination, likely included both types of tremors. 27 Suppose pure PT is associated with a ‘worse’ disease prognosis, while re-emergent tremor shares mechanisms with RT (which is linked to a more ‘benign’ disease course). In that case, the effects of these two tremor types may counteract each other, resulting in limited associations between PT and disease progression. 47 Further studies using EMG to help distinguish the two types of tremors, and to understand their association with PD prognosis and potential mechanism, are needed. Additionally, it is possible that the limited associations observed in our study are due to the relatively small sample size, indicating that larger studies may be necessary to confirm these findings.
Our study has several limitations: Since patients were only evaluated at prescheduled visits, it is only known that criteria became evident at some point during the interval between one visit and the next. Planned validation efforts should apply methods tailored specifically to interval-censored data. 28 Furthermore, as an exploratory study with relatively limited sample size and without multiple comparison corrections, we did not involve interaction effect or perform subgroup analysis among or regarding different types of tremors. Further studies were required to investigate possible interactions among different types of tremors with a larger sample size and multiple comparison corrections that help decrease the false discovery rate. Besides, although classifying PD based on tremor types may provide valuable insights into disease prognosis and mechanism and may assist in identifying potential biomarkers, this classification may not directly reflect or fully capture the underlying disease mechanisms or synuclein pathology. Future research may benefit from integrating a range of biomarkers across multiple domains to improve predictions of PD prognosis and deepen our understanding of the disease mechanisms. Besides, our grouping method may not achieve the best prediction of disease prognosis, and studies may need to incorporate additional information to improve predictive power and extend the clinical significance further.
This study identified an association between TD-PD motor subtype and RT presence with slower progression of PD. In contrast, the presence of action tremor, especially KT, was associated with faster progression. Different types of tremors are associated with distinct prognoses in PD, suggesting potential differences in brain damage patterns and underlying pathophysiological mechanisms, which need to be further investigated.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241305715 - Supplemental material for Association of motor subtype and tremor type with Parkinson's disease progression: An exploratory longitudinal analysis
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241305715 for Association of motor subtype and tremor type with Parkinson's disease progression: An exploratory longitudinal analysis by Yuke Zhong, Huahua Su, Ying Liu, Hang Liu, Guohui Liu, Zhihui Liu, Jiahao Wei, Junyi Wang, Yuchen She, Changhong Tan, Lijuan Mo, Lin Han, Fen Deng, Xi Liu and Lifen Chen in Journal of Parkinson's Disease
Supplemental Material
sj-docx-2-pkn-10.1177_1877718X241305715 - Supplemental material for Association of motor subtype and tremor type with Parkinson's disease progression: An exploratory longitudinal analysis
Supplemental material, sj-docx-2-pkn-10.1177_1877718X241305715 for Association of motor subtype and tremor type with Parkinson's disease progression: An exploratory longitudinal analysis by Yuke Zhong, Huahua Su, Ying Liu, Hang Liu, Guohui Liu, Zhihui Liu, Jiahao Wei, Junyi Wang, Yuchen She, Changhong Tan, Lijuan Mo, Lin Han, Fen Deng, Xi Liu and Lifen Chen in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by National Natural Science Foundation of China (grant number: 82371262, receiver: Lifen Chen), Natural Science Foundation of Chongqing (grant number: CSTB2023NSCQ-MSX0104, receiver: Changhong Tan), Kuanren Talent Program of The Second Affiliated Hospital of Chongqing Medical University (receiver: Xi Liu). Scientific and Technological Research Program of Chongqing Municipal Education Commission (grant number: KJQN202200417, receiver: Changhong Tan). The PPMI is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, the full list of PPMI funding partners can be found at ppmi-info.org/fundingpartners.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
