Abstract
Background
Deep brain stimulation (DBS) is effective in managing motor symptoms in select cases of Parkinson's disease (PD). Nonetheless, the ideal timing for surgery and the comparative outcomes of unilateral versus bilateral DBS procedures remain under-researched areas.
Objective
We aimed to compare the impact of unilateral and bilateral DBS on the motor manifestations of PD using standardized Unified Parkinson's Disease Rating Scale Part-III (UPDRS-III).
Methods
We conducted a retrospective analysis of PD patients who underwent multidisciplinary DBS screening which made a formal recommendation for surgical approach. We compared unilateral, bilateral “rapid” (less than 2 months apart), and bilateral “staged” (5–11 months apart) implantation approaches. The study included 90 patients, 48 patients, and 42 patients from the 3 groups, respectively. The primary outcome was the percentage improvement in baseline off UPDRS-III scores compared to medication-off/DBS-on conditions at 3–6 months and 10–14 months post-surgery. Mann-Whitney U tests were used to compare scores within groups and across follow-up periods. The Kruskal-Wallis test assessed differences among groups. Furthermore, multiple regression analyses were performed to adjust for confounding variables.
Results
UPDRS-III scores improved significantly from baseline at both follow-up intervals regardless of the type of DBS staging approach. The Kruskal-Wallis test revealed no significant differences in UPDRS-III percentage improvement among groups at 3–6 months (p = 0.125) and 10–14 months (p = 0.298) post-DBS.
Conclusions
Our study revealed that in a single experienced DBS center which employed multidisciplinary screening, assignment to unilateral and bilateral DBS, both rapid and staged, targeting the STN or GPi, effectively improved motor symptoms for up to 14 months.
Plain language summary
Deep brain stimulation is a surgical treatment that helps manage motor symptoms in select people with Parkinson's disease (PD). However, the best timing for surgery and whether it's better to have DBS implanted on one side of the brain (unilateral) or both sides (bilateral) from the beginning are still unclear. This study compared the effects of unilateral and bilateral DBS on movement problems in 180 PD patients. The patients were divided into three groups: 90 had DBS on one side, and 90 had DBS on both sides implanted in a staged manner, either quickly (named bilateral rapid, with surgeries done less than 2 months apart) or more slowly (named bilateral staged, with surgeries done 5–11 months apart). We measured their movement problems using a standard motor assessment after surgery at two different times: 3–6 months and 10–14 months; and we compared that to their baseline scores. We found that all groups showed significant improvement in their movement problems after DBS. There were no major differences in improvement between those who had one side done and those who had both sides done, whether quickly or slowly, up to 14 months after surgery. In conclusion, DBS, whether done on one side or both sides of the brain and regardless of the timing of staging between surgeries, significantly improved movement problems in PD patients for up to 14 months after surgery.
Keywords
Introduction
Parkinson's disease (PD) is the most rapidly growing neurological disease and it is anticipated that it will increase the socioeconomic burden on society as the global population grows and ages.1,2 Deep brain stimulation (DBS) in well screened PD populations has emerged as a safe and effective neuromodulation therapy both for several important motor symptoms and for enhancement of quality of life. 3 Despite the widespread use of this surgical approach, the optimal surgical strategy for unilateral versus bilateral approaches and the associated timing of staging of the procedure remains unknown.
The subthalamic nucleus (STN) and globus pallidus internus (GPi) are the primary targets used for DBS in PD, although occasionally the ventral intermedius nucleus of the thalamus is used in cases with isolated severe resting and intentional tremor. Perhaps the most common surgical approach is simultaneous bilateral DBS implantation, a procedure which is characterized by insertion of a DBS lead into each brain hemisphere during the same operation. There are, however, merits to unilateral or staged bilateral approach. Some of these alternative approaches have been shown to be associated with long-term motor improvement for up to 5 years.4–9 In a staged approach, a single lead is implanted during the initial DBS surgery and a contralateral lead is implanted only when symptoms in the contralateral hemibody worsen (i.e., a symptom such as dyskinesia or tremor emerges on the contralateral side and is disabling enough to consider a second sided DBS surgery).
Collective data has revealed that in DBS surgery the first lead provides the most benefit, especially given that the first lead usually targets the more affected side.6,9–11 Additionally, in persons with Parkinson's manifesting low off UPDRS motor scores (i.e., < 30) and those with predominantly unilateral disease, a single DBS lead may address the disability and avoid the potential for the known increased side effects of bilateral surgery.8,12,13
We conducted a retrospective analysis to evaluate motor outcomes across unilateral and bilateral operations and we a priori hypothesized that in cases pre-screened by a DBS multidisciplinary team for selection of the approach to DBS, the motor outcomes would be similar regardless of the approach. We assessed outcomes up to 14 months post-surgery.
Methods
Patient population
A retrospective study approved by the University of Florida (UF) Institutional Review Board (IRB) was conducted for all patients diagnosed with PD who underwent STN or GPi DBS surgery between January 2019 and September 2023. Patients were required to have a multidisciplinary evaluation (Figure 1) which included a discussion of one of three approaches to surgery (unilateral, staged bilateral or staged “rapid” bilateral implantation). Patient demographic data and standardized motor assessments were collected using the Unified Parkinson's Disease Rating Scale Part III (UPDRS-III) and were systematically extracted. Our inclusion criteria were: (1) Diagnosis of PD by a fellowship trained movement disorders neurologist per the Movement Disorders Society criteria 14 ; (2) DBS implantation, either unilateral or bilateral; (3) Surgeries performed at UF; (4) Availability of pre-operative baseline UPDRS-III scores in the medication-Off state; (5) At least one follow-up UPDRS-III score in the medication-Off and DBS-On state, conducted between 3–6 months and/or 10–14 months post-implantation surgery. Exclusion criteria were: (1) Prior DBS leads implanted at other institutions or other previous neurosurgical intervention (2) A diagnosis of atypical parkinsonism any time after DBS lead implantation.
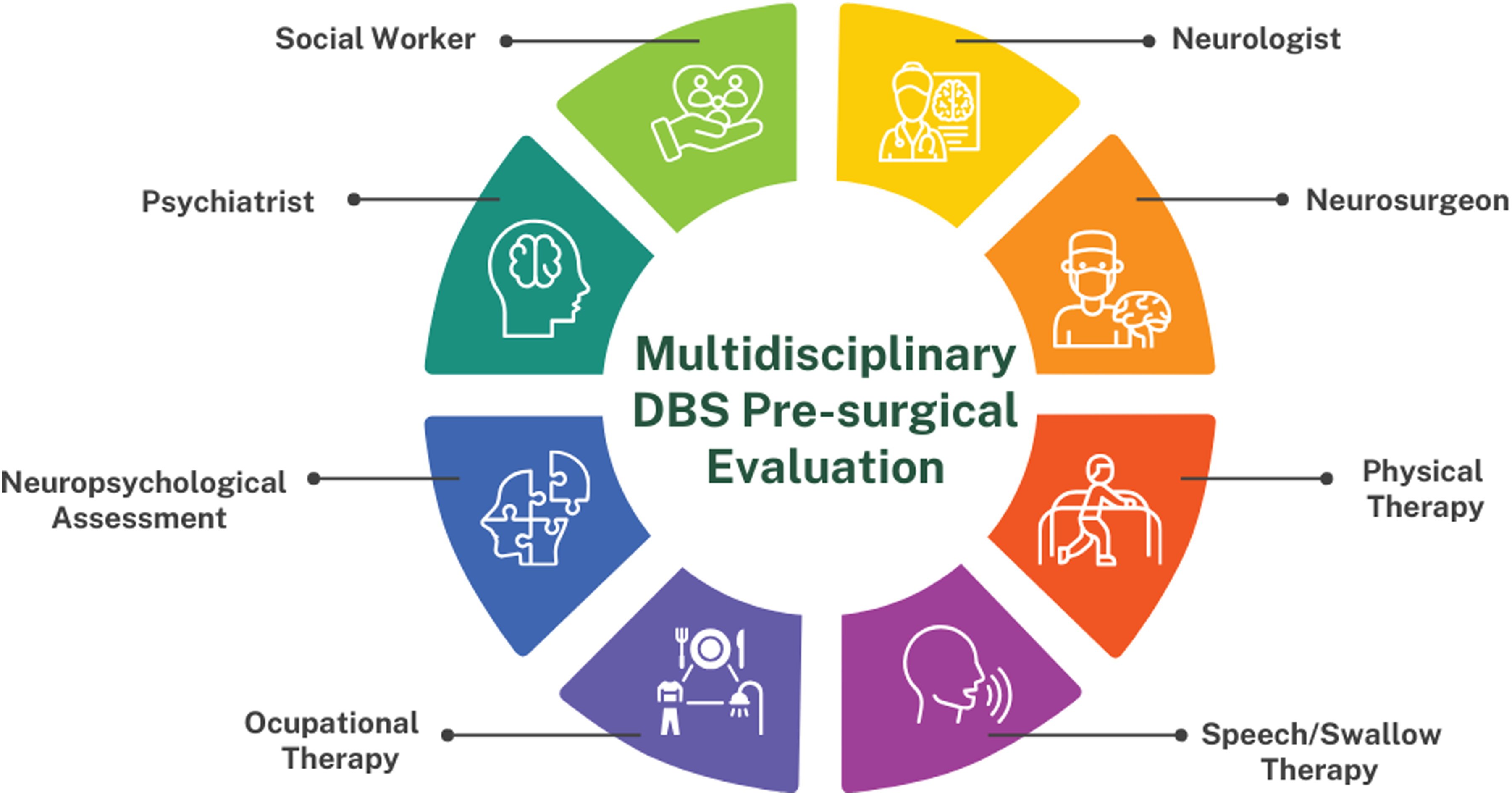
Multidisciplinary team evaluation process for DBS surgery at the University of Florida. A schematic representation outlining the involvement of professionals in the pre-surgical assessment process for deep brain stimulation (DBS) surgery at the University of Florida.
In this study, we categorized participants into three distinct groups based on the multidisciplinary recommendation for staging their DBS surgery. All patients included underwent the type of operation recommended by the multidisciplinary team. The unilateral DBS cohort consists of patients who underwent unilateral DBS surgery. We further identified two groups among patients who received bilateral lead implantations. The first group, termed “bilateral rapid,” includes patients who underwent bilateral surgeries with no more than 60 days between lead implantations. The second group, “bilateral staged,” encompasses patients who underwent bilateral lead implantations with an inter-surgery interval ranging from 5 to 11 months. Patients in both bilateral groups were required to have a medication-off/DBS-on UPDRS-III motor score recorded between 3–6 months and/or 10–14 months post-lead implantation. For the “bilateral rapid” group, the UPDRS-III scores were obtained considering the date of the second lead implantation, whereas for the “bilateral staged” group, the scores were based on the date of the first lead implantation.
Surgical procedure
All DBS surgeries were performed at a single center (University of Florida). Patients underwent a multidisciplinary team evaluation before DBS surgery (Figure 1). On the day of the surgery, an atlas-based anatomical mapping of the target location was performed on a pre-operative CT scan fused with a 3 T MRI image. Further guidance for lead implantation was obtained from the intraoperative microelectrode recordings and macrostimulation testing. The DBS lead was implanted under local anesthesia and a postoperative CT scan was performed and fused with the preoperative MRI image for confirmation of lead location. Pulse generator implantation surgery was scheduled within 2 weeks of lead implantation surgery. 15
Assessments
Clinical outcomes were compared between the three groups at two predefined time points: 3–6 months and 10–14 months after the implantation. The primary outcome measure was the UPDRS-III motor score. Evaluations were obtained preoperatively as part of our regular pre-surgical work up in the medication-off state, defined as withholding the intake of dopaminergic medications for at least 12 h. Postoperative clinical scales were obtained in the medication-off / DBS-on state.
Statistical analysis
All statistical analyses were conducted using Minitab 21 (Minitab, LLC, United States). Non-parametric Kruskal-Wallis tests were employed to identify statistically significant differences in the percentage improvement of UPDRS-III scores between the three groups at each time point and to assess potential baseline differences among the groups. Additionally, Mann-Whitney U tests were utilized to assess variations in UPDRS-III scores within each group across different time points.
Furthermore, a multiple regression analysis was conducted to control for potential confounding variables including demographics (gender, age at DBS surgery), clinical characteristics (disease duration before first surgery, baseline UPDRS Off medication score, pre-operative levodopa responsiveness, asymmetry index - calculated according to Pltonik et al. 16 ), and surgical factors (surgical target; unilateral vs. bilateral rapid vs. bilateral staged approaches). This analysis aimed to isolate the independent effect of the surgical approach on UPDRS-III score improvement while accounting for other relevant factors. We constructed two independent models, one for the 3–6-month follow-up end point, and another for the 10–14 month follow-up end point.
Results
A total of 90 patients (62 men, 28 women) who met our study criteria for the unilateral DBS cohort were included for analysis. The mean age (SD) at time of surgery was 68.3 (8.5) years old. Within this group, 54 patients (60%) received GPi DBS, and 36 (40%) received STN DBS. The mean disease duration (SD) at surgery was 9.7 (4.6) years. Mean (SD) baseline asymmetry index for this group was 0.30 (0.26).
A total of 48 patients (33 men, 15 women) who met our study criteria for the bilateral rapid DBS cohort were selected for comparison. The mean age (SD) at time of surgery was 60 (10) years old. The mean disease duration (SD) at first surgery was 11.9 (6.7) years. GPi was the target of choice in 35 patients (73%), while 13 (27%) received STN DBS. Mean (SD) baseline asymmetry index for this group was 0.18 (0.18).
Lastly, a total of 42 patients (28 men, 14 women) met the study criteria for the bilateral staged group. The mean age (SD) at time of surgery was 68.3 (7) years old. The mean disease duration (SD) at first surgery was 10.7 (3.9) years. GPi was the target of choice in 31 patients (74%), while 11 (26%) received STN DBS. Mean (SD) baseline asymmetry index for this group was 0.15 (0.13).
For the unilateral group, the mean (SD) pre-operative UPDRS-III score in the medication-off state was 34.3 (10.8). At 3–6 months post-DBS implantation, the mean UPDRS-III score in the Medication-off/DBS-on state was 23.2 (10.1). Subsequently, at 10–14 months post-implantation, the mean UPDRS-III score in the Medication-Off/DBS-On state was 25.5 (11.7). The mean UPDRS-III scores at 3–6 and 10–14 months were significantly lower compared to pre-DBS (p < 0.001 and p = 0.002, respectively).
For the bilateral rapid cohort, the mean (SD) pre-operative UPDRS-III in the medication-off state was 36 (9.9). Follow-up UPDRS-III Medication-off/DBS-on means (SD) were 24.3 (9.1) at 3–6 months after DBS implantation and 26.7 (11.25) at 10–14 months post-implantation. The mean follow-up UPDRS-III at 3–6 months and 10–14 months post-implantation were also significantly lower compared to pre-DBS in the bilateral group (p < 0.001 for both).
For the bilateral staged group, the mean (SD) pre-operative UPDRS-III in the medication-Off state was 40.6 (11.15). Follow-up UPDRS-III Medication-off/DBS-on means (SD) were 30.4 (10.4) at 3–6 months after DBS implantation and 26.5 (9.2) at 10–14 months post-implantation. The mean follow-up UPDRS-III at 3–6 months and 10–14 months post-implantation were also significantly lower compared to pre-DBS in the bilateral group (p < 0.001 for both).
Following unilateral lead placement, the median (IQR) percent improvement in UPDRS-III scores was 32.5% (20.2%–46.4%) at 3–6 months and 22.2% (4.35%-43%) at 10–14 months. In the bilateral rapid cohort, the median (IQR) improvement was 30.4% (16%-46.7%) at 3–6 months and 29.7% (13.4%-40.2%) at 10–14 months. For the bilateral staged group, the median (IQR) percent improvement was 20.85% (8%-39.5%) at 3–6 months and 30.2% (20%-45.3%) at 10–14 months (Figure 2). The Kruskal-Wallis test indicated no significant differences in the percentage improvement in UPDRS-III scores among groups at 3–6 months (χ²=4.159, p = 0.125) and at 10–14 months (χ²=2.419, p = 0.298) following DBS placement.
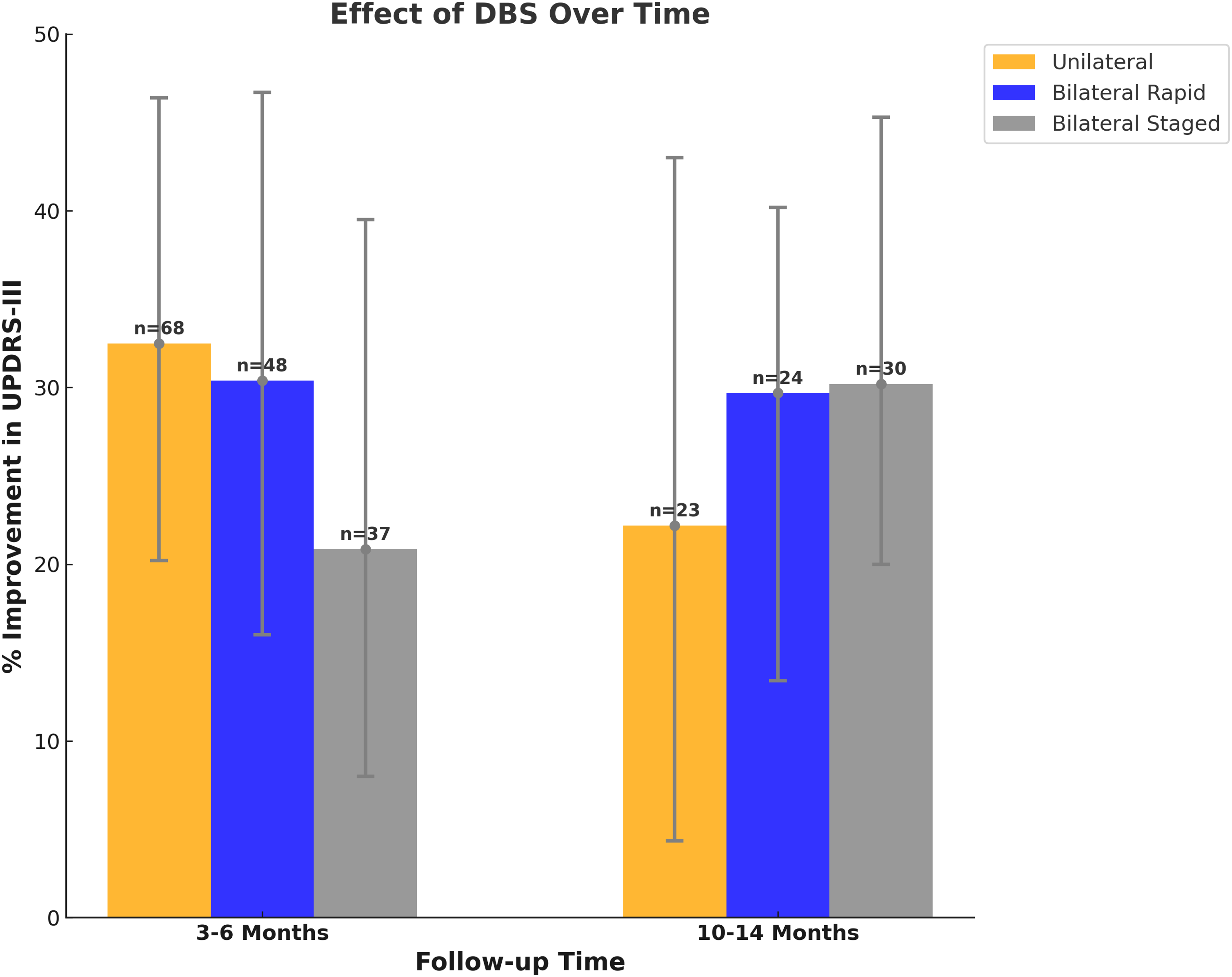
Effect of DBS over time. This figure illustrates the percentage improvement in UPDRS-III scores following deep brain stimulation (DBS) therapy over time, segmented by treatment approach: Unilateral (orange), Bilateral Rapid (blue), and Bilateral Staged (grey). Improvement is measured at two key intervals, 3–6 months and 10–14 months, post-treatment. The bars represent the median percentage improvement within each group, while the error lines indicate the interquartile range. Patient counts (N) for each group are displayed above the corresponding bars, highlighting the sample size for each observation. Notably, statistical analysis using the Kruskal-Wallis test revealed no significant difference in the percentage change across the three treatment groups, suggesting that each DBS treatment approach yields comparable improvement in UPDRS-III scores over the observed time frames.
Statistical analyses of baseline data (Table 1) indicated no significant differences in the percentage change following a Levodopa challenge (H = 4.54, p = 0.103) or in disease duration (H = 4.410, p = 0.110) across the groups. Notably, significant differences were observed in age (H = 24.22, p < 0.00001), baseline UPDRS scores off medications (H = 7.924, p = 0.019), and in the asymmetry index (H-statistic: 14.63, p-value: 0.0007). However, after adjusting for confounding covariates via multiple linear regression analysis (refer to Table 2), age and asymmetry index were not significantly associated with UPDRS outcomes at follow-up. This suggests that despite the possible selection bias of younger patients for bilateral procedures and patients with more asymmetry in motor symptoms for unilateral procedures, that difference did not significantly influence the outcomes analyzed (Supplemental Table 1). Conversely, baseline UPDRS-III scores in the medication-off state prior to DBS surgery were significantly associated with the percentage of improvement at both the 3–6 months (p = 0.001) and 10–14 months (p = 0.001) follow-up intervals. Additionally, targeting the subthalamic nucleus was significantly correlated with the percentage improvement at 3–6 months (p = 0.002), a significance that persisted at the 10–14 months follow-up (p = 0.011). Disease duration until the first surgery, gender, percentage change following a Levodopa challenge, and surgical approach did not independently influence UPDRS percentage improvement.
Baseline characteristics of patients in the study. The table presents baseline (pre-surgical) demographic characteristics, pre-surgical levodopa responsiveness, UPDRS-III scores (off-medication), asymmetry index, and surgical targets for all patients included in the cohort. Data are reported as means with standard deviations (SD), except for sex and target; the latter is shown as N (% of the subgroup). Age, baseline UPDRS-III off-meds and asymmetry index were significantly different between the groups. Dunn's post-hoc analysis comparing asymmetry index across groups was performed, revealing significant differences in the asymmetry index between the Unilateral group and each of the Bilateral Rapid and Bilateral Staged groups, indicating distinct asymmetry profiles. Conversely, the Bilateral Rapid and Bilateral Staged groups did not exhibit a significant difference in their asymmetry indices.

*p < 0.05 and **p < 0.001. DBS: deep brain stimulation; M: male; F: female; Meds: medications; LCT: levodopa challenge test.
Regression models coefficients and p-values.
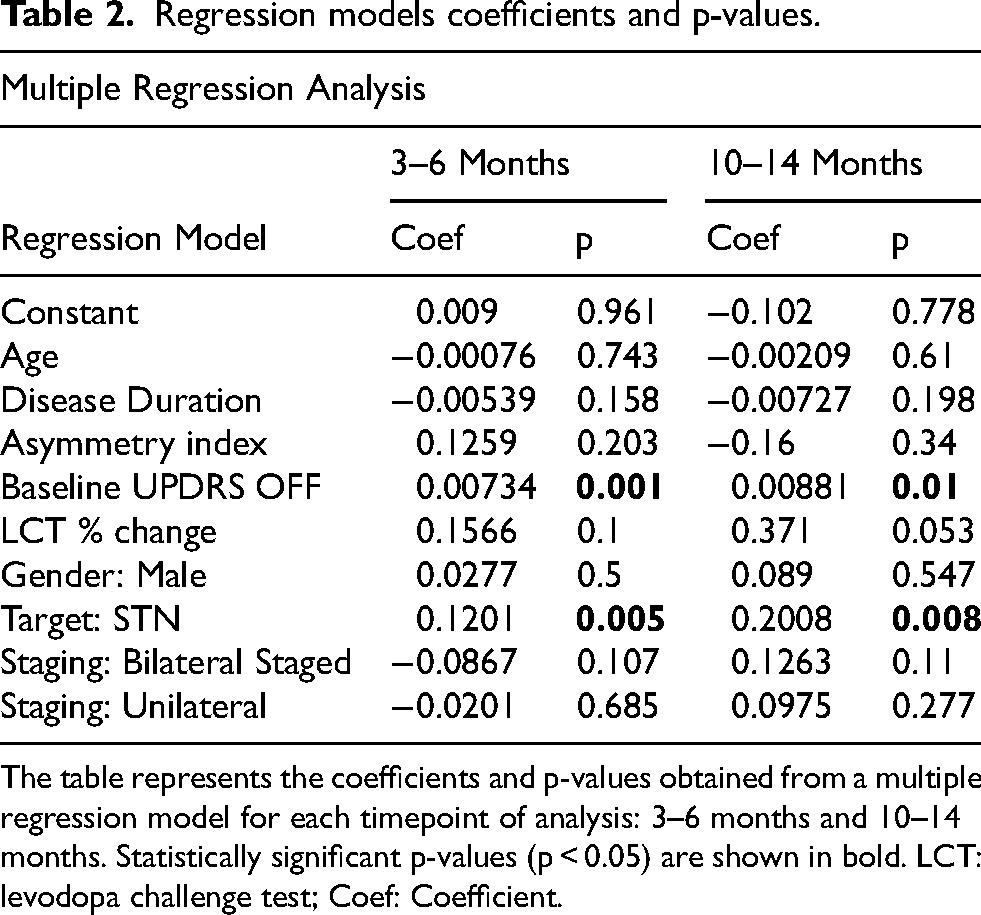
The table represents the coefficients and p-values obtained from a multiple regression model for each timepoint of analysis: 3–6 months and 10–14 months. Statistically significant p-values (p < 0.05) are shown in bold. LCT: levodopa challenge test; Coef: Coefficient.
Discussion
The results from this single center study revealed that when a multidisciplinary team was used to select between unilateral, bilateral rapid, and bilateral staged DBS, targeting either the STN or GPi, the motor outcomes were comparable when examined up to 14 months postoperatively. Despite the widespread use of bilateral simultaneous DBS implantation, recent data comparing it to unilateral or staged bilateral approaches remains limited. This data suggests that all three options may be viable and should be considered in the context of each patient's medical history, risk factors, and disabling features.
The most commonly employed option for DBS surgery worldwide as judged by the volume of literature on the topic is simultaneous bilateral implantation. This is employed in some centers regardless of age or asymmetry in motor symptom burden. Our observations are congruent with prior studies which reported positive effects on motor scores and quality of life in patients who undergo unilateral DBS surgery.4,6–8,10,14 In select cases, these benefits were significant enough to either postpone or eliminate the necessity for contralateral surgery.4,8,17
Taba and colleagues analyzed data from 44 patients included in the NIH's randomized STN vs. GPi COMPARE PD DBS trial, focusing on their decisions regarding a second DBS implant. Within this trial, all patients were given the option to have a contralateral DBS implant after 6 months of the unilateral implant. Out of the total, 21 patients did not undergo bilateral implantation and they were successfully treated for an average of 3.5 years. Interestingly, 14 (67%) had a GPi target. Patients who opted out of a second DBS surgery cited satisfaction with the motor improvements from the first procedure as their reason. Conversely, those who pursued a second DBS lead displayed significantly higher initial UPDRS-III motor scores and lower asymmetry indices. Logistic regression analysis indicated that patients initially treated at the STN were 5.2 times more likely to undergo bilateral DBS than those treated at the GPi. Moreover, the study found baseline asymmetry crucial in decision-making; for every 1% increase in asymmetry, the likelihood of opting for bilateral DBS decreased by 0.96. 9
In our study, we observed a median improvement of 32.47% in the medication-off/DBS-on UPDRS-III scores at 3–6 months in the unilateral DBS cohort. This was consistent with previous reports in the literature, ranging from 24% to 65%, as summarized by Hayashi et al. 18 While our study provides valuable insights, it is important to acknowledge its limitations. First, we did not analyze the specific reasons why the multidisciplinary team chose a unilateral or staged approach for each individual. Generally, a candidate who is recommended for bilateral staged treatment have significant bilateral disease with manageable comorbid conditions (including but not limited to balance, speech, and swallowing issues). Our dataset did not capture or analyze changes in medication following DBS surgery. Previous research by Slowinski et al. demonstrated a mean reduction of 21% in medication burden coincident with significant contralateral motor improvement after unilateral DBS. 8 Further research will thus be needed to elucidate the potential influence of medication adjustments on outcome. Additionally, our investigation did not explore ipsilateral motor improvement, despite emerging evidence suggesting its occurrence after unilateral STN and GPi DBS.18–21
Patients who underwent unilateral STN surgery demonstrated a significantly greater mean improvement in motor scores at 3–6 months and 10–14 months postoperatively compared to those receiving GPi stimulation at the same time point. This finding of greater short-term benefits with STN-DBS is consistent with prior studies by our group, as reported by Cernera et al. 22 for quality-of-life improvement and by Wong et al. 15 for tremor suppression. Notably, in the context of tremor suppression, this advantage diminished over time, leading to no discernible superiority of either brain target at 1-year follow-up. 15 It is noteworthy that our prior study also reported that in the case of staged bilateral DBS the overall improvement in quality of life was primarily attributed to the effects of the first lead. 22 Finally, this data should be interpreted cautiously as the targets for this study were not randomized and the UPDRS-III motor score does not capture perhaps the most important outcome of DBS surgery: reduction in motor fluctuations and dyskinesia.
The reduced intraoperative time associated with unilateral DBS implantation makes it a potentially attractive option, particularly for older patients who may be at increased risk from prolonged surgery. However, concerns exist regarding potential cost escalation and additional surgical risk due to the need for two separate procedures. Notably, Petraglia et al. conducted a small comprehensive study comparing simultaneous bilateral and staged unilateral DBS in PD patients. 12 Their findings demonstrated no significant differences in the 90-day postoperative rates of various complications, including infection, pneumonia, hemorrhage, and device-related issues. Additionally, no significant differences were observed in mean annualized costs between the groups, suggesting that operative risk and healthcare costs may not be primary factors influencing the choice between approaches. It is crucial to acknowledge that these findings may not be generalizable to all healthcare settings and their sample size was small. Thus, the data should be interpreted with caution.
Studies have demonstrably revealed an increased incidence of adverse effects following bilateral DBS as compared to unilateral approaches, including but not limited to cognitive decline, behavioral changes, gait impairment, speech impairment, and eyelid-opening apraxia.12,13,17,23–25 This finding may have significant implications for patient selection and for the timing of surgical procedures. At our institution, patients always undergo a multidisciplinary evaluation prior to DBS surgery to assess candidacy, target, approach, management, and co-morbidities which may impact outcome or post-operative approach. Surgical candidates with an increased risk for post-surgical deterioration in speech, gait, balance, swallowing, or neuropsychiatric function may require a second multidisciplinary evaluation before proceeding with the second lead implantation when employing a staged approach (Figure 3). This precaution has been employed by our group and other groups to minimize the risk of adverse effects. The results from our current study suggest that when appropriately applied, a prolonged staged approach can provide similar motor benefits to bilateral simultaneous DBS surgery.
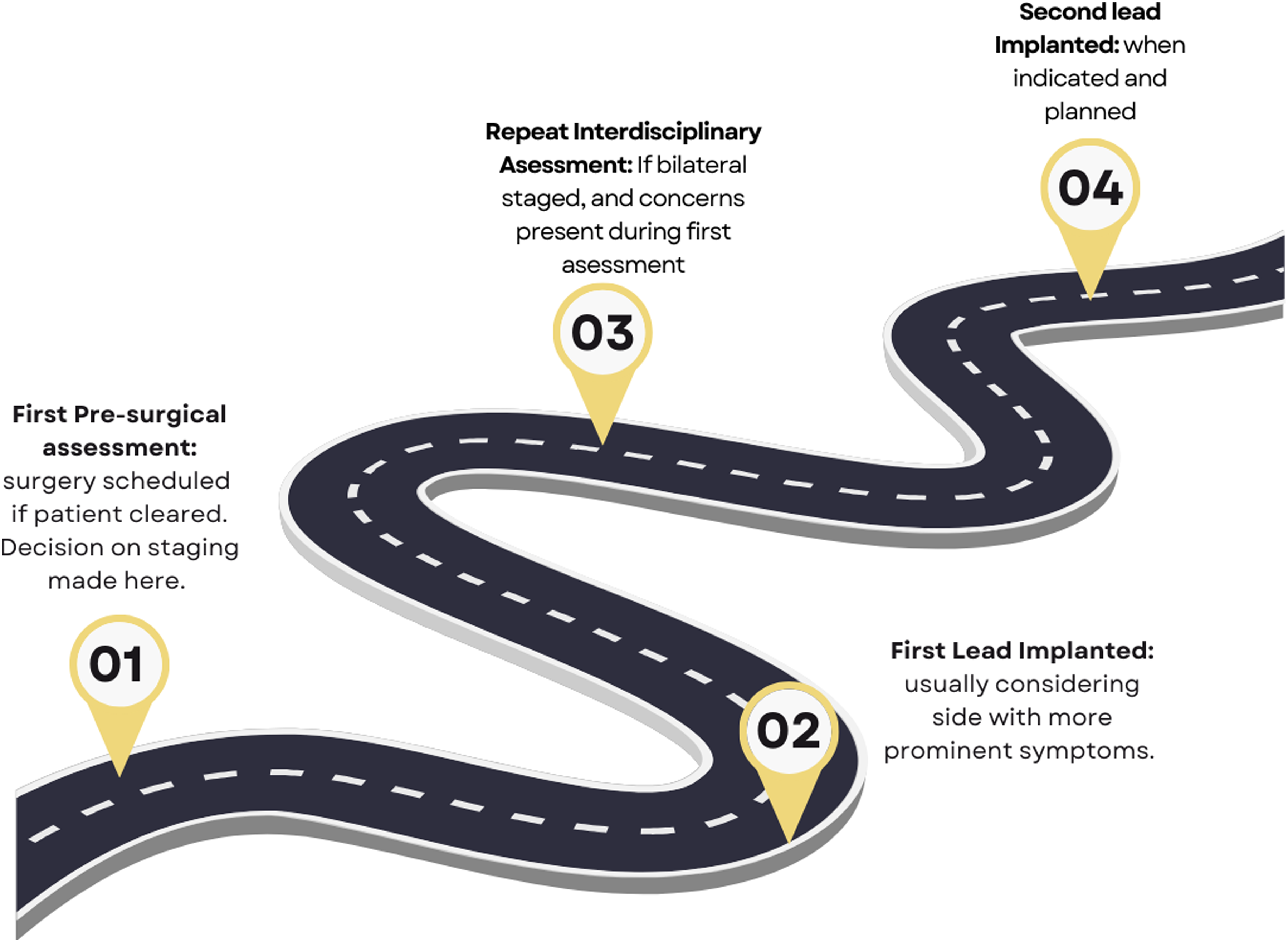
Staged bilateral DBS approach roadmap. Illustrates the staged approach to bilateral deep brain stimulation (DBS) with a focus on addressing concerns highlighted during the first multidisciplinary assessment. Reassessment is scheduled approximately 6 months after the first surgery before proceeding with the second surgery, when deemed necessary. This roadmap ensures comprehensive evaluation and patient-centered care in the DBS treatment process.
For patients with PD residing in resource-limited countries, access to advanced therapies such as DBS can be significantly hampered by socio-economic factors. In such settings, unilateral or staged bilateral DBS approaches could present a potentially viable and pragmatic solution. A growing body of evidence supports the effectiveness of unilateral DBS as an opportunity to expand access. This approach, when appropriately applied following multidisciplinary screening, not only aligns with emerging clinical evidence, but also may address economic constraints prevalent within resource-limited settings. Thus, the emphasis of a tailored and phased implementation of DBS interventions could possibly extend the benefits of neuromodulation therapies to a broader population, make outcomes more consistently positive and increase accessibility of technology.
This study had several important limitations. First, it was a retrospective design. Thus, clinical evaluations were not conducted in a blinded and controlled manner. Additionally, controlling for all potential confounding variables, such as medication adjustments during DBS programming, changes in cognition, and quality of life measures was not feasible due to the retrospective nature of data collection. Due to the retrospective nature of this study, there was also variability in the available follow-up data. Specifically, many patients presented to clinic on medication during their follow-up visits, which limited our sample size for group-level comparisons. Furthermore, our definition of bilateral DBS encompassed patients who underwent surgery with a staging interval of up to 2 months (bilateral rapid) and from 5–11 months (bilateral staged), which differed from the simultaneous bilateral approach described in the literature by different authors. A proportion of patients were lost to follow-up over time, resulting in unequal sample sizes at specific time points, and this may have affected the interpretation of the results. Finally, multidisciplinary team approaches and selection of surgical pathways based on detailed qualitative discussions following quantitative examinations by multiple specialties has not been and may not be easily standardized.
In summary, our study revealed that in a single experienced DBS center which employed multidisciplinary screening that assignment to unilateral, staged or rapidly staged cohorts were all associated with improved motor outcomes. Future studies should examine the safety of each approach.
Footnotes
Acknowledgments
ORCID iDs
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
Dr Hilliard has received consulting fees from AskBio.
Dr Okun serves as Medical Advisor the Parkinson's Foundation, and has received research grants from NIH, Parkinson's Foundation, the Michael J. Fox Foundation, the Parkinson Alliance, Smallwood Foundation, the Bachmann-Strauss Foundation, the Tourette Syndrome Association, and the UF Foundation. Dr Okun's research is supported by: R01 NS131342 NIH R01 NR014852, R01NS096008, UH3NS119844, U01NS119562. Dr Okun is PI of the NIH R25NS108939 Training Grant. Dr Okun has received royalties for publications with Hachette Book Group, Demos, Manson, Amazon, Smashwords, Books4Patients, Perseus, Robert Rose, Oxford and Cambridge (movement disorders books). Dr Okun is an associate editor for New England Journal of Medicine Journal Watch Neurology and JAMA Neurology. Dr Okun has participated in CME and educational activities (past 12–24 months) on movement disorders sponsored by WebMD/Medscape, RMEI Medical Education, American Academy of Neurology, Movement Disorders Society, Mediflix and by Vanderbilt University. The institution and not Dr Okun receives grants from industry. Dr Okun has participated as a site PI and/or co-I for several NIH, foundation, and industry sponsored trials over the years but has not received honoraria. Research projects at the University of Florida receive device and drug donations.
Dr Forghani has had a research collaboration/grant and has acted as consultant and/or speaker for Nuance Communications/Microsoft Inc., Canon Medical Systems Inc., and GE Healthcare. R.F. is also a co-investigator on a National Institutes of Health STTR grant subaward and a co-principal investigator on a National Science Foundation grant.
Dr Wong’s research is supported by NIH KL2TR001429.
Dr’s Sarmento, Daga, Wang, Srikar Lavu, de Araújo, and Aghili-Mehrizi have no financial disclosures to report.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
