Abstract
Background
Urinary tract infections (UTIs) have recently been linked to the onset of multiple synucleinopathies including Parkinson's disease (PD) and multiple system atrophy (MSA). UTIs are more common in people with PD or MSA, than in the general population and within these patient groups the incidence of UTIs is evenly distributed between men and women. UTIs are especially common during disease, but also in the years before clinical diagnosis.
Objective
The mechanisms by which UTIs may contribute to the development and progression of PD or MSA are not well understood. In this work, we evaluate the neuroinflammatory effects of recurrent UTIs on the brain.
Methods
In a humanized mouse model of ɑ-synuclein, we find that repeated administration of uropathogenic E. coli result in sustained UTIs, or a non-resolving chronic UTI phenotype with persistent bacteriuria. Using this model, we investigate the effects of repeated chronic UTIs on neuroinflammation and synucleinopathy in the brain.
Results
Recurrent UTIs lead to behavioral motor changes and are accompanied by persistent neuroinflammatory changes in multiple brain areas. Affected regions with microglial changes involve multiple lower brainstem areas responsible for sickness behavior, including the dorsal vagal complex, and the cingulate cortex.
Conclusions
These results suggests that recurrent UTIs can have lasting impact on the brain, and it warrants further investigation of the potential role of UTIs in the disease progression of synucleinopathies and related neurological disorders.
Plain Language Summary
This study explores how repeated urinary tract infections (UTIs) might influence the brain and contribute to the development of diseases like Parkinson's disease (PD) and multiple system atrophy (MSA). UTIs are common, especially in older adults and people with these neurodegenerative diseases, and they can worsen symptoms.
A novel mouse model is established by repeatedly infecting mice with a type of bacteria known to cause UTIs in humans. We find that repeated infections lead to persistent UTIs and, more importantly, causes long-term inflammation in the brain. The areas of the brain affected by this inflammation are known to be involved in movement, pain regulation and sickness behavior.
The findings suggest that chronic UTIs may not only be a symptom of PD and MSA but that they could also contribute to the progression of these diseases by causing ongoing brain inflammation. This inflammation might make the brain more vulnerable to further damage. Better understanding of these mechanisms could lead to new strategies for preventing or slowing the progression of these neurodegenerative diseases. The study highlights the need for more research to determine how managing UTIs could potentially protect the brain and improve outcomes for people at risk of or living with PD and MSA.
Keywords
Introduction
Peripheral infections are increasingly implicated in the onset and progression of neurodegenerative diseases. In the synucleinopathies, Parkinson's disease (PD) and multiple system atrophy (MSA), there is a long prodromal phase with peripheral symptoms that are typical of these diseases.1,2 It has been hypothesized that synucleinopathies, including subtypes of PD or MSA might originate from within these peripheral sites via inflammatory triggers.3,4 Some of these triggers are hypothesized to be infectious pathogens and could include bacteria and viruses. 4
Several bacterial and viral pathogens have been linked to the prodrome of synucleinopathies. 4 These pathogens can act as potential triggers of synucleinopathy in visceral barrier sites and subsequently contribute to the progression of the disease and cause neurological symptoms. Inflammation, whether generalized or in the brain, could thereby have lasting harmful effects via neural or humoral responses.
Our work and that of others recently indicated that urinary tract infections (UTIs) are implicated in the prodrome and the progression of synucleinopathies.5,6 UTIs are among the most common bacterial infections in humans. 7 They are known to cause cognitive and motor symptoms, especially in the elderly.8–10 UTIs are a leading cause of symptoms exacerbation and mortality in people living with PD and MSA. Autonomic dysfunction increases the risk for UTIs in these patients, but epidemiological studies suggest a link between UTIs and PD and MSA years before clinical diagnosis.5,6 This raises the possibility that UTIs could trigger disease onset.
However, the prevalence of UTIs and their relationship with synucleinopathies are not well understood as they remain under-reported and as the mechanisms behind UTIs and synucleinopathies are not yet fully elucidated.5,11 PD- and MSA-associated UTIs are equally common in men as in women as opposed to the general population where UTIs are more common in women.6,11–13 In PD and MSA, UTIs are also much more frequent,6,11 and this can lead to UTIs becoming chronic and recurrent over time. 6 In this vulnerable group of patients, recurrent chronic UTIs are a significant co-morbidity. There is a need for targeted disease prevention, and risk reduction strategies are warranted.
Given the relevance and the high prevalence of UTIs in PD and MSA, we recently developed an animal model of UTIs to study the onset and progression of synucleinopathy in MSA. 6 UTIs are known to cause central effects in animal models, which are likely mediated via systemic inflammation.8,10 However, it has not yet been investigated if recurrent UTIs can have lasting effects on the brain. Here, we build on our previous findings and developed an animal model of chronic recurrent UTIs. We investigated if recurrent UTIs could induce neuroinflammation in the brain. Via repeated superinfections with uropathogenic E. Coli (UPEC) we find that humanized ɑ-synuclein (ɑSyn) mice develop chronic UTIs. Humanized ɑSyn mice do not develop motor deficits or detectable brain pathology within their normal lifespan but recurrent UTIs lead to inflammation in the urinary bladder and cause behavioral changes months after the infections. Via whole brain histopathological analysis with unbiased artificial intelligence (AI)-models we observed persistent neuroinflammatory changes in the several brain regions. Affected regions with microglial changes involve multiple lower brainstem areas and the cingulate cortex.
Methods
Animals
In this study we used female transgenic mice from Jackson Laboratory FVB;129S6-Sncatm1Nbm Tg(SNCA)1Nbm/J and reference ID, RRID:IMSR_JAX:010710 (Jackson Laboratories stock no. 010710). These PAC-Tg(SNACAWT);Snca-/- mice are knock-out for the mouse allele and harbor the transgene encoding for the human ɑSyn. Mice were housed in the Van Andel Institute vivarium, with a maximum of 4 animals per cage, under a 12-h light-/dark cycle and had free access to water and food. The housing of the animals and all procedures were carried out in accordance with the Guide for the Care and Use of Laboratory Animals (United States National Institutes of Health) and were approved by the Van Andel Institute's Institutional Animal Care and Use Committee (PIL-21-09-007).
Mouse model of recurrent UTIs
Seven week-old transgenic humanized ɑSyn female mice were inoculated into the urinary bladder with [1 × 108 CFU/50 µL] of UPEC (human isolate UTI89 attHK022::COMGFP) or PBS (n = 15 per group) via transurethral catheterization under isoflurane anesthesia as previously described.6,14 This urinary bladder inoculation was repeated after 24 h (super infection protocol) as described in the literature to achieve a persistent bacteriuria and enhanced severity of infection.15,16 Super infections were repeated four times every 8 weeks and antibiotic treatment (trimethoprim/sulfamethoxazole water [270 and 54 μg/mL], respectively) was applied in the drinking water for 10 days, starting at 4 weeks after each infection to avoid kidney infection. Both groups experimental groups infected with UPEC and PBS were treated in parallel with antibiotics. Body weights were recorded periodically, and infection outcome was monitored from urine samples obtained by gentle suprapubic pressure and serially diluted and plated to quantify the number of colony forming units (CFU). 17 For each superinfection, urine collections were performed at 48 h, 2 weeks, and 4 weeks post-infection to determine the CFUs.
For infections in WT mice, seven weeks-old C57BL6/N female mice were inoculated into the urinary bladder with [1 × 108 CFU/50 µL] of UPEC or PBS (n = 13 per group) via transurethral catheterization under isoflurane anesthesia. When the mice did not develop acute UTIs, antibiotics were administered for 4 days instead 10 in accordance with the animal welfare committee suggestions. Both experimental groups, infected with UPEC and PBS, were treated in parallel with antibiotics.
Behavioral testing
At eleven months of age and nine months after the first super infection, humanized ɑSyn mice underwent motor performance testing using the CatWalk XT version 10.6 automated gait analysis system (Noldus Information Technology). This system employs a glass-floor walkway with fluorescence light beaming in the glass to track the animal's paws. It can detect natural gait, speed, coordination, and weight distribution. Animals were acclimated to the walkway over two days, with one session per day and were tested until they completed six compliant runs. A compliant run was defined as having less than 60% variability, and a total duration between 0.5 and 30 s. Runs were triggered as an animal entered the camera's field of view and automatically stopped once the animal exited the camera's field of view. Percent max speed variability was calculated using default settings in real-time.
Additionally, we evaluated the motor performance of humanized ɑSyn mice using the grip strength test. For this test we used a Chatillon DFE II Series Digital Force Gauge apparatus (CSC Force Measurement, model DFE2-010). The procedure involved placing the animal's forepaws on the wire mesh grid, and then gently pulling it backward by the tail until grip release. The test was repeated three times, and the maximum grip force recorded in each trial was averaged for each animal using the Animal Grip Strength System software (San Diego Instruments 2006–2009).
Tissue collection
After motor testing animals were deeply anesthetized with 130 mg/kg sodium pentobarbital (Sigma Aldrich) prior to tissue collection. Urinary bladders were isolated, and their wet weight recorded prior to perfusion. Urinary bladders were then fixed overnight in 70% ethanol for subsequent histological analysis. Brains were isolated after transcardial perfusion with saline, followed by fixation with 4% PFA. After overnight post-fixation with 4% PFA, brains were stored at 4°C in 30% sucrose in PBS until sectioning. Tissue from humanized ɑSyn mice was collected at 11 months. Tissue from C57BL6/N mice was collected at 15 months.
Immunohistochemistry
Paraffin embedded urinary bladders were sectioned at 6 µm and placed on glass mounting slides. Urinary bladder tissues were stained on glass mounting slides with 1 mL of incubation solution as described previously. 6 Slides were heated at 60°C in an oven for 30 min and rehydrated via serial rehydration in xylene, 100% ethanol, 90% ethanol, and 70% ethanol and distilled water. Antigen retrieval for urinary bladder was performed on paraffin-embedded urinary bladder tissue with the universal HIER antigen retrieval reagent (Abcam) for 30 min in a steam cooker at 95–100°C. For immunofluorescence analysis, sections were blocked with 10% donkey serum (Millipore-Sigma) in 0.1% Triton-X in PBS for 60 min at room temperature (RT).
For antigen detection in the urinary bladder, different concentrations of primary antibodies are listed in Table 1 and were used at RT (chromogenic staining) or 4°C (immunofluorescent staining) overnight. Sections were triple washed in 0.1% Triton-X in PBS and incubated with secondary antibody (Table 1) and 1:1000 DAPI for 2 h at RT after which the sections were washed again with 0.1% Triton-X in PBS. For immunofluorescence analysis, urinary bladder tissues were treated with Trueblack Lipofuscin Autofluorescence Quencher (Biotium) for 3 min to quench non-specific extracellular fluorescence and cellular autofluorescence. Slides were sealed with Vectashield Antifade Mounting Medium (Vector Laboratories). For chromogenic DAB immunoprecipitation and antigen detection, samples were blocked with 10% goat serum for 1 h after which they were incubated with primary antibody listed in Table 1 at RT overnight. Next day, slides were triple washed with 0.1% Triton-X in PBS and incubated with biotinylated anti-rabbit antibody (Vector Laboratories, BA-1000-1.5) for 2 h at RT, triple washed again and treated with Vectastain ABC kit (Vector Laboratories, PK-4000). Antigen detection was performed with Vector DAB (Vector Laboratories, SK-4100). In case of Nissl staining, slides were treated with 0.1% cresyl violet solution for 8 min before dehydration. After dehydration, slides were coverslipped with Cytoseal 60 mounting medium (Thermo Fisher Scientific). Immunofluorescence slides were viewed under a Zeiss LSM 880 confocal microscope.
Antibodies used for immunohistochemical analysis.
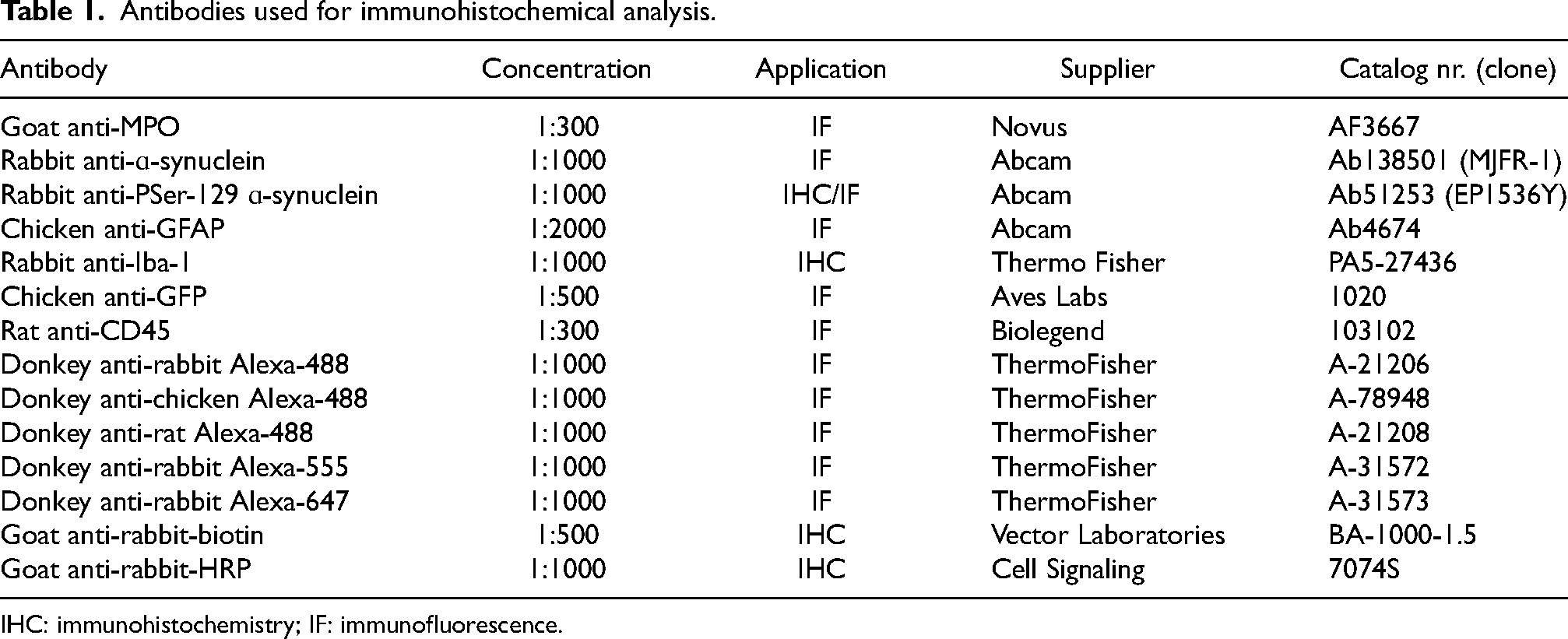
IHC: immunohistochemistry; IF: immunofluorescence.
Brain tissue was sectioned coronally at 40 µm thickness. For immunohistochemical analysis of brain tissue a series of free-floating tissue sections (every 240 µm) were blocked with 10% donkey or goat serum (Millipore) and 4% bovine serum albumin in 0.1% Triton-X in PBS for 2 h at RT. Samples were incubated with primary antibody listed in Table 1 at RT overnight for chromogenic staining. Next day, slides were triple washed with 0.1% Triton-X in PBS and incubated with biotinylated secondary sera at 1:1000. For the detection of the antibody with DAB, we used a standard peroxidase-based method (Vectastain ABC kit and DAB kit; Vector Laboratories). After dehydration, slides were coverslipped with Cytoseal 60 mounting medium (Thermo Fisher Scientific). Slides were scanned using a Leica CS scanner.
Immunohistochemical analysis
Automated analysis of neuroinflammation or synucleinopathy was performed using trained neural networks and Aiforia® technology. All images were acquired using a Aperio CS2 scanner (Leica) at 20x brightfield and at a 0.22 µm per pixel resolution. Tissue thickness was 40 µm. The digitized images were uploaded to Aiforia® Cloud for subsequent training and analysis. Training was performed by deep learning convolutional neural networks (CNN) and supervised learning.
For the analysis of neuroinflammation, a supervised, multi-layered CNN was adopted from Stetzik et al. 18 This model was also used for additional training. The AI model was trained on the most diverse and representative images from across multiple Iba-1 stained tissue slides and was capable of accurately detecting Iba1-stained microglia collected by multiple investigators. Whole slide images were loaded to the Aiforia® Cloud for new training data, and each feature layer was trained using tools in the Aiforia® Cloud platform. All analysis results, including the location of each microglia cell, were determined using the automated deep learning algorithm developed using Aiforia®. The training of brain regions was performed accordingly by manually annotating different brain regions in accordance to the Allen mouse brain atlas. The annotation of brain regions was based on the ability of the training supervisors to distinguish different brain areas in Iba-1 immunostained tissue. Analysis of brain regions was performed on serial sections and the counts of the number of microglial cells and their morphology is presented for the whole brain region. Annotation of the AP, NTS, and DMX was performed manually. Analysis of the AP, NTS, and DMX was performed for two tissue slides per animal. Microglial analysis of the AP, NTS, and DMX was performed using the automated deep learning algorithm developed for the detection of microglia using Aiforia®.
For the analysis of Pser129-ɑSyn pathology, a similar approach was used to train a multi-layered CNN for the recognition of Pser129-ɑSyn + inclusions. First, the parent feature layer was trained to recognize brain tissue that distinguishes between Pser129-ɑSyn immunostained tissue and glass slide background. Following this, the first daughter layer was trained to recognize different brain areas according to the Allen brain atlas. The annotation of brain regions was based on the ability of the training supervisors to distinguish different brain areas in Pser129-ɑSyn immunostained tissue. A second daughter layer was trained via object detection of Pser129-ɑSyn within different brain areas. The cell (inclusions) object detection diameter was set at 4 µm. Via object detection and segmentation, the AI model allows for precise measurement of Pser129-ɑSyn inclusion numbers and Pser129-ɑSyn inclusion diameter in whole brain. Annotation of the DMX was performed manually. Analysis of the DMX was performed for two tissue slides per animal. The area positive for Pser129-ɑSyn was determined via Aifoira®.
Immunofluorescence slides were scanned using the Axioscanner (Zeiss) at 20x for the urinary bladder. Analysis of the number of CD45 + leukocytes and MPO + neutrophils was performed via QuPath analysis. Single cells within a whole section of the urinary bladder were counted as the main readout.
Statistical analysis
For analysis of urinary bladder tissue, two tissue sections per individual animals were analyzed. The average of these two sections was used for statistical analysis. Statistical analysis for behavior was performed with Catwalk Noldus software for group statistics. The mean and standard deviation were calculated by CatWalk XT version 10.6 automated gait analysis system (Noldus Information Technology). All other statistical analysis was performed using GraphPad Prism 9. The type of analysis with post hoc correction for multiple testing is indicated in the text or in the legend of each figure.
Results
To study the effects of chronic UTIs in vivo and their potential link with ɑSyn, we superinfect transgenic humanized ɑSyn mice at multiple time points (Figure 1(a)). Humanized ɑSyn mice express levels of wild-type (WT) human ɑSyn comparable to mouse endogenous protein, but they do not express the endogenous mouse ɑSyn.6,19 The expression is driven by the complete human ɑSyn promoter. These transgenic mice do not develop any detectable motor deficits or synuclein pathology within their normal lifespan. 19 For superinfections, animals are injected twice, with an interval of 24 h, with 108 CFU of UPEC (Figure 1(a)). Infected mice are followed for 4 weeks, and bacterial titers are measured the first days after infection and at two and four weeks after infection in urine samples. Antibiotics are administered in the drinking water for 10 days to remove any infection that does not resolve spontaneously. Antibiotics were given to both infected as well as PBS treated mice. Eight weeks after the initial superinfection, animals are challenged with a subsequent superinfection. This process is repeated three times; mice were superinfected four times in total.
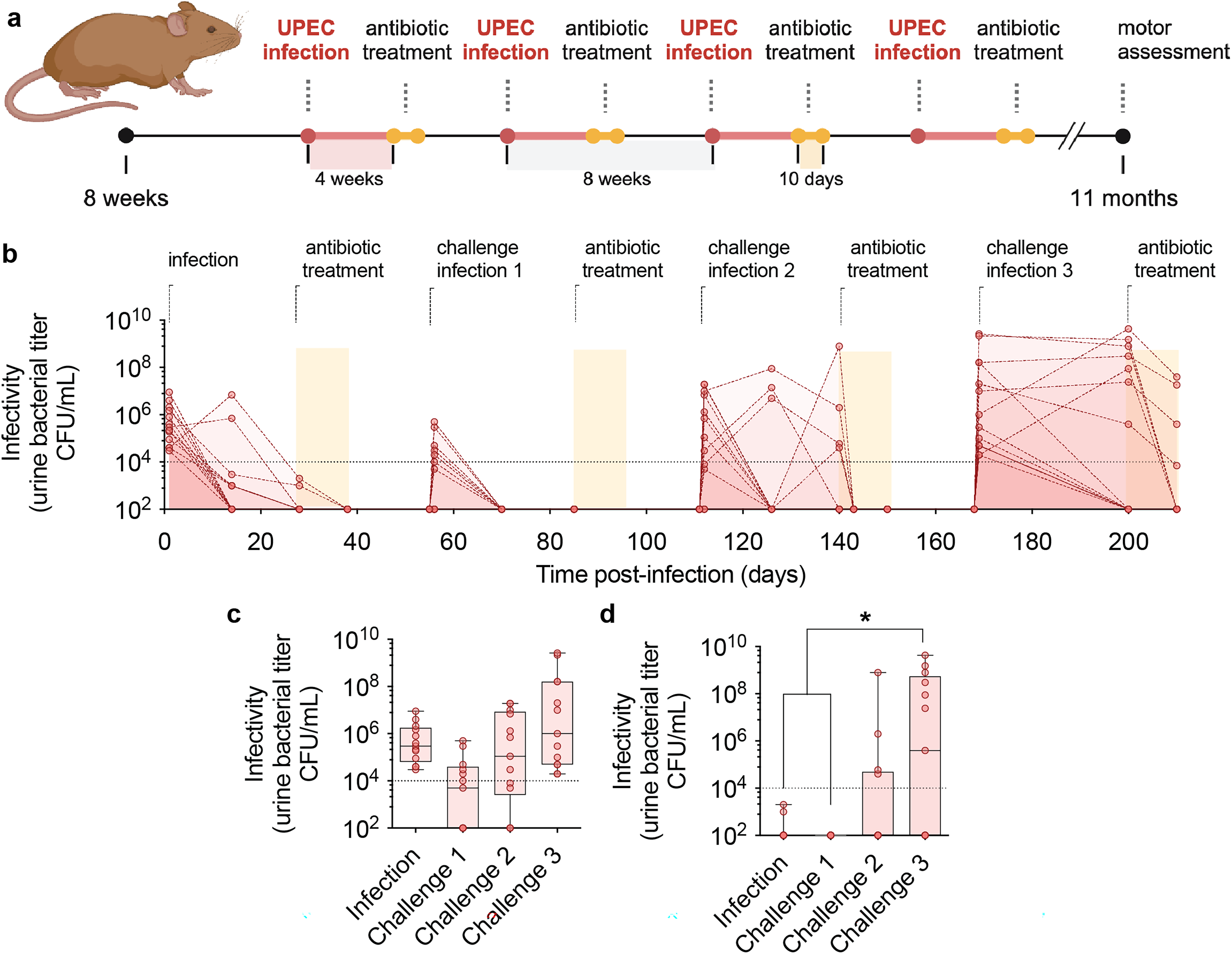
Recurrent superinfections in humanized ɑSyn mice cause chronic UTIs.
Via collection of urine the infectivity is assessed over time (Figure 1(b)). After the initial superinfection, animals develop acute UTIs with detectable bacterial titers at 48 h after UPEC administration (Figure 1(c)). However, at four weeks, no animals have a detectable infection (titers are below 104 CFU/mL), which shows that after the first superinfection all mice resolve the infection spontaneously (Figure 1(b)-(d)). After the second superinfection, the infection resolves spontaneously, with 6 out of 13 animals that already have infectious titers below 102 CFU/mL at 48 h (Figure 1(c)). Similar to the initial infection, none of the mice develop a chronic UTI after the challenge infection (Figure 1(d)). However, at the third and fourth superinfections, this response changes from a short, acute to a chronic phenotype (Figure 1(b)-(d)). At the fourth and final infection, 7 out of 13 animals develop chronic UTIs with infectivity that persists after 4 weeks of infection (Figure 1(d)). From the 7 animals that develop chronic UTIs, 3 of the mice show signs of bacteriuria even after treatment with antibiotics. This infection persists until the final time point of 11 months. None of the other animals show signs of bacteriuria. Thus, by performing repeated superinfection in transgenic humanized ɑSyn mice, a chronic UTI phenotype is observed in approximately half of the animals.
Due to the mixed background of the humanized ɑSyn, that have both an FVB/N and 129S6/Sv background, we do not have a comparable WT control to assess if chronic UTIs are due to this mixed strain background or due to the expression of human ɑSyn. However, by infecting WT C57BL6/N mice, we observe that C57BL6/N WT mice do not develop chronic UTIs (0 out of 12 animals develop chronic UTIs) using the same protocol. WT C57BL6/N mice spontaneously resolve repeated infections after three challenges (Supplemental Figure 1). This suggests that either the mouse strain background or the presence of human ɑSyn could influence the observed phenotypic shift.
During UTIs it has been shown that acute infections in experimental animal models can directly cause cognitive impairment as well as changes in locomotor activity or anxiety-like behavior. 8 Whereas acute UTIs thus have been shown to cause behavior changes, it is not known if chronic UTIs can cause motor symptoms typical of PD or MSA. To further assess if recurrent UTIs (rUTIs) can cause more permanent behavioral changes, we now examine motor performance two months after the last antibiotic treatment using a grip test and automated gait analysis. We find that rUTIs have no significant effects on general motor readouts including average walking speed, cadence, or grip strength (Figure 2(a)-(e)). However, we do observe significant effects on gait activity that involve base support and balance (Figure 2(f), (g)). Base support is a measure of posture and has been shown to be affected in acute PD models. 20 Animals with chronic UTIs develop significant motor impairments in front and hind paws and spend more time standing at all four paws during walking (Figure 2(h)).
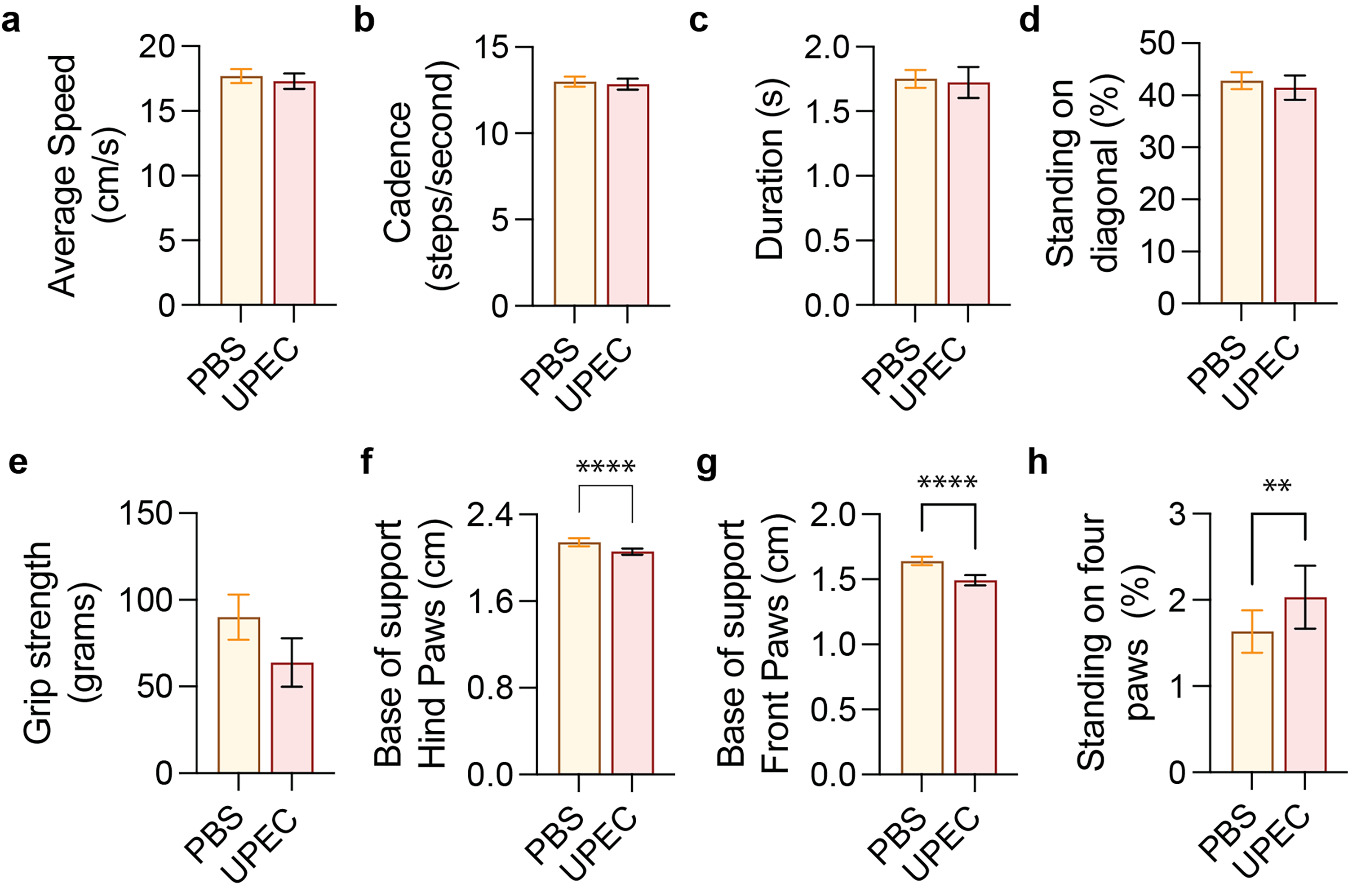
Behavioral changes after rUTIs in humanized ɑSyn mice.
To investigate the effects of rUTIs or chronic inflammation and high bacterial burden on the urinary bladder we isolate the urinary bladders at the final time point of 11 months for pathological analysis (Figure 3). Chronic inflammation of the urinary bladder can result in hypertrophy as repeated infections typically cause chronic inflammation with urothelial hyperplasia and thickening of the detrusor resulting in altered bladder function. 16 The urinary bladder of mice with rUTIs are significantly enlarged and hypertrophic compared to mice treated with PBS (Figure 3(a), (b)). The detrusor muscle furthermore shows significant thickening (Figure 3(c)), a histopathological sign that is also typical for humans with chronic cystitis. 21
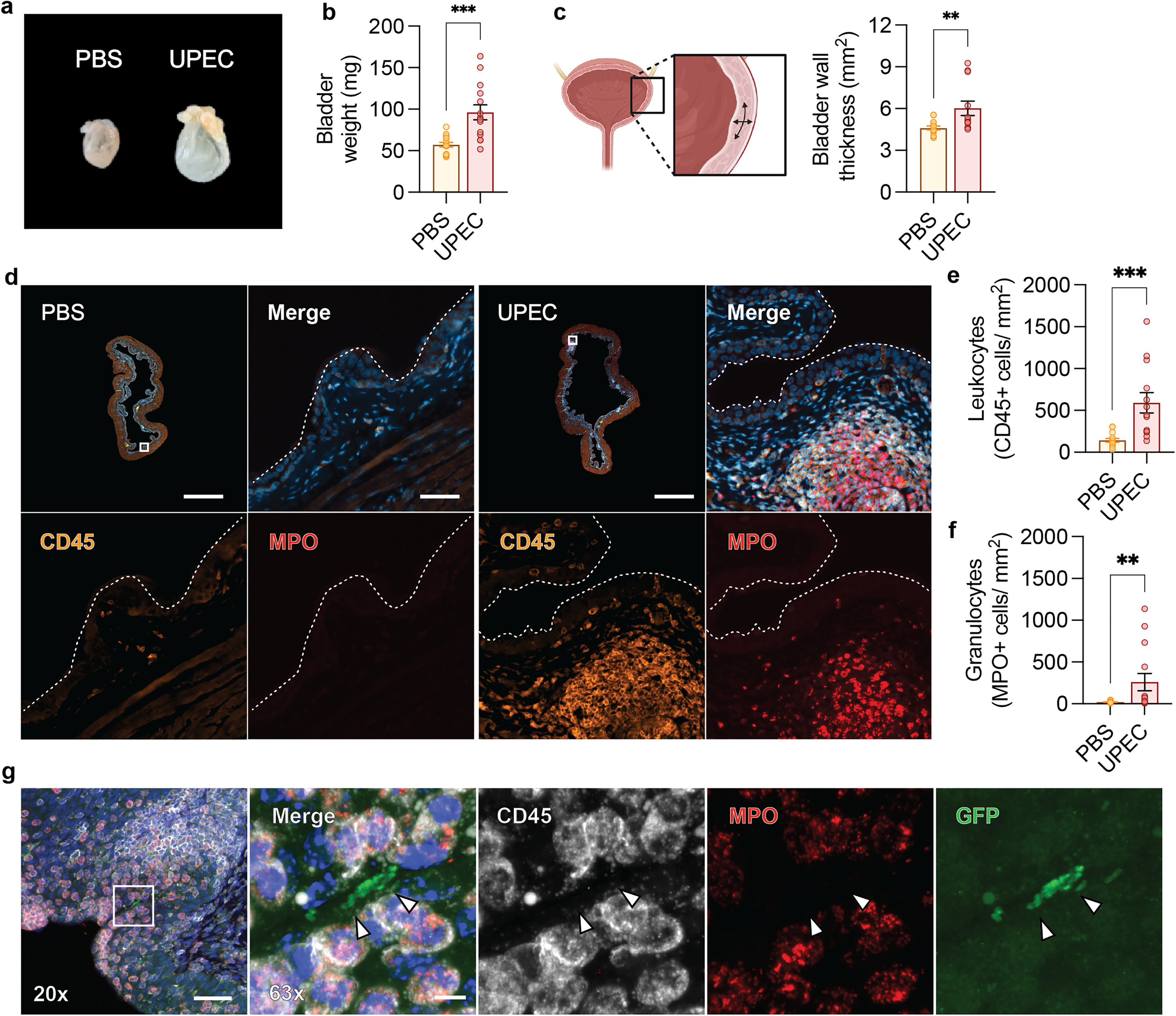
Bladder hypertrophy and inflammation after rUTIs.
Chronic cystitis is caused by uncontrolled bacterial replication in the bladder lumen and is associated with inflammation and submucosal lymphoid aggregates. To examine if there is a persistent immune response in humanized ɑSyn mice with chronic cystitis we perform immunohistological analysis of the urinary bladders for signs of inflammation or infection. In animals treated with PBS we do not observe any signs of inflammation or infection. However, in animals with rUTIs we observe an increased number of leukocytes via staining for the lymphocyte common antigen marker CD45 (Figure 3(d), (e)). This increase in cells is in part due to infiltrating neutrophils as shown by staining for the neutrophil marker myeloperoxidase (MPO) (Figure 3(d), (f)), indicating an ongoing innate immune response in the bladder of mice with rUTIs.
Ongoing inflammation is sometimes a sign of continued bacterial replication and since a small number of animals also develop persistent bacteriuria we further examine if UPEC can be detected in the bladder lumen. The human UPEC isolate used for the superinfections expresses the green fluorescent protein (GFP) marker, allowing to assess the presence of UPEC that is used for repeated superinfections. By staining for GFP we find that there are GFP-positive UPEC colonies on the urothelial surface surrounded by lymphoid aggregates (CD45+), mostly neutrophils (MPO+) (Figure 3(g)). Given the presence of neutrophils and that we previously described that during acute UTIs ɑSyn is released by neutrophils via extracellular traps we examined the presence of ɑSyn in the urinary bladder. By using an antibody that recognizes total ɑSyn we did not observe any detectable differences in ɑSyn expression in the urinary bladder between animals that were injected with PBS or UPEC (Supplemental Figure 2).
UTIs are known to trigger central effects via local cytokine production that can lead to systemic effects. In animal models of acute UTIs it has been shown that several cytokines can travel across the blood-brain barrier and cause acute inflammation in the brain.8,10 To further examine if recurrent or chronic UTIs can cause more permanent changes, we analyze the whole brain for the microglial marker ionized calcium binding adaptor molecule 1 (Iba-1) to assess neuroinflammation. For unbiased analysis of microglial cells numbers and their reactivity we further train an existing AI-based multi-layer deep learning model to recognize different brain areas in mouse brain based on the immunostaining with the Iba-1 marker (Figure 4(a)). The trained model recognizes several brain areas autonomously without referencing an existing brain atlas. Within the brain areas, the model identifies all cells positive for the Iba-1 marker, and by applying instance segmentation of the identified microglial cells the model further determines cell morphology as a measure of microglial reactivity or activation state (Figure 4(b)). By extracting the cellular data within these different brain areas, our model immediately extracts cellular and morphological data in an unbiased manner without manual annotations.
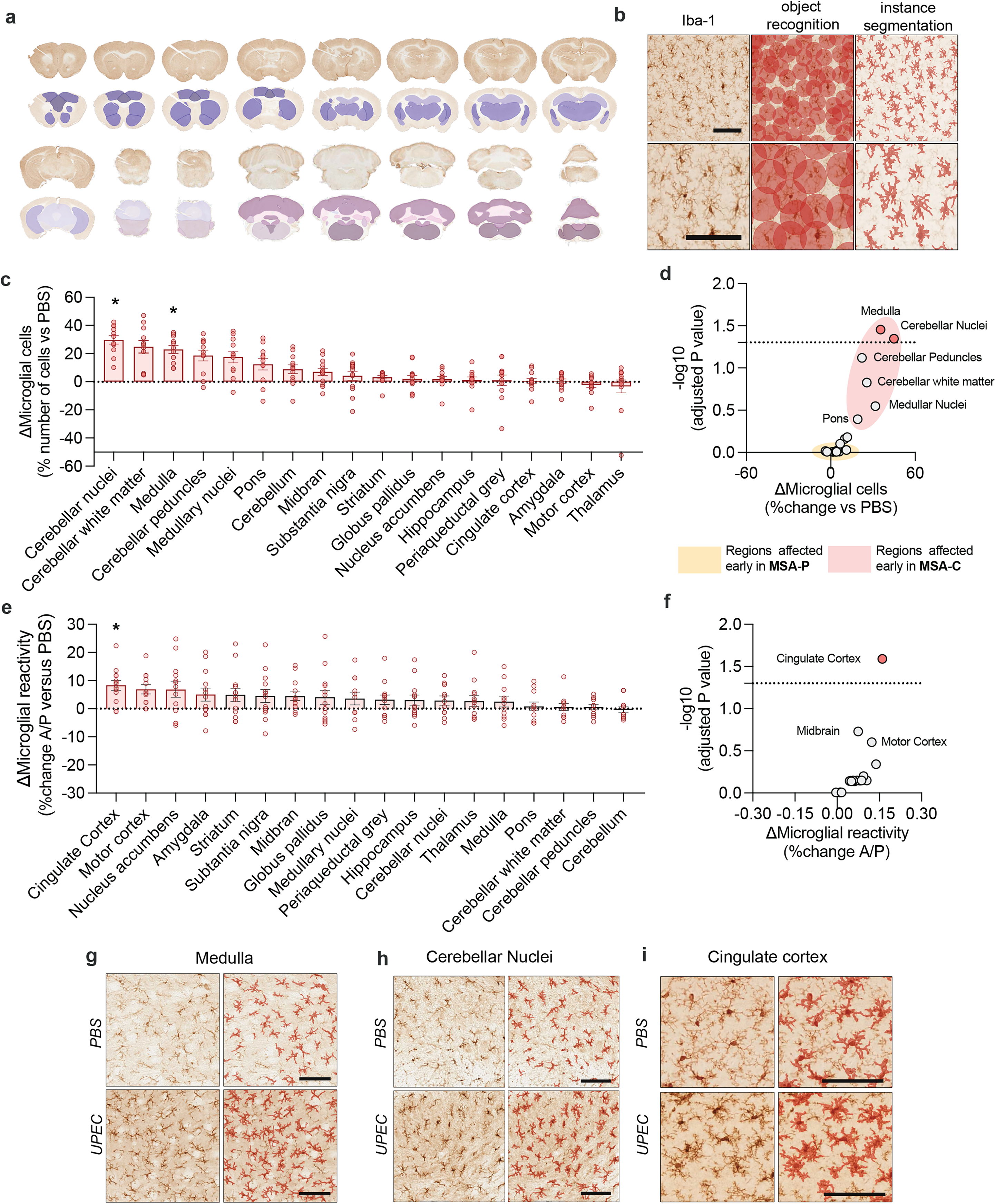
Neuroinflammation in distinct brain regions after rUTIs.
By ranking the regions based on the number of microglial cells, we find that regions in the brainstem and cerebellum have significantly higher numbers of microglial cells after recurrent UTIs compared to animals injected with PBS (multiple unpaired t-tests, adjusted *p < 0.05 after correction for multiple testing with Bonferroni-Dunn post hoc analysis, Figure 4(c)). When grouping brain regions based on the change in microglial numbers using the -log10 (adjusted p values), we find that in regions that have more microglial cells are exclusively regions that are early affected in the cerebellar form of MSA (MSA-C) (Figure 4(d)). Regions that are affected in the parkinsonian subtype of MSA (MSA-P) do not show a detectable increase in microglial numbers after treatment with recurrent UTIs.
Microglial reactivity can be a measure of neuroinflammation, as resting microglia have a relatively larger cell body perimeter versus reactive microglia that have a smaller cell body perimeter compared to their cellular soma. To quantify this comparison, we use the Iba-1 marker (Figure 4(b)). When ranking different brain regions according to the change in morphological parameters (area versus perimeter) we observe microglial reactivity in several brain regions (Figure 4(e)). After correcting for multiple testing, the cingulate cortex displays significant microglial reactivity after chronic UPEC treatment (multiple unpaired t-tests, adjusted *p < 0.05 after correction for multiple testing with Bonferroni-Dunn post hoc analysis, Figure 4(f)). Together, this figure shows that in defined brain areas, there are increased numbers of microglial cells (Figure 4(g), (h)) and reactive microglia (Figure 4(i)) in the months after recurrent and chronic UTIs.
Recently it was shown that sickness behavior is controlled by circumventricular structures in the hindbrain during peripheral inflammation.22,23 The anterior postrema (AP) and the nucleus solitarii (NTS) are two key areas in regulating the sickness response to bacterial endotoxins.22,23 As the AP has an incomplete blood brain barrier it can directly sense the humoral response via systemic mediators, such as endotoxins or cytokines. 24 The AP and the NTS are part of the dorsal vagal complex and are situated dorsally from the dorsal vagal nucleus (DMX), a region that is typically implicated in PD. Given the chronicity of our model and the significant microglial response particularly the hindbrain areas, we further examine if neuroinflammation might be affected in these specific areas. Since AI-assisted region annotations are not possible for these brain structures, due to a lack of clear reference structures, manual annotations of the NTS, AP, and DMX are performed (Figure 5(a)). Regions of interest are analyzed by our AI-based microglial detection model after manual region annotation to determine the number of microglial cells and their morphological parameters. The number of microglial cells in all regions of the dorsal motor complex (AP, NTS, and DMX) are significantly increased after chronic UTI (Figure 5(b)-(d), unpaired t-tests, *p < 0.05, **p < 0.01). The microglia in the AP are also reactive (Figure 5(e), unpaired t-tests, *p < 0.05), whereas the NTS and DMV do not show microglial reactivity. Together this show that peripheral inflammation after recurrent and chronic UTIs results in lasting microglial reactivity in the dorsal motor complex.
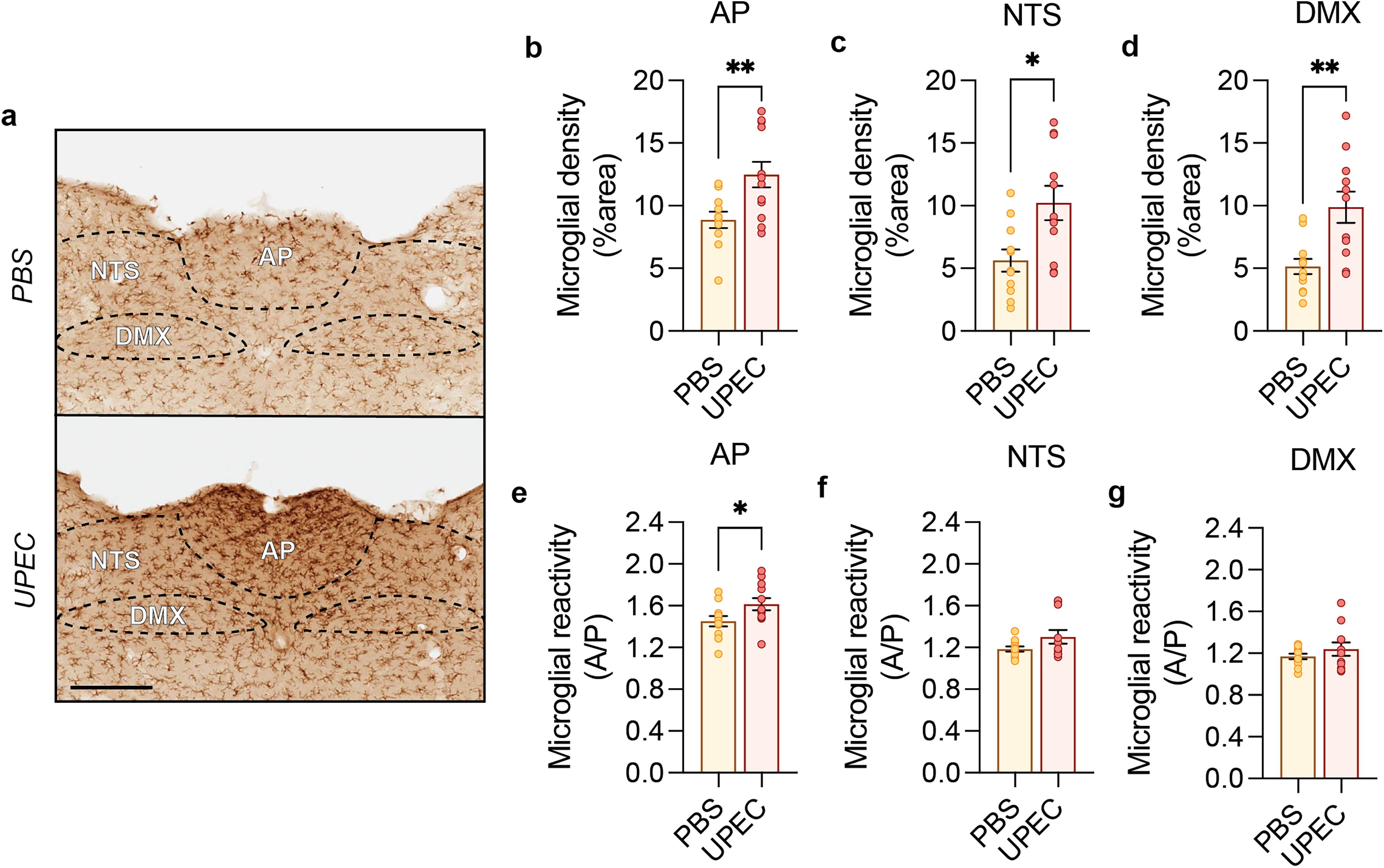
Microglial reactivity after rUTI in the dorsal vagal complex.
We further evaluate if similar neuroinflammatory changes could be observed in WT C57BL6/N mice. These mice do not show any chronicity of infection and furthermore lack detectable behavioral deficits up to 15 months (data not shown). Analysis of the microglial response in WT C57BL6/N mice at 15 months does not show any differences in the number of microglial cells (Supplemental Figure 3a) or microglial reactivity (Supplemental Figure 3b) in any of the brain regions analyzed after correction for multiple testing. This thus shows that in humanized ɑSyn the chronicity of infection results in persistent microglial changes, whereas in 15-month-old C57Bl6/N mice that do no develop chronic infections, no microglial changes are observed.
Neuroinflammation can disrupt cellular homeostasis and lead to neurodegeneration. We therefore further assess if the effects of repeated chronic UTIs in humanized ɑSyn mice can lead to a potential change in proteostasis, or expression and accumulation of ɑSyn protein. By using the pathological marker Pser129-ɑSyn, we perform whole brain analysis of ɑSyn pathology (Figure 6(a) and Supplemental Figure 4a). We train an assisted deep learning model to perform AI-based analysis for the detection of Pser129-ɑSyn inclusions within distinct brain regions. Based on the staining for Pser129-ɑSyn the AI model can autonomously detect and distinguish multiple brain regions in the mouse brain (Figure 6(a)). Following this, we performed Pser129-ɑSyn-based analysis to examine the number of inclusions in different brain regions via object detection and segmentation of Pser129-ɑSyn inclusions (Figure 6(b)). We define a Pser129-ɑSyn+ inclusion as an inclusion that on average is 35–100 µm2 in size (Supplemental Figure 4b).
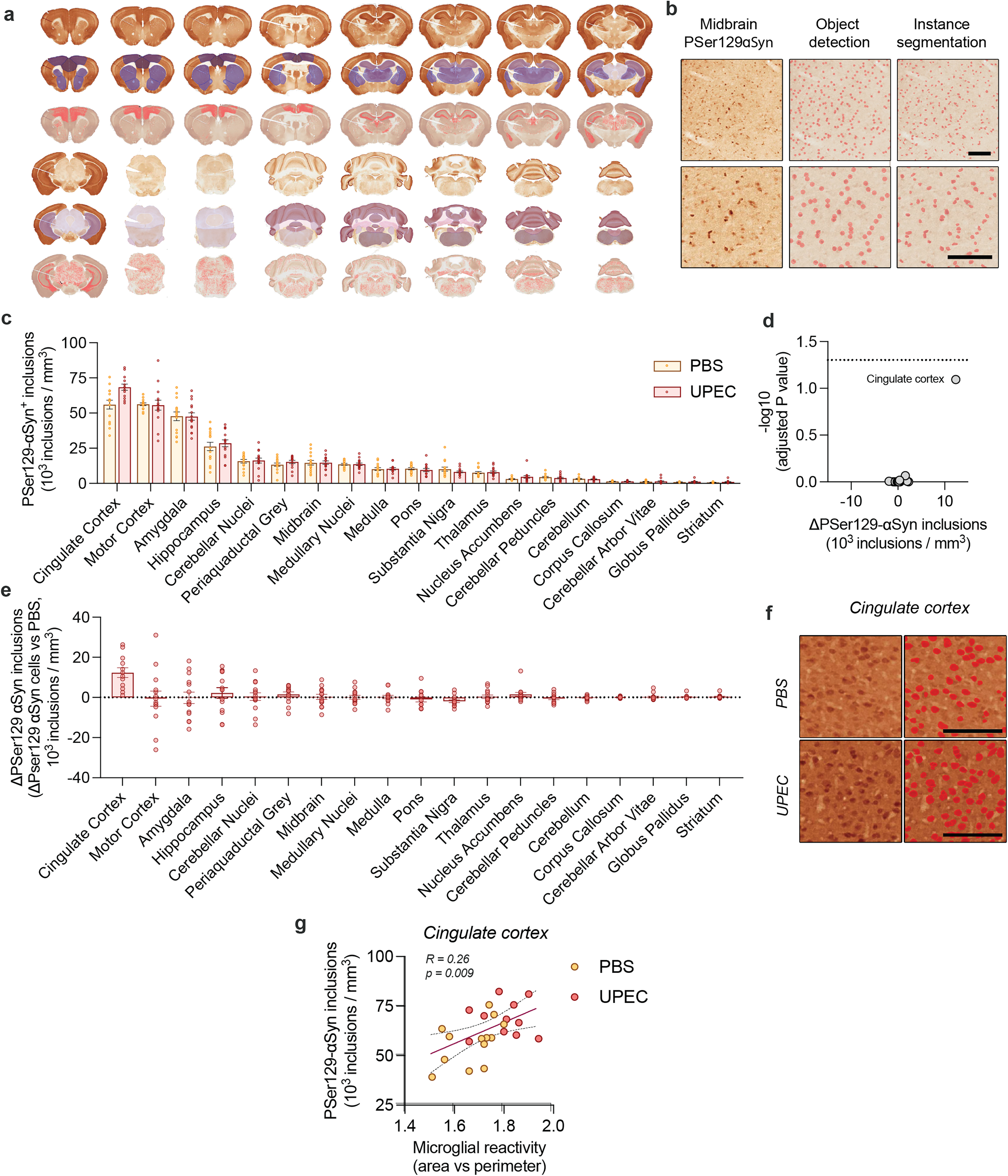
Whole brain analysis of Pser129-ɑSyn after rUTIs.
Transgenic humanized ɑSyn mice have similar levels of human ɑSyn expression in comparison to mouse ɑSyn expression in WT mice.6,19,25 In this model, we observe widespread Pser129-ɑSyn+ inclusions throughout the brain at 11 months of age in PBS treated control animals. Brain-wide analysis shows that areas typically involved in PD, such as cortical areas, the amygdala, and hippocampus have higher numbers of Pser129-ɑSyn+ inclusions compared to other regions (Figure 6(c)). After repeated UTIs we observe no detectable changes in Pser129-ɑSyn+ inclusions after correction for multiple testing (ranked, Figure 6(c)-(e)). However, by correlating the number of Pser129-ɑSyn+ inclusions with neuroinflammation in the cingulate cortex (Figure 5(f)), we do detect a significant correlation between microglial reactivity and Pser129-ɑSyn+ inclusions (R = 0.26, **p < 0.01, Figure 6(g)). When comparing Pser129-ɑSyn+ inclusion size, we do not find any differences between PBS and UPEC infected mice (data not shown).
As increased microglial numbers are also observed in the dorsal motor complex (Figure 5), we examine the potential effects of repeated UTIs on Pser129-ɑSyn+ inclusions in the dorsal motor complex. Manual annotation of the DMX (Figure 7(a)) allows to examine the Pser129-ɑSyn -positive area in the DMX. We find no detectable changes in Pser129-ɑSyn in the DMX after chronic UTIs (Figure 7(a)). Altogether, this shows that in the presence of microglial reactivity or neuroinflammatory changes no clear effects could be observed on Pser129-ɑSyn in humanized ɑSyn mice.
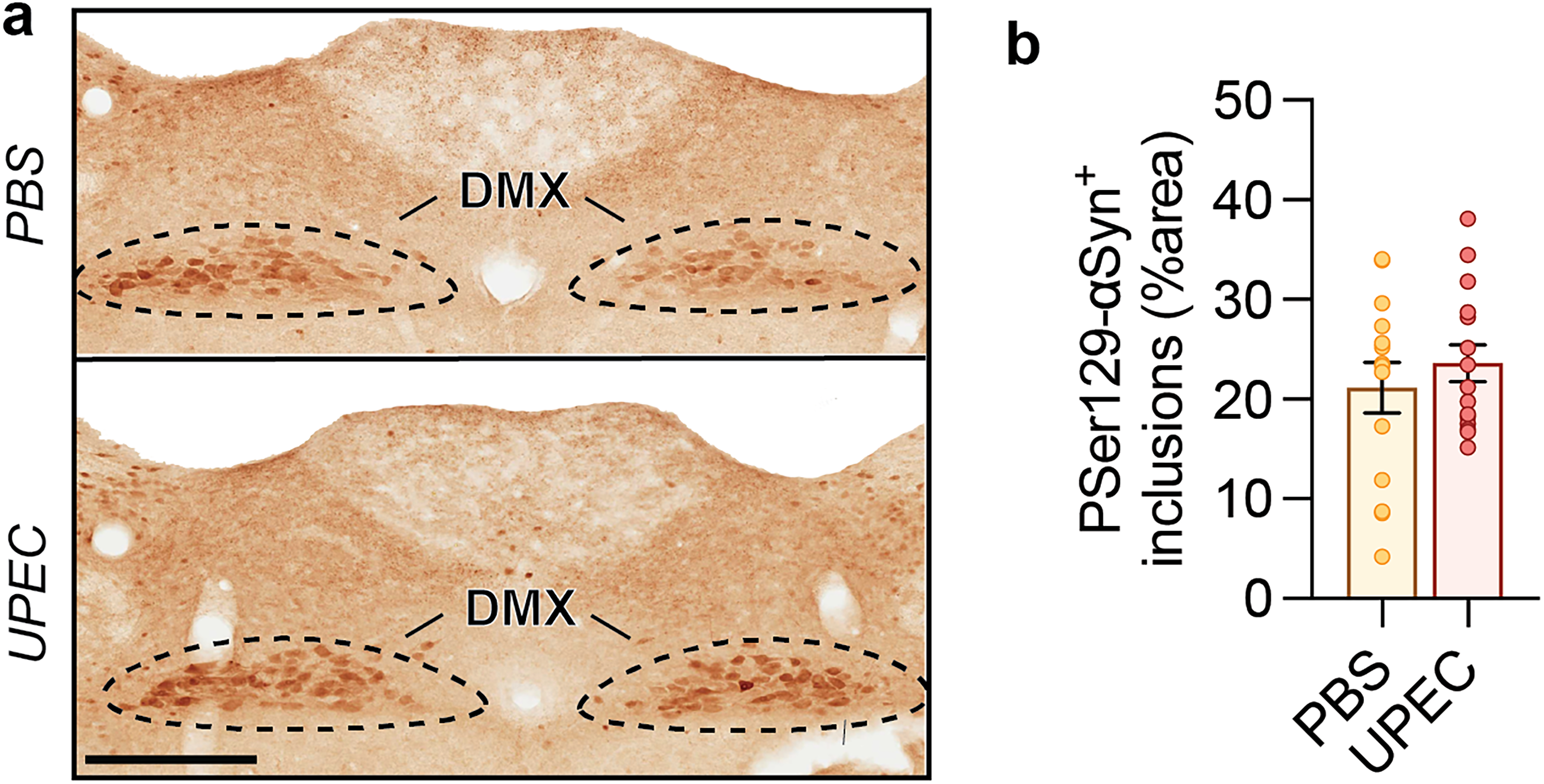
Pser129-ɑSyn analysis after recurrent chronic UTI in the dorsal motor nucleus of the vagal nerve.
Discussion
The reciprocal interactions between the brain and the immune system are even more evident as inflammation in the periphery is increasingly linked with synucleinopathy and brain function. Given the recent links between synucleinopathies and UTIs, we here investigated the potential mechanisms by which recurrent UTIs could be associated with brain inflammation and synucleinopathy.
To model and mimic recurrent chronic UTIs in mice, we performed repeated superinfection as this is an effective way of modeling chronic UTIs. 16 We focused on female humanized ɑSyn mice that overexpress ɑSyn under the control of the human ɑSyn promoter. We did not study male mice as this model urethral catheterization was initially established in female animals. After repeated superinfections, the mice developed chronic UTIs with a persistent bacteriuria (Figure 1). At the completion of the experiment and thus at 9 months after the initial superinfection, we observed significant hypertrophy and urothelial hyperplasia in the urinary bladder of infected mice (Figure 3). Mice with chronic cystitis showed persistent lymphoid aggregates and neutrophil infiltration surrounding regions with bacterial colonies in the bladder lumen. This inflammatory response and the bladder pathology are comparable to humans, as humans suffering persistent bacteriuria and chronic cystitis also exhibit bladder hyperplasia and inflammation. 21
The susceptibility towards chronic infections is known to be influenced by host strain background. 26 A limitation of this finding is that due to the mixed strain background of the transgenic mice, we could not evaluate if the development of chronic UTIs was due to the expression of human ɑSyn or due to the host strain background. Nevertheless, elevated susceptibility to chronic UTIs can be due to several different causes. During chronic UTIs, bacteria can evade host innate or epithelial immune mechanisms by surviving in the urothelial cytoplasm as intracellular bacterial colonies.27,28 They evade antibacterial clearance and avoid expulsion and exfoliation from urothelial cells during epithelial repair, leading to recurrent or chronic UTIs.27–29 Using the same infection paradigm, WT C57BL6/N mice did not undergo phenotypic conversion, and this thus suggests that the chronic phenotype is linked to the transgenic animal model. WT C57BL6/N mice furthermore did not develop any behavioral deficits up to 15 months and they did not show any microglial changes at the same timepoint. Although we did not further investigate the mechanism behind the observed change in our transgenic model from an acute to a chronic infection phenotype, we did observe signs of aberrant epithelial thickening, and this could likely have caused an impaired host defense against new or repeated infections.
To study the effects of recurrent chronic infections on the behavior of the humanized ɑSyn mice, we performed several motor performance readouts. From our analysis, we found that base support or posture is affected in the mice that had rUTIs (Figure 2). Altered base support or altered posture, is commonly affected in MSA and PD. An altered posture is seen when patients keep their feet closer together to make smaller movements while hunched. In aged people, UTIs can lead to falls and require hospitalization. After UTI, people with PD are also twice as likely to be hospitalized. 11 Typically, these effects are ascribed to reduced dopaminergic signaling or the loss of dopaminergic neurons in the basal ganglia that regulate motor control. To assess if these effects could have been observed in our model, we analyzed the integrity of the nigrostriatal pathway via stereological quantification of the total number of dopaminergic neurons. However, we did not find any signs of neuronal loss or neuronal atrophy within the substantia nigra (Supplemental Figure 5).
Although we cannot exclude that other brain regions might have been altered, due to neuroinflammation, it is possible that some of the motor deficits might therefore have been caused by general discomfort or pain. Pain is a hallmark of cystitis and can result in reduced locomotion in mice, leading to hunching or reduced rearing behavior. 30 In addition, the onset and pain associated with UTIs is often a source of anxiety in patients. 13 Bacterial endotoxins are also potent inducers of sickness behavior. Peripheral inflammation can cause reduced locomotion via endotoxins that are sensed by the NTS-AP axis. 22 The observation of the behavioral effects in the infected mice therefore should be further investigated, as it raised the possibility that these effects could have been caused by infection or discomfort rather than neurodegeneration of the affected central pathways. 30 In the absence of any overt signs of neurodegeneration, and in the presence of ongoing inflammation after rUTIs in the urinary bladder and in the brain (Figures 3 and 4), we believe that the behavioral effects in this prodromal model could thus have been caused by infection-associated pain or discomfort.
Given these pronounced peripheral and central inflammatory responses, we further sought out to determine what the effects of rUTIs could be on neuroinflammation and neuronal health. We found that rUTIs lead to chronic neuroinflammation in various brain regions. These effects were pronounced in regions of the cortex, the brain stem, and regions connecting to the cerebellum, including the medulla and the cerebellar nuclei. In the latter two regions, we find a significantly higher number of microglia after rUTIs. Manual analysis of the dorsal vagal complex also reveals significant microglial changes within this region. Since this study focused on the long-term effects of rUTIs on the brain, we did not study the effects of earlier inflammatory responses that might have been the result of acute UTIs. However, immediate, or early neuroinflammatory responses can lead to changes in microglial cell numbers via microglial proliferation, which can persist over time, even before the onset of widespread microgliosis. 31
Microglial proliferation also occurs in response to subtle or subclinical inflammatory stimuli, such as low-level neuronal damage or chronic low-grade neuroinflammation,32,33 which in this case could have been caused by rUTIs. UTIs can trigger neuronal damage or stress 8 and microglia can also proliferate to provide additional support to vulnerable neurons or to maintain tissue homeostasis when neuronal stress or dysfunction is detected. 33 An increase in microglial cell number in the absence of extensive reactivity could thus reflect an earlier inflammatory response or show preparatory changes in response to peripheral infectious insults.
Although the nature of these changes could be complex, some of these changes could be explained by microglial heterogeneity and their location within the rostro caudal neuroaxis. Microglial heterogeneity encodes region-specific sensitivities in a trigger-dependent manner. 34 For instance, microglia from the midbrain show a context dependent response after LPS administration and are at a constitutive state of alert after LPS treatment.34–36 Steady-state microglia in the cerebellum exist in a more immune-vigilant state and are metabolically more active since in these regions fewer microglia need to surveil larger brain volumes. 34 During aging, immune amplifying genes are strongly upregulated in white matter cerebellar microglia. 34 Our analysis shows that microglia especially in the caudal part of the neuroaxis (Figure 4(d)) are significantly increased in numbers (Figure 4(c)), and this might therefore indicate that microglia could be specifically triggered into a heightened immune-vigilant state after rUTIs within white matter caudal regions.
Another region of particular interest is the dorsal motor complex. This region is one of the earliest regions implicated in PD. 37 The dorsal motor complex is involved in regulating visceral pain 38 and sickness behavior22–24 via the AP, NTS and DMX that together integrate autonomic and humoral input during infection and inflammation. 39 We find significantly increased microglial numbers after chronic UTIs in all regions analyzed of the sensory dorsal motor complex (Figure 5). 40 Sustained microglial activation could thus potentially be priming the DMX for the development of age-related pathology or Lewy pathology.
In addition to these observations of increased microglial cell numbers, we found that rUTIs also caused microglia to become reactive in the cingulate cortex. During peripheral inflammation, circulating pro-inflammatory cytokines and other immune molecules can cross the blood-brain barrier or signal to the brain via neural and humoral pathways.40,41 Pro-inflammatory cytokines released during UTI, such as interleukins, can directly activate microglia in the brain or affect neuronal health in cortical regions during acute infection.8,40 These signals can stimulate microglia in various brain regions, including the cingulate cortex, after which they become reactive. We examined the presence of the interleukin-6 (IL-6) in the cingulate cortex, we could not detect a positive signal at the final time point (data not shown). We also assessed other commonly used marker for microgliosis, such as MHCII and CD68 but we could not detect any positive signal in the cingulate cortex or any other brain area examined (data not shown).
Microglia in the cingulate cortex can also communicate bidirectionally with neurons and other glial cells in response to peripheral inflammation.41,42 This neuroimmune communication can amplify microglial activation and contribute to the development of microgliosis. The cingulate cortex is an important region for the sensing of inflammatory pain.43,44 In conjunction, the NTS receives visceral pain input from the vagal nerve and relays this information to higher brain regions. 24 Cortical pain can be processed via visceral afferents that first reach the NTS.24,45,46 Microglia in these regions can rewire neuronal connections in response to neuronal activity. 42 Microglial changes in the NTS or the cingulate cortex resulting from rUTIs could therefore have functional consequences, such as alterations in synaptic plasticity, neurotransmission, locomotion and cognitive or emotional processing.
These microglial regional differences after rUTIs might influence selective neuronal vulnerability, or prime brain areas for subsequent insults as in the case for PD or different subtypes of MSA, such as in MSA-C. It is not yet studied how microglial priming after UTI might be involved in neurodegenerative disorders 47 but understanding these disease related mechanisms will be essential for elucidating the role of microglia in mediating the effects of peripheral inflammation during UTIs in synucleinopathies and thus warrants further investigations. Altogether, these results show the presence of persisting inflammatory response after rUTIs in specific areas of the brain.
In addition to determining the effects of rUTIs on central inflammation, we defined if these peripheral infections can also affect neuronal health or cause synucleinopathy. We previously demonstrated that in animals with viral vector-mediated oligodendroglial overexpression of ɑSyn, UTIs can aggravate synucleinopathy in the central nervous system. 6 Here, we investigated the effects of repeated chronic UTIs in naïve humanized ɑSyn mice that do not overexpress oligodendroglial ɑSyn. Via whole brain analysis of the pathological marker Pser129-ɑSyn, we did not find any significant changes in the detection of Pser129-ɑSyn. However, in the cingulate cortex we observed a strong positive correlation between microglial reactivity and Pser129-ɑSyn+ inclusions burden after rUTIs (Figure 5).
Even in the absence of clear synucleinopathy in this model, it is warranted to further explore the effects of UTIs in a progressive model of PD or MSA. Urinary symptoms are common in PD and MSA. However, UTIs are often classified as urinary dysfunction or other neurological causes that would thus mask an ongoing UTI. 11 Impaired bladder emptying, or impaired bladder function are typical of PD and MSA, and therefore could be a potential risk factor of UTIs. In people with PD or MSA, UTIs can lead to sudden deterioration,11,48 but the reasons behind this are not understood. UTIs can lead to delirium and precipitate or worsen existing neurological conditions, including dementia.11,48–51 This work thus shows that central effects can mediated via peripheral UTIs, causing a neuroinflammatory response with associated behavioral changes and that UTIs thus could potentially contribute to these effects.
In this study we only used female mice because of the chosen methodology. Studies have shown that a bias exist in the immune response and neuroinflammatory response. Women generally have a greater susceptibility to infectious diseases. 52 However, men generally exhibit more neuroinflammation than women in PD.53,54 This is reflected in higher microglial activation and reactivity during neuroinflammation. 55 This could differentially affect disease progression of PD after UTIs. Male sex is associated with severe symptoms and faster progression of PD. 56 A higher neuroinflammatory response could lead to a more severe disease course. It therefore remains to be tested how male mice would respond and if a differential response could influence the progression of disease after UTIs.
In conclusion, we show that rUTIs can become chronic in humanized ɑSyn transgenic mice. These rUTIs lead to behavioral impairment and neuroinflammatory changes in the brain. Given the relevance of UTIs during multiple stages of MSA and PD, this work further warrants the investigation and the role of UTIs in synucleinopathies. Future work will need establish the role of these common bacterial infections in disease onset and progression. Newly developed animal models that mimics these conditions could also be valuable tools to address new research questions and further assess the potential clinical relevance of UTIs in synucleinopathies. These advancements could ultimately help people who are currently suffering with PD or MSA and support preventive measure for those who are at risk of developing these diseases.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241289046 - Supplemental material for Chronic urinary tract infections cause persistent microglial changes in a humanized ɑ-synuclein mouse model
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241289046 for Chronic urinary tract infections cause persistent microglial changes in a humanized ɑ-synuclein mouse model by Gabriela Mercado, Ann-Céline Clabout, Vanessa Howland, Ehsan Arkin, Anna Barber Janer, Dieter Plessers, Jennifer A Steiner, Wanli W Smith, Tom Hannan, Patrik Brundin and Wouter Peelaerts in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We thank the KU Leuven Imaging Core for the use of the confocal microscope. We thank the Van Andel Institute staff from the Transpark Laboratory, Allison Lindquist, Carla Thomas, and the Vivarium at the Van Andel Institute (RRID:SCR_023211) for caring for the mice used in this study. We thank Dr Tom Hannan for kindly providing the UPEC strain used in this study.
ORCID iDs
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the FWO Flanders Post-Doctoral Fellowship (WP) and Farmer Family Foundation (PB).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: W.P. has received commercial support for research from Myrtelle Gene Therapeutics. W.P. has received support as a consultant from Roche. During the time that this study was being conducted PB became an employee of F. Hoffmann-La Roche and obtained stock in the company, although none of the data were generated by this company. He also has ownership interests in Acousort AB, Axial Therapeutics, Enterin Inc and Kenai Therapeutics. All other authors declare no additional competing financial interests.
Data availability
All data are available in the main text or the supplemental material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
