Abstract
Background
Multiple myeloma (MM) is virtually always preceded by monoclonal gammopathy of undetermined significance (MGUS). Elevated serum markers are used to classify MGUS patients into clinical risk categories. Previous research has indicated that the absence of a normal plasma cell signature in MGUS is linked to early progression.
Objective
To confirm that the presence of a “polyclonal plasma cell (PolyPC) signature” serves as a robust negative predictor of MGUS progression.
Methods
374 MGUS patients were enrolled, including 334 patients with stable disease and 40 patients who progressed to MM within 10 years. An oligonucleotide microarray analysis was performed on mRNA extracted from CD138-selected bone marrow plasma cells to evaluate gene expression profiles. The PolyPC signature was developed and validated to assess its role in predicting disease progression. Statistical analyses included Cox proportional hazards models to evaluate progression risk and receiver operating characteristic (ROC) curve analysis to determine the sensitivity, specificity, and overall predictive performance of the PolyPC score.
Results
Through this retrospective study, we developed PolyPC signature based on gene expression profiles of normal, uninvolved plasma cells to predict MGUS progression risk. ROC analysis demonstrated that this signature accurately predicted the risk of MGUS progression (C-statistic: 0.792). A PolyPC score ≤ 11.6 identified a subset of 89 patients with a 10-year progression probability of 31.5% (28/89), while the remaining 285 patients had a progression probability of only 4.2% (12/285) (p < 0.01). Sensitivity and specificity were 70% (28/40) and 81.7% (273/334). The external validation using the SWOG-S0120 dataset reinforces the robustness and clinical applicability of the PolyPC score in predicting MGUS progression to MM.
Conclusions
The strength of the PolyPC signature is a powerful negative predictor of MGUS progression. These findings support incorporating PolyPC into MGUS management to identify patients needing more frequent and intensive monitoring.
Keywords
Introduction
More than 176,000 patients are diagnosed with multiple myeloma (MM) globally each year. 1 MM is a malignancy of monoclonal antibody-secreting plasma cells in the bone marrow.2,3 This disease is almost always preceded by an asymptomatic phase known as monoclonal gammopathy of undetermined significance (MGUS) or smoldering multiple myeloma (SMM).4,5 MGUS is found in 3.2% of individuals aged 50 and older and in 5.3% of those aged 70 and older.6,7 Long-term retrospective studies have shown an annual 1.0% risk of MGUS to MM progression.6,8 MM patients who previously had MGUS have better overall survival rates than those without a known MGUS phase. 9 Early intervention in high-risk MGUS patients could potentially prevent or delay progression to MM and its associated organ damage, thereby improving survival. 10 However, this must be balanced against the risk of unnecessary treatment, as the overall risk of progression to MM is low. Identifying patients with a high risk of MM progression and who would benefit from early therapeutic intervention remains a challenge.
Currently, serum markers are used to stratify MGUS patients into clinical risk groups to estimate the risk of progression from MGUS to MM.10–12 This stratification has led to the development of clinical consensus guidelines but does not incorporate genetic or whole genome characteristics.13,14 Many genetic changes associated with myelomagenesis are present at the MGUS stage.15,16 Gene expression profiling (GEP) is a powerful tool for analyzing biological and clinical heterogeneity and has been used to define seven molecular subgroups in both MM and MGUS. 17 A 70-gene signature (GEP70) identifies high-risk MM with a median survival of 2.5 years in spite of intensive therapy with tandem stem cell transplants. 18 In our previous research, we developed the gene signature 36 (GS36) model to predict the risk of MGUS progression to MM. We found that the absence of a normal plasma cell signature in MGUS is associated with early progression. 19
Based on these findings, we hypothesized that the presence of a polyclonal plasma cell (PolyPC) signature serves as a robust negative predictor of MGUS progression. In this study, we aimed to validate the predictive utility of the PolyPC score by evaluating its ability to identify patients at low risk of progression to MM. Our goal was to establish the PolyPC signature as a clinically relevant biomarker that can enhance current risk stratification strategies and inform personalized monitoring for MGUS patients.
Materials and methods
Study population and eligibility criteria
From 2000 to 2018, a total of 374 patients with newly diagnosed MGUS were consecutively enrolled at the University of Arkansas for Medical Sciences. Diagnosis was based on standard clinical and laboratory criteria, including the presence of a serum monoclonal (M) protein <3 g/dL, bone marrow plasma cells <10%, and absence of CRAB features (hypercalcemia, renal insufficiency, anemia, bone lesions). MRIs were used in the diagnostic workup to exclude occult bone damage and ensure accurate classification.
Inclusion criteria for this study were: (1) confirmed diagnosis of MGUS, (2) availability of baseline bone marrow aspirates, and (3) patient consent for longitudinal follow-up and gene expression analysis. Exclusion criteria included prior diagnosis of MM or smoldering myeloma, presence of end-organ damage, or incomplete clinical or molecular data.
Patients were selected for GEP if high-quality CD138-selected bone marrow plasma cells were available and sufficient RNA yield was obtained for microarray analysis. GEP was successfully conducted in all 374 enrolled MGUS patients. Among this cohort, 174 patients contributed a total of 593 serial bone marrow samples over the course of follow-up.
During longitudinal monitoring, patients underwent routine clinical assessments, including serial bone marrow examinations, imaging studies (PET/CT and/or MRI), blood counts, and serum protein electrophoresis to evaluate monoclonal protein levels. Additional laboratory tests included serum lactate dehydrogenase (LDH), serum β-2-microglobulin (B2 M), and serum free light chains (SFLC). All procedures were approved by the Institutional Review Board of the University of Arkansas for Medical Sciences, and written informed consent was obtained from all participants in accordance with the Declaration of Helsinki.
Plasma cell selection and microarray processing
Plasma cells were isolated using anti-CD138 immunomagnetic beads that enrich mononuclear cell fractions from bone marrow aspirates. Post-selection, samples analyzed by microarray contained over 85% plasma cells, which was verified by two-color flow cytometry (CD38+ and CD45−/dim). 17 The average plasma cell purity was 89.6% and ranged from 85% to 100%. GEP was performed using the Affymetrix U133Plus2.0 microarray platform, which includes 54,613 gene probes. Affymetrix MAS5.0 signals were normalized.
Development of the polyclonal plasma cell (PolyPC) signature
We selected the probes associated with polyclonal plasma cells. To correct skewness, a log2 transformation was performed on the Affymetrix signals. The PolyPC score was developed by averaging the GEPs. The concordance statistic (C-statistic) was used to evaluate the PolyPC score's ability to predict MGUS progression within 10 years in the test datasets. An optimal cutoff point maximizing sensitivity and specificity on the ROC curve was determined to assess prediction accuracy and included sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV).
External validation with SWOG-S0120 dataset and comparison with mayo clinic risk stratification model
To validate the predictive capability of the PolyPC score, we utilized an independent dataset, SWOG-S0120 (GSE136400),20,21 comprising 57 MGUS patients with baseline GEP. Of these, 3 patients progressed to MM within 10 years, while the remaining 54 patients remained stable during the follow-up period. The PolyPC score was calculated for all patients based on the established methodology. A threshold score of ≤ 11.6 was applied, as determined in the initial model development phase. Sensitivity and specificity were calculated to assess the model's predictive performance.
To further evaluate the feasibility and clinical utility of the PolyPC score model, we compared its performance with the Mayo Clinic risk stratification model 10 using our dataset. Among the 374 MGUS patients included in the study, 337 had complete data available for serum M-protein concentration, M-protein isotype, and involved/uninvolved serum free light chain (FLC) ratio—parameters required to assign Mayo Clinic risk categories. For both models, we calculated and compared the C-statistic, sensitivity, and specificity in predicting progression from MGUS to multiple myeloma. These metrics allowed us to assess the discriminative ability and predictive accuracy of the PolyPC score relative to a widely accepted clinical risk model.
Statistical analysis
Baseline characteristics of MGUS patients were compared using Chi-square tests or Fisher's exact tests when expected frequencies were less than three. Time to MGUS progression was evaluated with Kaplan-Meier curves and compared using log-rank tests. Univariate Cox proportional hazard (CPH) models assessed individual risk factors for predicting MM progression. Significant risk factors from univariate models were further evaluated using multivariate Cox models. The optimal gene score cutoff for progression risk was determined with X-tile bioinformatics software (Yale University, version 3.6.1). Statistical analyses were performed with SPSS 12.0 (SPSS, Chicago, IL).
Results
Clinical characteristics
From 2000 to 2018, 374 MGUS patients were enrolled, all of whom had baseline GEP data available. The average age of the patients was 62.5 years, with a range of 27.3 to 91.5 years. Among these, 40 patients progressed to MM within 10 years following their MGUS diagnosis, with a progression time frame ranging from 2 months to 9.5 years. These patients were classified as the “progressing group”, while the remaining 334 patients, who did not progress during follow-up, were defined as the “stable group”. For the 334 patients in the stable group, the follow-up period ranged from 7 to 16 years, with a median follow-up of 12 years. The average age for the progressing group was 68.5 ± 9.6 years, while the stable group had an average age of 62.1 ± 12.2 years. The clinical characteristics of these cohorts are presented in Table 1. The progressing group exhibited a significantly higher proportion of patients with the IgG subtype (p = 0.037), elevated serum M-proteins (p < 0.001), abnormal FLC ratios (<0.1 or >10) (p < 0.001), and decreased levels of uninvolved immunoglobulins (p < 0.001).
Summary of demographics and baseline clinical characteristics of MGUS patients.
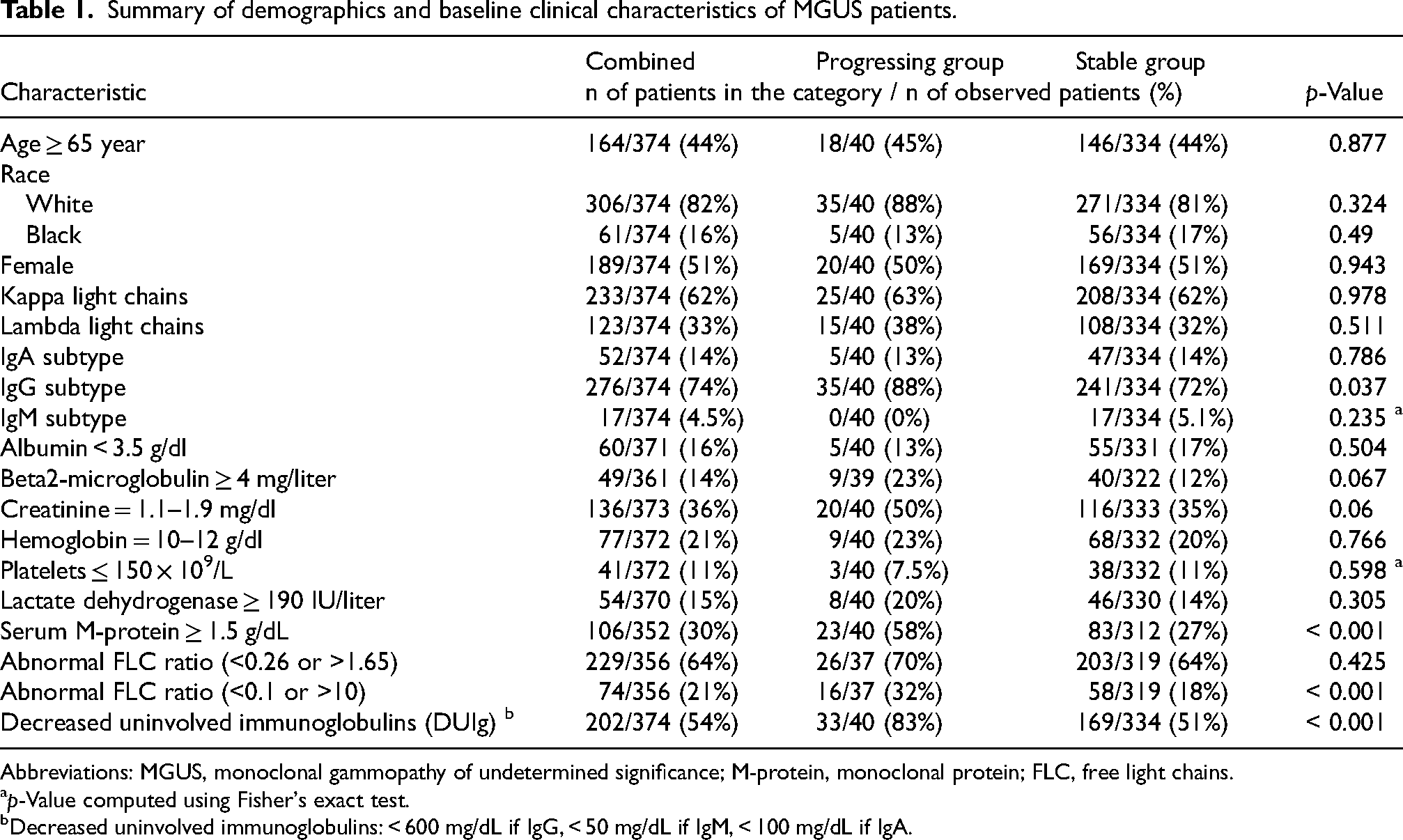
Abbreviations: MGUS, monoclonal gammopathy of undetermined significance; M-protein, monoclonal protein; FLC, free light chains.
p-Value computed using Fisher's exact test.
Decreased uninvolved immunoglobulins: < 600 mg/dL if IgG, < 50 mg/dL if IgM, < 100 mg/dL if IgA.
Defining polyclonal plasma cell (PolyPC) signature
We identified 33 probes that characterized the Polyclonal Plasma Cell (PolyPC) Signature (Table 2). These probes were selected based on their consistent overexpression in normal, uninvolved plasma cells compared to clonal plasma cells, using GEP data from baseline samples. Differential expression analysis was performed to identify genes highly enriched in the polyclonal compartment, with selection criteria including statistical significance (adjusted p < 0.05), fold-change thresholds, and biological relevance to immunoglobulin production and plasma cell function. The final set of 33 probes showed strong association with the presence of normal plasma cell activity and were used to construct the PolyPC score. A PolyPC score was developed by averaging the GEPs. PolyPC score ≤ 11.6 ROC analysis accurately predicted MGUS progression (C-statistic is 0.792) (Figure 1(a)). An optimal cut-off score of 11.6 was determined for risk assessment. Among 40 patients who developed MM within 10 years, 28 patients had a PolyPC score ≤ 11.6 or test positive (T+), yielding a sensitivity of 70.0%. For the remaining 334 stable patients, only 61 patients had PolyPC score ≤ 11.6, yielding a false positive rate of 18.3% or a specificity of 81.7%. Also, 89 patients were identified to have T+, while the remaining 285 patients tested negative (T-) or had their PolyPC score > 11.6. Among the 89 T + patients, 28 patients progressed to MM in 10 years, yielding a PPV of 31.5%. For the 285 T- patients, only 12 patients developed MM in 10 years, yielding a probability of MM progression of 4.2%, or a NPV of 95.8% (Figure 1(b)).
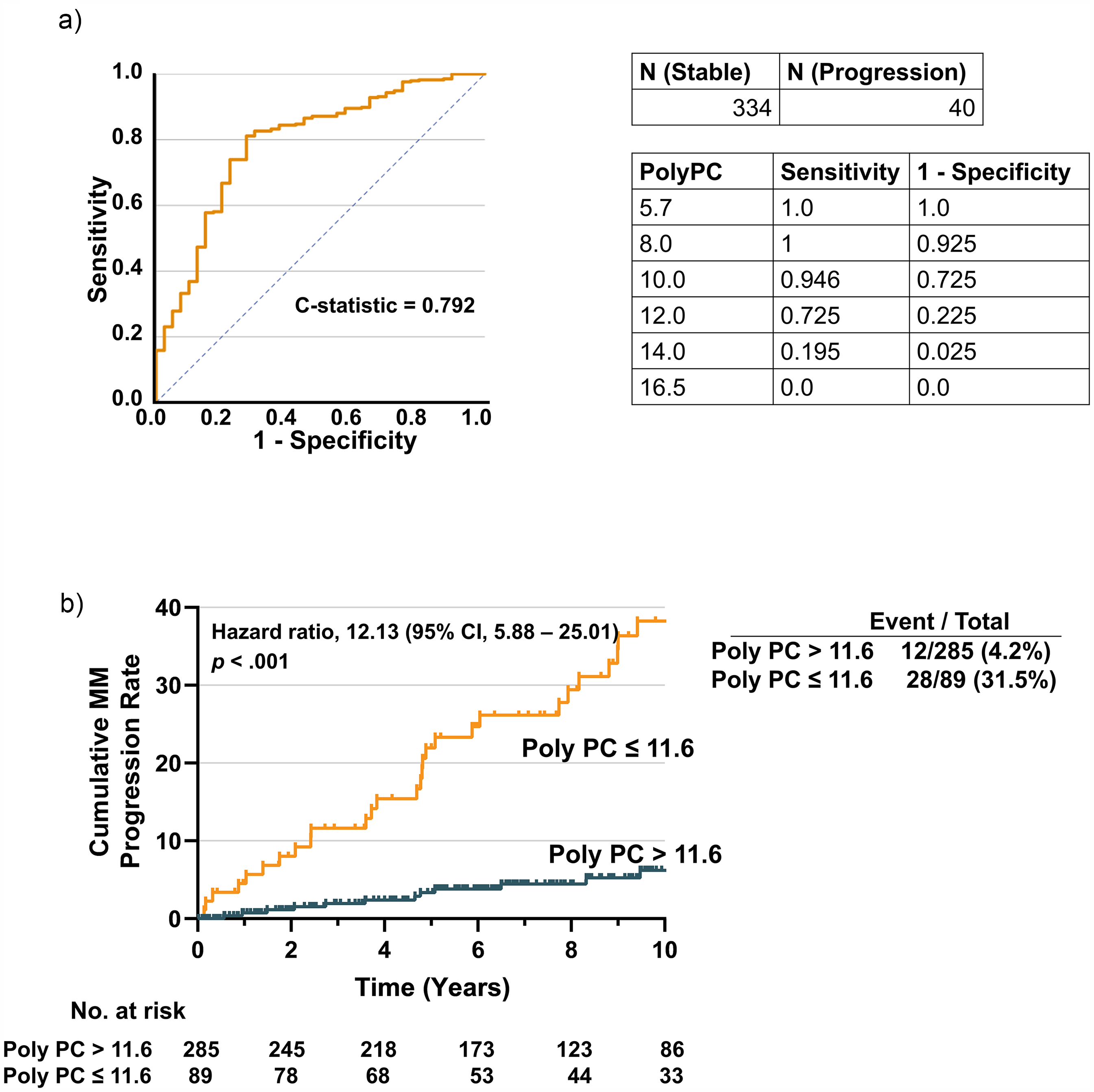
MGUS time-to-progression curves in the polyPC. (a) MGUS Time-to-progression Curves in the PolyPC. Receiver operating characteristic curve (ROC) based on PolyPC. The C-statistic of ROC is 0.792. (b) Time-to-progression curve based on PolyPC. Among 40 patients who developed MM within 10 years, 28 patients had a PolyPC score ≤ 11.6 or test positive (T+), yielding a sensitivity of 70.0%. For the remaining 334 stable patients, only 61 patients had PolyPC score ≤ 11.6, yielding a false positive rate of 18.3% or a specificity of 81.7%. Also, 89 patients were identified to have T+, while the remaining 285 patients tested negative (T-) or had their PolyPC score > 11.6. Among the 89 T + patients, 28 patients progressed to MM in 10 years, yielding a positive predictive value (PPV) of 31.4%. For the 285 T- patients, only 12 patients developed MM in 10 years, yielding a probability of MM progression of 4.2%, or a negative predictive value (NPV) of 95.8%.
PolyPC signature genes.

Abbreviations: PolyPC, Polyclonal Plasma Cell.
593 serial microarray analyses were conducted on CD138-selected bone marrow samples from 174 MGUS cases with baseline samples. The PolyPC score consistently decreased as patients transitioned from MGUS to MM (Figure 2(a)), whereas it remained stable in those without progression (Figure 2(b)).
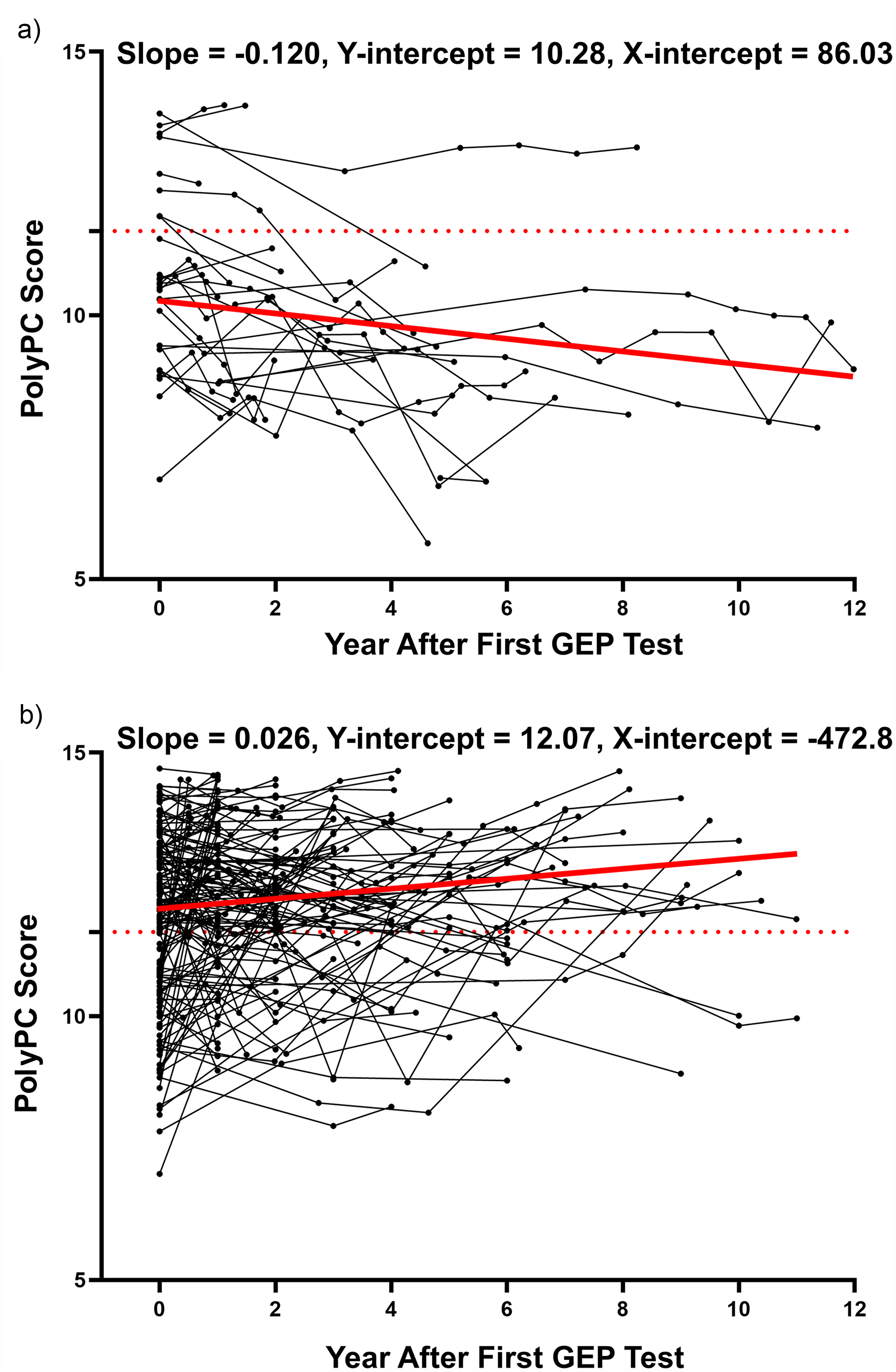
PolyPC score progression curves of stable group and progressing group. (a) A total of 593 serial microarray analyses were performed on CD138-selected bone marrow samples from 174 MGUS cases with a baseline sample. The PolyPC score invariably increased as the patients transitioned from MGUS into MM. Each line in the plot represents a unique patient and each point represents a unique PolyPC score at that time. (b) The PolyPC score remained stable in the group without progression to MM.
Next, we performed external validation using an independent dataset SWOG-S0120.20,21 In the S0120 datasets, we obtain 57 MGUS patients with baseline GEP (GSE136400). 3 patients progressed to MM within 10 years, while the other 54 MGUS had not progressed. Among 3 patients who developed MM, all had a PolyPC score ≤ 11.6, yielding a sensitivity of 100%. For the remaining 54 stable patients, only 9 patients had PolyPC score ≤ 11.6, yielding a specificity of 83.3% and a NPV of 80.0% (Figure S1).
We compared the performance of the PolyPC score with the Mayo Clinic risk stratification model in a subset of 337 MGUS patients with available data on serum M-protein concentration, M-protein isotype, and FLC ratio. 10 The PolyPC score demonstrated superior overall predictive accuracy, with a higher C-statistic (0.792) compared to the Mayo Clinic model (0.653). In identifying patients who progressed to MM, the PolyPC score achieved a sensitivity of 70%, substantially higher than the 24.3% sensitivity observed in the high-risk category of the Mayo model. However, this improvement in sensitivity was accompanied by a moderate reduction in specificity, with the PolyPC score yielding 81.7% specificity versus 97.7% for the Mayo model (Figure S2). These results highlight the enhanced ability of the PolyPC score to detect true progressors, while also illustrating the trade-off between sensitivity and specificity. The findings underscore the potential benefit of integrating gene expression–based models like PolyPC with established clinical parameters to improve MGUS risk stratification.
Identifying clinical and molecular parameters associated with MGUS progression
Each clinical characteristic was evaluated for its association with MGUS progression time using a univariate CPH model (Table 3). Increased risk of progression to MM was linked to serum M-protein ≥ 1.5 g/dL, abnormal FLC ratio (<0.1 or >10) and decreased uninvolved immunoglobulins. The PolyPC score had a very high hazard ratio (HR = 7.31, p < 0.001), alongside decreased uninvolved immunoglobulins (HR = 3.67, p = 0.002), high serum M-protein (HR = 2.84, p = 0.001), and abnormal FLC ratio (HR = 3.07, p = 0.001).
Univariate cox proportional hazards analysis of risk factors of MGUS progression.
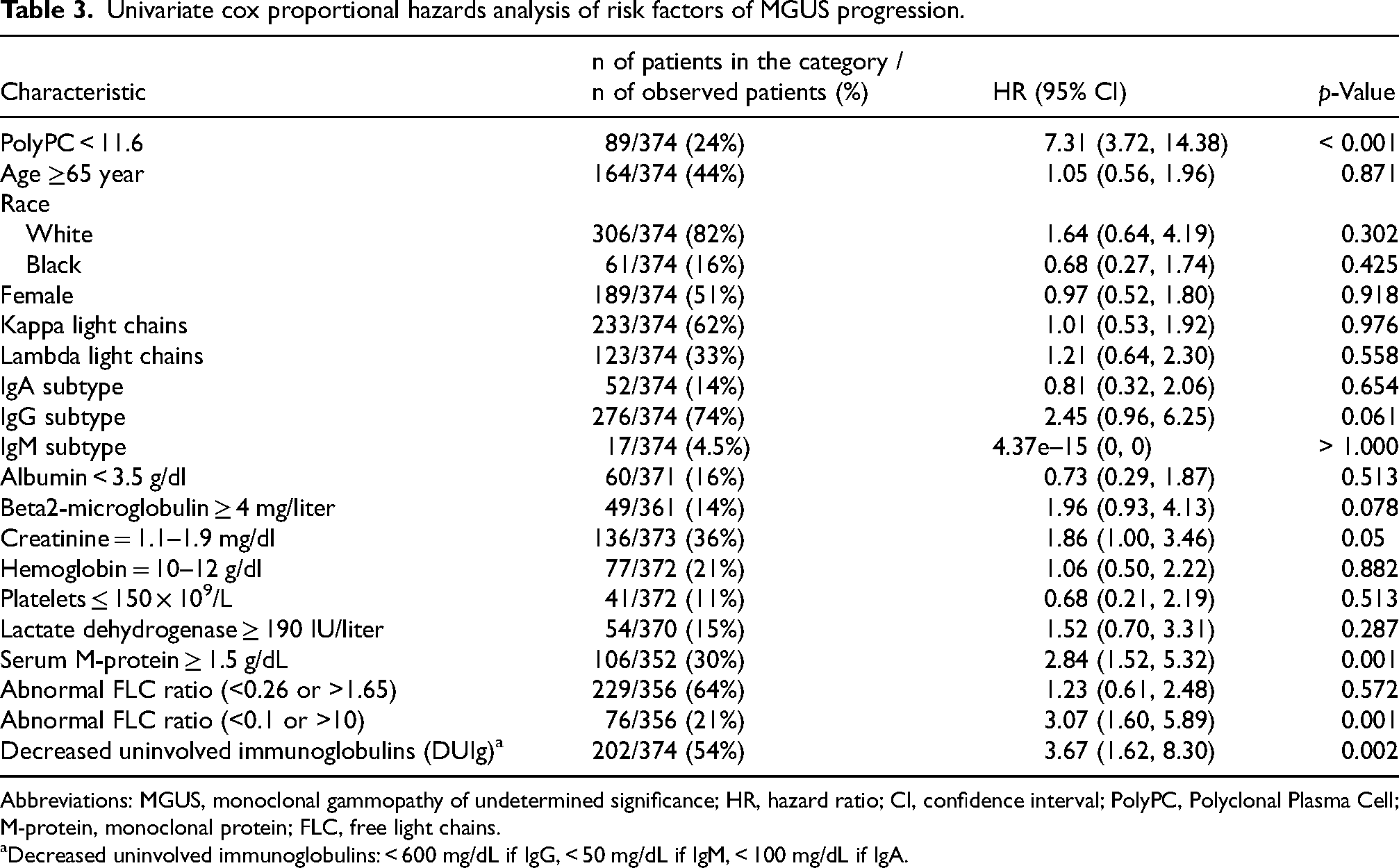
Abbreviations: MGUS, monoclonal gammopathy of undetermined significance; HR, hazard ratio; CI, confidence interval; PolyPC, Polyclonal Plasma Cell; M-protein, monoclonal protein; FLC, free light chains.
Decreased uninvolved immunoglobulins: < 600 mg/dL if IgG, < 50 mg/dL if IgM, < 100 mg/dL if IgA.
These significant factors were included in a multivariate CPH model, showing PolyPC (HR = 5.41, p < 0.001) as the only independent risk factor for progression (Table 4).
Hazard ratios (HRs) of MGUS progression predicted by risk factors using a multivariate cox proportional hazard model.
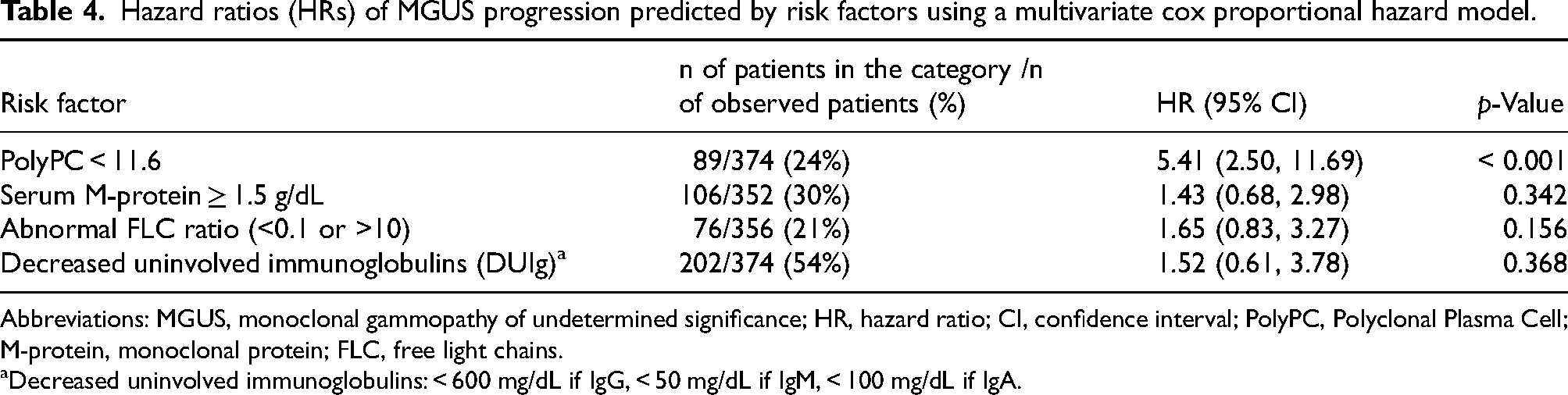
Abbreviations: MGUS, monoclonal gammopathy of undetermined significance; HR, hazard ratio; CI, confidence interval; PolyPC, Polyclonal Plasma Cell; M-protein, monoclonal protein; FLC, free light chains.
Decreased uninvolved immunoglobulins: < 600 mg/dL if IgG, < 50 mg/dL if IgM, < 100 mg/dL if IgA.
Discussion
Global GEP of highly purified plasma cells from 374 MGUS patients revealed that the Polyclonal Plasma Cell (PolyPC) score is a robust predictor of progression risk. Among the cohort, 40 patients progressed to MM within 10 years, while 334 remained stable. The PolyPC score predicted the 10-year probability of progression with a sensitivity of 70.0% and a specificity of 81.7%, enabling effective stratification of high-risk and low-risk patients. Additionally, the score achieved a high NPV of 95.8%, ensuring reliable identification of patients unlikely to progress. These findings emphasize the clinical value of the PolyPC score as a predictive tool for MGUS progression, supporting its potential integration into routine clinical practice for improved risk stratification and patient management.
Meanwhile, the external validation using the SWOG-S0120 dataset reinforces the robustness and clinical applicability of the PolyPC score in predicting MGUS progression to MM. The observed sensitivity of 100% highlights the model's capacity to accurately identify all patients at risk of progression within the cohort, while the specificity of 83.3% ensures reliable discrimination of low-risk patients. These findings also validated the accuracy of the PolyPC. The comparative analysis with the Mayo Clinic risk stratification model further underscores the clinical relevance of the PolyPC score. While the PolyPC model achieved higher sensitivity and overall predictive accuracy, the Mayo model demonstrated superior specificity. This trade-off suggests that each model captures distinct aspects of MGUS progression biology—clinical versus molecular—and that their integration may provide a more balanced and precise risk assessment.
MGUS is considered a premalignant plasma-cell disorder with a lifelong risk of progression to MM.22,23 Since MGUS progression to MM can cause permanent bone damage and immune dysfunction in MM, 24 early intervention to prevent such a progression might enhance long-term disease control and a potential cure. Therefore, treating MM at its earliest stages is advantageous. 25 Currently, the Dana-Farber Cancer Institute is conducting a clinical trial using Daratumumab to treat high-risk MGUS patients (NCT03236428). Identifying patients at increased risk of progression to MM is crucial to justify early therapeutic intervention. This study demonstrates that integrating genomic data from purified plasma cells in the bone marrow of MGUS patients can aid in clinical management.
At present, stratification of MGUS patients into clinical risk categories primarily relies on serum markers to estimate the likelihood of progression to MM. This approach has informed the creation of widely accepted clinical guidelines. 26 However, this approach lacks integration of genetic and genome-wide data, which could provide deeper insights into disease biology and improve risk prediction accuracy. GEP analysis of purified CD138+ plasma cells have proven to be a robust tool for predicting high-risk disease in MM.17,27
We identified 33 probes that characterized the PolyPC signature (Table 2). The genes predominantly encode components of immunoglobulins, which are critical for plasma cell function and antibody production. For example, genes such as IGLC1, IGKC, and IGLJ3 are involved in the constant and joining regions of light chains (kappa and lambda), playing a key role in the structural assembly of immunoglobulins. Similarly, IGHM, IGHG1, and IGHV3-23 encode regions of heavy chains, including constant and variable segments that are essential for antigen recognition and immune response. The expression levels of these genes reflect the activity of normal plasma cells and their capacity for immunoglobulin production, which is disrupted during the transition from MGUS to MM.
The PolyPC signature demonstrates significant potential as a predictive tool for identifying MGUS patients at risk of progression to MM. With a sensitivity of 70%, the model effectively identifies a substantial proportion of high-risk patients, although this also highlights an area for improvement. Due to the inherent complexity and heterogeneity of MGUS progression to MM, it is challenging for a single model to achieve 100% sensitivity. 11 However, the model's high NPV of 95.8% ensures accurate identification of low-risk patients, reducing unnecessary concern or interventions for those unlikely to progress. Additionally, the specificity of 81.7% underscores the model's robustness in minimizing false positives, which is crucial for avoiding overtreatment and optimizing resource allocation. Compared to other existing models, 26 the PolyPC signature strikes a strong balance between sensitivity and specificity, offering both predictive accuracy and clinical relevance. Nonetheless, certain limitations should be acknowledged. While the PolyPC score outperforms the Mayo Clinic model in sensitivity, it shows moderately reduced specificity, suggesting that integration with clinical features may further enhance precision. Moreover, the limited racial diversity in our cohort—82% of patients were White—may constrain the generalizability of the findings. Future studies should validate the model across broader, more diverse populations and in combination with established clinical parameters.
To further enhance the sensitivity of the PolyPC signature, future work should explore integrating the score with additional clinical and molecular markers, such as serum free light chain ratios, advanced imaging modalities, or comprehensive genomic profiling, to create a multi-modal predictive framework. Such approaches could refine risk stratification by overcoming the limitations of single-modality models, improving overall predictive performance. Validating the PolyPC signature in larger, independent cohorts with diverse clinical settings will be essential to establishing its generalizability and utility, positioning it as a valuable tool for personalized monitoring and timely intervention in MGUS patient management.
Conclusion
In conclusion, we developed and validated a PolyPC score with excellent accuracy in predicting the 10-year progression of MGUS to MM. The PolyPC score demonstrated a strong balance between sensitivity and specificity, with a high NPV ensuring reliable identification of low-risk patients. These findings highlight the potential of the PolyPC signature as a tool for improving risk stratification and guiding personalized management in MGUS patients. By identifying patients at higher risk of progression, the PolyPC score can help clinicians prioritize monitoring and intervention strategies, reducing unnecessary follow-ups for low-risk individuals. Future work should focus on integrating the PolyPC score with additional clinical and molecular markers to enhance its predictive power and generalizability. Additionally, larger validation studies in diverse populations will be essential to confirm its clinical utility. Incorporating such gene-based signatures into routine MGUS patient management represents a promising step toward precision medicine in hematologic malignancies.
Supplemental Material
sj-pdf-1-cbm-10.1177_18758592251344936 - Supplemental material for Polyclonal plasma cell (PolyPC) signature as a key indicator for predicting the progression of MGUS to multiple myeloma
Supplemental material, sj-pdf-1-cbm-10.1177_18758592251344936 for Polyclonal plasma cell (PolyPC) signature as a key indicator for predicting the progression of MGUS to multiple myeloma by Fumou Sun, Yan Cheng, Catherine Ma, Hongwei Xu, Clyde Bailey, David Mery, Timothy Cody Ashby, Daisy Alapat, Yong Li, Ken H Young, Samer Al Hadidi, Sharmilan Thanendrarajan, Carolina Schinke, Maurizio Zangari, Frits van Rhee, Guido Tricot, John D Shaughnessy Jr and Fenghuang Zhan in Cancer Biomarkers
Footnotes
Acknowledgments
We are indebted to the clinicians of the Myeloma Institute for Research and Therapy for referring patients to this study and to all the patients who have helped us in our pursuit of a cure.
ORCID iDs
Author contributions
Conception: John D. Shaughnessy, Jr, Fenghuang Zhan
Interpretation or analysis of data: Fumou Sun, John D. Shaughnessy, Jr
Preparation of the manuscript: Fumou Sun, Yan Cheng, Catherine Ma
Revision for important intellectual content: Fumou Sun, Yan Cheng, Catherine Ma, Hongwei Xu, Clyde Bailey, David Mery, Timothy Cody Ashby, Daisy Alapat, Yong Li, Ken H. Young, Samer Al Hadidi, Sharmilan Thanendrarajan, Carolina Schinke, Maurizio Zangari, Frits van Rhee, Guido Tricot, John D. Shaughnessy, Jr, Fenghuang Zhan
Supervision: John D. Shaughnessy, Jr, Fenghuang Zhan
Ethical approval and informed consent
The University of Arkansas for Medical Sciences Institutional Review Board approved the studies, and all participants gave written informed consent in line with the Declaration of Helsinki.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the National Cancer Institute U54CA272691-01 (F.Z., J.D.S., K.H.Y., Y.L., and T.C.A.), 1R01CA236814-01A1 (F.Z.), the Veterans Administration I01 BX006235 (F.Z.), the Leukemia Lymphoma Society LLS 6702-25 (F.Z.), the Paula and Rodger Riney Foundation (F.Z.), the Myeloma Solution Fund (F.Z.), and UAMS Winthrop P. Rockefeller Cancer Institute (WRCRI) Fund (F.Z.).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study can be obtained from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
