Abstract
Keywords
Introduction
Female reproductive system malignancies are classified in the latest WHO classification as tumours of the fallopian tube, tumours of the uterine corpus, tumours of the uterine cervix, tumours of the vagina, tumours of the vulva, gestational trophoblastic disease, and others. 1 Among these, cervical cancer, uterine cancer, and ovarian cancer are relatively common, while vaginal and vulvar cancers are relatively rare, and other types of cancers are extremely rare. Female reproductive system tumours have a significant impact on women's reproductive health and play an important role in global cancer mortality rates. 2 Cervical cancer is the fourth most common cause of cancer-related death among women worldwide, 3 with squamous cell carcinoma being the most common histological type. 4 Squamous ovarian carcinoma is a rare tumour with a tendency to spread locally and has a poor prognosis once it metastasises to the abdomen or distant sites. 5 Vaginal and vulvar cancers are most commonly squamous cell carcinomas that affect the entire reproductive system. 6
Despite the fact that there are numerous treatments for female reproductive system tumors that can significantly increase longevity, they can have a severe impact on fertility. 7 Prediction models are crucial for early identification and treatment, as well as for enhancing quality of life and survival rates, particularly in women's reproductive health.
The prognosis of various tumor histological types varies. Compared to non-keratinizing squamous cell carcinomas, keratinizing squamous cell carcinomas have a worse prognosis and a higher likelihood of becoming advanced tumors. 8 This study aims to investigate the prognosis of keratinizing squamous cell carcinoma of the female reproductive system, identify independent risk factors affecting patients’ prognoses using a competing risk model, and develop a nomogram. Unlike other studies on the ‘female reproductive system,’ this research primarily focuses on the prognosis of keratinizing squamous cell carcinoma of the female reproductive system. Furthermore, this study primarily relied on the Surveillance, Epidemiology, and End Results (SEER) database, which not only includes oncological characteristics but also incorporates sociodemographic attributes and various treatment modalities, allowing for a more comprehensive analysis of patient prognosis and offering a theoretical foundation for personalised clinical diagnosis and therapy.
Cancer patients face many alternative causes of death in addition to the disease itself, including suicide, heart disease, auto accidents, and others. These causes are typically regarded as competing risk events. 9 Traditional survival methods, such as the Cox proportional hazards model, treat the outcome as a single endpoint, which increases the likelihood of bias and overestimates cumulative incidence. In contrast, the competing risk model fully accounts for the effect of competing events. Therefore, using the competing risk model to examine the risk factors for patients with keratinizing squamous cell carcinoma of the female reproductive system can enhance predictive outcomes and align them more closely with clinical reality.10,11
Materials and methods
Data source
Using SEER*Stat software (version 8.4.0.1), patients with keratinizing squamous cell carcinoma of the female reproductive system are screened.Inclusion criteria: sex: female; primary sites: “C51.0”, “C51.1”, “C51.2”, “C51.8”, “C51.9”, “C52.9”, “C53.0”, “C53.1”, “C53.8”, “C53.9”, “C54.0”, “C54.1”, “C54.2”, “C54.9”, “C55.9” and “C56.9”; ICD-O-3 Hist/behave:8071/3; Data from 2004–2015. Exclusion criteria: Patients with unknown tumor size(n = 1170), Patients with incomplete variables(n = 391), Patients with unclear AJCC of tumors(n = 105). Patient identities in the SEER database are anonymous, and other information is public, so no ethics committee approval is required, and informed consent is not required. The screening process is shown in Figure 1.

Flow chart of patient selection.
Data selection criteria
Age, race, marital status, tumor site, AJCC stage, a summary of the stage, surgery, radiotherapy status, chemotherapy status, postoperative lymph node dissection (LNSur), surgery and radiotherapy (RadSur), income, cancer-specific death (CSD), overall survival (OS), and follow-up time are variables that have been filtered out. Age and tumor size were classified according to X-tile cut-off values, where age (≤77 years and >77 years), tumor size (≤27 mm, 28–45 mm, and ≥46 mm), Race is divided into white, black and other, and marital status is divided into married, single and other. Tumor sites are divided into Ovary, Uterus, Vagina, and Vulva. The primary endpoint was cancer-specific death.
Statistical analysis
All patients were randomly divided into training set (70%, 2623) and test set (30%, 1125). The definition of a competing event for a cancer-specific death was death from another cause. Utilising the cumulative incidence function (CIF), CIF curves were created, and Gray's test was performed to compare variables between subgroups over the course of 3, 5, and 8 years. Multivariate analysis using the Fine-Gray model and the cause-specific (CS) model. Next, a prognosis nomogram is created utiliing the factors that the competing risk model filtered out.12,13
The concordance index (C-index) and time-dependent area under the receiver operating characteristic curve (AUC) were used to assess the model's discrimination. The integrated discrimination improvement (IDI) and net reclassification improvement (NRI) were used to evaluate whether the model's predictive power had improved. 14 The accuracy of the nomogram was tested based on the calibration curves. 15 To assess the validity of the nomogram, decision-curve analysis (DCA) was performed.
Categorical variables were expressed as percentages. Interquartile ranges and the median were used to express continuous variables. The statistical analysis was conducted using R software (version 4.0.1) and SPSS Statistics software (version 27.0, Chicago, IL, USA), and statistical results were considered significant at p < 0.05.
Results
Basic patient information
Table 1 is the basic information of patients in the training set and test set. Finally, 3748 patients with keratinizing squamous cell carcinoma of the female reproductive system were included, the majority of them were ≤77 years old (n = 2997, 79.9%) and white (3118, 83.2%). The distribution of marriage and tumor size within subgroups is equal. Tumor sites were mostly located in Uterus (n = 1654, 44.2%) and Vulva (n = 1925, 51.4%), AJCC stage I (n = 1415, 37.8%) predominated, followed by stages II (n = 973, 25.9%), stage III (n = 933, 24.8%), and stage IV (n = 427, 11.4%). Tumor Summary of Stage was mostly carcinoma in situ (n = 1822, 48.6%) and regional invasion (n = 1629, 43.5%), and distant metastasis (n = 297, 7.9%) was rare. The vast majority opted for surgery and postoperative lymph node dissection, and more than half of the patients opted for surgery combined with radiotherapy (n = 2586, 68.9%). The patients who did not receive radiotherapy and chemotherapy were (n = 1951, 52.1%) and (n = 2508, 66.9%), respectively. The income of patients was mainly middle-income (n = 1956, 52.2%). The median follow-up time for all patients was 60.5 ± 51.5 months. The overall mortality rate was 1865 (49.8%), of which 708 (18.9%) were deaths from other causes and 1157 (30.9%) were cancer-related.
Baseline characteristics of patients.
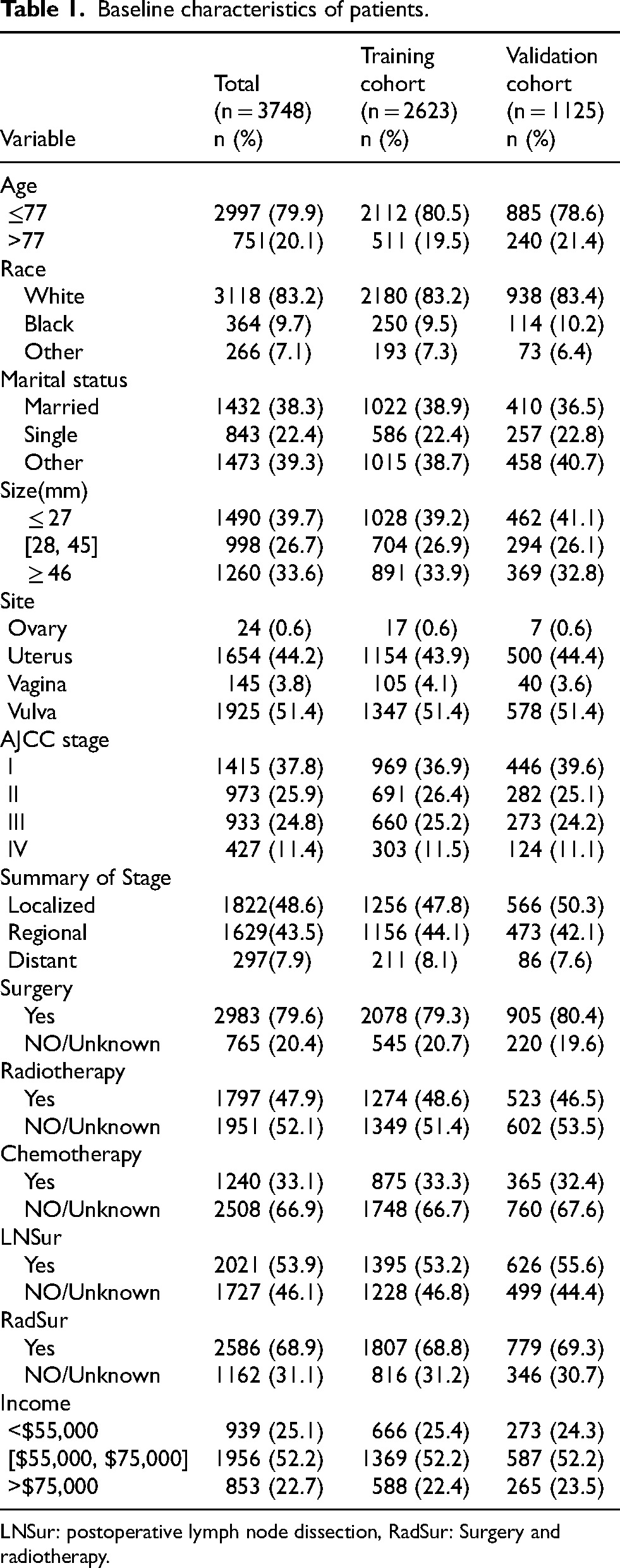
LNSur: postoperative lymph node dissection, RadSur: Surgery and radiotherapy.
Univariate survival analysis
Univariate analysis showed that age, marital status, tumor site, AJCC stage, summary of stage, surgery, radiotherapy status, chemotherapy status, postoperative lymph node dissection, surgery and radiotherapy significantly affected the prognosis of keratinizing squamous cell carcinoma of female reproductive system. The CIF results of each subgroup showed an upward trend in the cumulative incidence of CSD over 3, 5, and 8 years for most variables. Figure 2 displays the CIF curve. According to the figure, patients with stage IV of the AJCC, chemotherapy, radiotherapy, no postoperative lymph node dissection, tumors greater than 46 mm, and distant metastases had a greater cumulative incidence of CSD, with a statistically significant difference (Table 2).
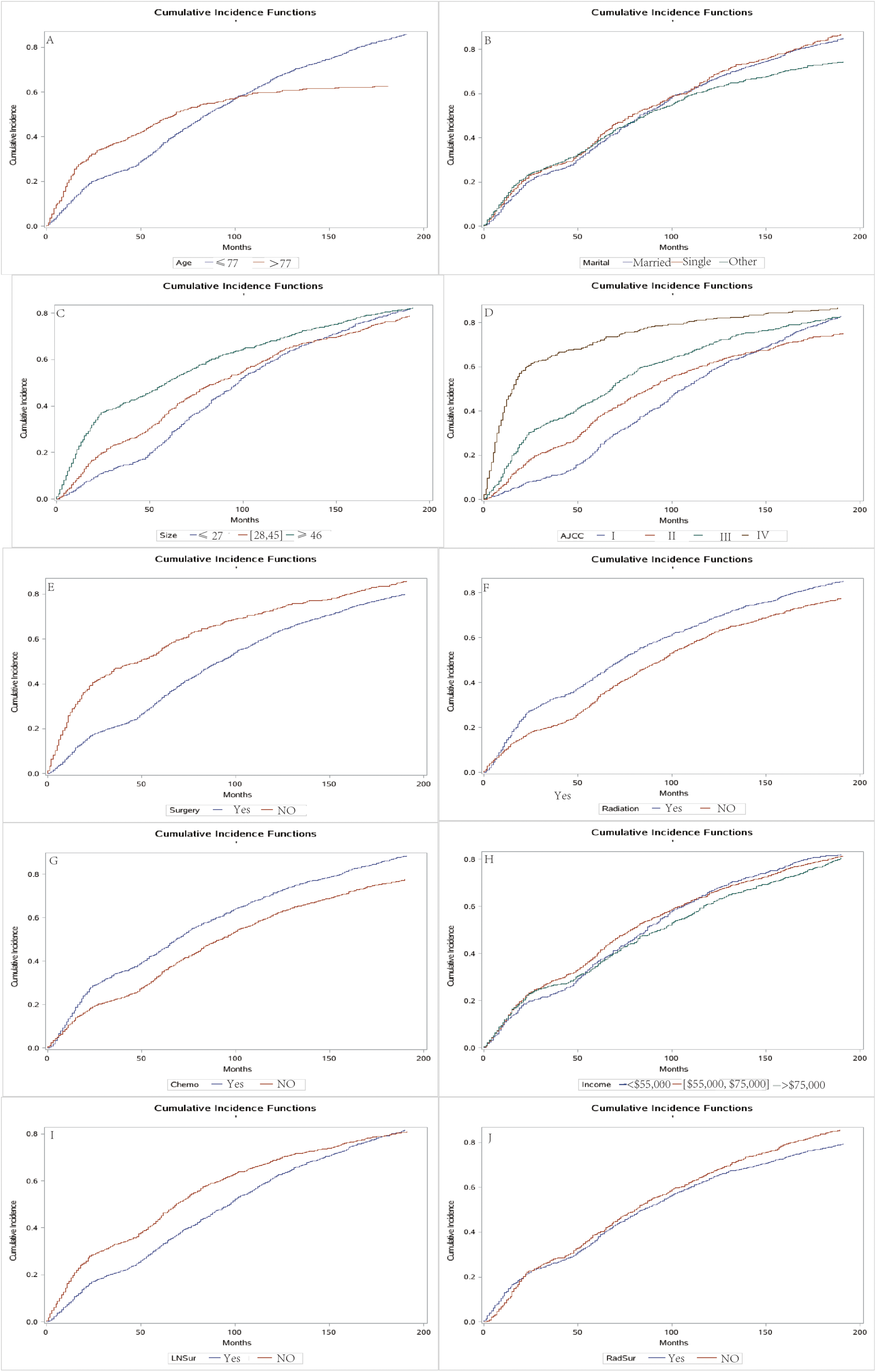
Cumulative incidence estimates of cancer-specific death in patients with keratinizing squamous cell carcinoma of female reproductive. Age (A), Marital status (B), Tumor size (C), AJCC (D), Surgery (E), Radiotherapy (F), Chemotherapy (G), Income (H), postoperative lymph node dissection (I), Surgery and radiotherapy (J).
Univariable competing risk analysis in patients with keratinizing squamous cell carcinoma of female reproductive system.
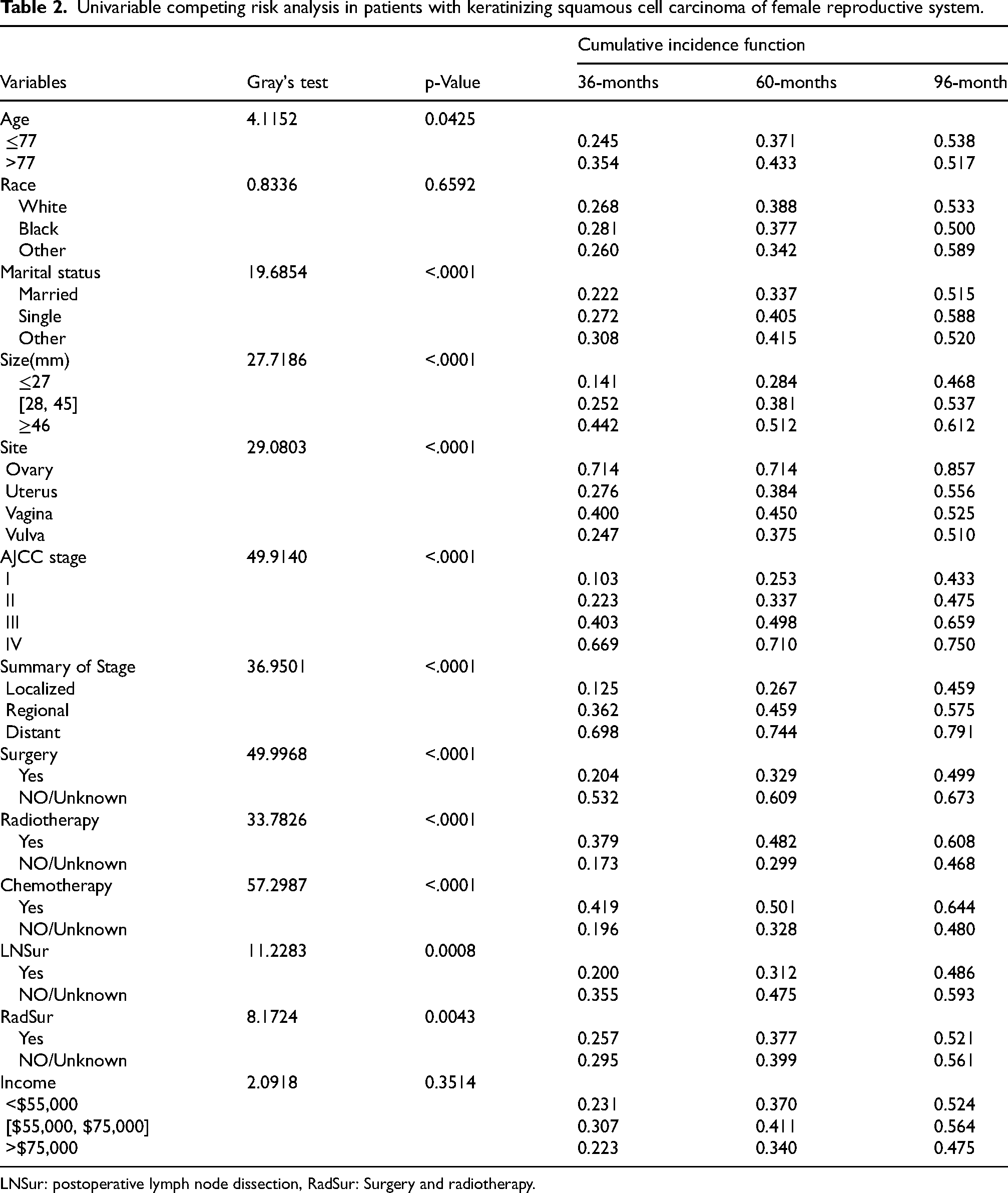
LNSur: postoperative lymph node dissection, RadSur: Surgery and radiotherapy.
Multivariable analysis
Incorporating statistically significant variables (p < 0.05) from univariate analysis into competing risk models and Cox regression models. The results of the Cox proportional hazards model demonstrated that age, race, tumor size, tumor site, AJCC stage, summary of stage, surgery, chemotherapy status, and postoperative lymph node dissection were risk factors influencing the prognosis of keratinizing squamous cell carcinoma of the female reproductive system. The findings of the Fine-Gray model demonstrated that age, marital status, AJCC stage, surgery, radiotherapy, postoperative lymph node dissection, surgery, and radiotherapy were independent risk factors influencing the prognosis of keratinizing squamous cell carcinoma of the female reproductive system. The results of the CS model revealed that age, tumor size, AJCC stage, surgery, radiotherapy, chemotherapy, postoperative lymph node dissection, surgery and radiotherapy, and income were independent risk factors affecting the prognosis of keratinizing squamous cell carcinoma of the female reproductive system (Table 3).
Multivariate analysis of keratinizing squamous cell carcinoma of the female reproductive system.
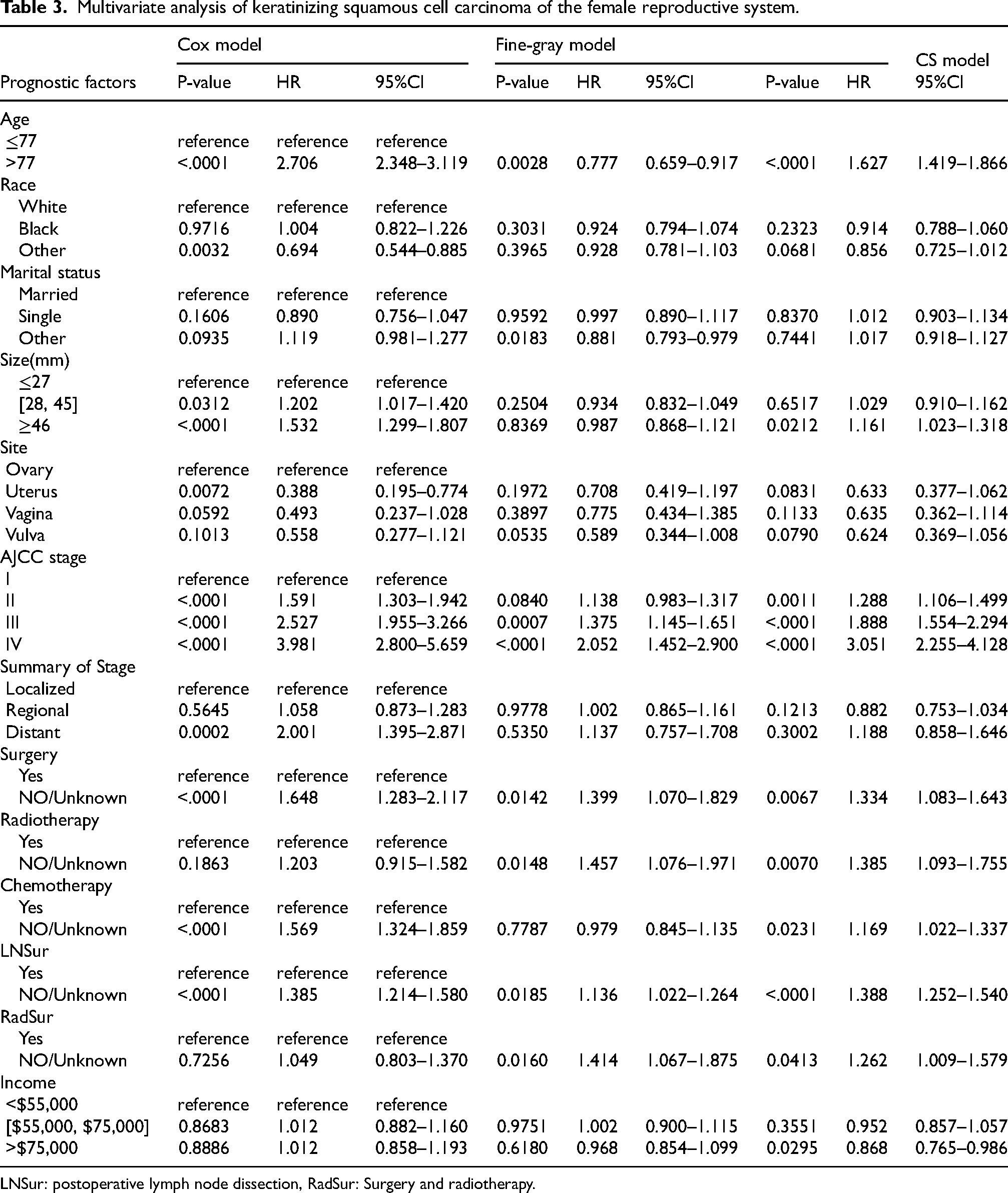
LNSur: postoperative lymph node dissection, RadSur: Surgery and radiotherapy.
Race, tumor site, and Summary of Stage were independent risk variables in the Cox proportional hazards model for keratinizing squamous cell carcinoma of the female reproductive system, but not in the competing risk model. The disparities between the competing risk model and the Cox proportional hazards model may be explained by competing risk events. Marital status, surgery and radiotherapy, and income were prognostic risk variables in the competing risk model but not in the Cox proportional hazards model.
Creation of a nomogram
Age, marital status, tumor size, AJCC stage, surgery, radiotherapy, chemotherapy, postoperative lymph node dissection, surgery and radiotherapy, and income were independent risk factors for CSD in keratinizing squamous cell carcinoma of the female reproductive system, according to multivariate analysis by competing risk model. Nomograms were created from these risk factors to forecast the probability of CSD at 3, 5, and 8 years (Figure 3). The top point of the nomogram represents each variable, and each variable point is combined together to get a total point, from which the chance of CSD at 3, 5, and 8 years is estimated.
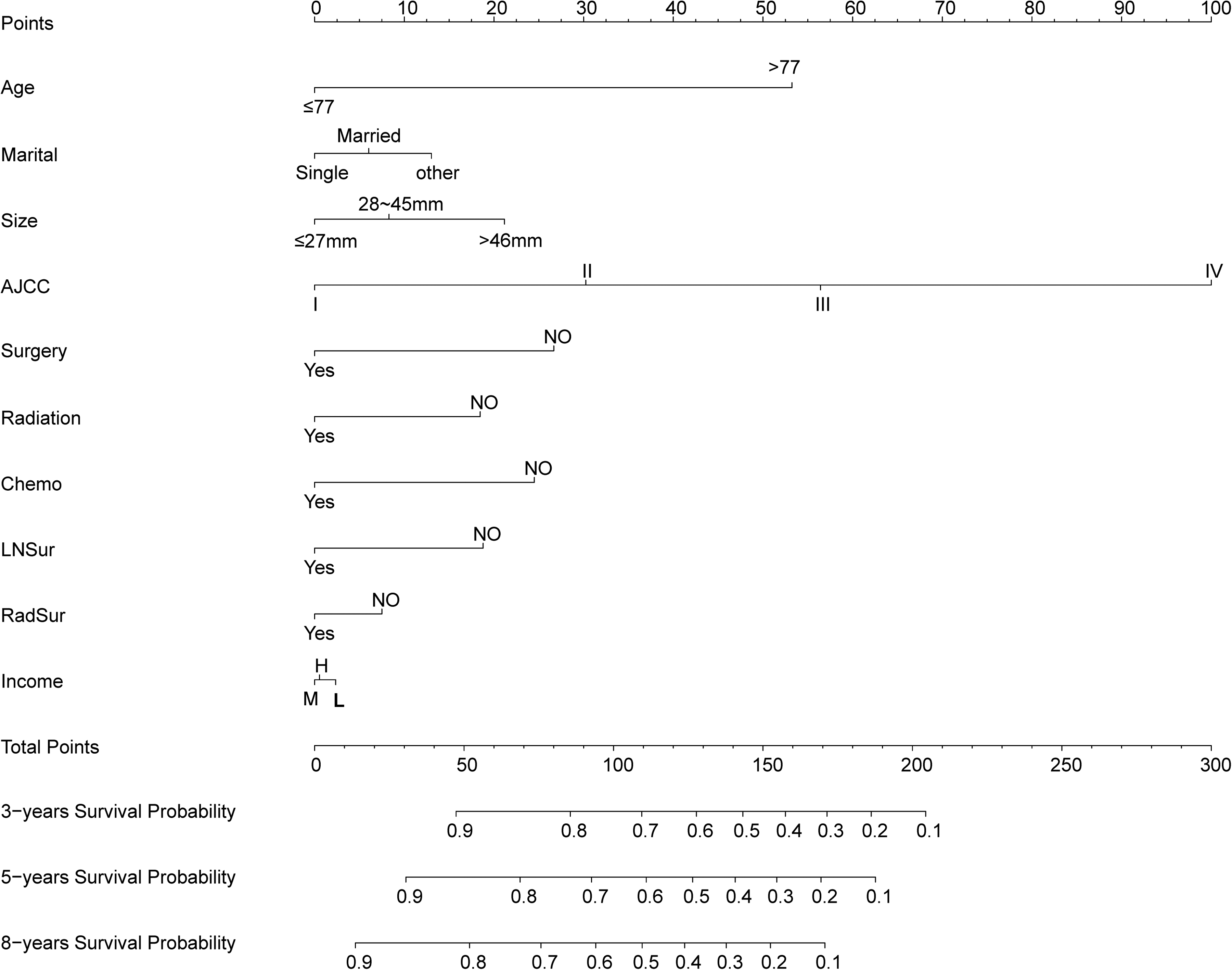
Nomogram for predicting 3-, 5-, and 8-year probabilities of keratinizing squamous cell carcinoma of female reproductive system. LNSur: Postoperative lymph node dissection, RadSur: Surgery and Radiotherapy.
Validation of nomogram
Its C-index was better than that of the AJCC staging system in both the training (0.752/0.682) and validation (0.736/0.672) sets. In the training and validation sets, the AUC values for the 3-, 5-, and 8-year CSD were 0.789 and 0.803, 0.787 and 0.787, 0.788 and 0.784, respectively; the comparable values for the AJCC staging system were 0.726 and 0.736, 0.711 and 0.699, 0.697 and 0.685, respectively. These findings indicated that the new model had better predictive power than the AJCC staging system (Figure 4). The NRI values for evaluating probability of CSD in the training set were 52.6% (95% CI = 40.72–62.23%) at 3 years, 58.2% (95% CI = 48.46–68.37%) at 5 years, and 63.4% (95% CI = 55.12–72.57%) at 8 years, and for the validation set were 50.8% (95% CI = 29.61–67.18%), 58.6% (95% CI = 42.51–70.76%), and 63.1% (95% CI = 47.75–74.49%), respectively. The training and validation sets’ respective IDI values for CSD at 3 years, 5 years, and 8 years were 10.9% and 10.8%, 11.5% and 11.2%, and 11.6% and 11.5%, respectively (p < 0.001). The NRI and IDI values were both higher than zero, indicating that the predictive power of the new model is better.
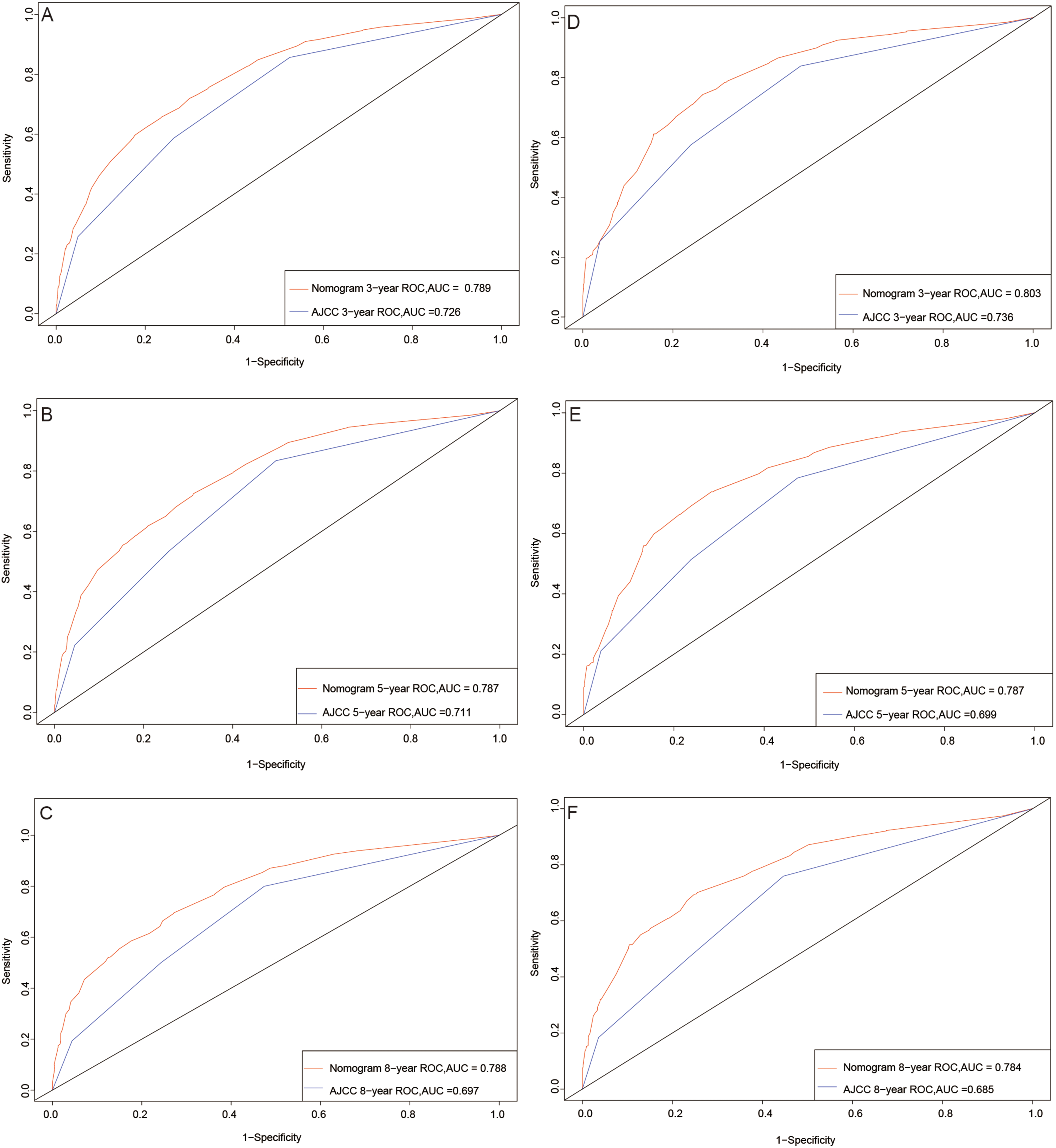
ROC curves. The area under the ROC curve (AUC) was used to evaluate the performance of the new nomogram. The training set (A, B, C) and validation set (D, E, F).
The calibration curve indicated that the overall slopes of the curves of the training and validation sets were very similar to the reference line, indicating that our model had a strong predictive ability (Figure 5). Figure 6 demonstrates that the nomogram we have constructed is successful, with the net advantage of this model in forecasting survival probability being greater than that of the AJCC system.
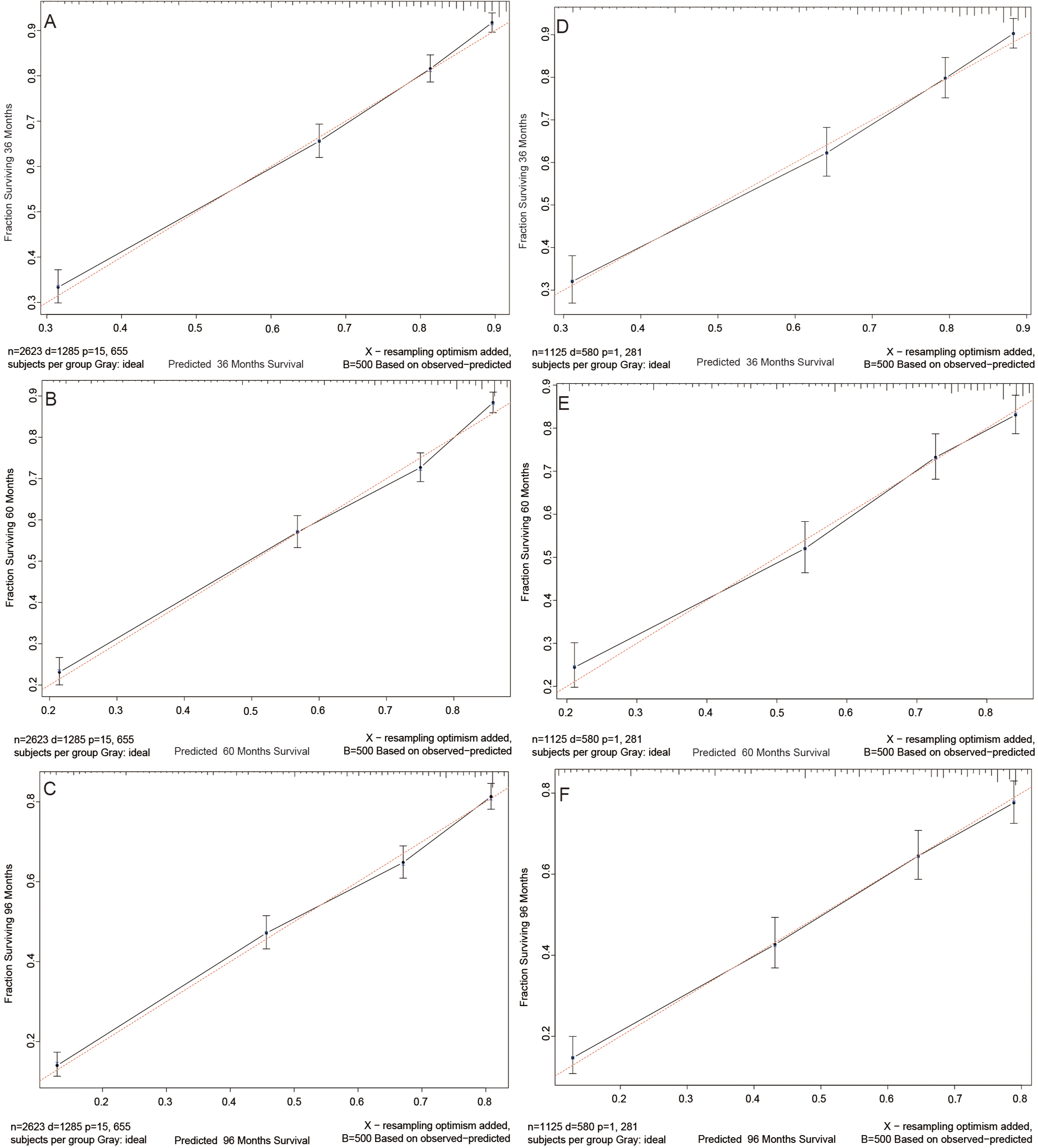
Calibration curves. 3-year, 5-year, and 8-year probabilities of cancer-specific death in the training set (A, B, C) and validation set (D, E, F).
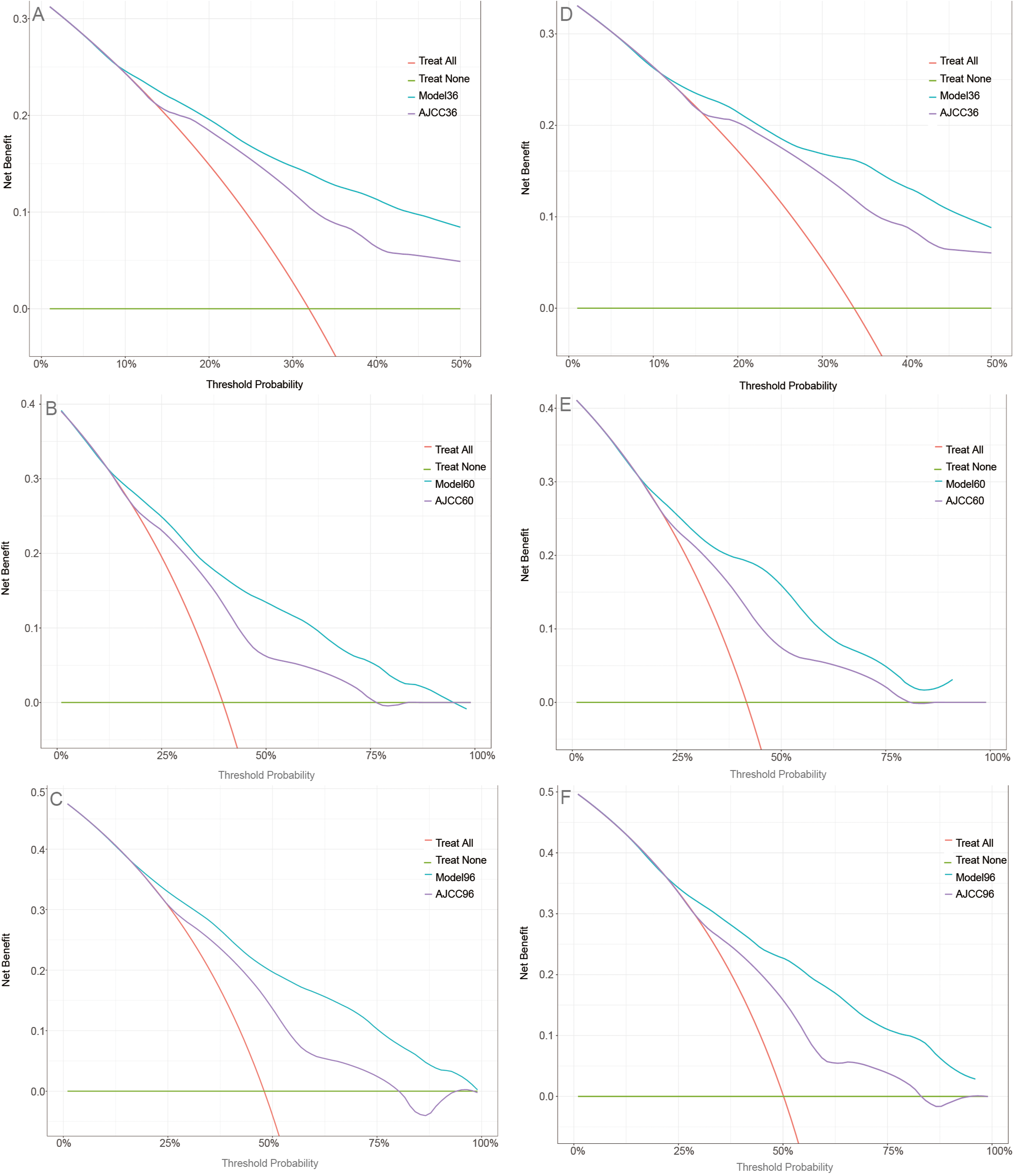
Decision curve analysis curves. Decision curve analysis of the training set (A, B, C) and validation set (D, E, F) for 3-, 5-, and 8-year Cancer-specific death probability.
Discussion
In young women, the ovary is the most common primary site for reproductive system cancers, followed by the cervix, vagina, and vulva. Squamous cell carcinoma is the most prevalent histological type, 16 and keratinizing squamous cell carcinoma is an independent predictor of survival in tumor patients. It can directly progress to invasive squamous cell carcinoma, resulting in a lower overall survival rate compared to non-keratinizing squamous cell carcinoma. 8 The epThe epidemiology, risk factors, and prognosis of keratinizing squamous cell carcinoma of the female reproductive system are not well characterised, as no systematic studies on this condition have been published.
To thoroughly examine the risk factors for keratinizing squamous cell carcinoma of the female reproductive system, this study employed a competing risk model. By accounting for both the tumor-related mortality and competing events, the model provides more reliable results. The findings of this study demonstrated that CSD in patients with keratinizing squamous cell carcinoma of the female reproductive system was independently predicted by age, marital status, tumor size, AJCC stage, surgery, radiotherapy, chemotherapy, postoperative lymph node dissection, combined surgery and radiotherapy, and income.
Both the Cox model and the competing risk model identified age as an independent risk factor. However, the Cox model indicated that patients aged >77 years had a higher risk of CSD (HR = 2.706, 95% CI = 2.348–3.119) compared to the competing risk model (HR = 1.627, 95% CI = 1.419–1.866), suggesting that the Cox model overestimated the risk associated with age. This overestimation may occur because age also contributes to competing mortality from other age-related diseases. Additionally, the Cox model estimated higher hazard ratios for AJCC staging and lower-middle income than the competing risk model due to the influence of competing risk events.
According to the Cox model, tumors in the uterus had a higher risk than tumors in other locations (HR = 0.388, 95% CI = 0.195–0.774), and distant metastasis (HR = 2.001, 95% CI = 1.395–2.871) was associated with a poorer prognosis. Black patients (HR = 0.694, 95% CI = 0.544–0.885) had a lower risk of cancer-specific death (CSD) compared to white patients. However, the competing risk model indicates that there are no significant differences in the effects of various racial groups on patient prognoses. Similarly, tumor site (ovary, uterus, vagina, vulva) and invasion status (localized, regional, distant) showed no significant impact on patient prognosis. These findings suggest that competing risk events may have caused the Cox model to overestimate the prognostic effects of race, tumor site, and summary stage.
The optimal tumor size cut-off values in this study were determined using X-tile software as 28 mm and 45 mm. Both models demonstrated that tumor size was a significant risk factor for patient prognosis. The Cox model indicated that patients with tumor sizes of 28–45 mm and ≥46 mm had significantly lower survival rates than those with ≤27 mm. However, the competing risk model suggested that only tumor sizes ≥46 mm were prognostic risk factors. Similarly, Huang et al. 17 reported a 1.62-fold increased risk of death in patients with vaginal cancer with tumor sizes >40 mm. This finding suggests that the presence of competing risk events may have caused the Cox model to overestimate the effect of tumor sizes of 28–45 mm on prognosis.
This study found that separated or divorced women (HR = 0.881, 95% CI = 0.793–0.979) with keratinizing squamous cell carcinoma of the reproductive system had a better prognosis than married women. Previous studies have suggested that being single, separated, or widowed is a risk factor for the prognosis of female reproductive system tumors compared to being married.18–23 This discrepancy may be due to bias in data selection, greater access to treatment options for women's health issues, or both. Therefore, additional external data verification is necessary.
Early-stage cancers are best treated surgically, and patients typically have a favourable survival rate. For vulvar squamous cell carcinomas less than 1 mm in size, extended surgical resection is recommended. Tumors larger than 1 mm require radical surgery and lymph node dissection, while advanced cases with lymph node or distant metastasis necessitate surgery combined with radiotherapy and chemotherapy.6,24 In this study, both models demonstrated that women with keratinizing squamous cell carcinoma of the female reproductive system who did not undergo surgery (HR = 1.399, 95% CI = 1.070–1.829), chemotherapy (HR = 1.169, 95% CI = 1.022–1.337), or postoperative lymph node dissection (HR = 1.136, 95% CI = 1.022–1.264) were at increased risk for poor outcomes. The competing risk model demonstrated that patients’ survival probabilities and prognoses could be significantly improved by radiotherapy or surgery combined with radiotherapy. According to Stecklein, radiotherapy alone improved overall survival in patients with locally advanced vulvar squamous cell carcinoma who were ineligible for surgery. 25 Orton et al. found that brachytherapy benefited patients with vaginal squamous cell carcinoma (HR = 0.80, 95% CI = 0.70–0.92), with the greatest benefit observed in tumors larger than 5 cm (HR = 0.68, 95% CI = 0.50–0.91). 26 Additionally, patients with tumors larger than 4 cm experienced notable benefits from radiotherapy and chemotherapy. 27 Moreover, advancements in treatment methods have shown that radiotherapy, chemotherapy, and combined therapies for ovarian and cervical cancers significantly improve patient survival rates.28–30 However, the Cox model underestimated the impact of radiotherapy and radiotherapy combined with surgery on patient prognosis.
After conducting a multivariate analysis of competing risks, independent risk factors influencing the prognosis of female reproductive system keratinizing squamous cell carcinoma were identified. A prediction model was subsequently developed to estimate the probability of CSD at 3, 5, and 8 years. The C-index, AUC, IDI, and NRI of our model are all superior to those of the AJCC staging system. The calibration curve demonstrates the accuracy of our model's calibration, while the DCA curve indicates that this model outperforms the AJCC evaluation method in terms of clinical validity. These findings suggest that our model has greater potential for application in clinical settings.
This study had several limitations. First, its retrospective design may have introduced selection and information biases. Second, the SEER database did not provide information on the location of positive lymph nodes or the extent and dose of chemoradiotherapy. Finally, prospective studies are required to validate the predictive power of the developed nomogram, which will be the focus of our future research.
Conclusion
For the first time, this study utilised the competing risk model to evaluate prognostic risk factors for keratinizing squamous cell carcinoma of the female reproductive system. The results can assist clinicians in making more informed clinical decisions, as the competing risk model is more precise and aligns better with actual clinical practice than the Cox model. Additionally, we developed a nomogram to predict the likelihood of cancer-specific death (CSD) at 3, 5, and 8 years. Compared to the AJCC system, our nomogram enables clinicians to more accurately estimate the likelihood of CSD.
Footnotes
Acknowledgments
We thank all SEER database staff and scientists. We are also very grateful to China Medical Education Association for their support.
Ethics approval and consent to participate
SEER database is freely available to the public and patient identities in it are anonymous, and other information is public, so no ethics committee approval is required, and informed consent is not required.
Authors’ contributions
RQC and HY designed the study; XKZ collected and analyzed the data; HY drafted the initial manuscript; QQZ revised the article critically; RQC and HY are co-first authors; LHD are the correspondence authors. All authors approved the final manuscript.
Funding
This research was funded by the Key Scientific Problems and Medical Technical Problems Research Project of the China Medical Education Association (grant number 2022KTZ009).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets generated and analysed during the current study are available in the SEER database, (![]() ). SEER is supported by the Surveillance Research Program (SRP) of the NCI Division of Cancer Control and Population Sciences (DCCPS). The aim is to inform the science of cancer surveillance and the collection, analysis, interpretation and dissemination of reliable population-based statistics. SEER releases a standard set of research data every spring based on the previous November's submission of data from the registries. The data we used is based on the November 2021 submission. We accessed these through the SEER*Stat software with additional approvals. The data that support the findings of this study are available from the SEER*Stat software but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available upon reasonable request and with the SEER Research Data Agreement.
). SEER is supported by the Surveillance Research Program (SRP) of the NCI Division of Cancer Control and Population Sciences (DCCPS). The aim is to inform the science of cancer surveillance and the collection, analysis, interpretation and dissemination of reliable population-based statistics. SEER releases a standard set of research data every spring based on the previous November's submission of data from the registries. The data we used is based on the November 2021 submission. We accessed these through the SEER*Stat software with additional approvals. The data that support the findings of this study are available from the SEER*Stat software but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available upon reasonable request and with the SEER Research Data Agreement.
