Abstract
Background:
The survival advantage of postoperative chemotherapy for high-risk stage II colon cancer (CC) patients remains unclear.
Objectives:
The purpose was to evaluate the survival of high-risk stage II CC cases treated with chemotherapy and to construct survival prediction models to predict the survival benefit from chemotherapy.
Design:
The study is a retrospective observational cohort study.
Methods:
Data on patients with stage II CC diagnosed from 2005 to 2019 who underwent radical surgery were obtained from the Surveillance, Epidemiology and End Results (SEER) database. A 1:1 propensity score matching (PSM) was applied to obtain two cohorts, chemotherapy versus no chemotherapy. A chi-square analysis was used to assess the differences before and after PSM in the above two groups. Kaplan–Meier survival analysis and Cox proportional hazards regression were applied to investigate the 5- and 10-year overall survival (OS) and cancer cause-specific survival (CSS). The predictive power of the constructed models was assessed by the concordance index (C-index) and calibration curves.
Results:
Of the 37,050 cases, 14,744 (39.8%) stage II CC were at high-risk and 29.2% of them received chemotherapy. Age, T stage, marital status, histologic grade, gender, and site independently influenced the reception of chemotherapy. The survival advantage of chemotherapy in the high-risk patients remained positive before and after PSM. The estimated 3, 5, and 10 years OS rates of chemotherapy group were 9.3, 10.7, and 15.6% higher than the nonchemotherapy group, respectively. Four nomograms predicting OS and CSS were established, with great discrimination (C-index between 0.627 and 0.691) and excellent calibration.
Conclusion:
Postoperative chemotherapy is beneficial for high-risk stage II CC patients, including the elderly patients (over 65 years of age). Our study developed nomograms to quantify the survival benefit of chemotherapy among high-risk stage II CC patients to develop personalized treatment recommendations and guide management decisions.
Introduction
Colon cancer (CC) remains a major health burden worldwide, with the third largest incidence among cancer types and increasing fatality rates each year, with an estimated 53,200 deaths in the United States in 2021. 1 Approximately 30–40% of CC patients are at stage II at diagnosis and are recommended to undergo radical surgery.2,3 There is a consensus that chemotherapy, a common treatment choice for CC, can prolong the survival of stage III CC patients after radical surgery and reduce the likelihood of tumor recurrence.4,5 The American Society of Clinical Oncology and the European Society of Medical Oncology guidelines also recommend postoperative chemotherapy for cases with stage II CC who are at high risk of recurrence.6,7 However, there are currently no clear criteria for defining patients at high risk. The current uniform view is that patients with stage II CC are considered to be at high risk of recurrence if they have at least one of the following clinical features: lymph node sampling <12; poor tissue differentiation (grade III or IV); vascular, lymphatic, or perineural invasion; tumor manifesting as obstruction or tumor perforation; and pT4 staging. All of these are usually considered to be features predictive of a poor outcome of stage II CC.6–8
An article published in 2011 studied CC patients from the Surveillance, Epidemiology and End Results (SEER) database diagnosed between 1992 and 2005 and concluded that chemotherapy had little impact on survival of stage II CC, for patients with and without unfavorable prognostic factors. This conclusion was also supported by two studies, both of which showed that chemotherapy did not improve survival.9–11 In contrast, an article that included patients recorded in the SEER database from 2005 to 2015 concluded that chemotherapy for stage II CC was effective, and two other large-scale recent studies11–13 concluded that all patients with stage II CC, both high and low risk, had significantly improved overall survival (OS) if given chemotherapy.
Chemotherapy’s efficacy in stage II CC remains debatable, especially for patients at high risk. If there is a benefit, whether the benefit of chemotherapy can be predicted by individualization to bring more accurate treatment to patients. There is also a certain value in evaluating the factors that affect the reception of chemotherapy for high-risk stage II cases and exploring other variables that influence survival of the above population which can also help us to further identify potential beneficiaries. It is known that the above questions need to be evaluated in large retrospective studies.
In this context, the present study examined a cohort of high-risk stage II CC cases recorded in the SEER database between 2005 and 2019, who underwent radical surgery. The aim of this paper is to investigate the impact of chemotherapy on survival and the independent variables affecting survival.
Methods
Study population
This article is a retrospective study. Data were extracted for patients with primary American Joint Committee on Cancer (AJCC) stage II colon adenocarcinoma diagnosed between 2005 and 2019 from a total of 18 cancer registries in the National Cancer Institute SEER Cancer Database. We submitted the data consent form to SEER management first and then collected the necessary data with SEER*Stat version 8.3.6. This study is based on publicly available data, and permission to access the data was obtained solely for research purposes. Data were obtained from the SEER database (approval number: 15617-Nov2017), a population-based cancer registry in the National Cancer Institute in the United States (https://seer.cancer.gov/). This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013) and the ethical approval for this study was exempted by the Medical Ethics Committee of the Shanghai East Hospital, College of Medicine, Tongji University (Shanghai, China), as SEER is a publicly available database, and the data extracted from SEER were identified as belonging to a non-human study. As a retrospective study, informed consent was not required from patients. The reporting of this study conforms to the STROBE statement. 14
Patient selection
All stage II CC patients who underwent primary tumor radical surgery after diagnosis between 2005 and 2019 were chosen. We screened patients using the following inclusion criteria, in the order shown in Figure 1: (1) no other prediagnostic CC tumors, (2) positive pathological diagnosis and histology type was adenocarcinoma, (3) age >18 years, and (4) complete radical surgery; and the exclusion criteria: (1) unidentified race, (2) undetermined pathological grade, (3) undetermined survival time, (4) undetermined marital status, and (5) unknown carcinoembryonic antigen (CEA) information.

The flow diagram of the selection process for the study.
Variables
The major objective variable for our research was OS, defined as the time from diagnosis to the date of death due to any cause. The time from diagnosis to death due to CC was defined as the secondary outcome variable, cancer cause-specific survival (CSS). Cancer cause-specific death (CSD) was defined as an event in which a patient’s cause of death was recorded as ‘death from CC’. The non-colon cancer cause-specific death (NCSD) is defined as an event in which a patient’s cause of death is recorded as ‘death from other causes’. The failure event was deemed to be a death attributed to CC. The competing event was patients who died of other causes, and vice versa. The poor prognostic characteristics identified from SEER data were T4 stage, poor/undifferentiated histology, and fewer than 12 lymph nodes examined. Cases who had at least one of the features were defined to be in the high-risk stage II CC. Age was divided into three categories: 18–49 years, 50–65 years, and >65 years. The SEER database coded data on the pretreatment serum CEA level as ‘Positive/elevated’, ‘Negative/normal; within normal limits’ and ‘Borderline; undetermined if positive or negative’, which were assigned as ‘negative’, ‘positive’ and ‘borderline’ separately in the article. The categories of histologic grade we obtained from the SEER database were ‘well differentiated; Grade I’, ‘moderately differentiated; Grade II’, ‘poorly differentiated; Grade III’ and ‘undifferentiated; anaplastic; Grade IV’ which were assigned as ‘grade I’, ‘grade II’, ‘grade III’ and ‘grade IV’ in the article. Marital status was classified as married or others (single, widowed, divorced, and separated). Tumor sites were classified as either the right-sided colon (including the cecum, ascending colon, hepatic flexure, and transverse colon) or the left-sided colon (including the splenic flexure, descending colon, and sigmoid colon). The number of lymph nodes sampled was classified as 0–11, 12, and chemotherapy was labeled ‘Yes’ or ‘No/Unknown’.
Statistical analysis
The baseline patients’ characteristics distributions by chemotherapy status were depicted by statistical description. The association between chemotherapy status and patient characteristics was assessed through the chi-square test and multivariable logistic regression. The differences in the distribution of the chemotherapy subgroups were assessed through the chi-squared test. A 1:1 propensity score matching (PSM) technique was used to address the selection bias due to nonrandomized treatment assignment, and the caliper of the nearest-neighbor method was set to 0.0001. It could eliminate a large number of the distributional differences and produce more similar treated and untreated groups, as would be expected to occur in a randomized trial. 15 The outcome variables of the PSM analysis were chemotherapy, and the independent variables including age, T stage, marital status, grade, gender, site, CEA, regional nodes examined, and race. The chemotherapy administration status was used to classify all patients and elderly group separately. Kaplan-Meier survival analysis technique was applied to compare the differences in OS between the subgroups and to describe the survival status within chemotherapy and nonchemotherapy group. For the survival analysis and prognosis evaluation, the cases were divided into two groups by random sampling and a 7:3 population was obtained, which were named training and validation groups separately. In the training cohort, grouping by chemotherapy status, the Kaplan-Meier method was used to draw the survival curve of each clinicopathological factor, and then the log-rank test was used for comparison. Univariate and multivariable Cox proportional hazard regressions were performed to identify the independent prognostic factors associated with OS and CSS, and hazard ratios (HRs) and 95% confidence intervals (CIs) for OS and CSS were presented. Clinicopathological variables with p < 0.05 in univariate analysis were chosen for multivariable analysis. The nomogram was built based on the results of multivariate analysis to predict the 5- and 10-years survival rates. The internal validity of the nomogram was measured by the concordance index (C-index), which was assessed by comparing the nomogram-predicted probability. The nomogram was further validated by comparing the nomogram-predicted probability of the patients in the validation cohort with their actual survival and the calibration plots were generated. Besides, cumulative incidence was calculated by a competing risk model and a cumulative incidence plot was constructed to describe the actual prognosis of different causes of death. The nomograms were created in R (version 3.6.2) and internally validated using 1000 bootstrap resamples. SPSS version 25 (IBM Corporation, Armonk, NY, USA) was used for all statistical analyses, and p values of 0.05 were considered statistically significant.
Results
Receipt of adjuvant chemotherapy
As depicted in Figure 1, we identified 99,180 stage II colon carcinoma patients diagnosed between 2005 and 2019 who underwent radical surgery. A total of 37,050 patients with stage II CC were included in this study, of whom 14,744 cases (39.8%) had high risk factors, including stage T4 tumor in 5339 cases (36.2%), poor differentiation in 6240 cases (42.3%), and lymph node harvested number less than 12 in 5979 cases (40.6%). Among all high-risk stage II CC patients (N = 14,744), 29.2% of them received adjuvant chemotherapy.
Characteristics of high-risk stage II CC patients
Table 1 demonstrates the demographic and clinical characteristics of high-risk stage II colon carcinoma patients. Among the 14,744 patients, the median age was 69 years, they were predominantly grade II (52.6%) and grade III (36.6%), predominantly Caucasian (81.6%), and the ratio of tumors originating from the right-sided colon to the left-sided colon was approximately 1.51:1. For the group younger than 50 years, it can be seen that the ratio of the population receiving chemotherapy versus those not receiving chemotherapy was close to 1.64:1, while for the group at the age between 50 and 65, the ratio of the population receiving chemotherapy versus those not receiving chemotherapy was close to 1:1. However, only 20.9% of older patients (>65 years) with any unfavorable prognostic features received chemotherapy.
Demographic information of chemotherapy subgroups before and after PSM.

CEA, carcinoembryonic antigen; PSM, propensity score matching.
Factors influencing the reception of chemotherapy
As illustrated in Table 2, multivariable logistic regression analyses indicated that age (p < 0.001), T stage (p < 0.001), grade (p = 0.030), marital status (p < 0.001), gender (p = 0.046), and site (p < 0.001) were independent factors that affected the outcomes of high-risk stage II CC cases receiving chemotherapy.
Univariate and multivariable analysis for the factors influencing the reception of chemotherapy.

CEA, carcinoembryonic antigen.
Survival advantage of chemotherapy
Applying Kaplan-Meier survival analysis to compare the difference in OS before and after PSM of high-risk stage II CC patients with and without chemotherapy (Figure 2) and statistically described (Table 3), it is obvious that chemotherapy improved the survival of high-risk stage II patients. Before PSM, patients with high-risk stage II CC who underwent chemotherapy had a higher chance of surviving than those who did not, with survival rates of 85.8, 76.7, and 60.7% at 3, 5, and 10 years, respectively. In the elderly group (>65 years), the above statement still holds, with OS rates of 82.7, 71.7, and 52.2%. After PSM, we were able to determine the exact impact of chemotherapy on survival. The estimated 3, 5, and 10 years OS rates of chemotherapy group were 9.3, 10.7, and 15.6% higher than nonchemotherapy group, respectively. And in the elderly group, these values were 12.0, 12.8, and 15.9%, respectively. The survival curves stratified by the variables were generated among the training cohort using Kaplan-Meier survival analysis (Figure 3) and compared using the log-rank test (Table 4). Age (p < 0.001), race (p < 0.001), pathological grade of tumor (p < 0.001), T-stage (p < 0.001), marital status (p < 0.001), CEA level (p < 0.001), and regional nodes examined (p = 0.001) were variables that affected the survival of high-risk stage II CC patients.

Overall survival grouped by chemotherapy administration status and age for high-risk patients (Kaplan-Meier). (a) Before PSM. (b) After PSM.
Overall survival rates by chemotherapy group.
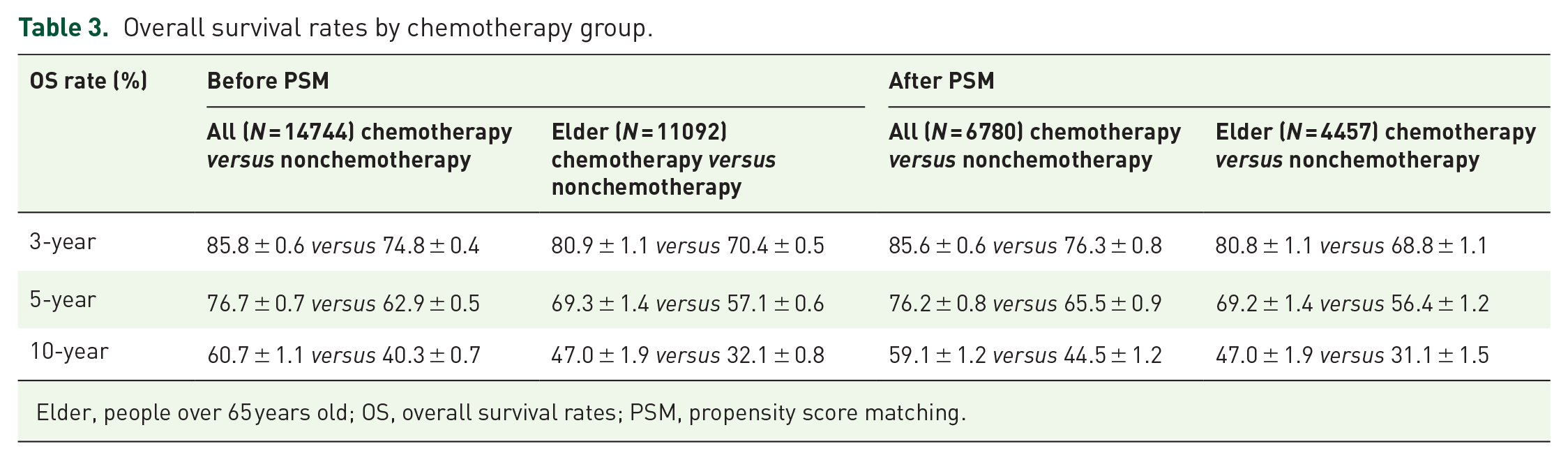
Elder, people over 65 years old; OS, overall survival rates; PSM, propensity score matching.

Overall Kaplan-Meier survival curves for high-risk stage II CC patients after PSM according to (a) age, (b) race, (c) gender, (d) site, (e) grade, (f) T stage, (g) marital status, (h) CEA, (i) regional nodes examined, and (j) chemotherapy.
5- and 10-year medium survival rates and univariate analysis of patients with stage II CC.

CEA, carcinoembryonic antigen; OS, overall survival rate.
Cumulative incidence of death and competing risk analysis
In addition to survival, we also focused on mortality. After PSM, among the two groups with or without chemotherapy, there were 1375 deaths in the population without chemotherapy, with an overall mortality rate of 40.6%, and the CSD rates at 3, 5, and 10 years were 14.7, 21.0, and 30.0%; in the population with chemotherapy, there were 913 deaths, the 3-, 5-, and 10-year CSD rates were 11.4, 18.2, and 26.5%. Among high-risk stage II CC patients, all-cause death, CSD, and NCSD were lower in those who received chemotherapy relative to those who did not, and the difference in NCSD between the chemotherapy and nonchemotherapy groups gradually increased with longer follow-up (Figure 4).
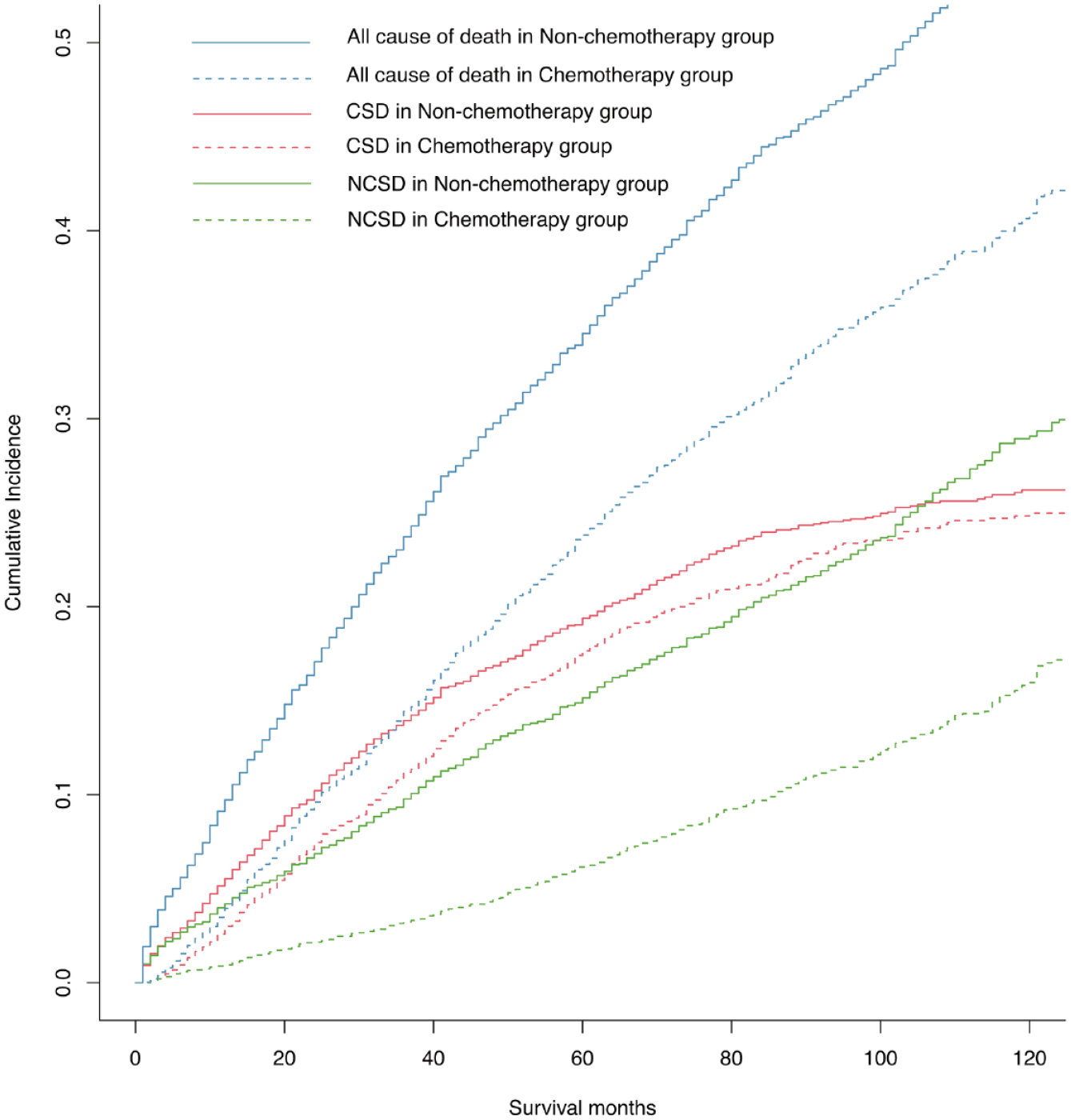
Competing risk analyses for high-risk stage II CC patients: CSD, NCSD, and all cause of death of receiving or not receiving chemotherapy subgroups.
Univariate and multivariable analysis for CSS and OS
Univariate and multivariable Cox regressions were performed to identify independent features affecting the prognosis of high-risk stage II CC patients (Table 5). All variables categorized as statistically significant in the univariate analysis were included in the multivariable analysis and used to construct nomograms to predict OS and CSS rates at 5 and 10 years. Multivariable analyses showed that age (p < 0.001), race (p = 0.007), T stage (p < 0.001), chemotherapy (p < 0.001), marital status (p < 0.001), CEA (p < 0.001), and regional nodes examined (p < 0.001) were correlated with OS, and the features above were also correlated with CSS.
Multivariable Cox analysis for OS and CSS among PSM population.

CEA, carcinoembryonic antigen; CSS, cancer cause-specific survival; OS, overall survival rate; PSM, propensity score matching.
Construction and validation of the nomogram
As shown in Figures 5 and 6, clinical prediction models for chemotherapy and nonchemotherapy were constructed to predict OS and CSS at 5 and 10 years. The C-index for predicting OS was 0.652 and 0.691 for chemotherapy and nonchemotherapy, respectively, and 0.627 and 0.631 for chemotherapy and nonchemotherapy for CSS, respectively. The calibration plots showed excellent correlations between the predicted survival probabilities and the observed outcomes (Figure 7).

Nomograms for comparing expected 5- and 10-year OS of high-risk stage II CC: (a) Chemotherapy-users. (b) Chemotherapy non-users.

Nomograms for comparing expected 5-, and 10-year CSS of high-risk stage II CC. (a) chemotherapy-users. (b) Chemotherapy non-users.

Calibration curve of the nomograms in training and validation group. (a) 5- and 10-year OS nomogram calibration curves of the chemotherapy group. (b) 5- and 10-year OS nomogram calibration curves of the nonchemotherapy group. (c) 5- and 10-year CSS nomogram calibration curves of the chemotherapy group. (d) 5- and 10-year CSS nomogram calibration curves of the nonchemotherapy group.
Discussion
The primary goal of the paper was to determine the extent of the survival benefit of adjuvant chemotherapy in high-risk stage II CC patients who underwent radical CC surgery and to effectively predict the OS and CSS of ‘chemotherapy users’ or ‘chemotherapy non-users’ by nomograms. The nomograms can be used to identify the population that will benefit from chemotherapy and it will guide clinicians and patients in selecting treatment regimens. In the present day, CC remains the second most common cause of cancer death in the United States. This is the first study to establish a predictive model to predict survival after radical surgery in high-risk stage II CC patients. We achieved comparability between groups through PSM to identify more scientific prognostic factors. Additionally, individual survival probabilities were predicted by examining both OS and CSS rates, thereby, the influence of unmeasured confounding factors relating to patient health condition is avoided.
Previous studies from various institutions have evaluated the survival of cases with stage II CC after chemotherapy. Some of them found that chemotherapy has a survival benefit.11,13 Others found that chemotherapy does not significantly improve survival.9,16 However, the more widely accepted view, as reflected in the clinical practice guidelines, is that chemotherapy clearly improves survival in high-risk stage II CC patients. Meanwhile, chemotherapy is not recommended for patients who have no unfavorable prognostic features.6,17 Therefore, we specifically addressed the high-risk population, analyzing 14,744 patients from SEER database. Both before and after PSM, we clearly observed that patients with one or more unfavorable prognostic factors clearly benefited from chemotherapy. After the PSM, which have frequently been used to eliminate selection bias and uneven distribution of factors, the estimated 3, 5, and 10 years OS rates of chemotherapy group were 9.3, 10.7, and 15.6% higher than nonchemotherapy group, with chemotherapy benefiting patients even more as time increased. In stage II CC, delayed initiation of chemotherapy and incomplete chemotherapy course are associated with higher cancer-specific mortality according to Bradley et al. 18 . Our study showed that among high-risk stage II CC patients, cancer-specific mortality was consistently lower in the chemotherapy population than in the nonchemotherapy population, as was non-cancer-specific mortality.
The benefit of chemotherapy was also evident in the elderly population, which is inconsistent with a previous study that analyzed older (age ⩾66 years) stage II CC patients with unfavorable prognosis from the SEER-Medicare database between 1992 and 2005. 9 O’Connor et al. concluded that any possible survival benefit from chemotherapy in older adults at high risk is likely to be less than 2% over 5 years, whereas the current study selected cases for a more recent decade (2005–2019). The results of the study suggest that chemotherapy benefited the elderly population. After the PSM, which have frequently been used to eliminate selection bias and uneven distribution of factors, the estimated 3, 5, and 10 years OS rates of elderly chemotherapy group were 12.0, 12.8, and 15.9% higher than elderly nonchemotherapy group. Clinicians tend to be more conservative in their treatment strategy because of advanced age, and elderly patients tend to prefer not to undergo chemotherapy because of their own preferences, a concern less common in younger CC patients. Our results provide more confidence in choosing chemotherapy for both young and elderly patients and provide some reference value for clinicians when choosing appropriate treatment options.
In terms of the reception of chemotherapy, of the 14,744 patients, approximately 29.2% underwent chemotherapy. For the group younger than 50 years, the ratio of the population receiving chemotherapy versus those not receiving chemotherapy was close to 1.64:1 and for the group at the age between 50 and 65, the ratio was close to 1:1. But for older patients (>65 years), only 16.7% of patients were treated with chemotherapy, clearly a more conservative reception of treatment among older patients. Also found in other studies, older patients receiving chemotherapy were less frequent in patients with stage III CC. 17 It can be seen that although the guidelines recommend that high-risk stage II CC patients receive chemotherapy,6–8 in actual clinical practice, the treatment decision is influenced by many other factors. In addition to age, the other factors include gender, tumor primary site, T stage, histologic grade, and marital status. There is some evidence that the reception of chemotherapy is physician-dependent. Hershman et al. 19 suggest that older women with breast cancer treated by younger private oncologists received more chemotherapy.
Unlike the previous study, 9 this paper added an analysis of CEA levels, a variable widely recognized for its impact on survival.20,21 For those who chose chemotherapy, the CEA level was an independent variable influencing whether patients benefited from chemotherapy, while the CEA level also independently affected OS and CSS in the high-risk stage II CC population. For patients with CEA-positive values, the HRs of OS for CEA-negative and CEA-borderline were 0.66 (0.60–0.74) and 1.09 (0.81–1.47), and the HRs of CSS were 0.51 (0.45–0.59) and 0.69 (0.43–1.09), respectively. This corroborates the detrimental effect of elevated CEA on the survival of CC patients, as found in other studies. 22
Although the median age of diagnosis for CC is 69 years, 23 many older adults are excluded from randomized chemotherapy trials due to age-related comorbidities, provider bias, or patient personal preference.24,25 The age distribution of the study population in the current study is more realistic, the median age of the 14,744 high-risk cases was 69 years, suggesting that the results obtained here can be extrapolated with some confidence to the older population.
However, the above results should be considered with caution due to several limitations of this study. First, regarding the definition of patient staging, the 7th edition of AJCC staging proposed in 2010 introduced the patient Group N1C, that is, patients with TXN1CM0 actually belong to stage III. Since the patient staging in this paper follows the 6th and 7th edition of AJCC staging, the cases in this article include a portion of patients who belong to stage III according to the 7th edition of staging. It is now widely accepted that patients with stage III CC benefit from chemotherapy.26,27 Therefore, the conclusion obtained in this paper that chemotherapy provides a survival benefit may be somewhat influenced by this factor. Second, regarding the definition of outcomes, the chemotherapy information in the SEER database inevitably causes confounding bias (sensitivity 72.1% according to one study) 28 ; for example, the chemotherapy records in the SEER database are classified as ‘No/Unknown’ and ‘Yes’. Although the group of patients with ‘Yes’ clearly received chemotherapy, it is not clear whether all patients with ‘No/Unknown’ refused chemotherapy. However, even so, if the group defined as ‘no chemotherapy’ in this paper is mixed with a portion of the chemotherapy population, our conclusion that chemotherapy is beneficial for survival still holds. Due to a lack of information, we could not specify the chemotherapy regimens and the completion of the chemotherapy. There is no information about patients’ comorbidity in the SEER data, which may influence chemotherapy treatment and survival in cancer patients. Another limitation of this study is the limited information on the actual numerical values of CEA levels and limited interpretation of elevated values, normal values, and a uniform cutoff value, although we believe that most US laboratories use similar and standardized reference values. In addition to the CEA included in this analysis, some molecular biomarkers that may affect the prognosis of patients with stage II CC were not included in this study,6,29–32 such as microsatellite instability and BRAF mutations, which may have led to a degree of bias.
Conclusion
We found that high-risk stage II CC patients can benefit from postoperative chemotherapy. Our study is applicable to patients of all ages, including elderly patients (over 65 years of age). Two clinical prediction models were developed to quantify the benefit of post radical surgery chemotherapy in high-risk stage II CC patients to guide personalized treatment recommendations and patient management.
Supplemental Material
sj-docx-1-tag-10.1177_17562848221137758 – Supplemental material for Adjuvant chemotherapy improves survival in high-risk stage II colon cancer: a retrospective cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848221137758 for Adjuvant chemotherapy improves survival in high-risk stage II colon cancer: a retrospective cohort study by Lin-Lin Liu and Zuo-Lin Xiang in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors acknowledge the great efforts of the Surveillance, Epidemiology, and End Results (SEER) Program in the creation of the SEER database. The interpretation and reporting of these data are the sole responsibility of the authors.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
