Abstract
Background
Heat shock factor 1 (
Objective
This study examined the expression and role of
Methods
Immunohistochemistry was performed to explore HSF1 level in 135 endometrial tissue specimens. The relationship between HSF1 level and EC patients’ clinicopathological characteristics was analyzed. Quantitative reverse-transcriptase polymerase chain reaction (qRT-PCR) and Western blotting were employed to explore HSF1 expression level in tissues
Results
EC tissues exhibited higher HSF1 expression level compared with normal endometrial and atypical endometrial hyperplasia tissues. High HSF1 expression level was associated with histological grade, muscular invasion, lymph node metastasis, and estrogen receptor (ER) expression level in EC tissues and cells. Kaplan–Meier analysis indicated that EC patients with elevated
Conclusion
The results demonstrated that
Introduction
Endometrial cancer (EC) is the most common gynecological cancer in several countries, and its incidence is rising globally. 1 Patients with endometrial carcinoma mainly face poor prognosis and high mortality due to the invasion and metastasis occurring in the early stage. Despite significant advances in surgery and chemotherapy over the recent decades, therapeutic failure and disease progression are still quite frequent.2,3 Therefore, there is an urgent requirement for proposing new biomarkers for EC, so as to develop more efficacious diagnostic and therapeutic strategies.
Heat shock factors (
The present study examined
Materials and methods
Patients and specimens
This study was approved by the Ethics Committee of the Fourth Military Medical University (Xi'an, China). For immunohistochemical analysis, 135 patients with endometrial lesions who underwent diagnostic curettage or surgical treatment at Xijing Hospital (Xi’an, China) between January 2011 and January 2013 were included in this study. There were 17 cases of normal endometrial tissue, 28 cases of atypical endometrial hyperplasia, and 90 cases of EC tissue. Patients’ ages ranged from 30 to 77 years, with a mean age of 54.6 (54.6 ± 9.3) years. Clinical data were obtained from electronic medical databases, and tumors were staged according to the International Federation of Gynecology and Obstetrics (FIGO) guidelines. None of the patients received chemotherapy or adjuvant treatments preoperatively. The study's protocol was approved by the Ethics Committees of Xijing Hospital, and informed consent was obtained from each patient before sample collection.
Immunohistochemistry
The 4-μm thick sections were fixed in 10% formalin, followed by embedded into paraffin. The slices were deparaffinized with xylene and dehydrated with graded alcohols. Endogenous peroxidase activity was quenched by 0.3% hydrogen peroxide, and antigen retrieval was performed using 0.01 mol/L citric acid buffer. Nonspecific binding was blocked using 10% normal goat serum at 37 °C for 30 min. The primary antibodies for HSF1 (1:250, ab52757; Abcam, Cambridge, UK) and ER (1:300, sc-8002; Santa Cruz Biotechnology Inc., Dallas, TX, USA) were incubated overnight at 4 °C, followed by incubation with anti-rabbit/mouse IgG-horseradish peroxidase (1:500; Zhongshan, China) at 37 °C for 30 min. Then, the sections were incubated with standard DAB reagents and lightly counterstained with hematoxylin. After serial dehydration, the slides were mounted for microscopic examination. Finally, the results were evaluated microscopically by two independent pathologists; in cases of conflicting opinions, a third independent pathologist was consulted for resolution. The intensity of HSF1/ER staining was scored as follows: 0 (no signal), 1 (weak), 2 (moderate), and 3 (strong). The percentage of positive cells was scored as 1 point for 1–25%, 2 points for 26–50%, 3 points for 51–75%, and 4 points for 76–100%. The intensity and percentage scores were multiplied to yield a final score ranging from 0 to 12 points. Tumors were classified as negative (−) for a score of 0 points, low expression (+) for scores ≤4 points, moderate expression (++) for scores 5–8 points, and high expression (+++) for scores ≥9 points. 12 In the present study, all the samples were grouped into the low-expression group (overall score, 0–4 points), the moderate-expression group (overall score, 5–8 points), and the high-expression group (overall score, 9–12 points).
Cell lines and cell culture
Human EC cell lines (HEC-1B, Ishikawa, and RL95-2) and human endometrial epithelial cells were purchased from the Cell Bank of Type Culture Collection of the Chinese Academy of Sciences (Shanghai, China). HEC-1B cell line was cultured in a Dulbecco's modified Eagle's medium (DMEM)/high-glucose (HyClone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin. Ishikawa and human endometrial epithelial cells were cultured in a DMEM (GIBCO, Waltham, MA, USA) supplemented with 10% FBS and 1% penicillin-streptomycin. RL95-2 cells were cultured in a DMEM/F-12 (GIBCO) supplemented with 10% FBS, 10 µg/ml insulin (Sigma-Aldrich, StLouis, MO, USA), and 1% penicillin-streptomycin (Invitrogen, Carlsbad, CA, USA). All cells were cultured in a humidified incubator at 37 °C with 5% CO2 atmosphere.
RNA interference
For the knockdown of
Quantitative reverse-transcriptase polymerase chain reaction (qrt-PCR)
TRIzol reagent (Invitrogen) was used to extract total RNA from different cells. The qRT-PCR was conducted using 2×SYBR-Green PCR Master Mix (Applied Biosystems, Waltham, MA, USA) and Premix Ex Taq reagent (TaKaRa, Shiga, Japan) on an Mx4000 Multiplex QPCR System (Stratagene, La Jolla, CA, USA). The cycling program was initiated at 95 °C for 3 min and 40 cycles of 94 °C for 30 s, 59 °C for 30 s, and 72 °C for 1 min, and a final step of 10 min at 72 °C. The results were normalized as the ratio of absolute mRNA copy numbers of
Western blot analysis
Cells were lysed in precooled radioimmunoprecipitation assay buffer (RIPA), containing protease inhibitor cocktail. Cell protein concentrations were determined by a BCA protein assay kit (Pierce, New York, NY, USA). Equal amounts of protein lysate were separated using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), followed by transferring onto polyvinylidene difluoride (PVDF) membranes (Millipore, Burlington, MA, USA). Subsequently, the membranes were blocked with 1% bovine serum albumin (BSA) for 2 h at room temperature and incubated with HSF1 (1:5000, ab52757, Abcam), ER (1:1000, sc-8002, Santa Cruz Biotechnology), HSP90 (1:1000, sc-69703, Santa Cruz Biotechnology), MMP2 (1:1000, sc-13595, Santa Cruz Biotechnology), MMP9 (1:1000, sc-393859, Santa Cruz Biotechnology), E-cadherin (1:1000, sc-8426, Santa Cruz Biotechnology), β-catenin (1:1000, sc-7963, Santa Cruz Biotechnology), and β-actin (1:5000, ab8227, Abcam) at 4°C overnight, followed by incubation with peroxidase-conjugated secondary antibody (Santa Cruz Biotechnology) at room temperature for 1 h. Protein expression levels were quantified using Quantity One software to measure the intensity of the protein bands.
Wound healing assay
Cells were cultured in 24-well plates at a density of 2 × 104 cells per well, and a small wound area was created using a sterilized pipette tip to generate a linear scratch. Then, the cells were twice rinsed with phosphate-buffered saline (PBS) and cultured in a fresh culture medium. The wound closure was monitored under a microscope at 0 and 48 h after the wounding. Photos were taken under a magnification of ×40 (Olympus, Tokyo, Japan), and the degree of cell migration was quantified by pixel through ImageJ software.
Cell migration and invasion assays
Migration and invasive abilities were evaluated by 24-well chamber with 8-μm pore size polycarbonate membrane filter (Corning Costar Corp., Dallas, TX, USA). Invasion chamber was identical to migration chamber, while the filter was pre-coated with 200 mg/ml Matrigel (BD Biosciences, San Jose, CA, USA). The upper chambers were filled with the serum-free DMEM, while the lower chambers were filled with standard DMEM containing 10% FBS as chemoattractant. About 3 × 105 cells were added to the upper chamber and allowed to migrate for 48 h. Afterward, the non-migration cells on the upper surface of the filters were removed gently by a cotton-tip swab. Cells on the lower surface of the chamber were fixed in methanol, stained with 0.1% crystal violet, and five random fields were counted and photographed.
Flow cytometric analysis of cell cycle progression and cellular proliferation
The Ishikawa cells were transfected with specific siRNAs (siNC, si
Flow cytometric analysis of apoptosis
The Ishikawa cells were transfected with specific siRNAs (siNC, si
Statistical analysis
SPSS 20.0 software (IBM, Armonk, NY, USA) was used to statistically analyze the data. Chi-square test or the Fisher's exact test, as appropriate, was used to evaluate the correlations between immunohistochemical expression and the clinical variables. Student's t-test and one-way analysis of variance (ANOVA) were employed to compare differences in Western blot analysis, wound healing assay, migration, and invasion. Survival curves were generated using the Kaplan-Meier method, and survival analysis was performed through the log-rank test. Two-sided
Results
HSF1 was upregulated in Ec tissues and it was correlated with the clinical parameters
HSF1 expression level in EC (n = 90), atypical endometrial hyperplasia (n = 28), and normal proliferative endometrial (n = 17) tissues was analyzed. It was found that HSF1 expression level was upregulated in cancer tissues compared with the matched normal endometrial tissues and atypical endometrial hyperplasia, and the positive staining was predominantly observed in the nucleus (Figure 1A and B). The correlation of HSF1 expression level with different endometrial tissues was presented in Table 1. The staining level was substantially higher in the EC group than that in other groups (Figure 1C). In the normal endometrium group, 58.8% (10/17) exhibited low HSF1 expression level, 29.4% (5/17) had moderate HSF1 expression level, and 11.8% (2/17) had high HSF1 expression level. Of the atypical endometrial hyperplasia group, 35.7% (10/28) exhibited low HSF1 expression level, 35.7% (10/28) had moderate HSF1 expression level, and 28.6% (8/28) had high HSF1 expression level. Of the EC group, 22.8% (20/90) exhibited low HSF1 expression level, 29.7% (27/90) had moderate HSF1 expression level, and 47.5% (43/90) had high HSF1 expression level. Significant associations were found among the normal endometrium group, atypical endometrial hyperplasia group, and EC group. The correlation of HSF1 expression level with EC patients’ clinicopathological features was presented in Table 2. HSF1 expression level was positively correlated with histological grade (
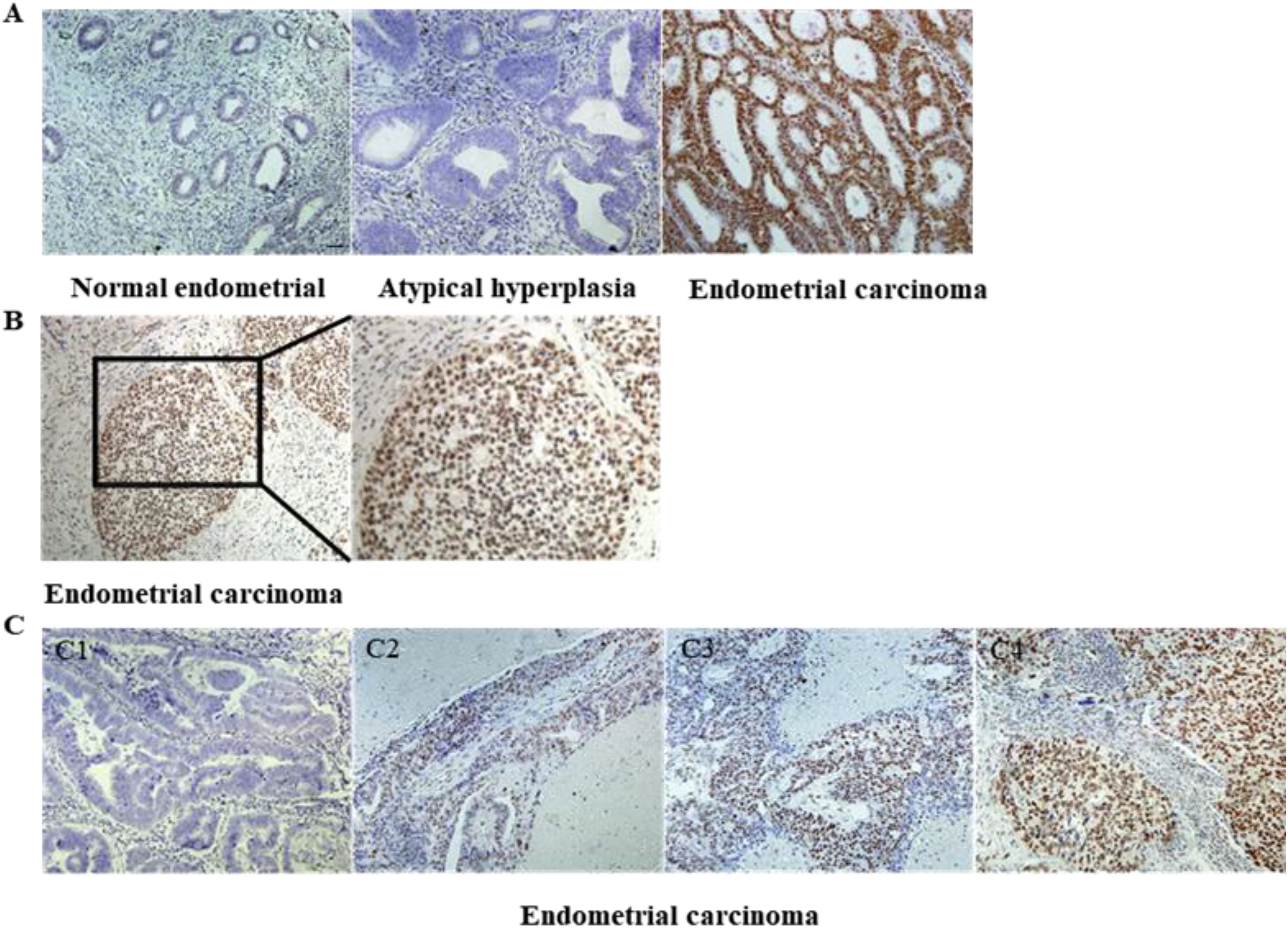
Immunohistochemical staining was employed to detect HSF1 protein in normal endometrial tissue, atypical endometrial hyperplasia, and EC. A: Immunohistochemical micrographs display that HSF1 expression level was strongly positive in EC tissues, while it was weakly stained in normal endometrial and atypical endometrial hyperplasia tissues (magnification, × 200). B: In EC, HSF1 expression level was primarily localized in the nucleus (magnification, × 200 and ×400). C: Immunohistochemical micrographs of HSF1 expression level in EC, which was categorized by staining intensity: (C1) negative, (C2) low, (C3) moderate, and (C4) high (magnification, × 200). Scale bar: 50 µm.
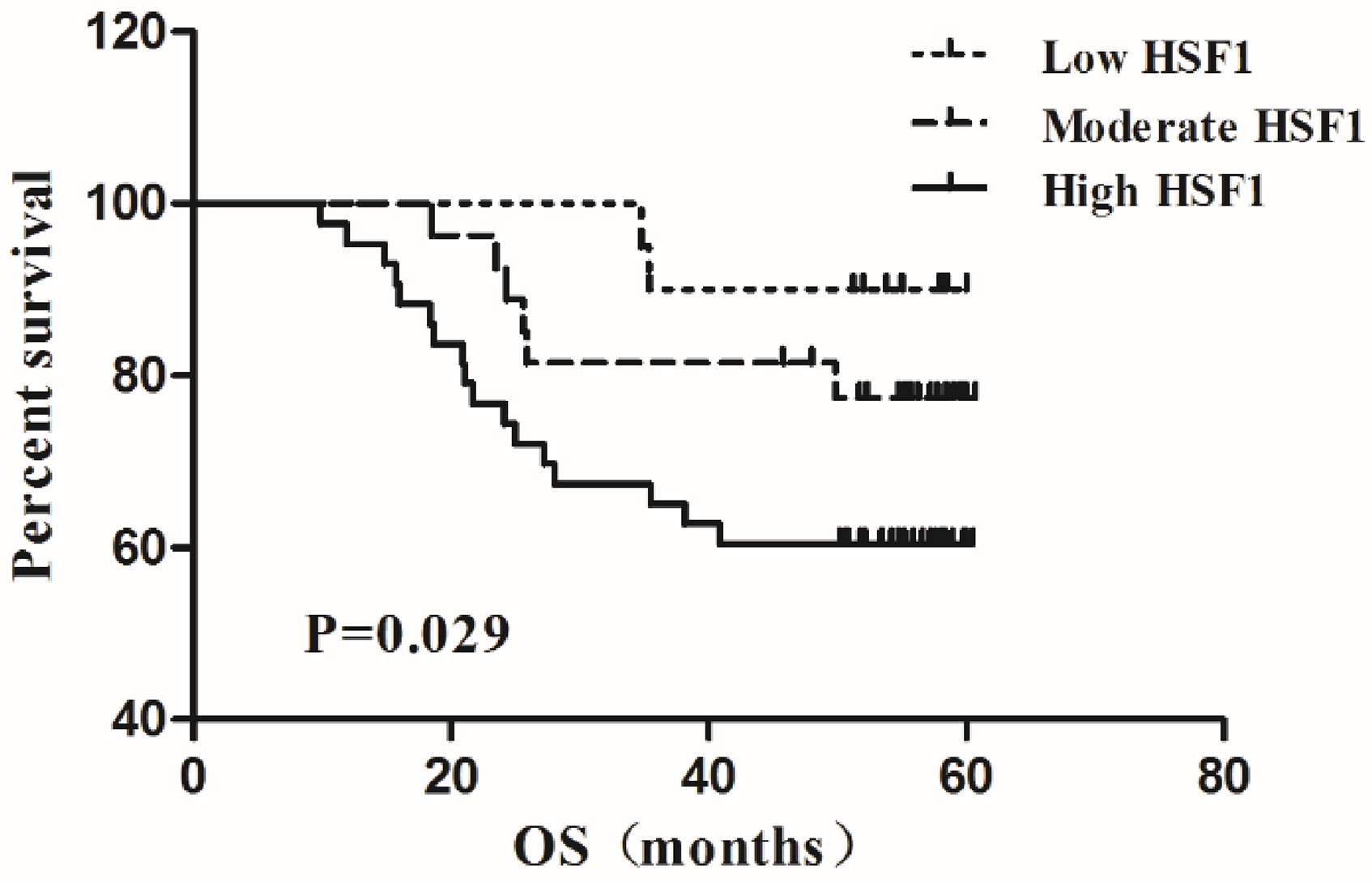
The 5-year survival curves according to the HSF1 expression level in EC patients indicated that those with high HSF1 expression level had significantly shorter overall survival (OS) compared with patients with moderate or low HSF1 expression level.
Expression level of HSF1 in different endometrial tissues.

Relationship between HSF1 expression level and EC patients’ clinicopathological features.
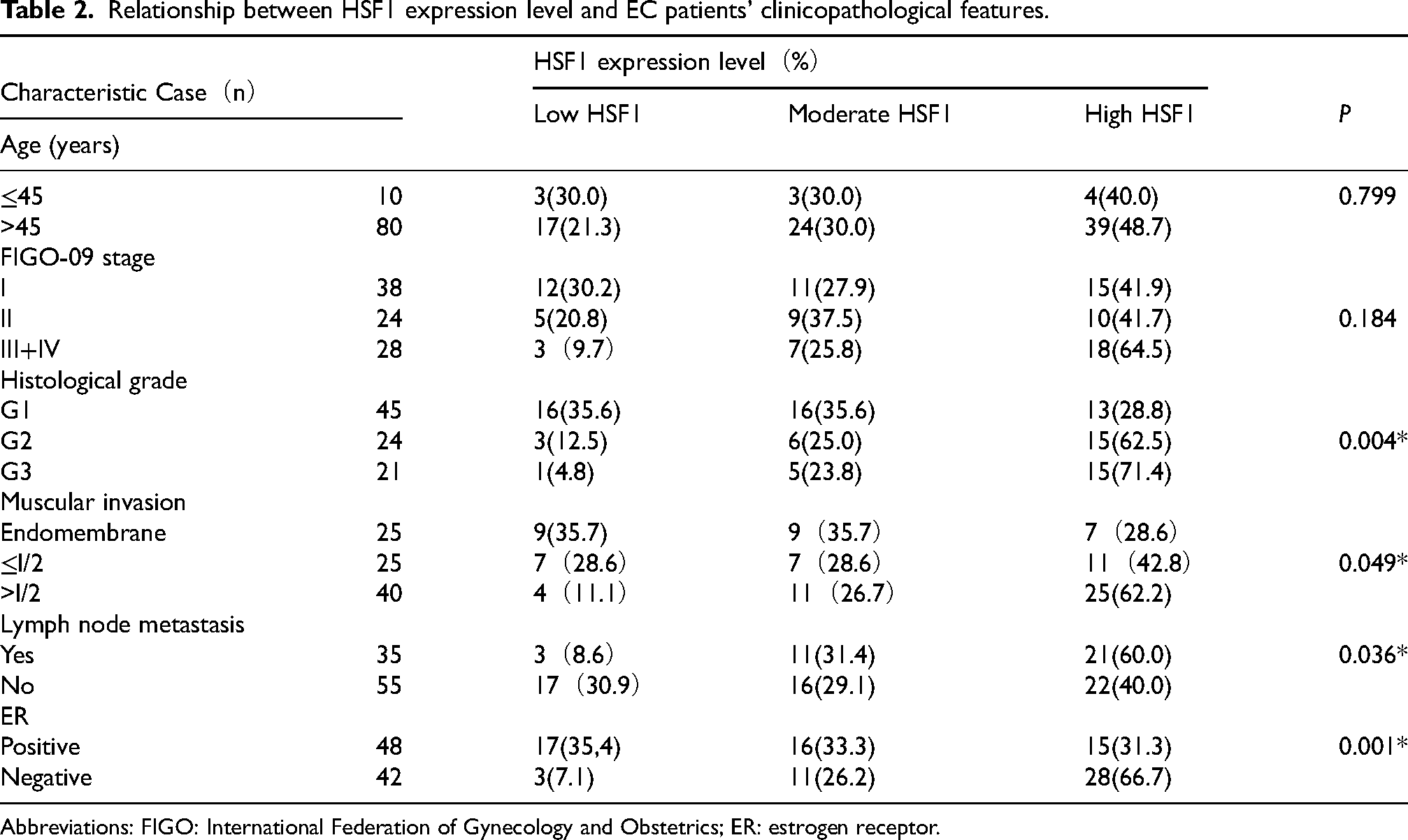
Abbreviations: FIGO: International Federation of Gynecology and Obstetrics; ER: estrogen receptor.
High HSF1 expression level in Ec cells was associated with low Er expression level
To explore HSF1 expression level, qRT-PCR, and Western blotting were employed to detect HSF1 mRNA and protein expression in human endometrial epithelial cells and three endometrial carcinoma cell lines. Human endometrial epithelial cells were used as the negative control. HSF1 and ER were detectable in all the cell lines. Among them, HSF1 expression level was the highest in Ishikawa cells (Figure 3A, C, and D), while ER expression level was the lowest (Figure 3B, E, and F).
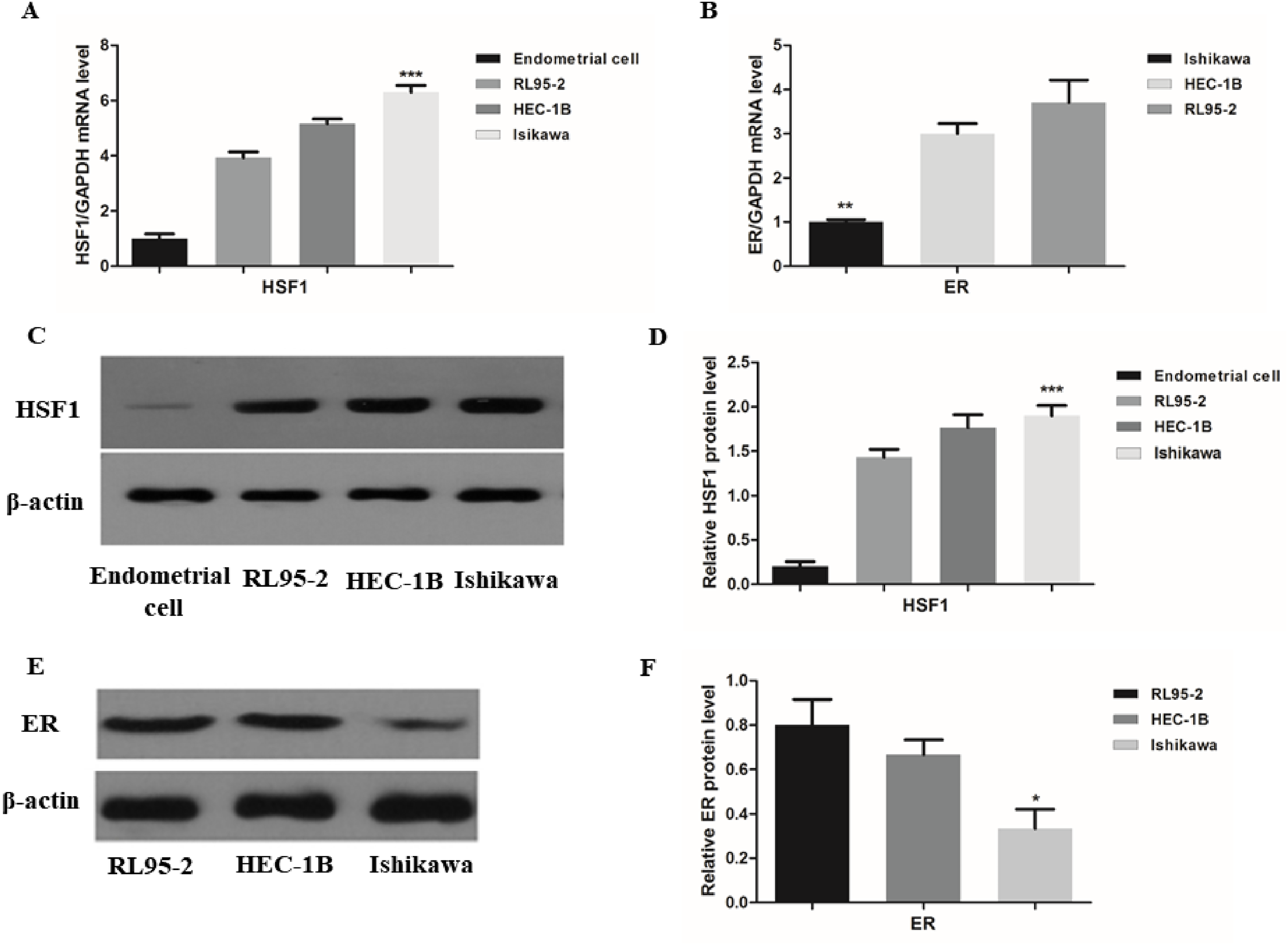
Expression levels of
Knockdown of HSF1 expression level inhibited invasiveness and migration of Ishikawa cells
To explore the potential function of
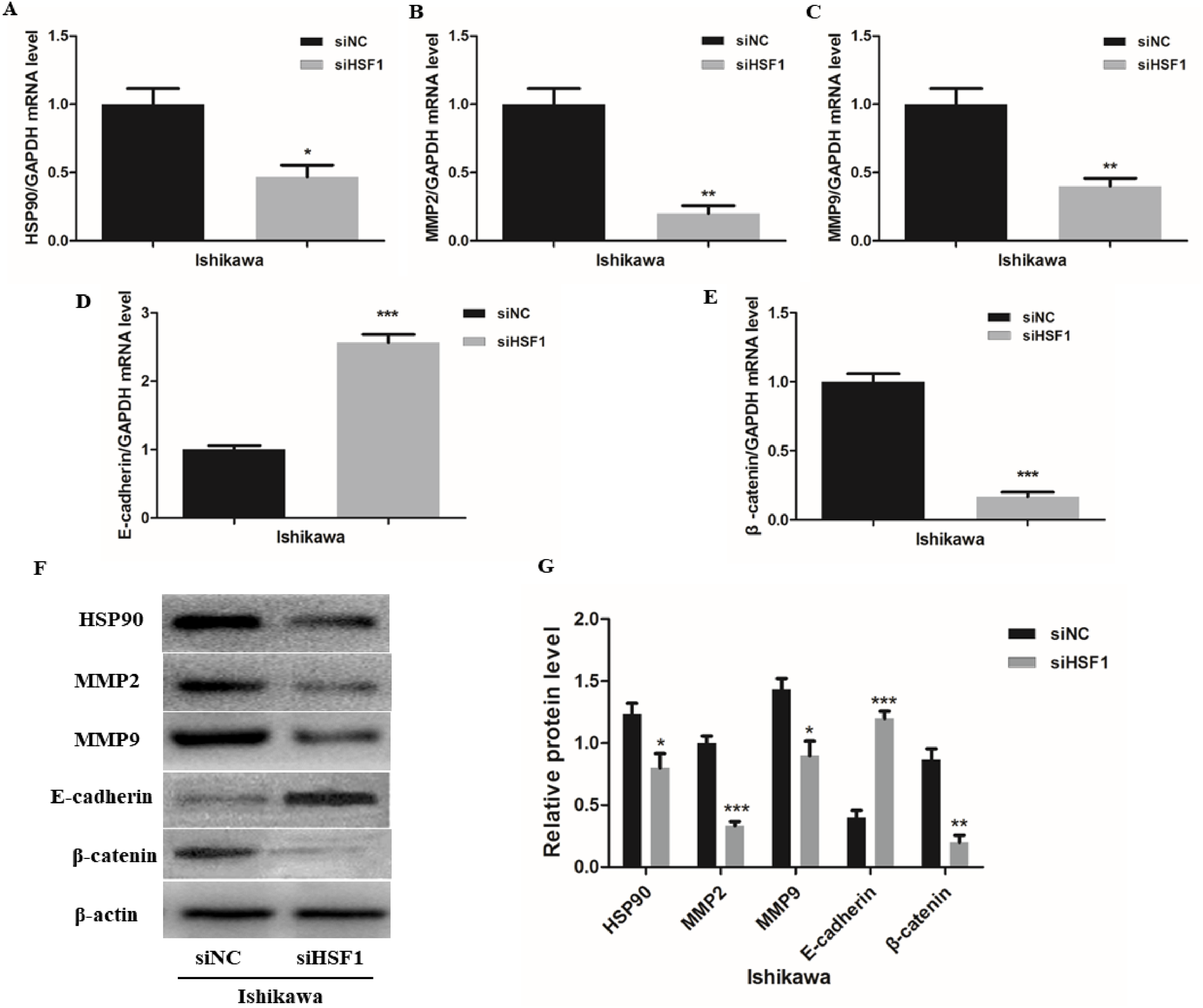
Knockdown of
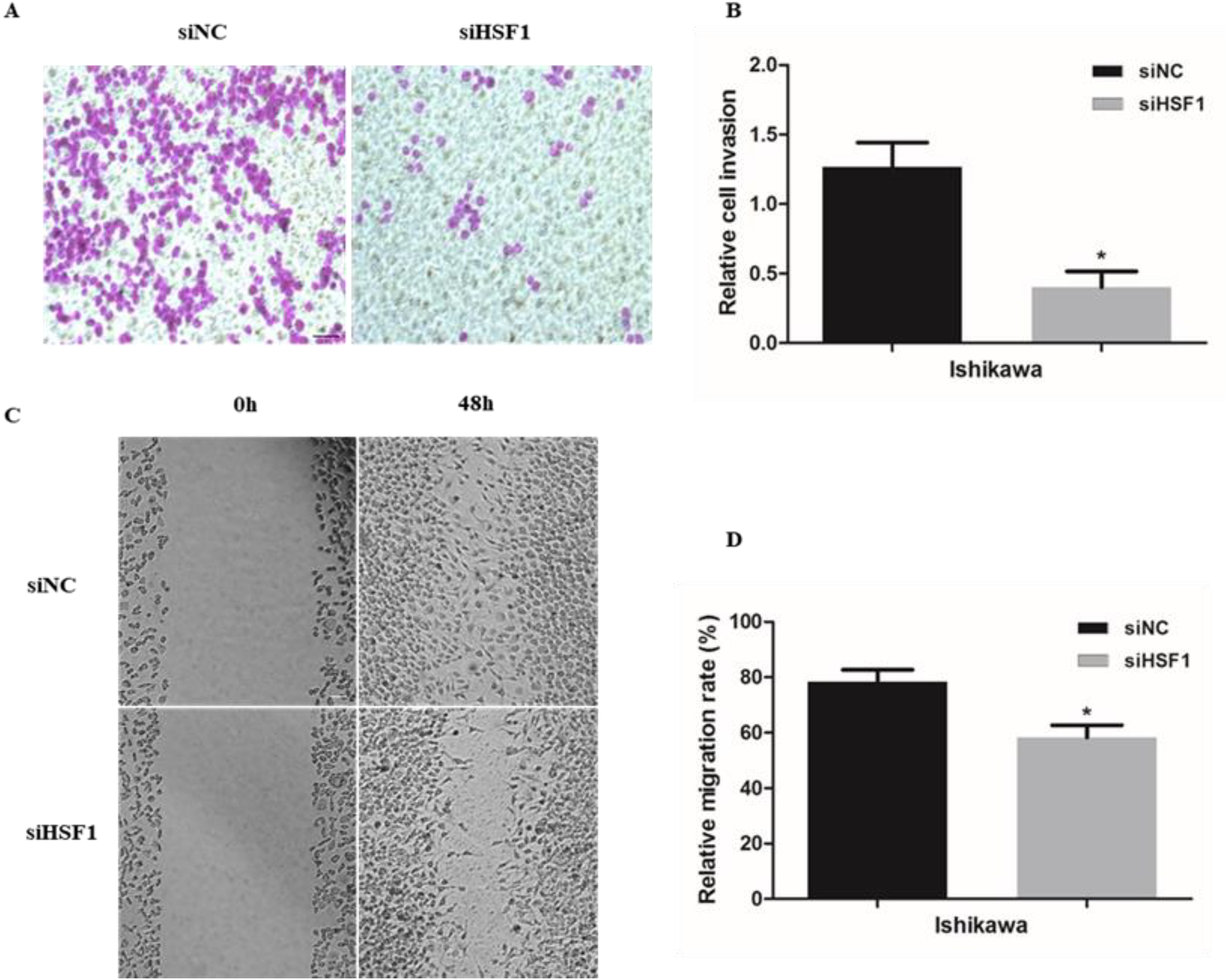
Cell invasion and migration were assessed in Ishikawa cells transfected with
Knockdown of HSF1 induced cell cycle arrest and apoptosis in Ishikawa cells
To assess the role of
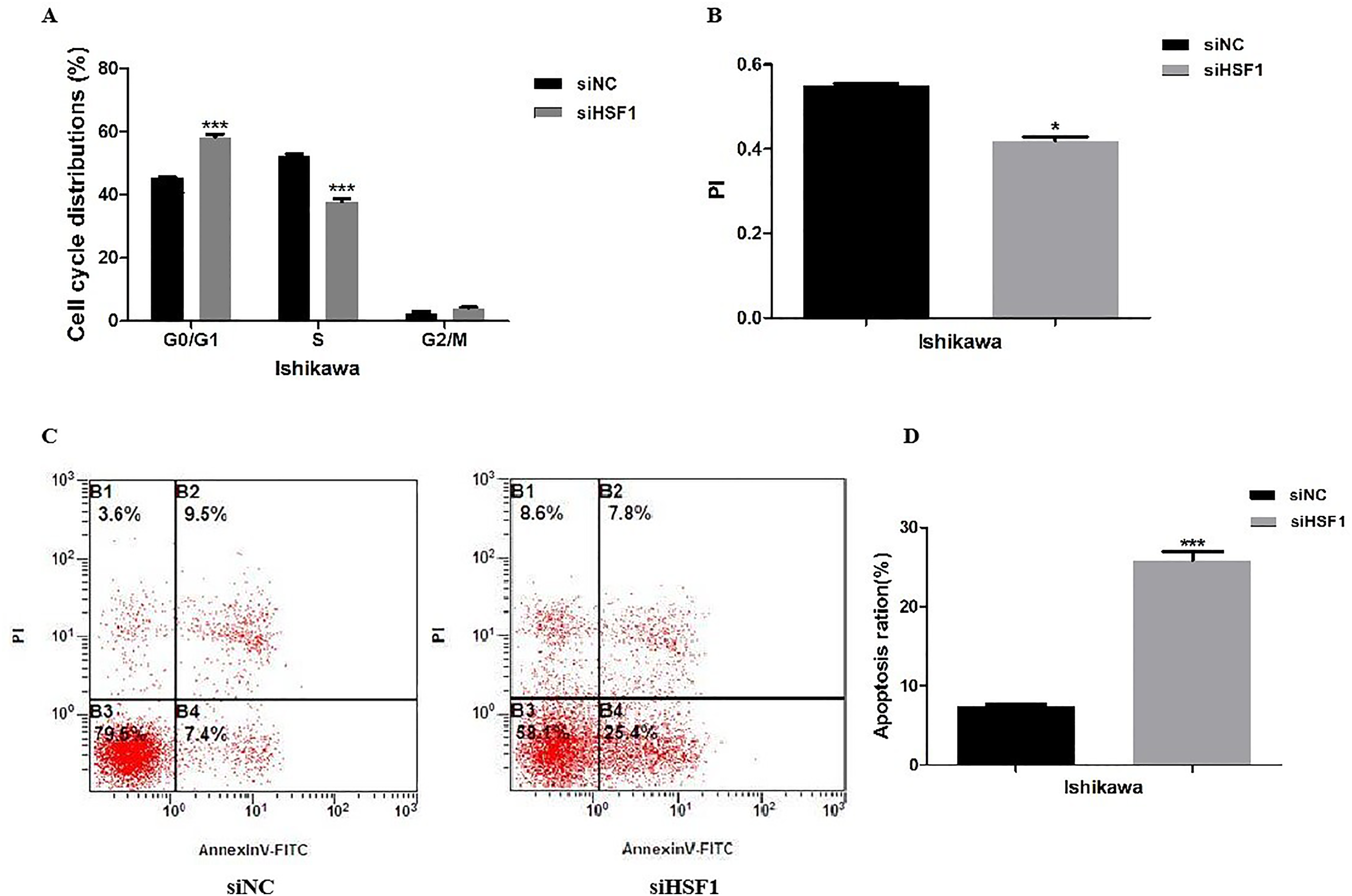
Knockdown of
Discussion
EC is a prevalent malignant tumor in obstetrics and gynecology, ranking first among malignant tumors of the female reproductive system in developed countries and second only to cervical cancer in China.1–3 However, the pathogenesis of EC has not yet been well documented. Existing literature suggests that
In the present study, the relationship between HSF1 expression level and 90 EC patients’ clinicopathological characteristics was assessed using immunohistochemistry. It was found that HSF1 expression level was positively correlated with histological grade, muscular invasion endomembrane, histological type, lymph node metastasis, and ER expression level. HSF1 is associated with aggressive disease and poor survival in EC. Engerud's study of 823 EC samples indicated that high HSF1 expression level was significantly associated with older age, non-endometrioid histological type, high tumor grade, and aneuploidy.
13
EC is typically categorized into type I (estrogen-dependent) and type II (non-estrogen-dependent) according to its main biological behavior.
15
Type I tumors typically express high levels of ER and progesterone receptor (PR) and are associated with superior prognosis. In contrast, type II tumors have low levels of ER and PR and are associated with poor prognosis.16–18
To elucidate the mechanism by which HSF1 functions as an oncogene in EC,
Invasion and metastasis are the major causes of EC patients’ poor prognosis.26,27 Therefore, it is essential in the ongoing research to explore the molecular mechanisms underlying the pathogenesis and progression of metastasis, emerging necessary for developing novel therapies to treat EC. The function of
Conclusions
This study further elucidated the molecular mechanisms underlying invasion and metastasis of EC. It was revealed that the expression level of
Footnotes
Acknowledgments
The authors thank the clinical staff who supported this study.
Ethics statement
This study was approved by the Ethics Committee of the Fourth Military Medical University. The patients/participants provided their written informed consent to participate in this study.
Author's contributions
Conceptualization: Haixia Liu, Xietong Wang, Biliang Chen, Junbin Yin, and Aihua Li.
Interpretation or analysis of data: Xiao Gu and Liang Shen.
Preparation of the manuscript: Haixia Liu, Jinlai Meng, and Yongzhong Gu.
Revision for important intellectual content: Jia Li and Yanhong Lv.
Supervision: Xietong Wang, Biliang Chen, Junbin Yin, and Aihua Li.
Funding
This study was supported by the Young Scientists Fund of the National Natural Science Foundation of China (Grant No. 81402133), Clinical Science and Technology Innovation Program of Ji’nan Province (Grant No. 202328072), and Medical Technology Innovation Incentive Program of Shandong Provincial Hospital Affiliated to Shandong First Medical University (Grant No. CXJL: ZQN – 202209).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The raw data that support the findings of this study are available from the corresponding author.
