Abstract
Background
Cisplatin (DDP) resistance is a major challenge in the management of non-small cell lung cancer (NSCLC). Panax notoginseng has anticancer effects on a variety of solid tumors, but data on NSCLC and DDP resistance are lacking.
Objective
To investigate the effect of Panax notoginseng on DDP resistance in NSCLC in vitro and in vivo and explore the mechanisms involved.
Methods
A 1 g/mL Panax notoginseng extract was prepared to treat the A549 and DDP-resistant A549/DDP cell lines. Cell proliferation was assessed using the CCK-8 assay, and apoptosis was measured via Annexin V-FITC/PI staining and flow cytometry. Glucose uptake, ATP production, and lactate levels were evaluated. Protein levels of p-AKT, GLUT1, HKII, and cleaved-caspase-3 were analyzed by Western blot. IGF1 was used to activate the Akt pathway. In vivo, A549/DDP cells were inoculated into nude mice to establish subcutaneous tumors, and tumor growth and apoptosis were assessed.
Results
Panax notoginseng inhibited A549/DDP cell proliferation, enhanced DDP-induced apoptosis, and reduced glucose uptake, ATP, and lactate levels (all p < 0.05). Combined treatment decreased p-AKT, GLUT1, and HKII expression while increasing cleaved-caspase-3(p < 0.05). IGF1 reversed these effects, indicating Akt pathway involvement (p < 0.05). In vivo, Panax notoginseng and DDP significantly suppressed tumor growth and increased apoptosis in tumors, confirming enhanced chemosensitivity (p < 0.05).
Conclusion
Panax notoginseng can improve the sensitivity of A549/DDP cells to DDP by inhibiting the effects of TRIM46 and Akt signaling pathways on glycolysis in vivo and in vitro.
Introduction
Lung cancer is a prevalent and deadly malignancy worldwide. Non-small cell lung cancer (NSCLC) is the main type of lung cancer, accounting for approximately 80–85% of cases. 1 NSCLC patients are often diagnosed at advanced stages, accompanied by metastasis to multiple organs, resulting in a low 5-year survival rate (less than 20%) and a high recurrence rate.1,2 Currently, besides early surgical resection, chemotherapy, particularly using cisplatin (DDP), is the primary treatment approach. However, DDP is prone to developing drug resistance, tremendously limiting its clinical efficacy. 3 Therefore, it is crucial to study and identify key molecules involved in the development of acquired resistance to DDP at different stages.
Traditional Chinese medicine (TCM) is an ancient practice dating back over 3500 years and has emerged as a promising therapeutic approach for lung cancer treatment. It can be used as an adjuvant or maintenance therapy alongside conventional treatments. Several clinical studies have confirmed the favorable efficacy of TCM in treating lung cancer. 4 Panax notoginseng, a traditional medicinal herb, has long been known for its analgesic properties and ability to improve blood circulation and prevent blood stasis. More recently, it has shown inhibitory effects on the proliferation and metastasis of gastric, esophageal, breast, and hepatocellular carcinomas.5,6 Nevertheless, there is limited research on its precise antitumor effects, and the existing studies lack systematic investigation. Notably, the role of Panax notoginseng in lung cancer remains unexplored both domestically and internationally.
The Warburg effect, also known as aerobic glycolysis, describes the phenomenon where tumor cells convert glucose into lactate even under well-oxygenated conditions rather than breaking it down through mitochondrial oxidative phosphorylation like normal cells. This metabolic alteration is characterized by increased glucose uptake and lactate production. Aerobic glycolysis is essential in cancer proliferation, growth, invasion, and treatment. Several researchers have found that aerobic glycolysis is involved in tumor cell resistance against sorafenib. 7 AKT plays a vital role in regulating glycolytic metabolism and is involved in the stimulation of glucose uptake and glycolysis processes. 8 For example, OTU deubiquitinase, Ubiquitin Aldehyde Binding 2 (OTUB2) is involved in aerobic glycolysis and promotes the development of NSCLC by activating the AKT/mTOR pathway and inducing the Warburg effect.9,10 Gaining a comprehensive understanding of targeted AKT activation and aerobic glycolysis process can provide insights into the pathogenesis of lung cancer, potential therapeutic strategies, and resistance mechanisms to DDP.
Tripartite motif-containing 46 (TRIM 46) is a member of the TRIM family. TRIM family proteins contain three conserved structural domains, namely the zinc finger structural domain, the B-box structural domain, and the coiled helix structural domain. TRIM family members are recently reported to be closely related to the process of tumorigenesis and progression. For example, TRIM15 and TRIM24 have been shown to participate in tumor development as oncogenes. 11 In the case of breast cancer, TRIM46 has been reported to promote tumor growth and chemoresistance. 12 Moreover, overexpression of TRIM46 has been shown to enhance lung adenocarcinoma cell proliferation, glycolysis, and resistance to DDP. This effect is achieved through the promotion of ubiquitination of PHLPP2 and upregulation of phosphorylated AKT (p-AKT). 13 However, the specific mechanism by which TRIM46 operates in NSCLC is still unclear.
The present study aimed to investigate the effect of Panax notoginseng on DDP resistance in NSCLC in vitro and in vivo and explore the mechanisms involved. The study could provide valuable insights into the mechanisms through which Panax notoginseng can benefit the chemotherapy of NSCLC.
Materials and methods
Preparation of Chinese medicine for Panax notoginseng
Crushed Chinese medication formula (100 g; Kangmei Pharmaceutical Co., Ltd., catalog number KMZH0522671) was placed in a round-bottomed flask. A 1:1 mixture of absolute ethanol and distilled water was added to soak the preparation for 30 min. The mixture was heated in a water bath, with stirring and filtration, for 60 min. In parallel, the Chinese medicine residues of Panax notoginseng were mixed with distilled water and decocted using the same process for 60 min. After filtration, the two decoctions were mixed, and the mixture was concentrated to a concentration of 1 g/mL using a water bath at 90°C. The resulting concentrated solution was stored at −20°C as a stock solution. The stock solution was diluted with distilled water to achieve the required concentrations. The solutions were filtered, sterilized, and stored at 4°C for use. 14 The characterization of the preparation is provided in the Supplementary Materials.
Cells and cell culture
The human NSCLC cell line A549 and its DDP-resistant counterpart A549/DDP were purchased from BioLeaf Biotech (Shanghai, China). A549 cells were cultured in RPMI-1640 medium (11875101, Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS, 10099141C, Gibco), and 1% penicillin/streptomycin solution (P/S, 100 U/ml, P1400, Solarbio, Shanghai, China). The cells were maintained in 5% CO2 at 37 °C. For A549/DDP cells, an additional concentration of 2 μg/mL of DDP was added to the cell culture medium.
CCK-8 assay
A549/DDP cells (3 × 103) were seeded onto 96-well culture plates and cultured at 37°C overnight. The cells were treated as needed, and at 0, 24, 48, and 72 h following treatment, Cell Counting Kit-8 (CCK-8) reagent mixed with a serum-free medium at a 1:10 volume ratio was added to each well (100 µL per well). The plates were incubated for 1 h at 37°C in a 5% CO2 incubator. The optical density (OD) value of each well was measured with a microplate reader (Bio-Rad Laboratories) at a wavelength of 450 nm.
Apoptosis assay
The cells subjected to treatment were digested and collected into responding centrifuge tubes. They were then washed once with PBS before being resuspended gently in 195 μL of Annexin V-FITC binding solution. Following this, 5 μL of Annexin V-FITC binding solution was added and mixed gently. The cell suspension was incubated at 4°C for 15 min while ensuring it was shielded from light. Then, 5 μL of propidium iodide staining solution was added and gently mixed, and the cells were further incubated at 4°C for 5 min in a light-free environment. Subsequently, flow cytometry (FCM, BD Biosciences) was employed to detect apoptosis.
Glucose uptake assay
After pre-treatment, the cell culture medium of each group was replaced with a low-serum culture medium containing 0.5% FBS. After continuing incubation for 1 h, the culture medium was removed and replaced with a tested mixture solution (Biovision, USA) that was formulated according to the kit instructions. The cells were then incubated for an additional 30 min. The cells were collected on ice, and a 1× analytical solution (provided in the kit) was introduced into the cells. Finally, glucose uptake was detected via FCM.
Lactate/ATP assay
Cells were grown in a 6-well plate (5 × 105 cell/well) for 24 h at 37°C 48 h after treatment. The cells’ lactate release and ATP content were determined by Lactic Acid assay kit (A020-1-2, Nanjing Jiancheng Bioengineering Institute, China) and ATP assay kit (A095-1-1, Nanjing Jiancheng Bioengineering Institute, China), respectively, following the manufacturer's instruction.
Western blotting assay
Treated cells were lysed with RIPA cell lysis solution (Shanghai Beyotime Biotechnology) on ice to extract proteins collected into centrifuge tubes. As determined by BCA protein concentration, an equal amount (20 μg) of protein was taken from each group and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) to separate the proteins. At the end of electrophoresis, the gel was transferred to a PVDF membrane, which was blocked in a 5% skimmed milk powder blocking solution for 2 h at room temperature. Afterward, this PVDF membrane was incubated with a primary antibody at 4°C overnight. The PVDF membrane was washed three times with TBST for 5 min each time, incubated with a secondary antibody (1:1000, Beyotime, Shanghai, China) for 1 h at room temperature, and after chemiluminescent detection by ECL (Bio-Rad Laboratories, Inc. Hercules, CA, USA), the PVDF membrane was scanned by a gel Imaging System to read the protein bands. The information on primary antibodies is as follows: Anti-TRIM46 (ab169044, 1:1000, Abcam, Cambridge, UK), Anti-GLUT1 (ab115730, 1:1000, Abcam), Anti-Hexokinase II (anti-HK2, ab209847, 1:800, Abcam), Anti-AKT (#9272, 1:2000, Cell signaling technology [CST], Danvers, MA, USA), Anti-p-AKT (#4060, 1:1000, CST), Anti-cleaved-caspase-3 (ab2302, 1:1000, Abcam), and Anti-GAPDH (#5174, 1:3000, CST). The secondary antibody was a sheep anti-rabbit HRP-conjugated antibody (#91196, 1:2000, CST). The captured images were processed using the Image J software (National Institutes of Health, Bethesda, MD, USA). In the software, the contrast and brightness were adjusted to ensure that the bands were clearly visible and to avoid over- or under-exposure of the bands. The appropriate tool was used to separate the bands from the background and measure the grayscale value of each band. To avoid bias, the bands for each sample were repeated at least three times, and the average gray value was calculated. Finally, the gray value of the target protein was compared with the gray value of the reference protein (GAPDH) to correct for potential variation in the experiment and obtain the relative expression level.
Tumor formation experiment in nude mice
The study was approved by the ethics committee of Wenzhou Traditional Chinese Medical Hospital (approval #WTCM-KT-2020002). Twenty-four male BALB/c nude mice (6–8-week-old, with a body weight of 20 ± 2 g) were purchased from Shanghai SLAC Laboratory Animal Co. (Shanghai, China). The mice were housed in a controlled environment with a temperature of 22 ± 1°C, humidity at 65–70%, and a 12-h light/dark cycle. The mice had free access to food and water. The 24 nude mice were randomly divided into four groups: control group, Panax group, DDP group, and DDP + Panax group. A549/DDP cells (5 × 106 cells) were subcutaneously inoculated into the axilla of each nude mouse. Subcutaneous tumors usually formed 7–10 days after inoculating the cells, and tumor growth was recorded by measuring tumor size starting on day 12. When the neoplasms grew to a size of 5 mm, the following treatments were administered. (1) The Panax group received Panax notoginseng powder at a dosage of 50 mg/kg per day via gavage. 15 (2) The DDP group received DDP at a dosage of 5 mg/kg per day via intraperitoneal (IP) injection. (3) The DDP + Panax group received Panax notoginseng powder (50 mg/kg per day) and DDP (5 mg/kg per day). (4) The control group received equal amounts of PBS via gavage and sterile physiological saline via intraperitoneal injection.
After the drug administration, the tumor diameter was measured every 3 days. The average volume of the neoplasms (mm3) was calculated using the formula: Volume = 1/2(Length × width)2, and the growth curves were plotted accordingly. Three weeks after treatment, the nude mice were executed. The tumor blocks were stripped, weighed, and partially fixed in paraformaldehyde, while the remaining tumor tissues were preserved in liquid nitrogen.
TUNEL assay
The paraformaldehyde-fixed tumor tissues were embedded into wax blocks and made into paraffin sections with a thickness of 4–5 μm. Sections were dewaxed, hydrated, and immersed in trypsin for digestion for 15 min and washed three times with PBS for 3 min. To detect apoptotic cells, 50 μL of TUNEL reaction mixture solution from the TUNEL Staining Kit (Roche, Indianapolis, IN, USA) was added to the specimens, and the reaction was carried out for 1 h at room temperature while being protected from light. The specimens were then subjected to DAB staining, dehydrated step by step, vitrified by dimethyl benzene, and sealed with neutral balsam. Finally, the sections were photographed under a microscope (Nikon, Tokyo, Japan). The captured images were processed using the Image J software (National Institutes of Health, Bethesda, MD, USA). In the software, the contrast and brightness were adjusted to ensure that the bands were clearly visible and to avoid over- or under-exposure of the bands. The appropriate tool was used to separate the bands from the background and measure the grayscale value of each band. To avoid bias, the bands for each sample were repeated at least three times, and the average gray value was calculated. Finally, the gray value of the target protein was compared with the GAPDH to correct for potential variation in the experiment and obtain the relative expression level.
Statistical analysis
Experimental data were presented as mean ± standard deviation, and all experiments were conducted in triplicate. The statistical significance of differences between the two groups was determined using the Student's t-test using SPSS statistical software. Meanwhile, one-way ANOVA was utilized to compare the differences between groups. p-values <0.05 were considered to indicate a statistically significant difference.
Results
Panax notoginseng improves the sensitivity of A549/DDP cells to DDP
A549/DDP cells were initially treated with different concentrations of Panax notoginseng to observe the changes in cell proliferation. As shown in Figure 1(a), all groups significantly inhibited the proliferation of A549/DDP cells after 48 h of treatment. Hoewever, it should be noted that a high concentration of Panax notoginseng exhibited some toxic effects on cells. Therefore, subsequent experiments utilized concentrations of 100, 200, and 400 mg/L of Panax notoginseng.
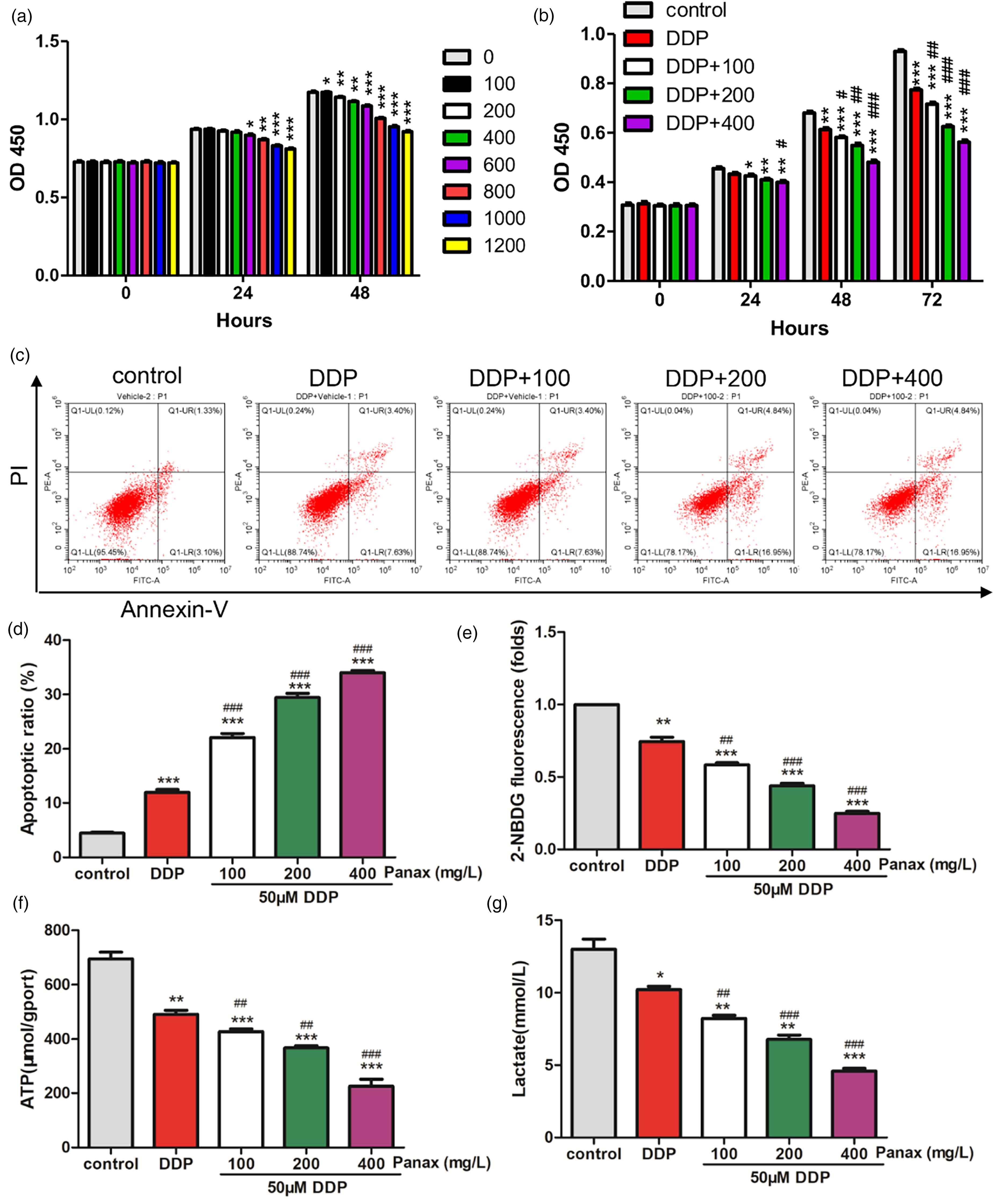
Panax notoginseng can improve the sensitivity of A549/DDP cells to DDP. a. A549/DDP cells were treated with different concentrations of Panax notoginseng (0, 100, 200, 400, 600, 800, 1000, and 1200 mg/L), and CCK-8 detected the changes in cell proliferative ability. *: p < 0.05, **: p < 0.01, ***: p < 0.001 vs. control. B-H. DDP (50 μM) alone and in combination with Panax notoginseng (0, 100, 200, 400 mg/L) co-treated A549/DDP cells, CCK-8 detected the changes in cell proliferative ability (b), and FCM detected apoptosis (c-d), glucose uptake (e), ATP production (f), and lactate production (g). *: p < 0.05, **: p < 0.01, ***: p < 0.001 vs. control. #: p < 0.05, ###: p < 0.01, ####: p < 0.001 vs. DDP.
Next, A549/DDP cells were co-treated with 50 μM of DDP alone and in combination with Panax notoginseng (100, 200, and 400 mg/L) to explore the role of Panax notoginseng in DDP resistance of lung cancer cells. The results showed that DDP significantly inhibited the proliferation of A549/DDP cells (Figure 1(b)). Additionally, different concentrations of Panax notoginseng, when combined with DDP, significantly increase the ability of DDP to inhibit cell proliferation. This suggests that Panax notoginseng could enhance the sensitivity of A549/DDP cells to DDP. In terms of apoptosis, compared to the control group, treatment with DDP alone led to a significantly higher apoptosis rate. The combination of DDP and Panax notoginseng further increased the apoptosis rate significantly, with a dose-dependence effect of Panax notoginseng (Figure 1(c) and (d)).
Considering the significant role of glycolysis in tumor development, 13 we further explored whether Panax notoginseng could affect lung cancer cell metabolism. As shown in Figure 1(e) to (g), compared to the control group, treatment with DDP alone caused a decrease in glucose uptake, as well as ATP and lactate production. The combination of DDP and Panax notoginseng further amplified these effects significantly and exhibited a concentration-dependent manner.
Panax notoginseng improves the sensitivity of A549/DDP cells to DDP by inhibiting the Akt signaling pathway
The abnormally activated AKT signaling pathway can promote chemotherapy resistance in malignant tumors. 16 In light of this, DDP treatment was observed to decrease the protein levels of p-AKT, GLUT1, and HKII while elevating cleaved caspase-3 expression. Furthermore, the combination of DDP and Panax notoginseng elevated the effects of DDP on these proteins’ expression when compared to DDP treatment alone (Figure 2(a)). These results suggested that Panax notoginseng treatment can elevate A549/DDP cells’ sensitivity to DDP via the Akt signaling pathway.
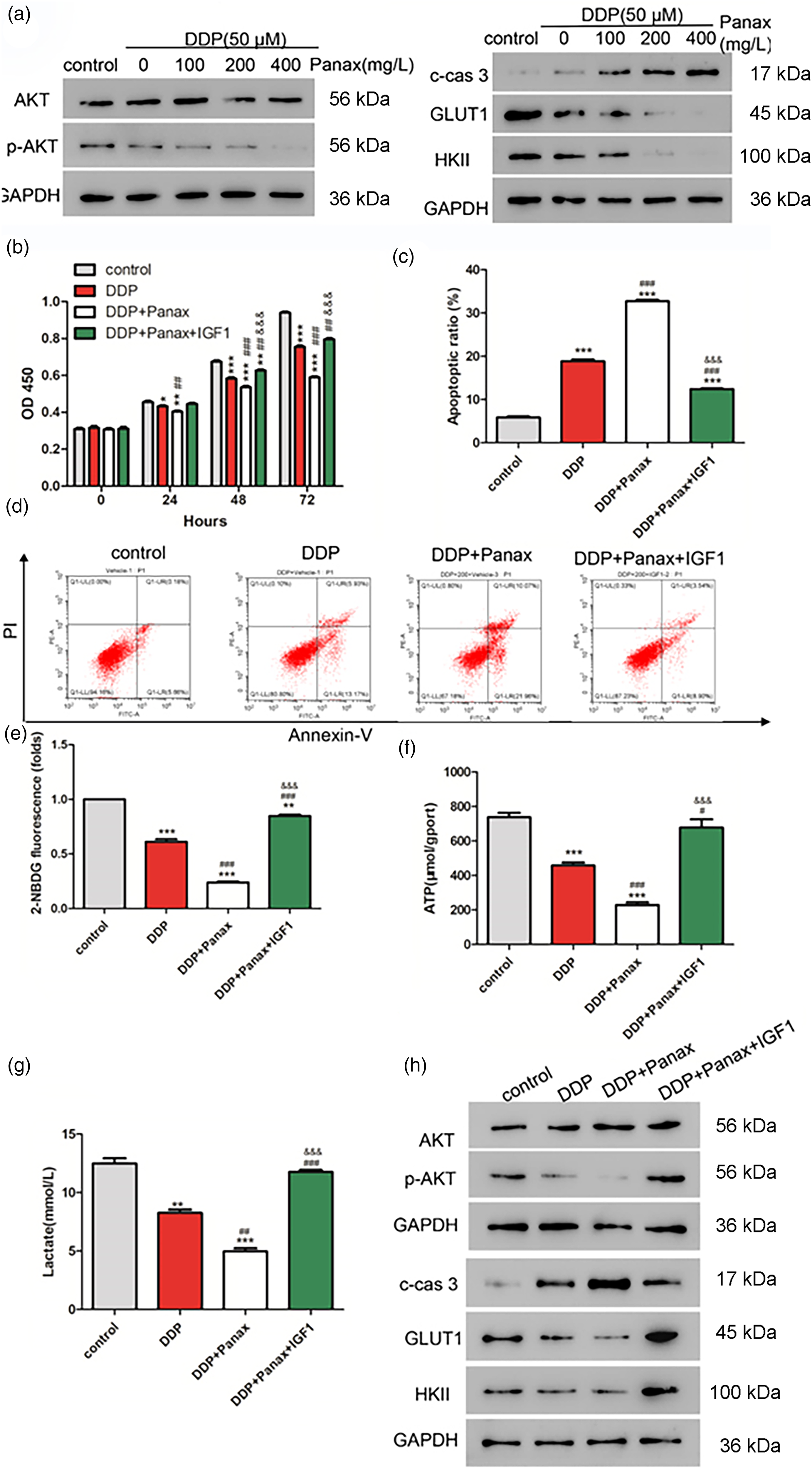
Panax notoginseng improves the sensitivity of A549/DDP cells to DDP by modulating the Akt signaling pathway. A549/DDP cells were treated with DDP (50 μM) alone and in combination with Panax notoginseng (200 mg/L) and IGF1 (AKT agonist, 50 ng/mL). a. Protein expression changes in p-AKT, c-cas3, GLUT1, and HKII were detected by Western blot. Changes in cell proliferative ability (b) and apoptosis (c-d) were detected by CCK-8. Glucose uptake (e), ATP production (f), and lactate production (g) were detected by FCM (h). Changes in the protein expression of p-AKT, c-cas3, GLUT1, and HKII were detected by western blot. *p < 0.05,**: p < 0.01,***:p < 0.001 vs. control. #:p < 0.05,##:p < 0.01,###:p < 0.001 vs. DDP. &&&:p < 0.001 vs. DDP + Panax.
IGF1, an agonist of the Akt signaling pathway, was utilized to verify whether the Akt signaling pathway is involved in the function of Panax notoginseng. First, the results of the cell proliferative ability assay showed that the IGF1 treatment reversed the decrease in proliferation induced by DDP and Panax notoginseng (Figure 2(b)). Next, we found that apoptosis induced by DDP and Panax notoginseng could also be neutralized by IGF1 (Figure 2(c) and (d)).
Metabolism-related indices were next examined. As expected, IGF1 treatment reversed the reduction in glucose uptake, production of ATP, and lactate induced by DDP and Panax notoginseng (Figure 2(e) to (g)). Furthermore, DDP and Panax notoginseng treatment could inhibit the protein expression of p-AKT, GLUT1, and HKII while promoting the protein expression of cleaved-caspase-3. Notably, all these effects could be reversed by IGF1 (Figure 2(h)). In summary, Panax notoginseng exerts its function by regulating the Akt signaling pathway.
Impact of Panax notoginseng involved in TRIM46 on the proliferation of A549 cells
To verify whether Panax notoginseng was involved in the effects of TRIM46, we co-treated cells with TRIM46 overexpressing lentivirus in combination with 5 μM DDP and 200 mg/L Panax notoginseng. The cell proliferation assay showed that TRIM46 overexpression could reverse the decrease in proliferation induced by DDP, but this effect was counteracted by Panax notoginseng (Figure 3(a)). We also found that TRIM46 could partially neutralize apoptosis induced by DDP, and the addition of Panax notoginseng further increased apoptosis (Figure 3(b) and (c)).
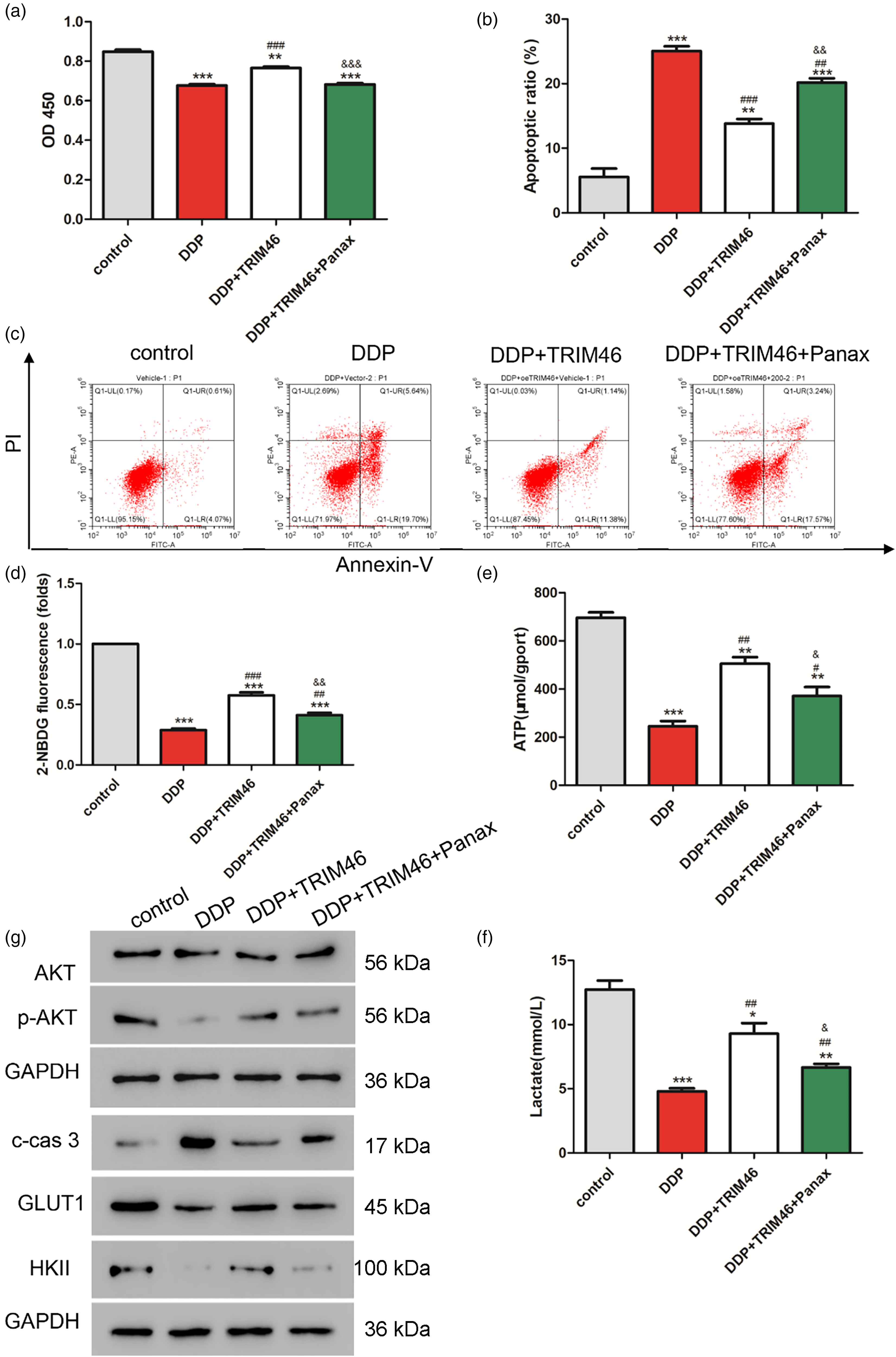
Effects of Panax notoginseng involved in the TRIM46 on the proliferation of A549 cells. TRIM46 overexpressing lentivirus was co-treated with 5 μM DDP and 200 mg/L Panax notoginseng in A549 cells. CCK-8 was used to detect the changes in cell proliferative ability (a). FCM was used to detect apoptosis (b-c), glucose uptake (d), ATP production (e), and lactate production (f). Western blot was performed to detect protein expression changes in p-AKT, c-cas3, GLUT1, and HKII (g). *: p < 0.05, **: p < 0.01, ***: p < 0.001 vs. control. #: p < 0.05, ###: p < 0.01, ####: p < 0.001 vs. DDP. &: p < 0.05, &&: p < 0.01, &&&: p < 0.001 vs. DDP + TRIM46.
Moreover, glucose uptake, ATP, and lactate production were significantly lower in all treatment groups compared to the control group. Furthermore, TRIM46 overexpression can alleviate the inhibitory effect of DDP on cell metabolism. However, the addition of Panax notoginseng can effectively reverse this alleviation effect (Figure 3(d) to (f)).
Meanwhile, DDP treatment inhibited the activation of the Akt pathway (Figure 3(g)). However, TRIM46 overexpression was able to reverse these inhibitory effects. Notably, the presence of Panax notoginseng could neutralize the effects of TRIM46 overexpression on the Akt pathway. These findings suggest that Panax notoginseng may interfere with the regulatory function of TRIM46 in modulating the Akt pathway.
Panax notoginseng enhances the sensitivity of A549/DDP cells to DDP in vivo
To establish subcutaneous tumorigenic models, the axilla of nude mice was inoculated with A549/DDP cells. As shown in Figure 4(a) and (b), both Panax notoginseng alone and DDP exhibited inhibitory effects on tumor growth. However, the combination treatment of Panax notoginseng and DDP resulted in the lowest tumor growth and volume. TUNEL staining was performed to assess cell apoptosis within the tumor tissue. The results demonstrated that while both Panax notoginseng and DDP alone induced a certain degree of apoptosis, the combination treatment group exhibited significantly higher apoptotic cell numbers compared to the other groups (Figure 4(c) and (d)). These findings indicated that Panax notoginseng significantly enhanced the chemosensitivity of A549/DDP cells to DDP.
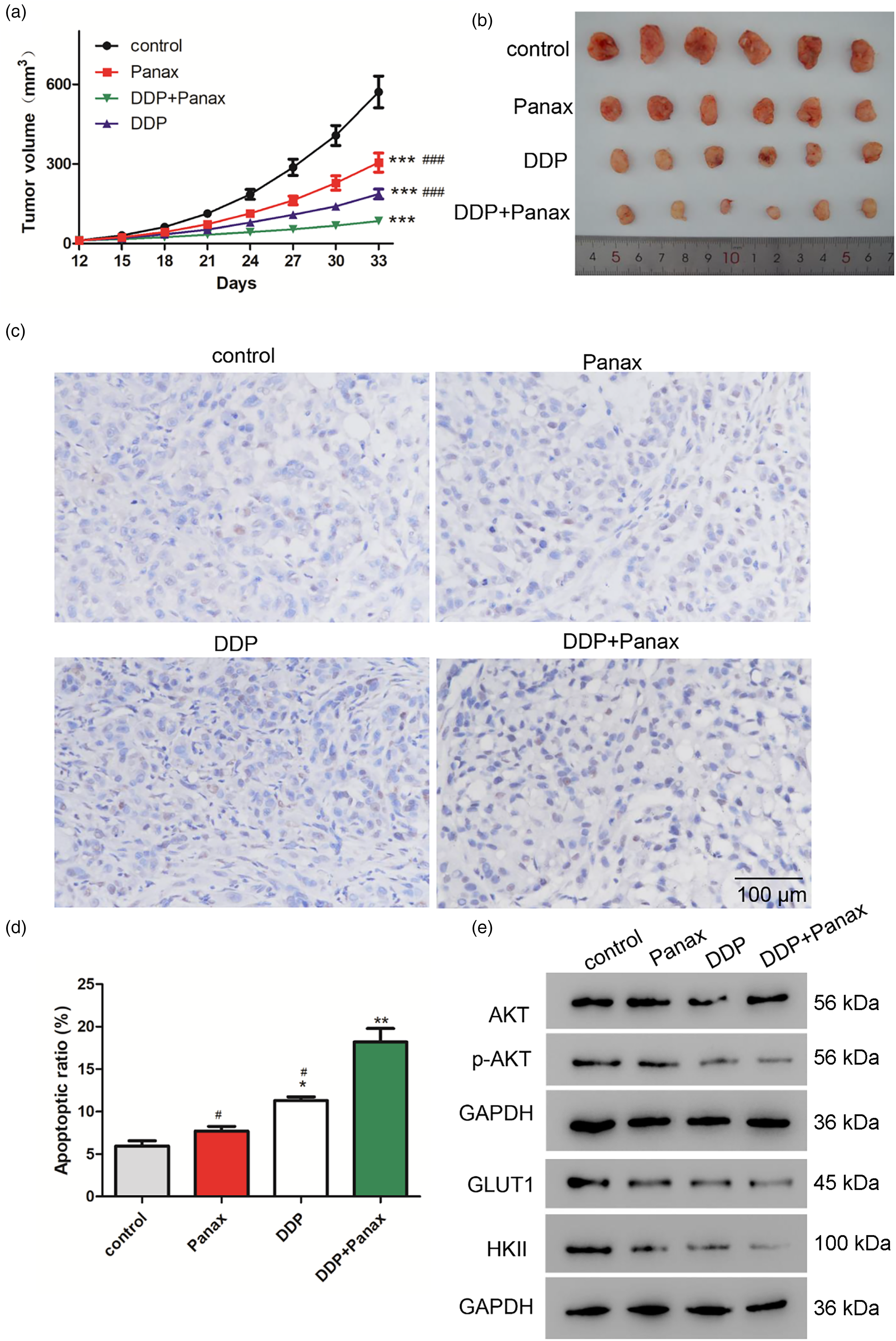
Panax notoginseng enhances the sensitivity of A549/DDP cells to DDP in vivo. (a) Volume records of tumors in each group (n = 6). (b) Picture records of tumor size in each group. (c-d) TUNEL assay for apoptosis. (e) Protein expression level of p-AKT, GLUT1, and HKII in tumor samples of each group. *: p < 0.05, **: p < 0.01 vs. control. #: p < 0.05, ####: p < 0.001 vs. DDP + Panax.
Furthermore, protein expression levels of p-AKT, GLUT1, and HKII in both Panax notoginseng and DDP groups were significantly lower compared to the control group. Additionally, the lowest protein expressions were observed in the group receiving combined Panax notoginseng and DDP treatment (Figure 3(e)). In conclusion, Panax notoginseng may inhibit the growth of DDP-resistant tumors by inhibiting the Akt signaling pathway.
Discussion
In this current study, the combination treatment of Panax notoginseng and DDP exhibited significant inhibition of cell proliferation, promotion of apoptosis, and reduction in aerobic glycolysis levels in A549/DDP cells. Furthermore, the combined treatment effectively suppressed the proliferation of subcutaneous neoplasms in nude mice. These findings highlight the ability of Panax notoginseng to enhance the sensitivity of DDP-resistant lung cancer cells to DDP by inhibiting the effects of TRIM46 and the Akt signaling pathway on glycolysis.
As one of the most commonly used traditional Chinese medicines, Panax notoginseng has significant therapeutic effects in inhibiting tumor proliferation and metastasis. In recent years, studies have shown that Panax notoginseng can effectively alleviate the side effects caused by DDP in the treatment of malignant tumors. For instance, the total saponins of Panax notoginseng have been found to increase HIF-1α/BNIP3 and inhibit mitochondrial apoptosis pathways,17,18 thereby reducing DDP-induced acute kidney injury.18,19 Additionally, Panax notoginseng can enhance the cytotoxicity of cisplatin against malignant tumors by affecting gap junctions. 20 This study provides evidence for the inhibitory effects of Panax notoginseng on inhibiting the proliferation of NSCLC and found its ability to enhance the sensitivity of NSCLC to DDP, thus providing a theoretical foundation for investigating drug resistance in tumors. Moreover, various extracts of Panax notoginseng, including Rg3, Rh2, Rg5, and saponin compounds, have been found to inhibit tumor proliferation. However, further exploration and verification are required to determine which specific component of Panax notoginseng was responsible for the observed effects in this experiment. The fingerprinting of the Panax notoginseng preparation (Supplementary Materials) was consistent with previous studies. 21
A fingerprinting and network pharmacology study of Panax notoginseng showed that AKT is among the possible targets of Panax notoginseng. 22 Abnormal activation or overexpression of AKT has been observed in various cancers, including ovarian, lung, and pancreatic cancer, and is associated with increased cancer cell proliferation and survival. Consequently, targeting AKT has emerged as a potential pathway for preventing and treating cancer.20,23 In recent years, numerous studies have demonstrated that various effective components of Panax notoginseng can exhibit antitumor activity by inhibiting the AKT signaling pathway. For instance, Panax notoginseng saponin R1 has been shown to slow down the progression of breast cancer by targeting the YBX3/PI3 K/Akt axis. 24 Similarly, Panax notoginseng Rh4 has been found to inhibit AKT in esophageal cancer by targeting, indicating a potent anti-esophageal cancer effect. 25 In this study, it was found that Panax notoginseng can enhance the inhibitory effect of DDP on the Akt signaling pathway in NSCLC cells. Caspase-3 is a key protease related to the process of apoptosis, and the expression level of cleaved caspase-3 can reflect the level of cell apoptosis. 26 In this study, it was observed that the combination treatment of Panax notoginseng and DDP could promote apoptosis in A549/DDP cells.
Changes in energy metabolism not only provide cancer cells with ATP for cellular energy but also generate important metabolic intermediates that support macromolecular biosynthesis and play a crucial role in promoting cell proliferation, invasiveness, and resistance to chemotherapy. As a result, targeting glycolysis has emerged as a potential treatment strategy for cancer.27,28 Furthermore, aerobic glycolysis has been implicated to exert an essential effect in the resistance of lung cancer cells to chemotherapeutic drugs. 29 This experiment demonstrated that Panax notoginseng treatment could reduce aerobic glycolysis in DDP-resistant lung cancer cells. Moreover, some studies have shown that drug-resistant cells have greater glucose uptake. For example, DDP-resistant ovarian cancer cells have an increased need for glucose and are more sensitive to glucose deprivation. Our study found that Panax notoginseng-treated A549/DDP cells could reduce glucose uptake. Additionally, we found a decrease in the protein expression of GLUT1, a glucose transport mediator, as well as key rate-limiting enzymes for glycolysis, such as HK II. Meanwhile, Panax notoginseng Rh4 has been demonstrated to inhibit aerobic glycolysis in esophageal cancer cells by targeting AKT. 25 Consistent with previous studies, our findings indicate that Panax notoginseng treatment can reduce aerobic glycolysis in cisplatin-resistant lung cancer cells by inhibiting the AKT pathway.
This study presents novel findings that demonstrate the inhibitory effect of Panax notoginseng on the expression of TRIM46. TRIM46 is closely associated with glucose metabolism and has been implicated in promoting inflammation in human retinal capillary endothelial cells induced by high glucose through various mechanisms.30,31 Moreover, TRIM46 has been shown to enhance glycolysis, thereby promoting cell proliferation and resistance to DDP in LUAD. 25 Consistent with prior research, our study also revealed that TRIM46 can counteract the inhibition of glycolysis caused by DDP treatment. However, this effect was effectively reversed by the administration of Panax notoginseng.
This study has limitations. It was a study in cells and animals, lacking translational evidence in humans. Only DDP resistance was investigated, while NSCLC can develop resistance to several drugs. Although some mechanisms were investigated, the investigations remain superficial and do not provide a complete picture of what actually occurs.
To summarize, our study demonstrates that Panax notoginseng can enhance the sensitivity of NSCLC cells to DDP both in vitro and in vivo. This effect is attributed to the inhibition of TRIM46 and AKT signaling pathways. These findings provide novel molecular insights and potential therapeutic strategies for the clinical management of NSCLC. Future studies should characterize the exact pathways responsible for the effects of Panax notoginseng on DDP resistance. Since Panax notoginseng is already widely used in humans for a number of indications, a trial of Panax notoginseng in patients with NSCLC treated with DDP could be undertaken.
Supplemental Material
sj-docx-1-cbm-10.1177_18758592241303377 - Supplemental material for Panax notoginseng improves the sensitivity of non-small cell lung cancer to cisplatin by inhibiting Akt signaling
Supplemental material, sj-docx-1-cbm-10.1177_18758592241303377 for Panax notoginseng improves the sensitivity of non-small cell lung cancer to cisplatin by inhibiting Akt signaling by Lizhen Pan, Dandan Zhang, Qiqi Shao, Maohao Cheng, Zhicheng Liao, Lingpei Yu, Yuanyuan Wang, Pengcheng Jia and Jizhou Zhang in Cancer Biomarkers
Footnotes
Ethical statement
Not applicable.
Author contributions
Lizhen Pan, Dandan Zhang, and Qiqi Shao carried out the studies, participated in collecting data, and drafted the manuscript. Maohao Cheng, Lingpei Yu, and Jizhou Zhang performed the statistical analysis and participated in its design. Yuanyuan wang, Pengcheng Jia, and Zhicheng Liao participated in the analysis or interpretation of data and drafted the manuscript. All authors read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Natural Science Foundation of Zhejiang Province (LY20H290001) and Zhejiang Chinese Medical University Special research projects of affiliated hospitals (2022FSYYZZ29).
Declaration of conflicting interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availablity
All data generated or analyzed during this study are included in this published article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
