Abstract
Introduction
Colorectal cancer (CRC) is a common type of digestive tract tumor, with the third highest prevalence of all cancers globally, and the third highest mortality rate. 1 According to 2020 data, there are approximately 1.95 million newly diagnosed cases of CRC globally, of which more than 900,000 people have died as a result of the disease, 2 a figure that highlights the enormous threat that the disease poses to human life and health, as well as the heavy financial and emotional burdens it imposes on patients and their families. Studies have shown that long-term intake of a high-fat and low-fiber diet is strongly associated with the development of CRC. 3 In addition, the incidence of familial colonic polyposis is significantly higher in patients than in other populations. The American Cancer Society evaluated and analyzed the prognosis of patients with different stages of CRC and found that even among patients with the same pathological stage, their prognosis for recovery varied. 4 In view of this, there is an urgent clinical need to find sensitive and effective serum markers for accurate prognostic assessment and timely interventions, which are crucial for improving patients’ quality of life. It has been established 5 that the development of CRC is the result of signaling pathway activities involving multiple complexes rather than a single factor. Therefore, combining the detection of multiple biomarkers is particularly important for the early identification of CRC and the accurate assessment of its prognosis.
High mobility group protein 1 (HMGB1) is a crucial pro-inflammatory factor that sustains the body in a chronic inflammatory state. By binding to the glycosylated end product receptor, HMGB1 is able to activate multiple signaling pathways that contribute to cancer development. 6 Studies have shown 7 that HMGB1 is expressed at high levels in colon cancer (CRC) cells and tissues, which is capable of enhancing tumor proliferation, invasion and metastasis, and thus promoting tumourigenesis and progression. Therefore, HMGB1 has the potential to be a biomarker for predicting tumourigenesis and progression, which is instructive for early diagnosis of colon cancer. Amyloid A (SAA) is a stable acute time-phase reactive protein produced by hepatocytes, which usually reflects the inflammatory state of the body. Several studies have found8,9 that chronic inflammation induced by the occurrence and development of CRC leads to a sustained increase in serum inflammatory factor SAA concentration and maintains a high level. Tumor-specific growth factors (TSGF) are a class of substances secreted by tumor cells during formation and growth that are closely related to tumor growth and are relatively specific for tumors. Previous studies have shown a high degree of sensitivity and specificity, suggesting that TSGF is important in the preoperative diagnosis of malignant tumors as well as in the postoperative review of tumor recurrence. 10 However, there are fewer reports on the relationship between TSGF and the prognosis of CRC patients. In this context, this study intends to investigate the expression of serum HMGB1, SAA, and TSGF in CRC patients and the value of prognostic assessment, with a view to providing a reference for clinical diagnosis and treatment of CRC and prognostic assessment.
Materials and methods
Basic information
A total of 60 patients with colorectal cancer (CRC) were enrolled in the study between January 2018 and December 2020 in our hospital, forming the CRC group; meanwhile, another 60 patients with inflammatory bowel disease (IBD) were selected into the IBD group, as well as 60 healthy medical check-ups were categorized into the control group. The differences between the three groups in terms of basic clinical information such as age, gender, and body mass index (BMI) were not statistically significant (P > 0.05), and the detailed data are shown in Table 1.The patients participating in the study had to fulfil the following criteria: (1) all members of the CRC group were diagnosed with CRC by pathological examination and were in TNM stages I to III; (2) all patients in the IBD group complied with the established clinical diagnostic and treatment guidelines; (3) the age range was between 20 and 75 years old, had complete clinical records and follow-up data, and were cooperative; (4) the study was approved by the Medical Ethics Committee of our hospital; and (5) the participants had signed an informed consent form. Patients with the following conditions will be excluded from the study: (1) patients with major organ (e.g., heart, liver, lung, kidney, etc.) failure; (2) patients with autoimmune diseases or with other types of malignant tumors; (3) patients with coagulation dysfunction or experiencing severe infections; (4) patients who have already received anti-tumor therapy, anti-inflammatory therapy, or who have had a recent surgery; ((5) Patients with mental illness or people with abnormal behavior; (6) Patients who have failed to complete surgery or drug treatment as prescribed. See Table 1 below.
Comparison of clinical data of the three groups of subjects.

Sample collection
On the second day of the subject's admission, a 3 to 5 ml sample of fasting venous blood was drawn. The blood samples were allowed to stand for some time and then processed by centrifugation at 3000 rpm for 10 min, with a radius of 10 cm in the centrifugation operation in order to isolate the patient's serum. Subsequently, the extracted serum was stored at 20 degrees Celsius and kept aside.
Detection Methods
Serum HMGB1 and SAA levels were detected by enzyme-linked immunosorbent assay (ELISA): 5 mL of fasting venous blood was taken from the subjects, set the rotational speed at 3000 r/min, centrifuged for 15 min, and the upper layer of serum was put into EP tubes, stored in the refrigerator at −80°C for testing, the serum HMGB1 and SAA levels of the subjects were detected by ELISA in the groups, and the kits were purchased from Xiamen Huijia Bio-technology Co. The test was repeated 3 times, and the final result was taken as the average of the 3 times. The serum TSGF level was measured by the following method: 5 mL of fasting venous blood was taken from the subjects, the rotational speed was set at 3000 r/min, the centrifugation time was 15 min, and the upper layer of serum was put into the EP tube, stored in the refrigerator at −80°C and waited for the measurement, and the serum TSGF level was measured by the spectrophotometric colorimetric assay, and the reagent kit was purchased from Fujian Newland Bio-technology Co. The kit was purchased from Fujian New World Biotechnology Co.
Follow-up method
After treatment acceptance, all patients were started on follow-up. They were followed up every 3 months by telephone enquiry or outpatient review to count their survival during the 3 years after treatment, while patients who died from non-tumor-related causes were excluded.
Observation indicators
(1) Compare serum HMGB1, SAA, and TSGF levels in each group, and compare serum HMGB1, SAA, and TSGF levels in CRC patients with different clinical characteristics. (2) Follow-up for 8–36 months, group CRC patients according to prognosis (good prognosis group, bad prognosis group), and compare serum HMGB1, SAA, and TSGF levels between the two groups. (3) ROC was used to analyze the predictive value of serum HMGB1, SAA, TSGF and their combined assays on the prognosis of CRC patients.
Statistical processing
SPSS 26.0 software was used to process the data. Measurement data such as serum HMGB1, SAA, and TSGF levels were expressed as (
Results
Comparison of serum HMGB1, SAA, and TSGF levels among groups
Serum HMGB1, SAA, and TSGF levels were significantly higher in the CRC group than in the IBD group (P < 0.05), and serum HMGB1, SAA, and TSGF levels were significantly higher in the IBD group than in the control group (P < 0.05). See Table 2 and Figure 1.
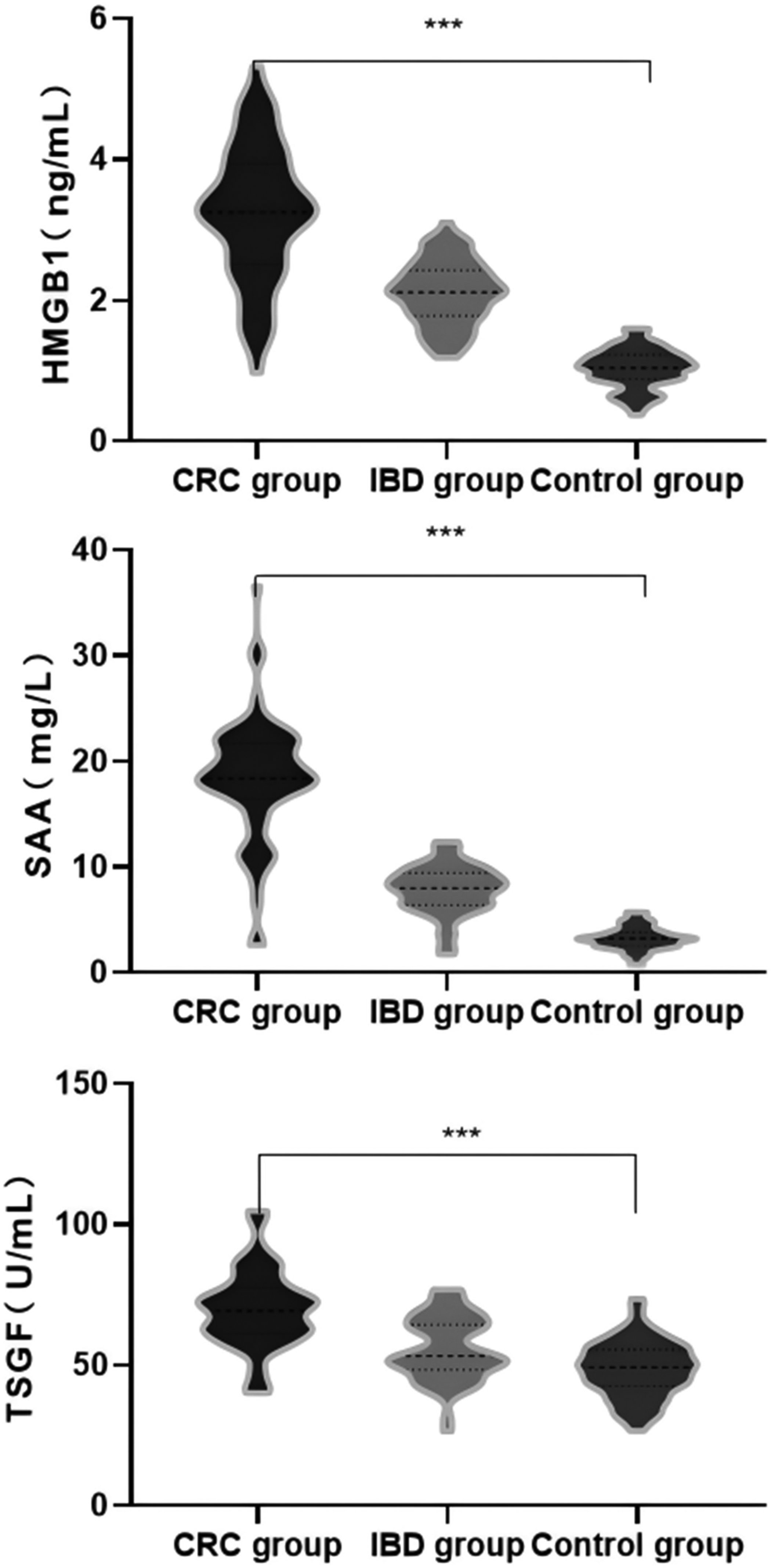
Comparison of HMGB1, SAA and TSGF across groups.
Comparison of serum HMGB1, SAA, and TSGF levels among groups (
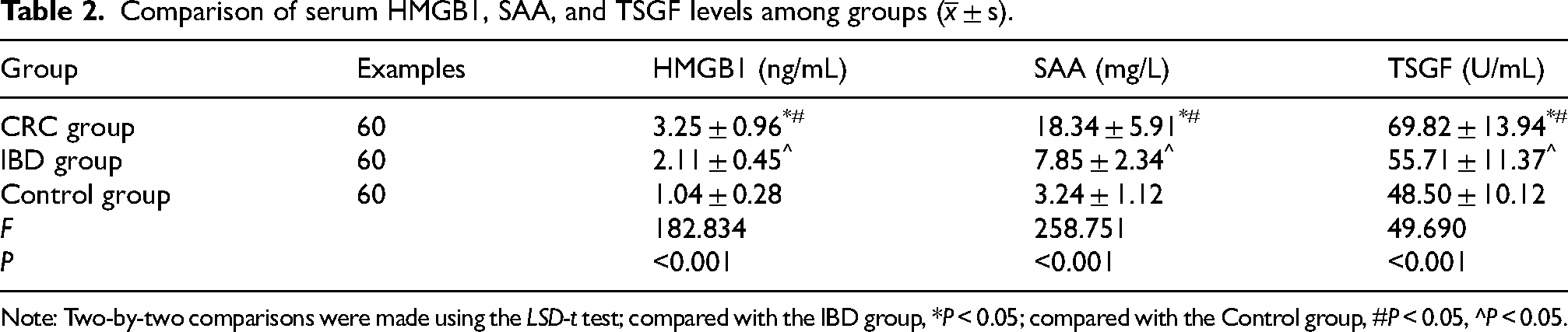
Note: Two-by-two comparisons were made using the LSD-t test; compared with the IBD group, *P < 0.05; compared with the Control group, #P < 0.05, ^P < 0.05.
Comparison of serum HMGB1, SAA, and TSGF levels in CRC patients with different clinical characteristics
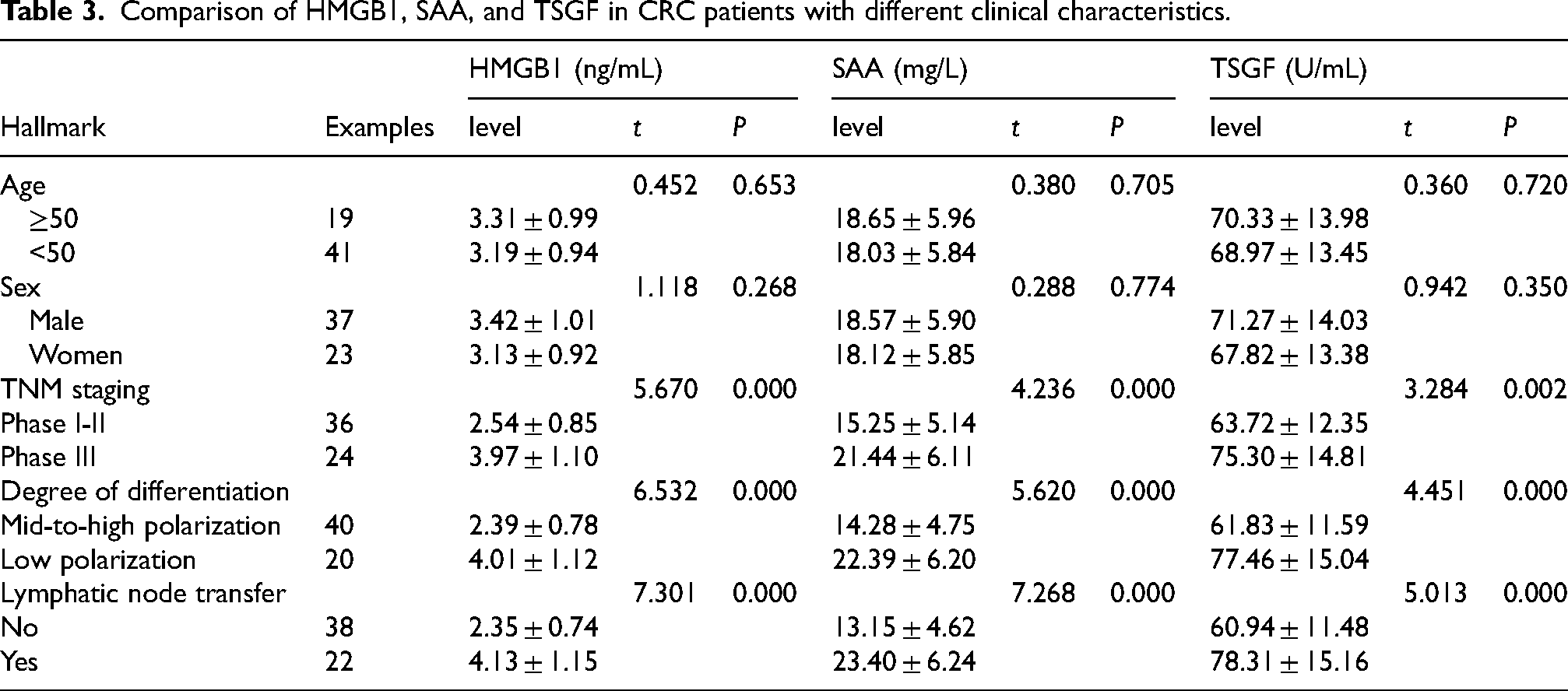
Comparison of serum HMGB1, SAA and TSGF levels in CRC patients of different ages and genders did not show statistically significant differences (P > 0.05). Serum HMGB1, SAA and TSGF levels were significantly higher in patients with TNM stage III than in patients with TNM stage I to II in the CRC group (P < 0.05), and serum HMGB1, SAA and TSGF levels were significantly higher in poorly differentiated than in middle or highly differentiated patients (P < 0.05).The levels in patients who developed lymph node metastasis were significantly higher than those in middle or highly differentiated patients (P < 0.05) were higher than those of middle and high differentiated patients (P < 0.05), Serum HMGB1, SAA, and TSGF levels were significantly higher in those who developed lymph node metastases than in patients without lymph node metastases (P < 0.05). See Table 3.
Comparison of HMGB1, SAA, and TSGF in CRC patients with different clinical characteristics.
Effect of serum HMGB1, SAA and TSGF levels on the prognosis of patients With CRC
Follow-up ranged from 8 to 36 months, and all 60 CRC patients completed the final follow-up, with a mean follow-up of (26.4 ± 6.3) months and 18 deaths. Kaplan-Meier survival curve analysis showed that the median survival of the HMGB1 high expression group (above its median of 3.25 ng/mL) was 27 months, which was lower than that of the HMGB1 low expression group (36 months) and the difference was statistically significant (χ2 = 9.185, P < 0.05); the median survival of the SAA high-expression group (above its median of 18.34 ng/mL) was 28 months, which was lower than that of the SAA-low-expression group (36 months), and the difference was statistically significant (χ2 = 7.960, P < 0.05); the median survival of the TSGF high-expression group (above its median of 69.82 U/mL) had a median survival of 28 months, which was lower than that of the low TSGF expression group (37 months), and the difference was statistically significant (χ2= 5.894, P < 0.05). See Figure 2.
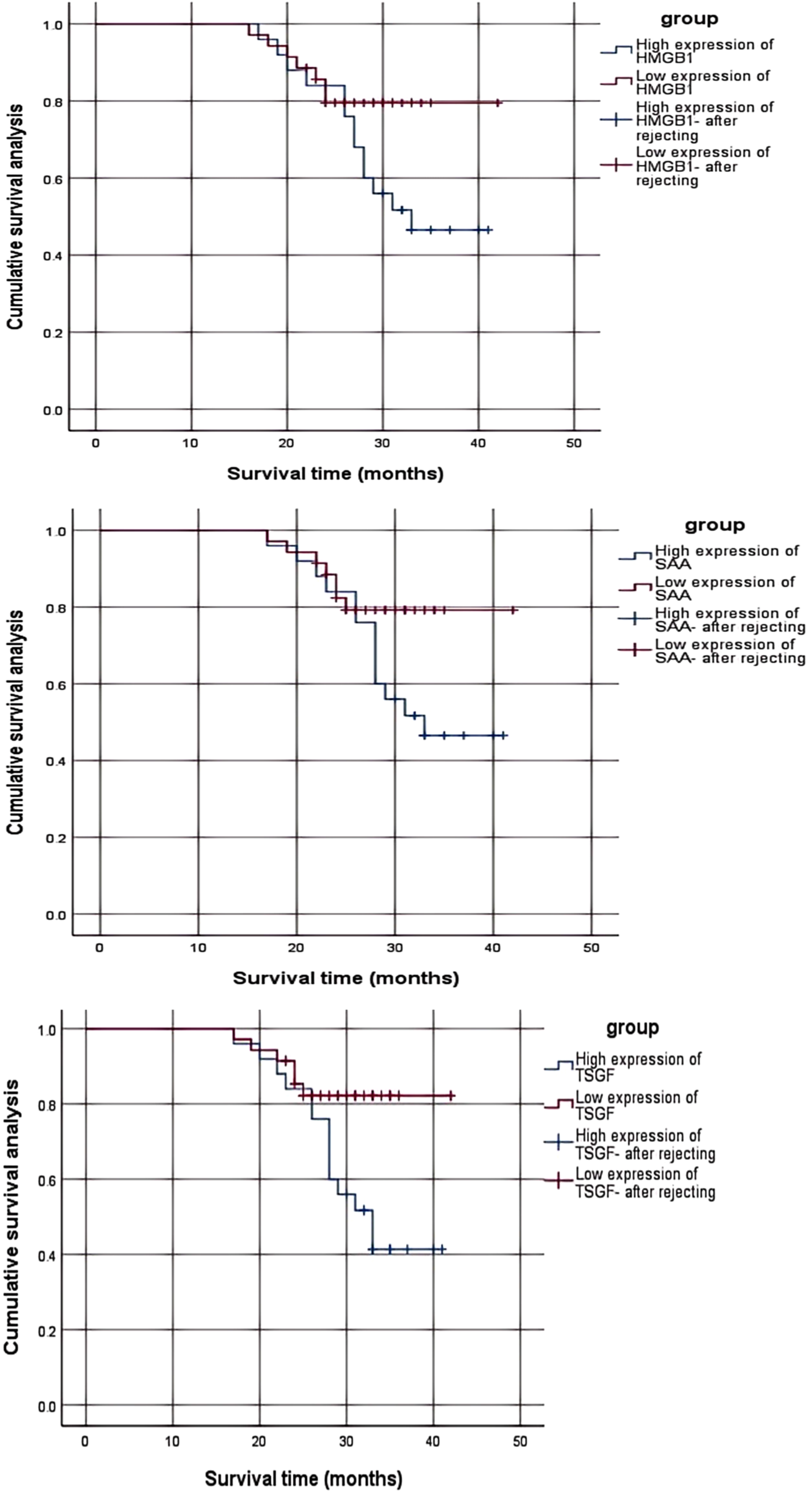
Survival curves for different serum HMGB1, SAA and TSGF expression levels.
Univariate and multifactorial Cox regression analyses of prognostic factors in patients with CRC
The results of univariate analysis showed that TNM stage III, low tumor differentiation, lymph node metastasis, and high expression of HMGB1, SAA, and TSGF were all factors affecting the prognosis of CRC patients (P < 0.05), and after further correcting for the effects of confounding factors such as pathological stage, degree of tumor differentiation, and lymph node metastasis by Cox regression analysis, the high expression of HMGB1, SAA, and TSGF remained significantly associated with the CRC patients’ prognosis significantly (P < 0.05), see Table 4.
Multifactorial Cox regression analysis affecting the prognosis of CRC patients.
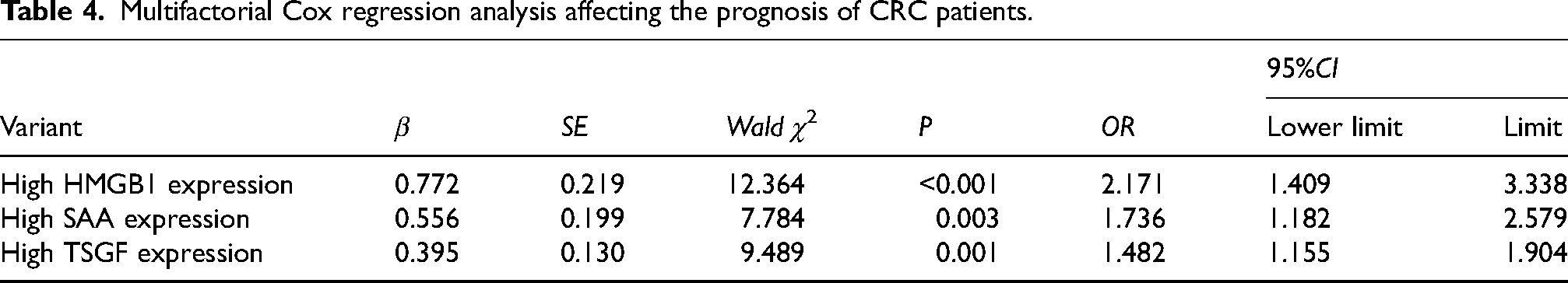
The value of serum HMGB1, SAA, and TSGF in predicting prognosis in patients with CRC
The ROC curve showed that the area under the curve (AUC) of serum HMGB1, SAA, and TSGF for predicting prognosis in CRC patients was 0.790, 0.774, and 0.733, respectively, and the AUC of the combined assay for predicting prognosis in CRC patients was 0.904, which was higher than that of the assay alone (Z = 2.536, 2.420, and 2.218, P = 0.013, 0.020, 0.031). According to the optimal critical value (the optimal critical value is at the maximum value of the Jordon index), when HMGB1 was higher than 3.50 ng/mL, the sensitivity was 76.12% and the specificity was 83.45%; when SAA was higher than 21.90 mg/L, the sensitivity was 62.71% and the specificity was 90.69%; when TSGF was higher than 74.44 U/mL, the sensitivity was 57.35% and specificity was 75.64%. See Table 5 and Figure 3.
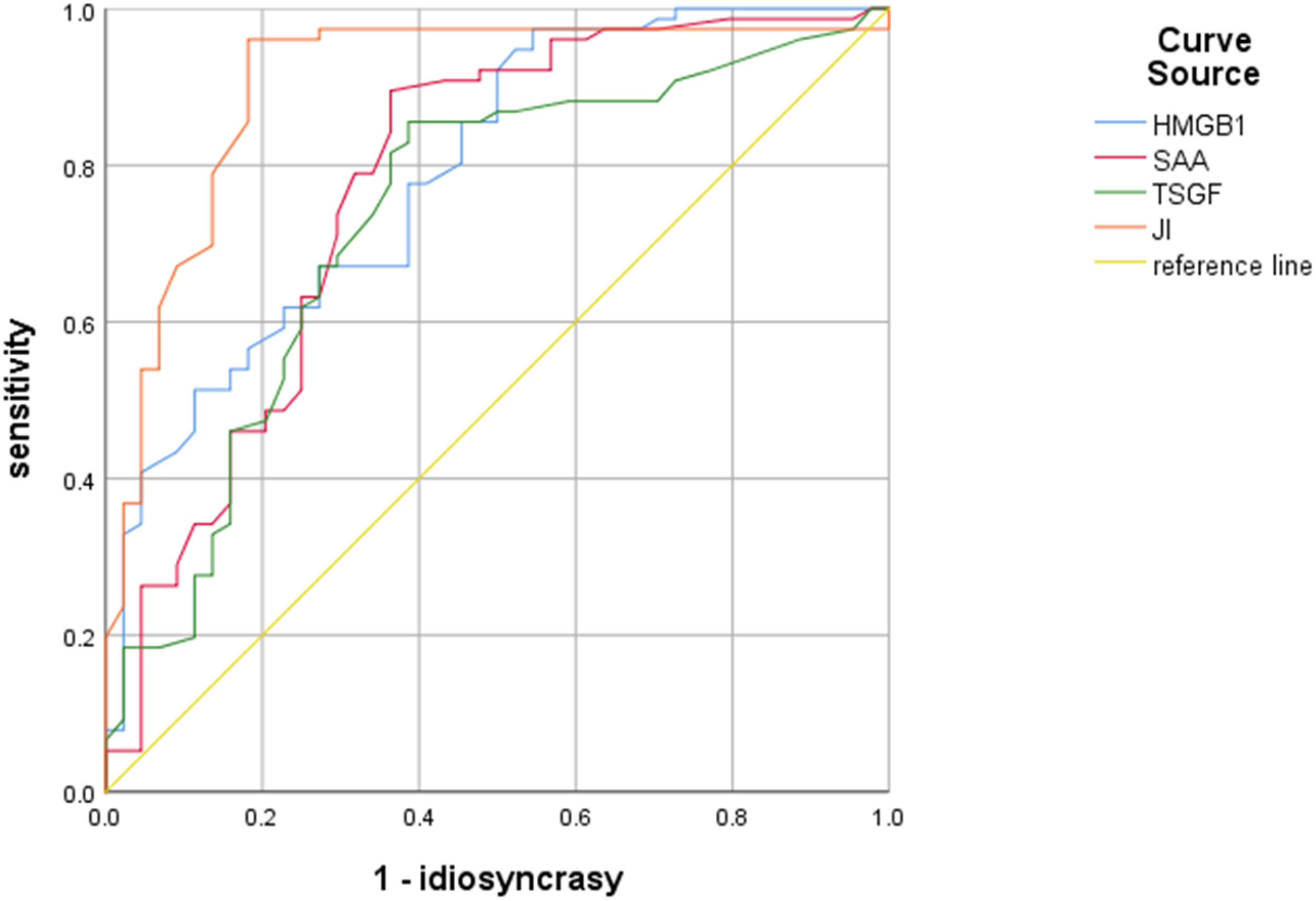
ROC curves of serum HMGB1, SAA, and TSGF for predicting prognosis in CRC patients.
Value of serum HMGB1, SAA, and TSGF in predicting prognosis in patients with CRC.

AUC is the area under the ROC curve and is used to assess the accuracy of a diagnostic test or prediction; the higher the AUC value, the more accurate the test or prediction. An AUC between 0.5 and 0.7 indicates low diagnostic value, between 0.7 and 0.9 indicates moderate diagnostic value, and above 0.9 indicates high diagnostic value.
Discussion
The development of colorectal cancer (CRC) has been associated with a number of factors, including prolonged sedentary lifestyle, overweight or obesity, and continued consumption of processed and unprocessed red meat. Being physically active and consuming nutrients such as fish, plant foods, folic acid, vitamin D and calcium can help reduce the risk of developing this disease. 11 Colorectal cancer is a malignant disease that occurs in the epithelial cells of the colonic mucosa. Due to the abundant blood supply of the intestinal tract and good lymphatic return, coupled with the fact that colorectal cancer cells are prone to remote metastasis through the blood and lymphatic circuits, by the time most patients with colorectal cancer are diagnosed, the optimal time to treat the disease has already been missed, resulting in its poor outcome. 12 Therefore, the search for biomarkers that can assess the condition of colorectal cancer patients at an early stage and predict their prognosis is of great significance for the development of appropriate treatment plans and the improvement of patient survival.
HMGB1 is a member of the high mobility group (HMG) of proteins that bind non-specifically to the narrow grooves in the double helix structure of DNA, thereby playing a role in the structure and transcription of DNA. The intranuclear localization of this protein and its affinity for DNA are regulated by phosphorylation and acetylation. HMGB1 plays a key role in the transcription of numerous tumor-associated genes. The C-terminal region of HMGB1, known as the C-box, is a key site for its interaction with the receptor for the end products of late glycosylation, a process that has an impact on the migratory capacity of cancer cells. When tumor cells undergo necrosis, they release HMGB1 into the surrounding microenvironment, which induces an inflammatory response and repair mechanisms, which in turn promotes tumor cell survival, growth and spread. 13 The expression level of HMGB1 is high in many types of tumor tissues and correlates with the TNM stage of the tumor, thus it can be used as an independent indicator to predict poor prognosis in tumor patients. 14 Studies have shown 15 that HMGB1 plays an important role in the transmission of a variety of tumors during their genesis, growth and invasion. Especially in colorectal cancer (CRC) patients, it has been demonstrated that there is overexpression of HMGB1 protein. Lee et al 16 human study further confirmed that HMGB1 is expressed in the nucleus of both normal and tumor cells, but the expression level of HMGB1 in tumor cells is significantly higher than that of normal cells, which suggests that, in colorectal cancer tissues, the HMGB1 expression position is related to the infiltration degree of T lymphocytes. Therefore, the intracellular expression position of HMGB1 can be used as a prognostic marker for patients with advanced CRC. 17
There is a strong link between tumourigenesis and inflammation, with the latter contributing to the formation and progression of the former. Sustained chronic inflammatory stimuli may lead to cellular hyperplasia and activation of cellular cascade reactions, which in turn cause permanent DNA damage.SAA, a marker that can reflect the level of inflammation in the serum, may drive the disease towards a metastatic state by contributing to the abnormal cellular hyperplasia and causing damage to the cellular DNA, which ultimately triggers the metastasis and invasion of malignant tumours. Some studies have pointed out that SAA is closely associated with the occurrence, 18 development and metastatic deterioration of colorectal cancer, and has a certain reference value for the diagnosis of colorectal cancer at different stages, but also pointed out that it has a low sensitivity and specificity, and is susceptible to the influence of the body's inflammatory response. Regarding the relationship between SAA and colorectal cancer, a study by Wu Yuchen et al 19 showed that inflammatory factors CRP and SAA combined with CEA, CA242, and CA50 could improve the diagnostic efficiency of colorectal cancer, especially in determining the diagnostic significance of stage IV colorectal cancer accompanied by distant metastases. Literature reports indicate that the level of inflammatory mediator SAA is elevated in patients with various cancers, including rectal cancer, lung cancer, and renal cell carcinoma, among which SAA demonstrates high specificity and sensitivity in colorectal cancer detection. 20 This suggests that SAA serves as an inflammatory indicator during the formation and development of colon cancer and helps to assess the degree of inflammatory response to the disease.TSGF is an internationally recognised substance associated with malignant tumours, and there have been several reports confirming that TSGF exhibits high levels of expression in a wide range of malignant tumours, including lung, liver, and colorectal cancers.TSGF is capable of stimulating malignant tumour angiogenesis, promoting the growth of malignant tumours and the massive proliferation of their surrounding capillaries and release into the peripheral blood, a process that has no significant effect on the proliferation of non-tumour blood vessels.
In this study, we found that serum HMGB1, SAA, and TSGF levels were all significantly higher in CRC patients compared with those in IBD patients and the control population, and serum HMGB1, SAA, and TSGF levels were significantly higher in patients with TNM staging stage III, poorly differentiated patients, and those who developed lymph node metastasis in the CRC group, which is basically in line with previous reports.21,22 This conclusion suggests that serum HMGB1, SAA and TSGF are all highly expressed in CRC and play an important role in the occurrence, infiltration and metastasis of CRC, i.e., they are closely related to the biological behaviors of tumor cells such as migration and differentiation. The reasons for this may lie in the following: (1) HMGB1 is produced by activated immune cells in the serum of CRC patients, and the necrosis of tumor cells leads to their release of HMGB1 when the body tissues are injured; in addition, tumor cells or immune cells may also actively secrete HMGB1. (2) Serum amyloid A (SAA) is produced by the liver under the stimulation of inflammatory factors, such as interleukin-6 or interleukin-1, and by the liver. SAA is a protein produced by the liver under the stimulation of inflammatory factors such as interleukin-6 or interleukin-1, which is closely related to tumor formation, disease progression and inflammatory response, and therefore, its level increases significantly after CRC development. (3) After the occurrence of CRC, the levels of a variety of tumor-specific growth factors (TSGF) related to the formation and growth of CRC rise, which promotes the generation of CRC blood vessels and peripheral micro vessels, which enables TSGF to enter the blood circulation along with the blood vessels, leading to an increase in the level of TSGF in the serum; at the same time, with the continuous generation of CRC blood vessels and peripheral capillaries, they can gradually penetrate the mucosal layer and metastasize along lymphatic vessels and blood vessels, which ultimately led to the fact that patients with invasion of the plasma membrane layer, lymph node metastasis and hepatic metastasis had higher TSGF levels than patients without invasion of the plasma membrane layer, lymph node metastasis and hepatic metastasis.
In this study, we further found that serum HMGB1, SAA, and TSGF prediction all have high application value in assessing the prognosis of CRC patients, in which the sensitivity of HMGB1 in predicting the prognosis of CRC patients is higher than that of SAA and TSGF, and the specificity of SAA in predicting the prognosis of CRC patients is higher than that of HMGB1 and TSGF, but there is no significant difference in comparing all three of them. The analysis of the mechanism may lie in (1) HMGB1 is one of the key factors regulating cellular autophagy, which can lead to tumor chemotherapy resistance by regulating cellular autophagy, 23 which in turn affects the effectiveness of CRC treatment and leads to poor prognosis of patients. (2) Inflammatory response plays an important role in the development of CRC, and SAA is a protein synthesized by the liver stimulated by inflammatory factors such as interleukin-6 (IL-6) or interleukin-1 (IL-1), so detecting its level can reflect the progression of CRC disease to a certain extent, 24 and play a role in the assessment of CRC prognosis. (3) TSGF is a collective term for glycosides and metabolites associated with malignant tumor growth. TSGF can be measured in the early stage of malignant tumor formation, and it only plays an important role in malignant tumor vasculopoiesis, while it has no significant effect on non-tumor vasculopoiesis, so detection of its level can be used to assess the condition and prognosis of CRC patients. When serum HMGB1, SAA, and TSGF levels are higher than 3.50 ng/mL, 21.90 mg/L, and 74.44 U/mL, respectively, the risk of poor prognosis is high, so these patients should be alerted to the regression of the disease, and the survival of patients should be improved through the development of an active and effective treatment plan. In this study, we found that the AUC of the combined assay for predicting the prognosis of CRC patients was 0.904, which was higher than that of any of the individual assays, suggesting that the combination of HMGB1, SAA, and TSGF assays has a higher efficacy in predicting the poor prognosis of CRC patients than the individual assays, suggesting that the combination of the three assays has a certain degree of complementarity in predicting the poor prognosis of CRC patients, which is useful for evaluating the prognosis of CRC patients, formulating relevant interventions, and improving the survival of patients with CRC. This is of great significance in assessing the prognosis of CRC patients, formulating relevant interventions and improving the prognosis of CRC patients. Inflammation plays an important role in the development of CRC, HMGB1 is a pro-inflammatory factor, and SAA can reflect the inflammatory state of the human body. TSGF is a collective term for glycosides and metabolites related to the growth of malignant tumors, which plays an important role in the vascular proliferation of malignant tumors, but has no significant effect on the proliferation of non-tumorous blood vessels, and has a relatively specific effect on tumors. 24 Therefore, it is important to analyze the above indicators in the prediction of the prognosis of CRC patients. Therefore, analyzing the role of the above indicators in predicting the prognosis of CRC patients is feasible and complementary, and in clinical practice, it can play a role in evaluating the condition and prognosis of CRC patients through the combined detection of the above indicators.
Conclusion
Serum HMGB1, SAA, and TSGF expression was significantly higher in colon cancer patients than in patients with inflammatory bowel disease and healthy populations, and was associated with TNM stage, degree of differentiation, and the presence or absence of lymph node metastasis. Detection of serum HMGB1, SAA, and TSGF expression levels can help to assess the prognosis of colon cancer patients. However, this study only revealed the expression status of serum HMGB1, SAA, and TSGF in colon cancer patients and their relationship with prognosis, in which the specific mechanism of action still needs to be further explored, with a view to providing new ideas and directions for the treatment of this disease in the future. In addition, given that this study is a retrospective study, it may not be possible to control for confounding factors affecting the results of the study; the patients were only admitted to our hospital and health checkups, and it is a single-center study with authentic information, but there may be regional differences. In addition, the number of samples included in this study was limited, and the sample size should be increased to conduct randomized multi-center controlled studies with other hospitals in order to obtain more generalizable findings.
Footnotes
Author contribution
First Author: Kaifeng Wang
Data analysis and Interpretation Conception
Corresponding Author: Junxing Huang
Revision for all important intellectual content of the manuscript Supervision of research and writing process.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used in this research study will be made available upon request.
