Abstract
Objective
Increasing evidence suggests that circular RNAs (circRNAs) play a major role in tumorigenesis and cancer progression. This study aimed to identify aberrant expression of hsa_circ_0003829 in oral squamous cell carcinoma (OSCC) and to explore its clinical significance.
Methods
We conducted a prospective clinical study to examine the expression pattern of hsa_circ_0003829 in 60 paired OSCC and normal clinical samples and in cell lines using real-time quantitative polymerase chain reaction. We also evaluated the diagnostic value of hsa_circ_0003829 in OSCC based on receiver operating characteristic (ROC) curve analysis, and examined the relationships between hsa_circ_0003829 expression and clinicopathological features in patients with OSCC. We further used bioinformatics software CircInteractome (https: //Circinteractome.nia.nih.gov/) to predict circRNA–microRNA interactions.
Results
Hsa_circ_0003829 was significantly downregulated in OSCC compared with adjacent normal tissues. The area under the ROC curve was 0.81. Low expression levels of hsa_circ_0003829 in OSCC tissues were negatively correlated with lymph node metastasis status and TNM stage.
Conclusions
Downregulated expression of has_circ_0003829 suggests that this may be a key circRNA in OSCC, and may serve as a prospective biomarker for the diagnosis of OSCC.
Keywords
Introduction
Oral squamous cell carcinoma (OSCC) accounts for >90% of cases of oral cancer, and is the sixth most common malignancy worldwide. 1 Despite recent advancements in cancer diagnosis and clinical therapy, including surgery, chemotherapy, radiation therapy, and biologic targeted therapy, the 5-year survival rate of patients with OSCC remains approximately 60%. 2 In addition, patients are frequently diagnosed at an advanced stage, leading to a worse prognosis. There is thus an urgent need to identify new biomarkers and explore molecular-targeted therapies to improve the prognosis of patients with OSCC.
Circular RNAs (circRNAs) are a unique type of endogenous non-coding RNA. Unlike linear RNAs, circRNAs form a covalently closed cyclic structure by back-splicing, and thus lack 5′ caps and 3′ poly A tails. 3 CircRNAs are characterized by a stable and conserved structure that enables them to resist degradation by RNA exonucleases. 4 CircRNAs were previously thought to be functionless by-products of misspliced RNAs; however, additional functional circRNAs have recently been identified by high-throughput sequencing and bioinformatics analysis, 5 and have been shown to function as micro RNA (miRNA) or protein sponges and to regulate splicing and transcription, thus playing a critical role in the pathophysiology of human diseases. 6 , 7 Emerging evidence has revealed that circRNAs participate in tumorigenesis and cancer progression, such as in esophageal squamous cell carcinoma (ESCC), gastric cancer, colorectal cancer, and hepatocellular carcinoma.8–11 The properties of circRNAs make them potentially ideal cancer biomarkers. However, the role of circRNAs in the development of OSCC is largely unknown.
We previously conducted high-throughput sequencing of eight pairs of OSCC and adjacent normal tissues to screen differentially expressed circRNAs. 12 In the current study, we selected hsa_circ_0003829 from the resulting database of differentially expressed circRNAs and analyzed its expression in OSCC by real-time quantitative polymerase chain reaction (qRT-PCR). We also generated a receiver operating characteristic (ROC) curve to assess the diagnostic value of hsa_circ_0003829 in OSCC, and examined the correlations between hsa_circ_0003829 expression in OSCC tissues and clinicopathological factors. We aimed to determine if hsa_circ_0003829 could be used as a molecular biomarker for the diagnosis of OSCC.
Materials and methods
Clinical specimens
We conducted a prospective clinical study based on OSCC tissues and adjacent normal tissues obtained from patients with OSCC undergoing surgery at the Department of Oral and Maxillofacial Surgery, Peking University Shenzhen Hospital, from May 2018 to May 2020. No patients received chemotherapy or radiotherapy before surgery. All samples were confirmed by experienced clinicians, and were stored at −80°C immediately after collection. This study was approved by Ethics Committee of Peking University Health Science Center (IRB00001053 08043), and each patient signed an informed consent form.
Cell culture
Human oral keratinocyte (HOK) cells and OSCC cell lines (SCC25, SCC15, and CAL27) were cultured in Dulbecco’s modified Eagle medium (Gibco, Grand Island, NY, USA) containing 10% fetal bovine serum (Gibco). HOK cells were obtained from the Chinese Academy of Sciences Cell Resource Center (Shanghai, China). SCC25, SCC15, and CAL27 cells were obtained from the College of Stomatology, Wuhan University (Wuhan, China). Cells were maintained at 37°C with 5% CO2 in a humidified chamber.
RNA extraction and qRT‐PCR
Total RNA was extracted using TRIzol reagent (Takara, Japan) according to the manufacturer’s protocol. cDNA was synthesized using PrimeScript™ RT Reagent Kit (Takara, Japan) by reverse transcription and qRT-PCR was then performed using SYBR® Premix Ex Taq (Takara) with a LightCycler® 96 System (Roche, Switzerland). All procedures were carried out according to the manufacturer’s protocols. The relative expression levels of genes were calculated using the 2−ΔΔCt method and normalized to β-actin. The primer sequences were: hsa_circ_0003829 forward: 5′-CCATAAATTGAAGCAAACTTTAAG-3′, reverse: 5′-CATTGTGGTTTATGTGTGAATCT-3′; β-actin forward: 5′-AAACTGGAACGGTGAAGGTG-3′, reverse: 5′-AGTGGGGTGGCTTTTAGGAT-3′.
Statistical analysis
Data analyses were performed using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA), and figures were created using GraphPad Prism 7.0 (GraphPad Software Inc., San Diego, CA, USA). Continuous data were described as mean ± standard deviation (experimental data) or mean ± standard error (clinical data). Differences between groups were compared by Student’s t-tests. P < 0.05 was considered statistically significant.
Results
Patients
Sixty pairs of OSCC and matched adjacent normal tissues were obtained. The clinical characteristics of the patients are presented in Table 1.
Correlation between hsa_circ_0003829 expression and clinicopathological characteristics in 60 patients with oral squamous cell carcinoma.

SEM, standard error.
Hsa_circ_0003829 was downregulated in OSCC tissues and cell lines
Hsa_circ_0003829 is a transcription product of the CASC4 gene located at chromosome 15: 44671887-44673174. It includes two exons and has a spliced sequence length of 270 bp (Figure 1). We detected the relative expression levels of hsa_circ_0003829 in 60 paired tumor and adjacent normal tissues and in OSCC and normal cell lines by qRT-PCR. Hsa_circ_0003829 expression was significantly downregulated in OSCC clinical samples (n = 60, P < 0.001) (Figure 2a), and in SCC15, SCC25, and CAL27 cell lines compared with HOK cells (all P < 0.01) (Figure 2b).
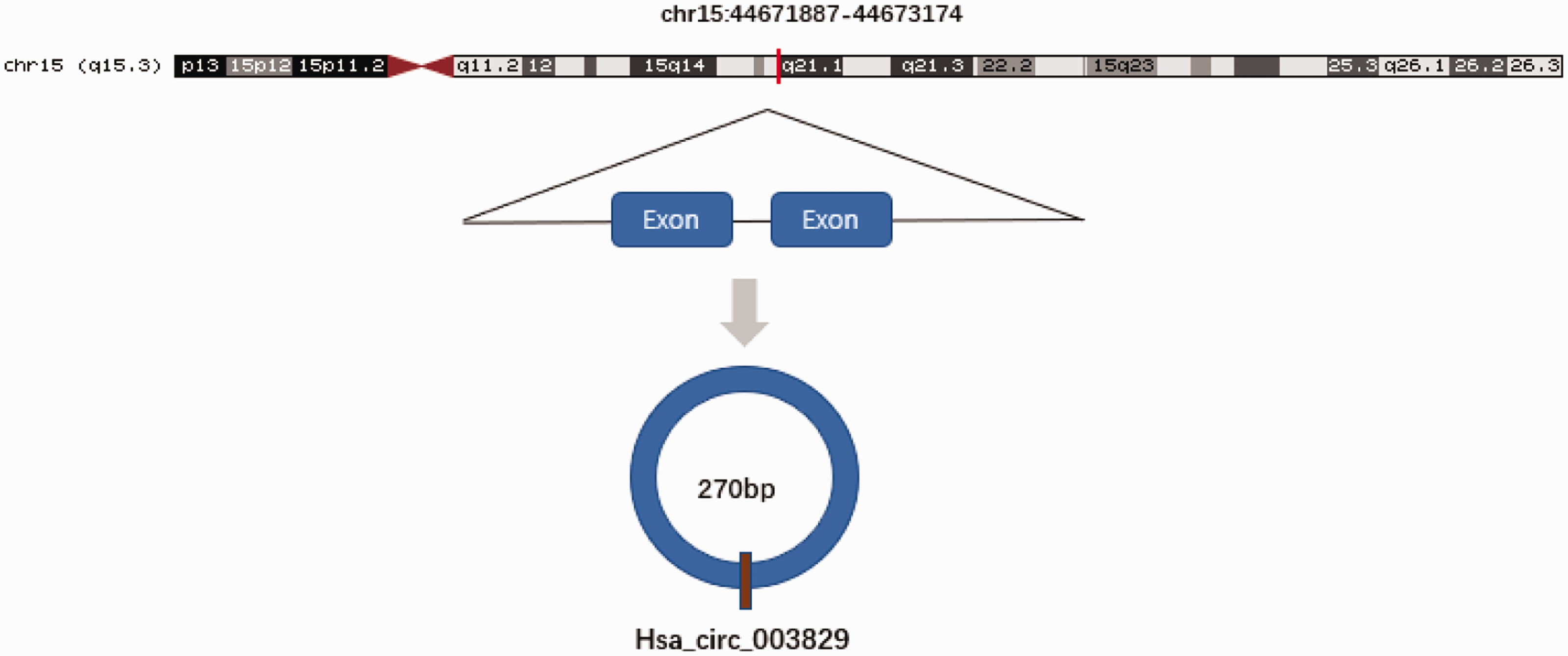
Hsa_circ_0003829 is encoded by chromosomal region 15q3, comprising two exons.
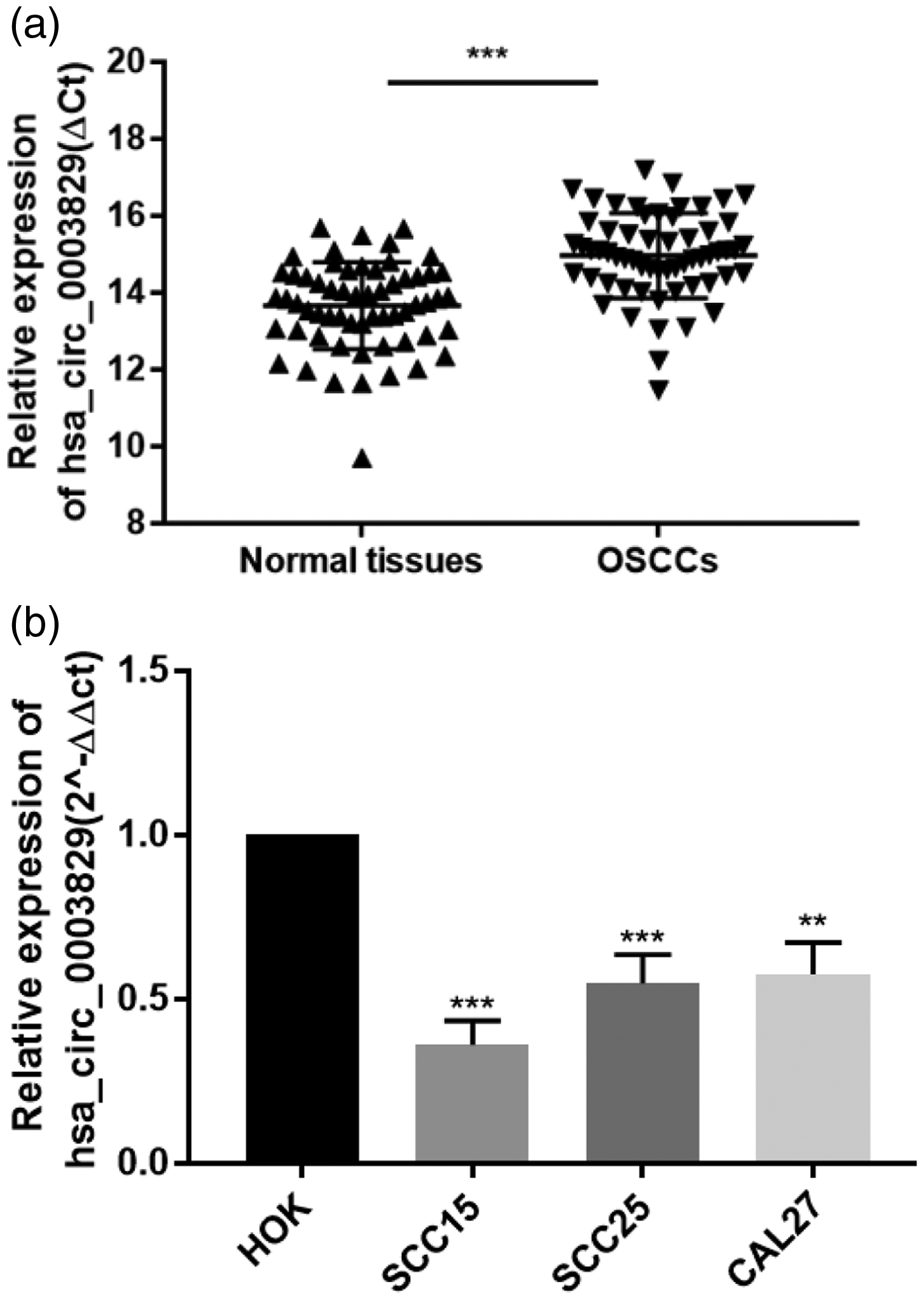
Expression of hsa_circ_0003829 in clinical specimens and cell lines. Hsa_circ_0003829 levels (a) in 60 paired oral squamous cell carcinoma and adjacent normal tissues, and (b) in cell lines, using real-time quantitative polymerase chain reaction. OSCC, oral squamous cell carcinoma. **P < 0.01; ***P < 0.001.
Correlations between hsa_circ_0003829 expression levels and clinicopathological factors
We analyzed the relationships between the expression levels of hsa_circ_0003829 and various clinicopathological factors in patients with OSCC. Hsa_circ_0003829 expression levels were significantly negatively related to lymphatic metastasis (P = 0.036) and TNM stage (P < 0.001) (Table 1, Figure 3), but were not associated with other clinicopathological features, such as sex, age, or tumor differentiation.
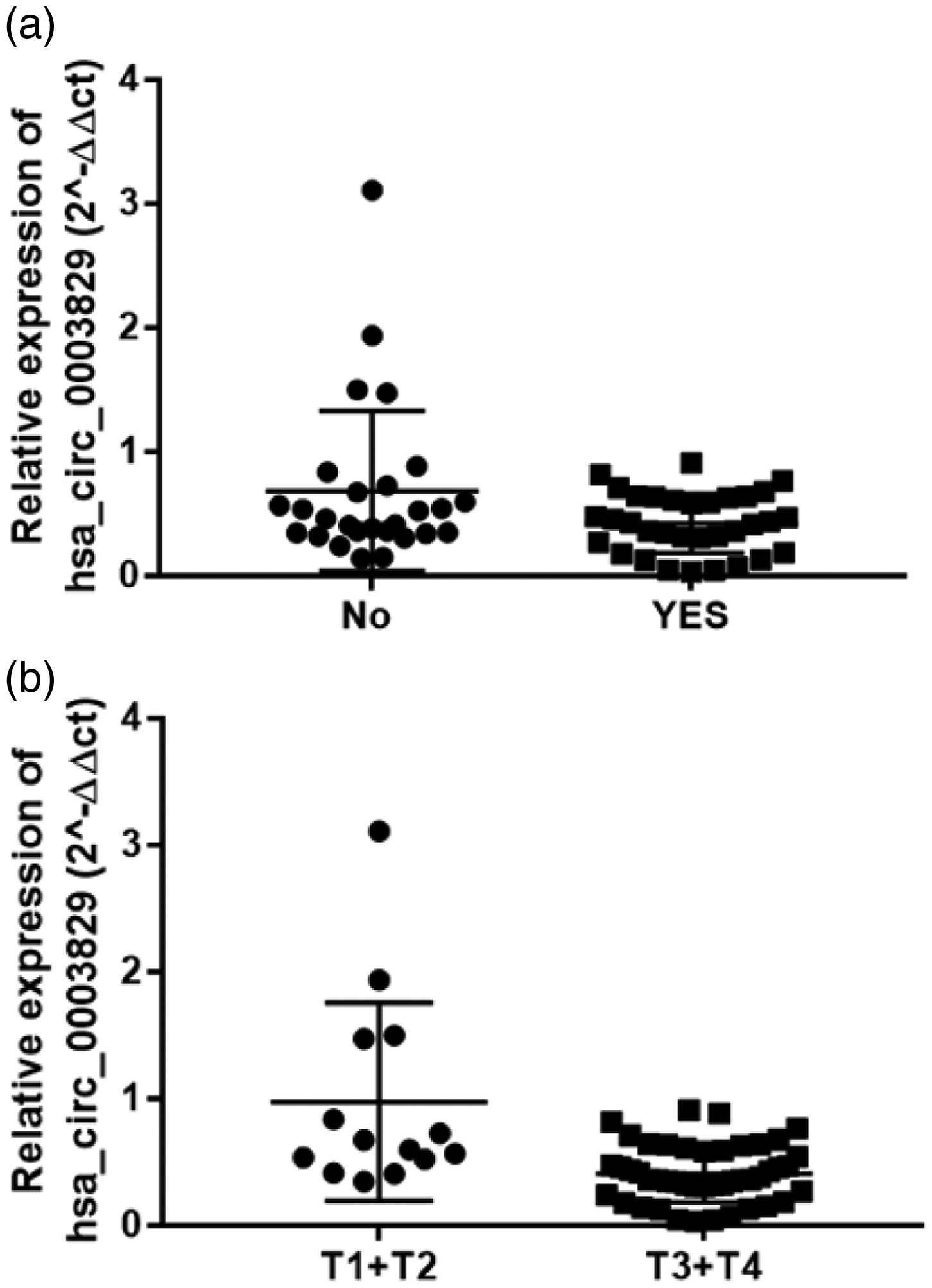
Correlation between hsa_circ_0003829 expression and clinicopathological parameters in 60 paired oral squamous cell carcinoma samples. Low expression of hsa_circ_0003829 was significantly associated with (a) lymph node metastasis and (b) advanced TNM stage.
Potential diagnostic value of hsa_circ_0003829
An ROC curve was created and analyzed to evaluate the potential clinical diagnostic value of hsa_circ_0003829 in patients with OSCC (Figure 4a). The area under the curve was 0.81, and the sensitivity and specificity of hsa_circ_0003829 were 70% and 80%, respectively (Figure 4b). These data suggested that hsa_circ_0003829 might serve as a potential biomarker for the diagnosis of OSCC.

Diagnostic value of hsa_circ_0003829 in oral squamous cell carcinoma. (a) The area under the curve was 0.81. (b) The cutoff of hsa_circ_0003829 was 14.57. The sensitivity and specificity were 0.70 and 0.80, respectively. ***P < 0.001. OSCC, oral squamous cell carcinoma; Sens, sensitivity; Spec, specificity.
Prediction of circRNA–miRNA interactions
We predicted potential target miRNAs of hsa_circ_0003829 using the Circular RNA Interactome database (https:// Circinteractome.nia.nih.gov/). Eight miRNAs were predicted to pair with hsa_circ_0003829: hsa-miR-1208, hsa-miR-1257, hsa-miR-145, hsa-miR-198, hsa-miR-516b, hsa-miR-580, hsa-miR-587, and hsa-miR-7 (Table 2).
Eight miRNAs predicted to bind with hsa_circRNA_0003829.

Discussion
OSCC is the most common malignant tumor in the head and neck region. 13 It is highly malignant and prone to lymph node metastasis, resulting in a poor prognosis and dismal survival rates. 14 Consequently, many studies have investigated the biological mechanisms underlying tumorigenesis and progression of OSCC, with the aim of discovering novel tumor biomarkers to facilitate an early diagnosis and thus improve prognosis.
Numerous circRNAs have recently been found to be aberrantly expressed in tumors, and might play crucial roles in biological processes. CircRNAs are widely expressed in eukaryotes and are remarkably stable owing to their closed loop structure, 15 making them promising novel biomarkers for cancer diagnosis. A recent study 16 revealed that the upregulation of circGSK3β was associated with metastasis and a poor prognosis in patients with ESCC, suggesting its potential to function as a diagnostic and prognostic biomarker for this type of cancer. Su et al. 17 postulated that hsa_circ_0055538 participated in OSCC progression via the p53 signaling pathway, and may thus be a prospective diagnostic biomarker. Similarly, Dou et al. 18 suggested that decreased expression of the key circRNA hsa_circ_0072387 in OSCC could make it a potential diagnostic biomarker and therapeutic target in OSCC. Furthermore, Zhu et al. 19 revealed that the hsa_circRNA_100533-miR-933-guanine nucleotide-binding protein axis affected the biological behavior of OSCC cells via a mechanism of competing endogenous RNAs (ceRNAs), suggesting that hsa_circRNA_100533 may also be a novel diagnostic biomarker and therapeutic target for OSCC.
Multiple studies have demonstrated that circRNAs can associate with related miRNAs, and the circRNA–miRNA axis can play a vital role in the pathogenesis of human diseases by regulating gene expression. 20 Hansen et al. 21 reported the sponge function of circRNAs, whereby circRNAs bind to specific miRNAs, thus competing with the binding of miRNAs to their target genes and leading to altered gene expression. 21 We explored the ability of hsa_circ_0003829 to bind to miRNAs using the bioinformatics program CircInteractome (https: //Circinteractome.nia.nih.gov/), and identified eight candidate miRNAs that were predicted to interact with hsa_circ_0003829. These data suggest that hsa_circ_0003829 may act as a ceRNA to bind these specific miRNAs, and this interaction may play an important role in the regulation of OSCC progression. However, further studies are needed to verify these hypotheses.
OSCC usually progresses rapidly, and its prognosis is closely related to tumor stage. 1 Lymph node status is the single most important prognostic factor in head and neck squamous cell carcinoma. 22 In the US, approximately 50% of patients with OSCC already have regional or distant metastasis at the time of initial diagnosis. 23 The current study indicated that decreased expression of hsa_circ_0003829 was significantly associated with lymph node metastasis and TNM stage in OSCC patients, suggesting that hsa_circ_0003829 may have potential value in OSCC diagnosis. However, the molecular mechanisms delineating the relationship between hsa_circ_0003829 and lymph node metastasis need further investigation.
Hsa_circ_0003829 may exert tumor-suppressive effects and may be a promising potential diagnostic biomarker for OSCC. Further studies are planned to explore the molecular biological functions of hsa_circ_0003829 to improve our understanding of its role in OSCC.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the basic Research Program of Shenzhen Innovation Council of China [grant nos. JCYJ 20180228175511141 and SZBC2017023] and Sanming Project of Medicine in Shenzhen [SZSM 201512036, Oral and Maxillofacial Surgery Team, Professor Yu Guangyan, Stomatology Hospital Peking University].
