Abstract
Background:
The etiology and risk factors of congenital vertebral anomalies are mainly unclear in isolated cases. Also, there are no reports on the risk factors for different subgroups of vertebral anomalies. Therefore, we assessed and identified potential maternal risk factors for these anomalies and hypothesized that diabetes, other chronic diseases, smoking, obesity, and medication in early pregnancy would increase the risk of congenital vertebral anomalies.
Methods:
All cases with congenital vertebral anomalies were identified in the Finnish Register of Congenital Malformations from 1997 to 2016 for this nationwide register-based case–control study. Five matched controls without vertebral malformations were randomly selected. Analyzed maternal risk factors included maternal age, body mass index, parity, smoking, history of miscarriages, chronic diseases, and prescription drug purchases in early pregnancy.
Results:
The register search identified 256 cases with congenital vertebral malformations. After excluding 66 syndromic cases, 190 non-syndromic malformations (74 formation defects, 4 segmentation defects, and 112 mixed anomalies) were included in the study. Maternal smoking was a significant risk factor for formation defects (adjusted odds ratio 2.33, 95% confidence interval 1.21–4.47). Also, pregestational diabetes (adjusted odds ratio 8.53, 95% confidence interval 2.33–31.20) and rheumatoid arthritis (adjusted odds ratio 13.19, 95% confidence interval 1.31–132.95) were associated with mixed vertebral anomalies.
Conclusion:
Maternal pregestational diabetes and rheumatoid arthritis were associated with an increased risk of mixed vertebral anomalies. Maternal smoking increases the risk of formation defects and represents an avoidable risk factor for congenital scoliosis.
Level of evidence:
III
Keywords
Introduction
Disturbance during the vertebrae development (third to eighth gestational weeks) leads to vertebral malformations. Treatment and prognosis of these deformities can vary from asymptomatic, incidentally found and later only observed structures to difficult deformities of spine such as severe scoliosis and kyphosis resulting into pulmonary insufficiency 1 and spinal cord defects. 2 Fully segmented hemivertebrae often require hemivertebrectomy at an early age and represent the most common indication for surgery in congenital scoliosis.3,4 Then again, segmentation defects might require surgical interventions, such as anteroposterior spinal fusion or growth guidance distraction instrumentation (vertical expandable titanium prosthetic rib, VEPTR). 1
The reported prevalence of congenital vertebral malformations varies between 5 and 10 per 10,000 births. However, the actual incidence might be even higher when asymptomatic people remain easily without diagnosis. 5 Recently, a total prevalence of 2.2 per 10,000 births was reported in a population-based study with an increasing trend. 6 Due to this increasing trend, we aimed to evaluate the maternal risk factors for congenital vertebral defects.
Etiology of congenital vertebral malformations appears to be a combination of genetics with environmental features, and maternal factors have been identified in roughly 20% of pregnancies associated with these anomalies. 7 Previous studies have reported that maternal risk factors include anticonvulsant medication,7,8 gestational diabetes,9,10 maternal insulin-dependent diabetes mellitus,9,11 hyperthermia, 5 and maternal alcohol use. 5 Tobacco smoke includes multiple potential inhaled toxic oxides including carbon monoxide, which might represent the most significant teratogen. Animal studies have demonstrated a dose-correlated association with carbon monoxide exposure during pregnancy and congenital vertebral defects, including vertebral formation defects. 12 However, there are no human data on maternal risk factors of these anomalies.
Our objective was to identify potential maternal risk factors for formation and segmentation defects and mixed vertebral anomalies. We hypothesized previously identified maternal risk factors, such as diabetes (hyperglycemia), smoking, maternal obesity, chronic diseases, and prescription medications, in early pregnancy would increase the risk for congenital vertebral anomalies.
Material and methods
Finnish legislation makes it compulsory to the health personnel to gather certain information and statistics for medical registers. The high coverage and accuracy of these data is validated in many national and international reviews.13,14 Finnish Institute of Health and Welfare (THL) provides and maintains the Finnish Register of Congenital Malformations (FRM), the Medical Birth Register, and the Register of the Induced Abortions. A medical geneticist evaluates, classifies, and codes all the recorded diagnoses at FRM and if diagnosis is unclear, more information is requested from the involved hospitals. 15 Reimbursed maternal prescription medicine purchase data are gathered to the Reimbursement for Medicine Expenses and long-term illnesses to the Register of Entitlements both maintained by the Social Insurance Institution of Finland (Kela). This study is based on the data from registers mentioned above and a more detailed description of the data collection protocol is available in previous publications.15,16
The diagnoses in the registers are coded according to the International Statistical Classification of Diseases and Health-Related Problems (ICD) by the World Health Organization (WHO)—both 9th and 10th revisions (ICD-9 and ICD-10) were used during our study period from 1997 to 2016. Register search with codes either 7561 (ICD-9) or Q76 (ICD-10) for congenital anomalies of spine was performed, and all matches were included in the study. From the Medical Birth Register, five controls without congenital vertebral deformities were randomly selected for each case matched for residency and time of birth or termination of pregnancy (±1 year). Live-born controls were selected for the terminated fetuses.
Classification of congenital vertebral anomalies was performed as proposed by McMaster and Ohtsuka: 17 failure of vertebral formation (hemivertebra), failure of vertebral segmentation (bony bar), and mixed vertebral anomalies (combination of different types of anomalies). The written diagnosis in the FRM was used for the classification of vertebral anomalies as different types of vertebral anomalies do not have specific ICD codes.
The maternal risk factors analyzed included age at delivery, body mass index (BMI), parity, history of miscarriages, chronic diseases, medical purchases, and smoking. Maternity health clinics are attended by over 99.7% of pregnant women in our country. 18 At first prenatal visit in healthcare, approximately 8–10 weeks in pregnancy maternal weight were recorded systematically, although the data on maternal BMI were collected to the Medical Birth Register since 2004 and available from all birth hospitals since October 2005. Smoking was defined only if mother was currently smoking during the first trimester. Maternal pregestational diabetes was diagnosed either before conception or during pregnancy and contained both type 1 and 2 (ICD-10 codes E10, E11, O24.0, and O24.1). The Register of Entitlements to Reimbursement of Medicine Expenses was used to cross-check pregestational diabetes (codes 105 and 215). All mothers with pregestational diabetes were also using insulin in our cohort. Therefore, insulin usage and pregestational diabetes were not analyzed separately. Gestational diabetes group included both women with diagnosed gestational diabetes (O24.4) and an abnormal glucose tolerance test result.
Maternal medication was initially screened in the third-level Anatomical Therapeutic Chemical (ATC) Classification system by WHO and those groups with at least five mothers with drug purchases of interest among cases or controls were selected for analysis. The analyses were mainly performed in the fourth level of the ATC. Exception was made with oral corticosteroids (H02), as they have been previously identified as a risk factor for birth defects in some studies. 19 Also, insulins (A10A) were analyzed in the third level of ATC. Long-term illnesses included in the analyses were diabetes, connective tissue diseases and rheumatoid arthritis (later rheumatoid arthritis, Kela code 202), and psychotic disorders and other severe mental disorders (later psychotic disorders, Kela code 112).
Statistics
Potential risk factors were evaluated with Fisher’s exact test, and then univariate logistic regression models were chosen to suit for case–control studies. Multivariable models were built up with all significant factors in the univariate model included. Then, non-significant factors were gradually removed one by one. Maternal age and smoking, which are the known risk factors for various congenital malformations, were included in all multivariable models. Sensitivity analyses were performed due to missing values in BMI (missing before 2004/2005) and diabetes, when multiple imputation (n = 50) was performed with fully conditional specification (FCS) method. BMI, case or control status, delivery method, diabetes, infertility treatment, parity, psychotic variables, rheumatoid arthritis, smoking, and year of delivery or termination were taken into account in multiple imputation process. The number of missing values was negligible for all other variables than diabetes and BMI.
All statistical tests were performed as two-sided, with a significance level set at 0.05. The analyses were performed using SAS System, version 9.4 for Windows (SAS Institute Inc., Cary, NC, USA).
Ethical considerations
The approval of the Institutional Review board was acquired before conducting this study. Also, the Finnish Institute for Health and Welfare (THL) and Kela (Social Insurance Institution of Finland) gave authorization to use their register data.
Results
In total, 256 cases diagnosed with congenital vertebral anomalies were identified in the FRM. After excluding 66 malformations associated with known syndromes as defined by EUROCAT, 20 190 non-syndromic cases including 74 formation defects (hemivertebra), 4 segmentation defects (unilateral bar), and 112 with mixed anomalies were included in the study. Cases with congenital vertebral malformations were compared with 950 matched controls. Separate risk factor analyses were not performed for segmentation defects due to the small number of cases.
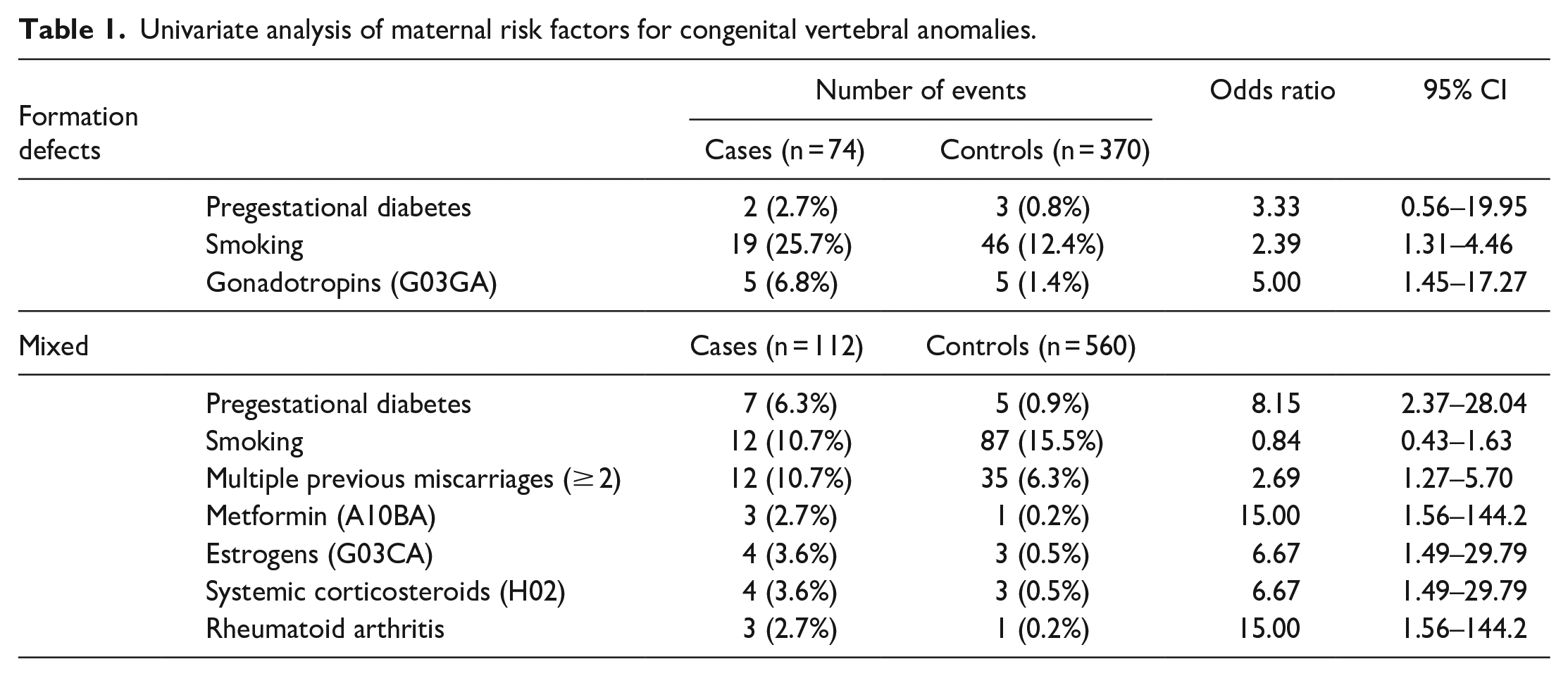
In univariate analyses, maternal smoking (odds ratio (OR) 2.39, 95% confidence interval (CI) 1.31–4.46) and gonadotropins (OR 5.00, 95% CI 1.45–17.27) were the significant risk factors for vertebral formation defects. Also, maternal pregestational diabetes (OR 8.15, 95% CI 2.37–28.0), multiple miscarriages (OR 2.69, 95% CI 1.27–5.70), estrogens (OR 6.67, 95% CI 1.49–29.8), and rheumatoid arthritis (OR 15.0, 95% CI 1.56–144.2) were associated with increased risk for mixed vertebral anomalies (Table 1). No significant associations were observed with gestational diabetes, other chronic diseases, maternal age, parity, and BMI.
Univariate analysis of maternal risk factors for congenital vertebral anomalies.
In multivariable analysis, maternal smoking was a significant risk factor (adjusted odds ratio (aOR) 2.33, 95% CI 1.21–4.47) for vertebral formation defects (Table 2). Maternal pregestational diabetes (aOR 8.53, 95% CI 2.33–31.20), multiple miscarriages (aOR 3.10, 95% CI 1.42–6.80), estrogens (aOR 6.99, 95% CI 1.53–31.90), and rheumatoid arthritis (aOR 13.2, 95% CI 1.31–133.0) were significantly associated with mixed vertebral anomalies (Table 3).
Analysis of maternal risk factors for vertebral formation defects.
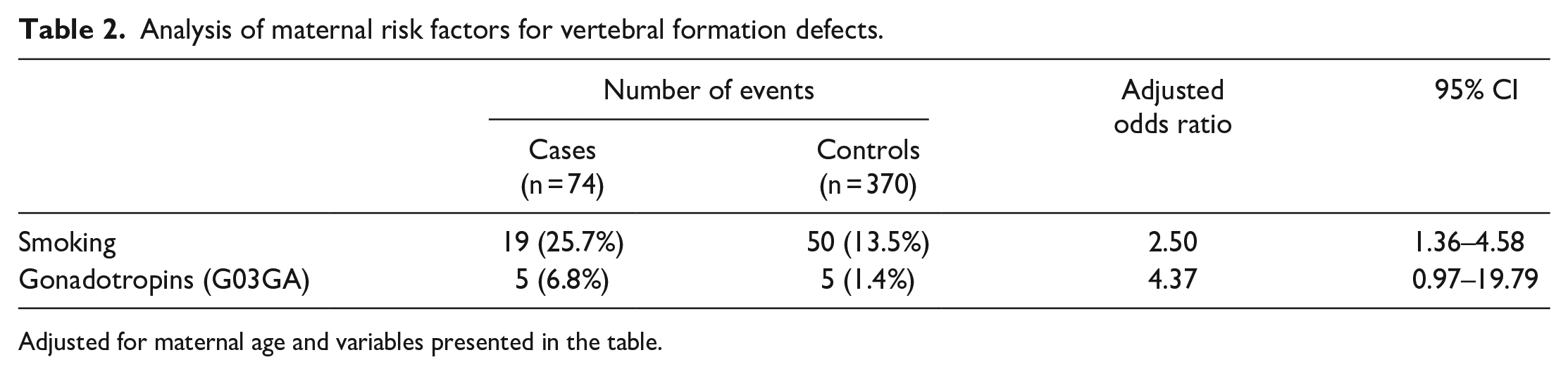
Adjusted for maternal age and variables presented in the table.
Analysis of maternal risk factors for mixed vertebral anomalies.
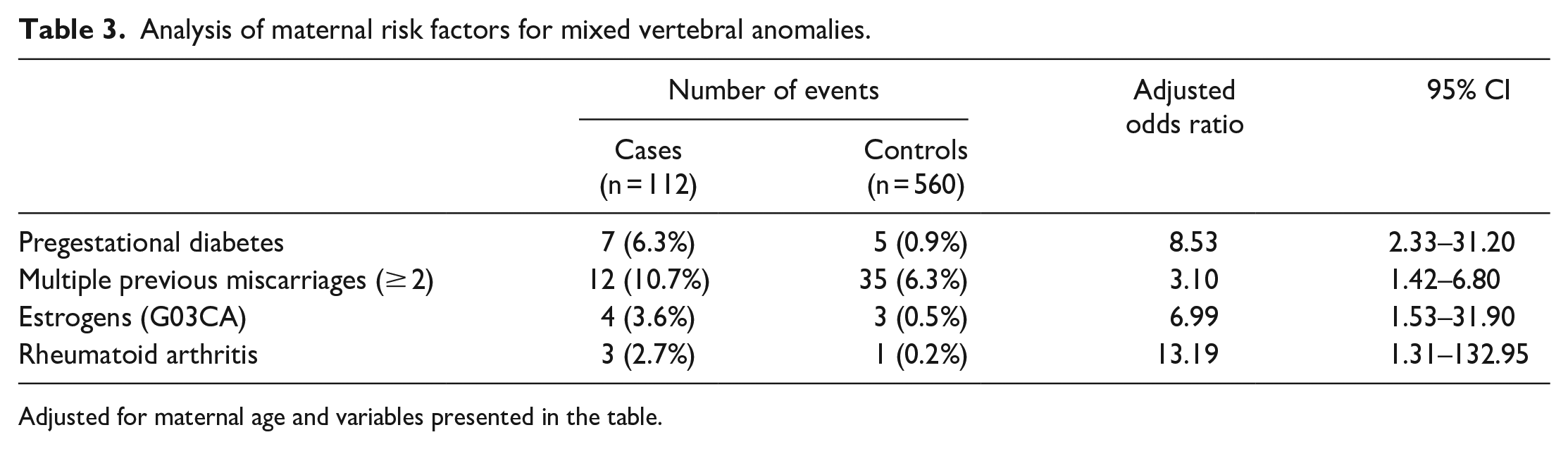
Adjusted for maternal age and variables presented in the table.
Sensitivity analyses with imputed values were performed, which did not have significant impact on the risk factor profile (Appendix).
Discussion
This population-based case–control study has revealed that maternal smoking during the first trimester of pregnancy is associated with a 2.5-fold increased risk of hemivertebrae. Maternal pregestational diabetes is a significant risk factor for mixed congenital spine deformities but was not associated with an increased risk of formation defects.
Previous studies have demonstrated an association between maternal smoking and several congenital anomalies, such as congenital heart defects, gastrointestinal anomalies, orofacial clefts, and neural tube defects. 21 Experimental studies have proposed that smoking may play a role in the development of congenital spine deformities.22,23 To the best of our knowledge, this is the first human study to reveal a direct association between maternal smoking and risk of vertebral formation defects. Tobacco smoke includes multiple potential inhaled toxic oxides, including carbon monoxide, which might represent the most significant teratogen. Animal studies have demonstrated a dose-correlated association with carbon monoxide exposure during pregnancy and congenital vertebral defects. 12 In that study, pregnant females were exposed for 7 h to 200, 400, or 600 ppm CO. Observed anomalies included wedged, hemi- and missing vertebrae; and these were located in all regions of the spine. There were no defects in the controls and a 77% incidence of congenital malformations at 600 ppm (p < 0.0001).
Previous studies have confirmed that maternal pregestational diabetes increases the risk of several congenital malformations.9,16,24 Also, there are substantial evidence suggesting that the primary teratogen causing embryonic maldevelopment is hyperglycemia. 25 Previous reports have identified an increased risk of spine malformations with maternal diabetes.9,11 Contrary to the Swedish study, we observed no association with gestational diabetes and congenital vertebral defects. In the current study, pregestational diabetes was significantly associated with mixed vertebral anomalies but not with formation defects. It is possible that the deviations in the glucose metabolism associated with gestational diabetes are not strong enough to affect the organ development at early pregnancy.
Previously, rheumatoid arthritis has been linked with an increased risk of congenital defects.26,27 In this study, we observed an association with rheumatoid arthritis and mixed vertebral anomalies. However, this observation was based on a limited number of exposed mothers.
An association with maternal use of exogenous sex hormones in early pregnancy and various congenital malformations has been observed in several studies.16,28,29 An association was observed with estrogen and mixed congenital vertebral defects in the current study. However, the risk of congenital vertebral malformations was not associated with assisted reproductive technology. As in previous studies, it remains elusive whether this association is attributed to the medication itself, maternal and paternal factors related to subfertility or assisted reproductive technology.16,30
Previously, a number of genes have been associated with vertebral defects, including PAX1, DLL3, SLC35A3, WNT3A, TBX6, and T (Brachyury). 31 These were sequenced in a group of 50 congenital vertebral malformations with three having mutation in T (Brachyury) gene. 32 Interestingly, these patients also had maternal pregnancy exposure histories of diabetes, valproic acid, and clomiphene. 31 Therefore, it appears that both environmental and genetic factors are likely to contribute to the occurrence of congenital vertebral defects. 7
Strengths and limitations
The strength of our study is that the registries used in this study are validated with population-based coverage.13,14 Furthermore, all the diagnoses were confirmed and double-checked by a medical geneticist before entering to the registry. Also, the nationwide data were prospectively collected by the universally accessible healthcare system.
There are some limitations to our study, mainly a relatively small sample size accompanied with reduced statistical power. Also, the data were collected for maternal prescription drug purchases only. Therefore, a limitation is caused by the lacking information on the dosages and whether the drugs were actually taken. Furthermore, maternal BMI was systematically recorded for the latter half of the study period only. This limitation was evaluated using imputed data with sensitivity analyses and these resulted in similar findings as with non-imputed data. Moreover, the data on illicit drugs, over-the-counter medication, and herbal medications during pregnancy were not available since they are not collected in the medical registers. It should also be noted that our risk factors are valid in the Finnish population but may not be exactly reproducible in other populations.
In conclusion, maternal pregestational diabetes is a significant risk factor for mixed congenital vertebral anomalies. Also, multiple miscarriages, estrogens, and rheumatoid arthritis were also associated with an increased risk. Maternal smoking, which is a voluntarily modifiable factor, more than doubled the risk of formation defects. Therefore, expecting mothers and those planning to conceive should be informed of this risk.
Supplemental Material
sj-docx-1-cho-10.1177_18632521241235027 – Supplemental material for Maternal risk factors for congenital vertebral formation and mixed defects: A population-based case–control study
Supplemental material, sj-docx-1-cho-10.1177_18632521241235027 for Maternal risk factors for congenital vertebral formation and mixed defects: A population-based case–control study by Susanna Heiskanen, Ilkka Helenius, Johanna Syvänen, Teemu Kemppainen, Eliisa Löyttyniemi, Matti Ahonen, Mika Gissler and Arimatias Raitio in Journal of Children’s Orthopaedics
Supplemental Material
sj-pdf-2-cho-10.1177_18632521241235027 – Supplemental material for Maternal risk factors for congenital vertebral formation and mixed defects: A population-based case–control study
Supplemental material, sj-pdf-2-cho-10.1177_18632521241235027 for Maternal risk factors for congenital vertebral formation and mixed defects: A population-based case–control study by Susanna Heiskanen, Ilkka Helenius, Johanna Syvänen, Teemu Kemppainen, Eliisa Löyttyniemi, Matti Ahonen, Mika Gissler and Arimatias Raitio in Journal of Children’s Orthopaedics
Footnotes
Author contributions
S.H., A.R., M.G., J.S., and I.H. contributed to study conception and design. A.R., M.G., J.S., and I.H. involved in acquisition of data. S.H. and A.R. drafted the article. All authors participated in analysis and interpretation of data, and critical revision of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
The approval of the Institutional Review board at the Turku University Hospital was obtained. Also, the Finnish Institute for Health and Welfare (THL) and Kela (Social Insurance Institution of Finland) gave permission to use their register data in this study. Register studies do not require ethical board approval.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research grants from Clinical Research Institute HUCH received by S.H., J.S., I.H., and A.R. In addition, A.R. and I.H. have received research grants from Päivikki & Sakari Sohlberg Foundation, and Emil Aaltonen Foundation, respectively, and I.H. received scientific funding to Institution from The Finnish Pediatric Research Foundation, Stryker, Medtronic, and Nuvasive. For the remaining authors, none was declared.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
