Abstract
Objective
We aimed to identify risk factors for macrosomia in women with high-risk pregnancies, which will allow for early identification and timely intervention and improve the health of mothers and infants.
Methods
A retrospective case–control study was conducted of pregnant women and neonates born at Shenmu Hospital in China between 1 January 2020 and 31 December 2023. Cases comprised neonates with macrosomia (birth weight ≥4000 g), and controls comprised a group of normal-weight neonates (birth weight 2500–3999 g). Conditional logistic regression analysis was performed to identify independent predictor variables.
Results
In total, 10,253 cases with complete maternal and neonatal-related data were included in this study, with 1100 cases in the macrosomia group and 9153 in the control group. Male neonates (odds ratio = 1.636), gestational age (odds ratio = 1.726), maternal height ≥165 cm (odds ratio = 3.648), prepregnancy body mass index ≥28 kg/m2 (odds ratio = 2.733), excessive weight gain during pregnancy (odds ratio = 2.817), ≥2 pregnancies (odds ratio = 1.368), ≥2 prior deliveries (odds ratio = 1.285), gestational diabetes (odds ratio = 3.227), excess amniotic fluid (odds ratio = 4.592), scarred uterus (odds ratio = 0.693), gestational hypertension (odds ratio = 0.750), oligohydramnios (odds ratio = 0.262), and umbilical cord anomalies (odds ratio = 0.727) were all associated with an increased risk of macrosomia.
Conclusions
Women with high-risk pregnancies who have a height of ≥165 cm, ≥2 previous deliveries, prepregnancy obesity, and a scarred uterus should be provided with individualized prenatal care and delivery plans; for these women, monitoring weight gain, blood glucose, and amniotic fluid should be intensified along with targeted interventions to provide optimal care for this high-risk population.
Introduction
Newborns with a birth weight ≥4000 g are referred to as macrosomic. Macrosomia poses many health risks for mothers, including increased incidence of cesarean section, postpartum hemorrhage, and severe vaginal tears, as well as many risks for infants, including potential shoulder dystocia, clavicle fracture, brachial plexus injury, respiratory distress, hypoglycemia, and increased early mortality. 1 A cross-national study of 115 million live births in 15 countries reported that neonates with birth weight >4500 g had higher early mortality than those with birth weight of 2500–3999 g. 2
The incidence of macrosomia varies in different countries and regions with incidence rates ranging from 0.76% in Japan 3 and 3.22% in South Korea 4 to 7.8% in the United States. 5 In China, the incidence of macrosomia increased from 6.0% in 1994 to 7.3% in 2014, 6 among the highest in Asian countries. These differences may be related to a variety of factors such as obesity rates, racial composition, living habits, and nutritional status, which vary widely worldwide. 7
Studies have increasingly shown that early life growth and development have a nonnegligible impact on health later in life, 8 and some studies have shown that macrosomia increases the risk of cardiovascular disease, becoming overweight or obese, thyroid disease, and cancer as well as intellectual and psycho-behavioral problems in newborns. 9
Therefore, identifying the risk factors of macrosomia, and providing targeted and timely interventions to reduce the incidence of macrosomia, which is a major public health concern, are goals that are addressed in this study.
Materials and methods
Study design and data sources
This study was a retrospective case–control study that analyzed data from 1 January 2020 to 31 December 2023 of mothers and neonates at Shenmu City Hospital maternity and neonatal wards in China. The study protocol was approved by the local ethical committee (NO: KYLL-2025-004). Cases in the macrosomia group, comprising neonates with a birth weight of ≥4000 g, were compared with controls, comprising neonates with a birth weight of 2500–3999 g. Inclusion criteria were singleton live births and neonate birth weight of ≥2500 g, whereas neonate-mother pairs that included stillbirths, multiple births, neonate birth weight of <2500 g, and an incomplete medical history were excluded.
Data collection and measurements
By reviewing the electronic medical records, we collected maternal data on age (years), height, educational attainment, pregnancy, delivery number, number of previous cesarean sections, prepregnancy body mass index (BMI), weight gain during pregnancy, gestational hypertension, gestational diabetes mellitus, anemia, hypothyroidism during pregnancy, history of placenta previa, placental abruption, premature rupture of membranes, abnormal amniotic fluid, umbilical cord abnormality, scarred uterus, intrauterine distress, assisted reproduction, and advanced age (years) at pregnancy. Neonate information that was collected included sex, gestational age (weeks), and season of birth.
Macrosomia was defined as a condition in which a neonate has a birth weight of ≥4000 g. We considered maternal age of <35 years as normal maternal age and ≥35 years as advanced maternal age. To categorize BMI before pregnancy, we used the following cut-off values from the Guidelines for the Prevention and Control of Overweight and Obesity in Chinese Adults: emaciation (BMI < 18.5 kg/m2), normal (18.5 ≤ BMI < 24 kg/m2), overweight (24 ≤ BMI < 28 kg/m2), and obesity (BMI ≥ 28 kg/m2). We evaluated pregnancy weight gain by subtracting the prepregnancy weight from the predelivery weight and compared this with the American Institute of Medicine recommended standards. The ideal range of pregnancy weight gain is 12.5–18.0 kg (BMI < 18.5 kg/m2), 11.5–16.0 kg (18.5 ≤ BMI < 25 kg/m2), 7.0–11.5 kg (25 ≤ BMI < 30 kg/m2), and 5.0–9.0 kg (BMI ≥ 30 kg/m2). Each BMI group below the lowest value of the ideal range was considered as insufficient weight gain, that in the ideal range was considered as reasonable weight gain, and that above the ideal range was considered as excess weight gain. Anemia during pregnancy was assessed using the criteria provided by the World Health Organization with the standard cut-off value for anemia during pregnancy (at sea level) being hemoglobin level <110 g/L. 10 Gestational diabetes mellitus was assessed using a 75-g glucose tolerance test at any time during pregnancy, with the diagnosis being made if blood glucose levels reached any of the following three parameters: fasting blood glucose ≥ 5.1 mmol/L, ≥10 mmol/L one hour after sugar intake, and ≥ 8.5 mmol/L two hours after sugar intake. Gestational hypertension refers to a first instance of systolic blood pressure being ≥140 mmHg and/or the diastolic blood pressure being ≥90 mmHg or diastolic blood pressure being ≥90mmHg, with or without proteinuria or edema and other symptoms after the twentieth week of pregnancy in a patient with no prior diagnosis of hypertension. Umbilical cord abnormalities include a single umbilical artery, umbilical cord around the neck, umbilical cord around the body, umbilical cord of abnormal length, and umbilical cord true knot or false knot. Abnormalities in the amniotic fluid include amniotic fluid volume exceeding 2000 mL at any point during pregnancy or amniotic fluid volume <300 mL in late pregnancy, as defined in the ninth edition of Obstetrics and Gynecology from the People’s Health Publishing House. 11 Seasons of birth include the traditional seasons of spring, summer, fall, and winter.
Statistical analysis
Analyses were performed using the Statistical Package for Social Science (SPSS) software version 27. An independent sample t-test was used to compare the two groups with counting information expressed as cases (ratio), and a χ2 test or Fisher’s exact probability method was used to compare the two groups.
Univariate analysis of risk factors was performed using conditional logistic regression in order to determine the strength of the association. Multivariate conditional logistic regression analysis was carried out for variables that were significant in the univariate analysis. Odds ratio with 95% confidence intervals (CIs) were calculated for risk factors and complications. A P value of less than 0.05 was deemed statistically significant.
Results
Maternal and neonatal characteristics
During the study period, 10,819 pregnant women were hospitalized and delivered in the obstetrics department of Shenmu City Hospital. There were 29 cases of stillbirth, 102 cases of twin births, 429 cases of delivery of newborns weighing <2500 g (including 24 cases of twin births), and 30 cases of incomplete medical history data. In total, 10,253 cases of maternal and neonatal data were used in the analysis and included 1100 participants in the macrosomia group and 9153 in the control group. The incidence of macrosomia was 10.2% (1100/10790), which is slightly higher than previously reported rates.
In the macrosomia group, the mean maternal age (years) at delivery was 30.39 ± 4.01 years, with 14.36% (158/1100) of mothers having advanced maternal age, which was significantly higher than the maternal age (years) of the control group (P = 0.005). The average gestational age was 39.60 ± 1.06 weeks and average birth weight was 4178.68 ± 346.39 g, with a maximum weight of 6000 g. Male neonates accounted for 64.09% (705/1100) of the macrosomia group and were significantly higher than those in the control group with a male-to-female ratio of 1.78:1 (P < 0.01). Other parameters, including mean height, BMI before pregnancy, and weight gain during pregnancy, were also significantly higher among the cases than among the controls (Table 1). Maternal educational status was not significantly different between the groups.
Maternal and neonatal characteristics.

BMI: body mass index.
Maternal and neonatal risk factors for macrosomia
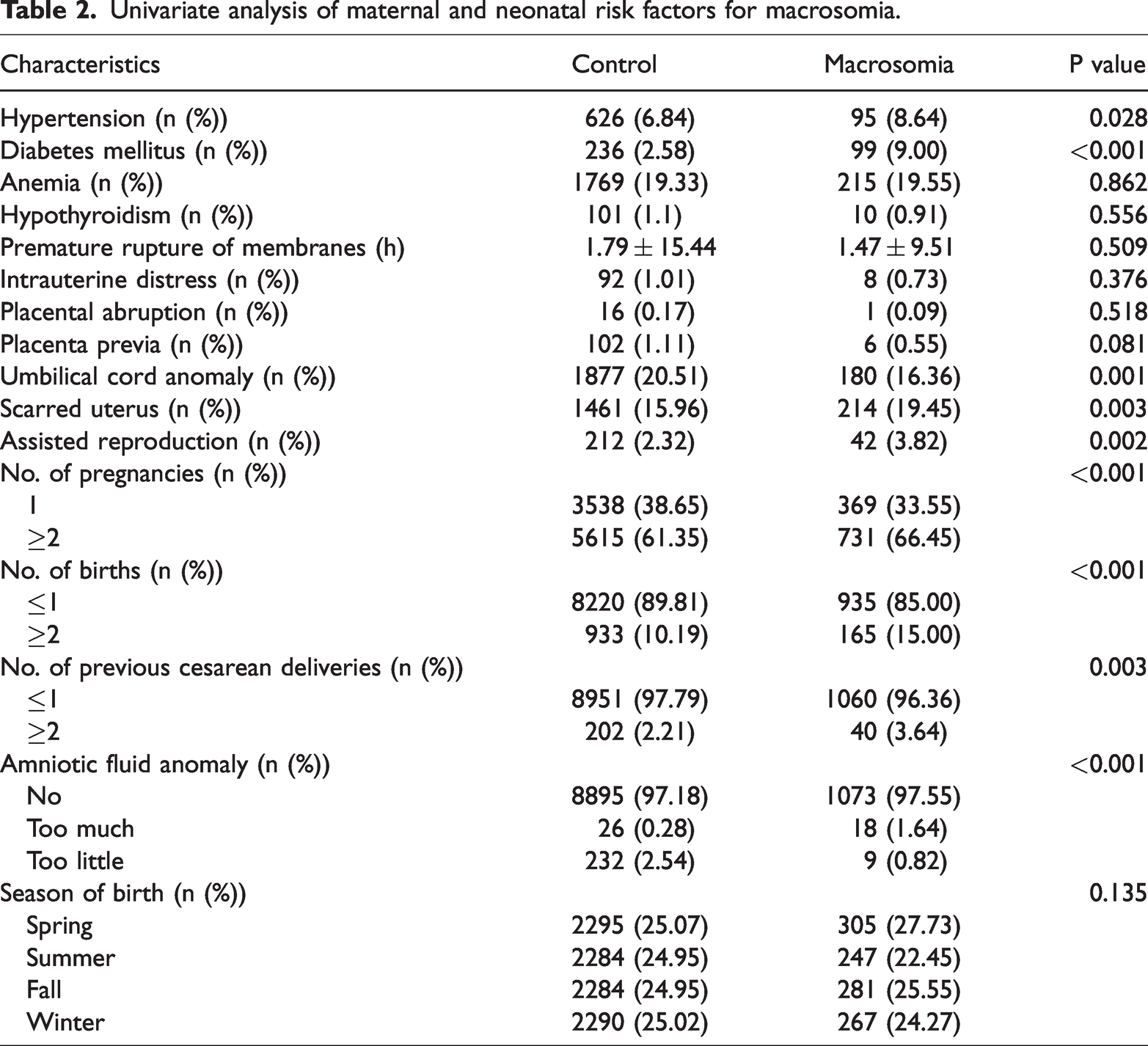
Univariate analysis of factors affecting the occurrence of macrosomia showed significant differences between the two groups in terms of number of pregnancies, number of deliveries, number of previous cesarean deliveries, gestational diabetes mellitus, gestational hypertension, umbilical cord anomalies, amniotic fluid anomalies, keloidal uterus, and assisted reproduction. However, there was no difference in the educational status as well as the prevalence of anemia during pregnancy, hypothyroidism during pregnancy, placenta previa, placental abruption, premature rupture of membranes, and intrauterine distress. Further, the season of birth of newborns were not significantly different between the two groups (Table 2).
Univariate analysis of maternal and neonatal risk factors for macrosomia.
Multifactorial analysis of factors influencing the occurrence of macrosomia
The outcome variable of whether the child was macrosomic was used as the dependent variable Y, and the factors significantly associated with the occurrence of macrosomic children were set as independent variables included in the logistic regression model. The analysis showed that the effects of neonatal sex, gestational age (weeks), mother’s height, BMI before pregnancy, weight gain during pregnancy, the number of pregnancies, the number of births, keloidal uterus, gestational hypertension, gestational diabetes mellitus, umbilical cord abnormality, and amniotic fluid abnormality on the occurrence of macrosomia babies were statistically significant. The risk factors for macrosomia were male sex of the newborn, higher maternal height, BMI before pregnancy, excessive weight gain during pregnancy, higher gestational age (weeks), ≥2 previous pregnancies, ≥2 previous deliveries, gestational diabetes mellitus, and excessive amniotic fluid. A scarred uterus, gestational hypertension, excess amniotic fluid, and umbilical cord abnormality were protective factors for macrosomia, as shown in Table 3.
Multivariate analysis of maternal and neonatal risk factors for macrosomia.

CI: confidence interval; OR: odds ratio.
Discussion
This case–control study was conducted to investigate the risk factors for macrosomia in a Chinese population. We found that neonatal sex, gestational age (weeks), maternal height, prepregnancy BMI, gestational weight gain, gestational diabetes mellitus, number of pregnancies and births, as well as keloidal uterus and umbilical cord abnormalities were all associated with increased macrosomia risk.
This risk for male newborns was approximately 1.636 times higher than that for female newborns (odds ratio = 1.636, 95% CI 1.423–1.881, P < 0.01), consistent with results from previous studies,12,13 and may be related to the fact that the Y chromosome promotes fetal growth and testosterone secretion in males that leads to an increase in protein synthesis. We also found that the risk of macrosomia increased by approximately 0.726 times for every 1-week increase in gestational age. A prior study conducted in the United States showed that the risk of having a birth weight of more than 4500 g increases from 1.3% at 39–40 weeks of gestation to 2.9% when the gestational age exceeds 41 weeks, 14 which may be caused by the fetus obtaining excess nutrients from the placenta for a prolonged period of time. 7 Genetic factors such as maternal height also have a significant effect on the occurrence of macrosomia. We found that the risk of delivering a macrosomic child in women with a height of 155–159.9 cm, 160–164.9 cm, and ≥165 cm was approximately 2.406, 2.887, and 3.648 times higher than that in women with a height <155 cm, respectively, also consistent with prior reports.7,15 This could be in part because taller pregnant women tend to have a larger pelvic structure, providing fewer physical restraints on fetal growth. BMI before pregnancy and weight gain during pregnancy are equally important risk factors for the development of macrosomia. In this study, the risk of delivering a newborn with macrosomia in normal-weight, overweight, and obese pregnant women was approximately 1.901 times, 2.679 times, and 2.733 times higher than that in normal-weight pregnant women. Similarly, the risk of delivering a newborn with macrosomia in overweight pregnant women is approximately 2.138 times higher than that in pregnant women who have an average amount of weight gain during pregnancy. There has been significant research on the association between maternal BMI, weight gain during pregnancy, and macrosomia. For example, one prior study pointed out that weight gain of >15 kg in the first 6 months of pregnancy is an important risk factor for the development of macrosomia. 7 This is principally because over nutrition and excess calorie intake during pregnancy leads to endocrine abnormalities in pregnant woman and the fetus, especially in cases of maternal obesity. This may exacerbate the phenomenon of maternal insulin resistance, leading to increased serum blood glucose levels in mothers which can stimulate fetal insulin secretion, and the combined effect of maternal hyperglycemia and fetal hyperinsulinemia results in excess fetal growth.
Gestational diabetes mellitus is widely recognized as an independent risk factor for macrosomia. Some studies have shown a positive correlation between maternal blood glucose levels and the incidence of macrosomia. The probability of fetal macrosomia is approximately 50% for women with combined maternal hyperglycemia and gestational diabetes mellitus, 30% in those with gestational diabetes mellitus alone, and 20% in those with mildly impaired glucose tolerance. 16 We found that the risk of macrosomia in pregnant women with gestational diabetes mellitus was approximately 3.227 times (95% CI 2.449–4.254) higher than that in women without gestational diabetes mellitus. This is because the nutrient-insulin -insulin-like growth factor (IGF) metabolic axis is central to the regulation of fetal growth. IGF-1 is a primary regulator of fetal growth in the second trimester and the neonate in the early postnatal period; further, it positively correlates with neonatal birth weight. Additionally, higher IGF-1 levels have been found in the umbilical cord blood of mothers with gestational diabetes mellitus.
In addition, we found an association between the number of births and the occurrence of macrosomia, with the risk of macrosomia increasing with the number of births, and the probability of delivering a macrosomic child in second-trimester mothers and mothers with ≥2 births was approximately 1.368 and 1.285 times higher than that in primigravid mothers, respectively. This is consistent with the findings of previous studies6,17 and has biologic plausibility because multiple pregnancies may contribute to adaptive changes in the reproductive system, such as increased uterine size and laxity of the uterine cavity, which may contribute to an increased risk of macrosomia. It is also possible that in addition to biologic factors, women with multiple pregnancies may rely on their past experiences of labor and delivery and have fewer regularly scheduled obstetric examinations. In this study, it was observed that abnormalities in amniotic fluid volume had a significant effect on the occurrence of macrosomia. Specifically, the risk of delivering a newborn with macrosomia from pregnant women with excess amniotic fluid was 4.592 times higher than that in pregnant women with normal amniotic fluid, which has been reportedpreviously. 18 Inversely, insufficient amniotic fluid was a protective factor against the occurrence of macrosomia, although having insufficient fluid can be associated with other neonatal complications. This may be because of the fact that excess amniotic fluid may lead to a more fluid-rich environment for the fetus, promoting over nutrition and leading to increased weight gain. Dilatation of the intrauterine volume with excess amniotic fluid also diminishes the natural restriction of fetal growth and development and increases the risk of macrosomia.
Notably, this study confirmed that a scarred uterus, umbilical cord anomalies, and gestational hypertension were protective factors against the development of macrosomia. It is possible that a scarred uterus may affect the efficiency of nutrient exchange between the mother and the fetus by altering the location of placental attachment and blood flow, which in turn may affect the efficiency of material exchange between the mother and the fetus. 19 There are fewer studies on the relationship between umbilical cord anomalies and macrosomia; however, prior research examining if the umbilical cord is undercoiled or overcoiled showed an association with various adverse pregnancy outcomes but not specifically a decrease in fetal macrosomia. 20 Umbilical cord abnormalities may impede the efficient transfer of nutrients from the placenta to the fetus, leading to reduced fetal nutritional access, thereby increasing the risk of intrauterine growth restriction and adversely affecting neonatal weight. Many studies have shown that gestational hypertension increases the incidence of macrosomia; however, there are conflicting reports.21,22 One potential mechanism by which gestational hypertension could be protective against macrosomia is that it may trigger placental ischemia, causing fetal hypoxia and developmental malformations. Thus, because of the inability of the mother to meet the adequate supply of nutrients needed for fetal growth and development, fetal cardiomyocytes may undergo compensatory hyperplasia, leading to cardiac abnormalities, and ultimately a series of adverse pregnancy outcomes, including low birth weight.
Strengths and limitations of the study
The limitations of this work include the fact that the indicators included in the study were factors related to maternal pregnancy; however, most factors were analyzed on the basis of count data and lacked quantitative analysis, such as blood glucose, D-dimer, triglycerides, IGF-1, and maternal vitamin D levels. Further, we did not evaluate several other factors affecting weight gain during pregnancy, such as dietary content and exercise.
Conclusions
In summary, our study showed that the incidence of macrosomia was approximately 10.7% in 2020–2023 at Shenmu Hospital. Male sex of the newborn, mother’s height ≥165 cm, prepregnancy obesity, excessive weight gain during pregnancy, older gestational age (weeks), ≥2 previous pregnancies, ≥2 previous deliveries, gestational diabetes, and excess amniotic fluid were all risk factors for macrosomia. In contrast, a scarred uterus, gestational hypertension, low amniotic fluid, and umbilical cord anomalies reduced the risk of macrosomia. Given that the incidence of macrosomia is affected by the above factors, individualized antenatal care and delivery plans that include intensive monitoring of maternal weight gain, blood sugar, and amniotic fluid levels are necessary to allow for timely and targeted interventions for high-risk mothers.
Footnotes
Acknowledgments
I sincerely thank Fu Yong Jiao and Na Gao for the selfless help and support provided throughout the process of writing the manuscript.
Author contributions
Rui Xue: research design, data collection, data organization, and thesis writing. Zhongming Yun: research guidance and thesis revision.
Data availability statement
Not applicable. Please contact the author for data requests.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by grants from Yulin City Science and Technology Association Youth Talent Support Program Project (
