Abstract
Purpose:
The purpose of this study is to compare pediatric hip aspiration in the operating room under general anesthesia or via bedside aspiration under moderate sedation and delineate the anesthetic time required.
Methods:
A database query conducted at two academic institutions identified all patients under the age of 17 who underwent hip aspiration between 2000 and 2017. At one institution, aspiration was performed in the operating room under general anesthesia. Patients were kept anesthetized until cell count was complete. At the second institution, aspiration was performed in the emergency room at bedside under sedation. The medical record was reviewed for demographic data, hip aspiration results, diagnoses, treatment, and anesthesia time.
Results:
A total of 233 patients (233 hips) with a mean age of 7.2 years were identified. Seventy-five patients underwent aspiration in the operating room, and 158 patients underwent bedside aspiration. Patients with a negative aspiration averaged 87 min under anesthesia when performed in the operating room and 29 min under sedation when performed at bedside. Patients with a negative aspiration performed in the operating room after 5 pm averaged 99 min under anesthesia, and 73 min under anesthesia when performed between 7 am and 5 pm (p < 0.01). Seventy-eight (49%) patients who underwent bedside aspiration did not require operative intervention and therefore avoided general anesthesia.
Conclusion:
Pediatric hip aspiration performed in the operating room results in prolonged anesthesia times while synovial fluid is transported and processed. Anesthesia times are significantly longer after 5 pm. Bedside aspiration resulted in significantly less anesthesia exposure, with half of patients undergoing bedside aspiration avoiding general anesthesia altogether.
Level of evidence:
Level III.
Introduction
Pediatric patients presenting with a hip effusion and suspected septic arthritis represent one of the few pediatric orthopedic emergencies. Septic arthritis has variable presentations but is typically associated with hip effusion and elevated inflammatory markers. The broad differential diagnosis in this situation includes transient synovitis, sacroiliitis, pyomyositis, osteomyelitis, Lyme disease, neoplasm, and juvenile idiopathic arthritis.1–3 Prompt and accurate diagnosis is imperative as the long-term consequences of missed septic arthritis include cartilage and femoral head destruction, deformity, joint contracture, gait abnormalities, and hip dislocation. 4
Evaluation of suspected septic arthritis classically begins with a thorough physical examination. This includes evaluation of hip range of motion and ability to weight bear through the affected limb. If these maneuvers raise suspicion, the next step is typically application of the modified Kocher criteria: white blood cell count >12,000, inability to bear weight, fever > 38.5°C, and erythrocyte sedimentation rate >40 (and/or C reactive protein > 20). The probability of septic arthritis may be as a high as 93% when all four of these criteria are present and 2% when all four are absent.5–7 Aspiration of hip synovial fluid is recommended for all cases where there are concerns for septic arthritis; specifically, when a hip effusion is identified in the presence of elevated inflammatory markers. Advanced imaging modalities, such as doppler ultrasound and magnetic resonance imaging (MRI), are not able to discern between septic arthritis or other common causes of hip effusion.8,9 Thus, hip aspiration, cell count, and culture are the gold standard approach to diagnose septic hip arthritis.
At many centers, pediatric hip aspiration is performed in the operating room (OR) under general anesthesia. If the aspiration is grossly purulent, irrigation and debridement (I&D) is carried out immediately. If not, some institutions will keep the patient anesthetized until a preliminary cell count is available. If the cell count is high, I&D is then carried out. If the cell count is low, the procedure is ended, and the child is awakened. While this practice allows for immediate surgical intervention in the setting of septic arthritis, a large cohort of children with negative aspirations are subjected to prolonged general anesthesia. In multiple observational studies, exposure to general anesthesia during the early years of development has been associated with learning disabilities, attention deficit hyperactivity disorder (ADHD), and impaired memory.10–12
Current literature suggests that hip aspiration in the emergency department may be as safe and efficacious as aspiration performed in the OR.13–15 There is a paucity of literature, however, comparing the time spent under anesthesia between these respective approaches. Therefore, the aims of the present study were to compare the mean time children spent under anesthesia and risk factors for prolonged anesthetic between hip aspiration in the OR compared to hip aspiration at the bedside in the emergency room. We hypothesized that aspirations performed at the bedside in the emergency room would result in a decrease in the total anesthetic time compared to aspirations performed in the OR for pediatric patients.
Materials and methods
After an institutional review board (IRB) approval, a retrospective review was completed for all pediatric hip aspirations performed for suspected septic arthritis at two academic institutions. All patients who underwent hip aspiration over a 17-year period (2000–2017) were included.
Surgical technique
At the institution performing aspiration in the OR (Institution A), all hip aspirations were performed by fellowship-trained pediatric orthopedic surgeons. Antibiotics were held before the procedure to reduce possible suppressive effects on culture results. Fluoroscopic guidance was used intraoperatively to visualize the needle entering hip joint. Aspiration was performed and contrast dye was then injected into the joint to confirm intra-articular needle placement. Synovial fluid was then taken to the laboratory for immediate white blood cell count and gram stain. Aerobic, anaerobic, and fungal cultures were all incubated for a total of 24 days, with results being reported daily when positive and at 5, 14, and 24 days, respectively. The patient was maintained under anesthesia until the initial cell count was available. A decision was then made to either end the procedure or perform formal I&D of the hip. If aspiration fluid was frankly purulent based on the attending physician’s opinion, the decision to proceed with I&D was made before the synovial fluid cell count was determined. A drain was placed in all patients who underwent debridement and remained in place for at least 48 h.
At the institution performing aspiration at bedside (Institution B), aspiration was either performed by fellowship-trained pediatric orthopedic surgeons or fellowship-trained radiologists. No aspirations were performed in the OR. Antibiotics were held prior to the procedure. Ketamine was used for sedation. Sedation was performed by trained emergency room physicians. Patients were prepped with chlorhexidine, and a sterile field was created with drapes or sterile towels. Fluoroscopic or ultrasound guidance was used to visualize needle placement. Synovial fluid was obtained and processed in the same manner to Institution A. The patient was awoken following the completion of the procedure while awaiting initial cell count. If cell count raised suspicion for septic arthritis, the patient was then transported to the OR for formal I&D. A drain was placed in all patients undergoing I&D in a similar manner to Institution A.
Clinical outcomes
“Anesthesia time” was defined as time under general anesthesia in the OR. “Sedation time” was defined as time under ketamine sedation in the emergency department. Time under anesthesia in the OR was determined using the anesthesia start and end times documented in the electronic anesthesia medical record. Time under sedation in the emergency department was determined in a similar fashion using sedation start and end times. Surgical time was documented using incision start and final closure times. Aspiration results, final diagnosis, and treatment were confirmed by reviewing notes from the primary treatment team. Time spent in the MRI scanner under anesthesia was excluded from this analysis. Final follow-up was ascertained through the most recently documented musculoskeletal examination.
Statistical methods
The patient cohort and surgical data were assembled using electronic medical records and diagnostic codes. All statistical tests were two-sided and a p value <0.05 was considered significant. Microsoft Excel was utilized to track data and perform statistical calculations.
Results
A total of 305 patients were initially identified. Seventy-two of these patients were excluded as their hip aspiration was performed prior to arrival at either institution. Two hundred thirty-three patients with a mean follow-up of 34.2 months underwent hip aspiration and were identified for inclusion in the analysis. Seventy-five patients underwent hip aspiration in the OR under general anesthesia, and 158 patients underwent bedside aspiration under ketamine sedation. There were 97 females and 136 males with a mean age of 7.2 years (range: 0–17 years) and mean body mass index of 18.2 kg/m2 (range: 10.9–23.4 kg/m2). Demographic data for each cohort is described in Table 1.
Baseline demographics in patients undergoing aspiration in the operating room (Institution A) versus patients undergoing aspiration at bedside (Institution B).
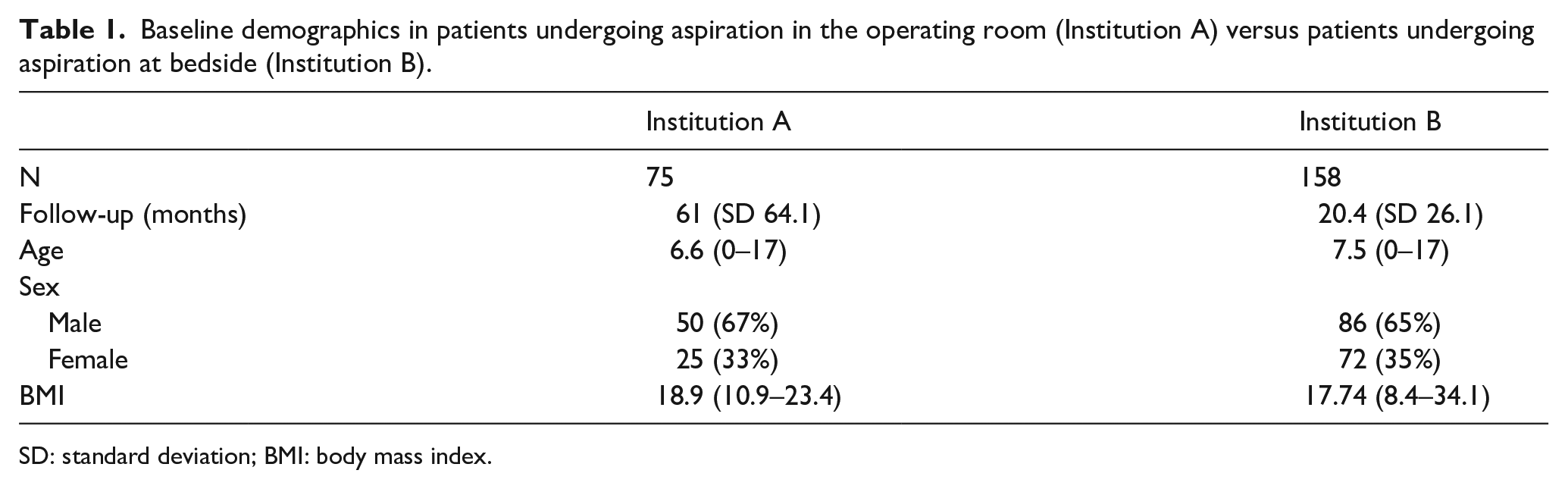
SD: standard deviation; BMI: body mass index.
Aspiration results
In total, aspiration was frankly purulent or resulted in a white blood cell count of greater than 50,000/mm3 in 110 (47%) patients. Nineteen (8%) aspirations resulted in a dry tap (4 [5%] OR aspirations and 15 [9%] bedside aspirations). These 19 patients were included in our negative aspiration cohort, and none of the 19 underwent I&D. Positive synovial fluid cultures were identified in 70 (30%) patients. Synovial fluid aspirations did not demonstrate bacterial growth in 162 (70%) patients at the end of the incubation period.
Final diagnosis and treatment
Final diagnoses included 100 (43%) isolated septic arthritis of the hip, 66 (28%) transient synovitis, 15 (6%) osteomyelitis, 15 (6%) Lyme arthritis, and 13 (6%) osteomyelitis and septic arthritis (Figures 1 and 2). One hundred seventeen (50%) patients underwent operative I&D, while 47 (20%) received antibiotic therapy alone after receiving an alternative diagnosis to septic arthritis. Less common treatments are described in Table 2. Seventy-eight (49%) patients who underwent bedside aspiration did not require operative intervention and therefore avoided general anesthesia altogether.
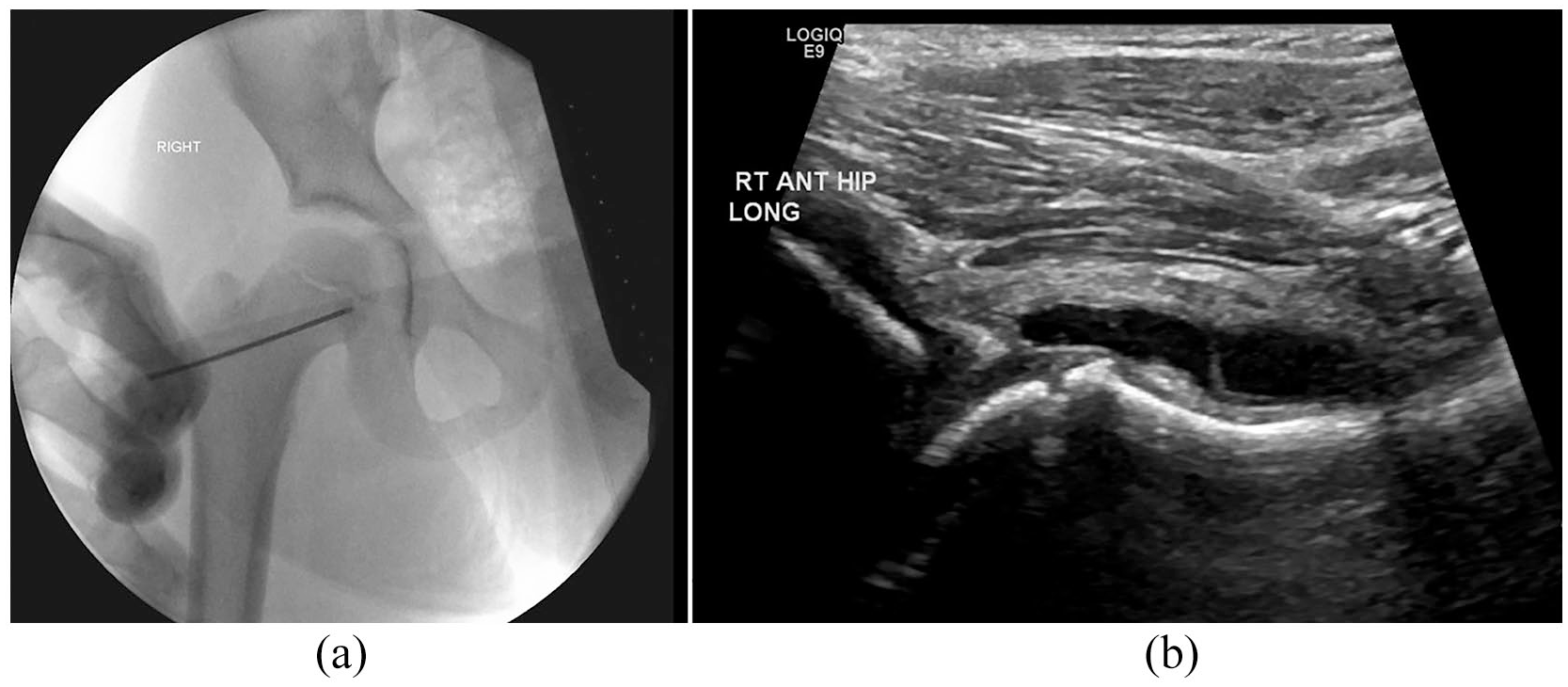
AP radiograph (a) and US image (b) of a 6-year-old male who spent 2 h 36 min under anesthesia for aspiration, irrigation, and debridement. Aspiration was positive for MSSA, and the patient was diagnosed with septic arthritis.
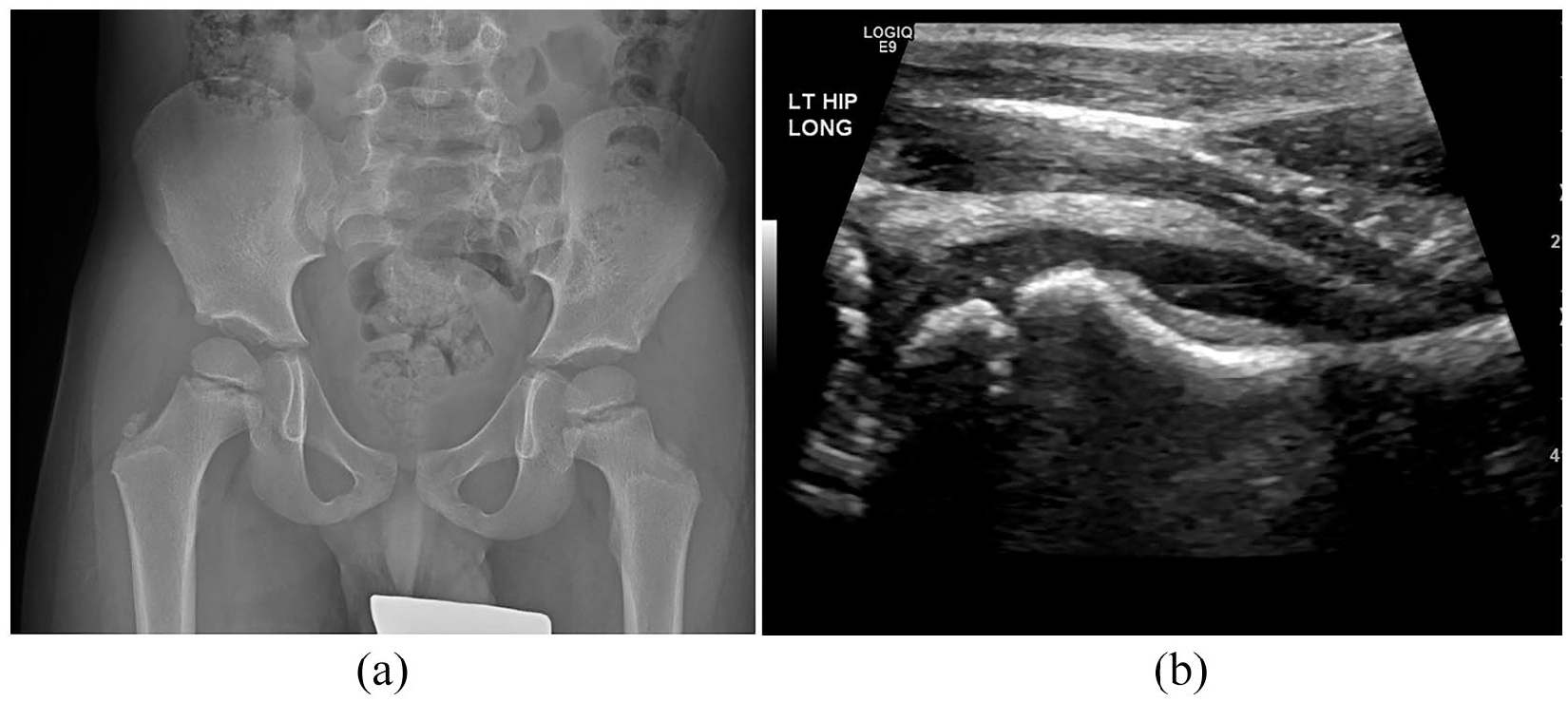
AP radiograph (a) and US image (b) of a 5-year-old male who spent 1 h 18 min under anesthesia. Aspiration yielded negative results, and the patient was diagnosed with transient synovitis 2 days later.
Final treatment for all included patients.

I&D: irrigation and debridement; IVIG: Intravenous immunoglobulin.
Mean time under general anesthesia
Patients with a negative aspiration performed in the OR averaged 87 min under anesthesia. Patients undergoing negative aspiration in the OR averaged 73 min under anesthesia when the procedure occurred before 5 pm, and 99 min under anesthesia when the procedure occurred after 5 pm (p < 0.01; Table 3). Patients with a negative aspiration performed at bedside averaged 0 min under anesthesia (p < 0.01) and 29 min under ketamine sedation. There was no change in mean time under sedation after 5 pm.
Mean time under anesthesia for pediatric patients who underwent hip aspiration in the operating room (Institution A) versus at the bedside (Institution B).
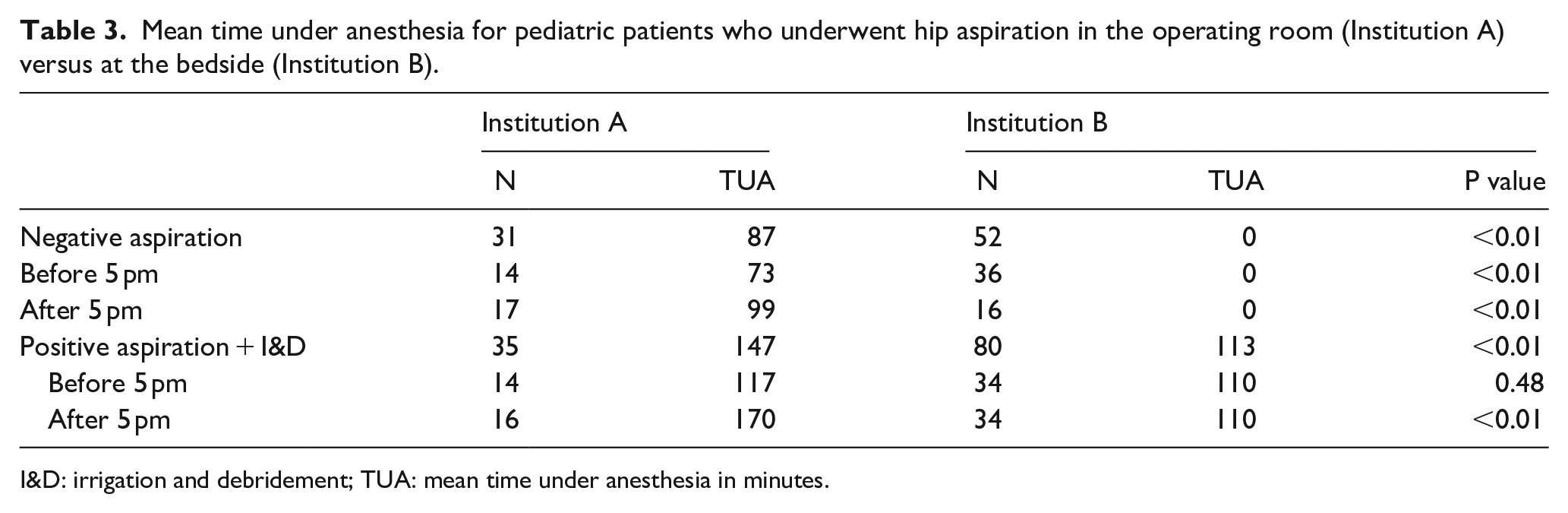
I&D: irrigation and debridement; TUA: mean time under anesthesia in minutes.
Patients with a positive aspiration (elevated cell count and/or frank purulence) in the OR who underwent subsequent I&D had a mean time under anesthesia of 147 min. Patients with a positive aspiration at bedside prior to I&D had a mean time under anesthesia of 113 min (p < 0.01) and mean time under sedation of 26 min. Patients with a positive aspiration in the OR and subsequent I&D before 5 pm averaged 117 min under anesthesia, while those patients with a positive aspiration at bedside and subsequent I&D before 5 pm averaged 110 min under anesthesia. Patients with a positive aspiration in the OR and subsequent I&D after 5 pm averaged 170 min under anesthesia, while those patients with a positive aspiration at bedside and subsequent I&D after 5 pm averaged 115 min under anesthesia (p < 0.01; Table 3). The mean time under sedation for patients with a positive aspiration at bedside remained 26 min both before 5 pm and after 5 pm.
In addition to time under anesthesia, surgical time was also analyzed. Time spent waiting for cell count to return was included in the surgical time of patients who underwent aspiration in the OR. Patients who underwent aspiration in the OR with subsequent I&D had a mean surgical time of 79 min before 5 pm and 95 min after 5 pm, while patients who underwent aspiration at bedside with subsequent I&D had a mean surgical time of 58 min before 5 pm (p = 0.01) and 58 min after 5 pm (p < 0.01).
Subgroup analysis of the cohort who underwent I&D after positive aspiration in the OR demonstrated that 16 (50%) patients proceeded directly to I&D after frank purulence was encountered, and 16 (50%) patients were held under anesthesia while awaiting cell count. Operative reports were unclear in the remaining three patients. Those patients who proceeded directly to I&D after aspiration spent an average of 115 min under anesthesia, while those patients who waited for the return of cell count prior to I&D spent an average of 180 min under anesthesia (p < 0.01). Time under anesthesia among those patients who waited for the return of cell count results was significantly prolonged after hours (p = 0.02, Table 4). The surgical time in patients who proceeded directly to I&D was 67 min and the surgical time in patients who waited for the return of cell count was 113 min. Those who proceeded directly to I&D had a similar surgical time to the cohort of patients who underwent aspiration at bedside (67 min versus 58 min, p = 0.18). These data suggest that the mean time in the OR waiting for cell count results varied from 33 to 89 min, depending on the time of day.
Time under anesthesia for patients undergoing aspiration in the operating room and subsequent I&D before and after typical work hours.

I&D: irrigation and debridement; TUA: mean time under anesthesia in minutes.
The longest anesthetic time in this study was 335 min, 110 min lengthier than the next patient. This patient was taken to the OR after hours with a subsequent positive aspiration and I&D. His anesthetic record specifically demonstrates a delay awaiting aspiration results of 103 min, as well as prolonged OR setup time after the positive results became known.
Discussion
Differentiating septic arthritis from other less-emergent diagnoses remains a challenge in the pediatric population. Historically, patients undergoing aspiration in the evaluation for septic arthritis have been taken to the OR because it was assumed that a debridement procedure would be performed in most cases. However, in this study, only 50% of patients ultimately required operative I&D as part of their treatment plan. Limiting or reducing anesthetic time is therefore a significant opportunity for care improvement in the management of pediatric septic hip arthritis. Many institutions have successfully transitioned to performing aspirations for pediatric patients in the emergency department, with some utilizing only local anesthesia. 16 This raises the question: does aspiration in the OR unnecessarily subject 50% of patients to general anesthesia?
OR efficiency has been shown to vary according to the time of day with decreased OR efficiency occurring after hours. 17 In this study, patients with a negative aspiration in the OR were subjected to an average of 87 min of general anesthesia, compared to only 29 min of sedation when performed at bedside. Negative aspirations performed in the OR after business hours took 26 min longer than when the procedure was performed between 7 am and 5 pm (p < 0.01). Similarly, those patients who underwent positive aspiration in the OR followed by I&D after 5 pm averaged 53 min longer under anesthesia than patients who underwent this operation during normal operating hours (p < 0.01). This suggests that the benefit of transitioning to bedside hip aspiration may be magnified when performed after hours.
Patients who required operative I&D after aspiration in the OR were anesthetized for an average of 147 min. Patients spent 115 min under anesthesia when I&D was performed immediately after encountering frank purulence which was not statistically different than those patients who underwent bedside aspiration and subsequent I&D (113 min, p = 0.81). A significant delay did occur in those patients with a positive aspiration performed in the OR when there was a wait for the return of initial cell count both before 5 pm (148 min; p = 0.02) and after 5 pm (204 min; p < 0.01). This suggests that delays in obtaining the cell count leads to prolonged anesthetic times which were exacerbated by almost an hour after 5 pm. Therefore, even among those patients for whom anesthesia is required for debridement, eliminating the added time under anesthesia waiting for cell count would be beneficial, as children are at increased risk for learning disabilities with longer cumulative duration of anesthesia exposure. 12 The largest of such studies to date, the Mayo Anesthesia Safety in Kids (MASK) study, demonstrated similar concerning trends in behavior and fine-motor skills in children with multiple anesthesia exposures. 18 Performing aspiration prior to induction of anesthesia for operative I&D would also decrease the time to antibiotic administration, which is known to correlate strongly with improved outcomes when treating infection.19–22 This has been demonstrated in patients with sepsis, pneumonia, meningitis, urinary tract infection, and intra-abdominal infection. While there is a relative paucity of data pertaining to antibiotic administration delay in pediatric septic arthritis, a benefit from timely antibiotic administration can be inferred from these relevant data.
Hip aspiration at bedside in the emergency room offers several advantages from a resource perspective compared to the OR aspiration strategy. Bedside aspiration in the setting of non-septic hip effusion may be therapeutic as it relieves capsular distention, leading to rapid pain relief and shorter hospital stays regardless of the ultimate diagnosis. 11 Furthermore, some data suggest that serial ultrasound-guided aspirations alone may be a safe method of definitive treatment of septic arthritis of the hip joint although debridement remains the common standard of care if the child’s medical condition allows. 23
Technique articles have been published to properly educate residents and staff physicians to safely perform a bedside hip aspiration under ultrasound guidance.16,24,25 These illustrate a step-by-step approach for patient positioning, aspiration, use of ultrasound, and evaluation of need for sedation. Sedation, in the experience of Thapa et al., 16 for instance, was often deemed unnecessary with the help of child life services. Even if sedation is required, overall time exposed to these medications is limited. To illustrate, the average time under ketamine sedation during bedside aspiration in our cohort was 29 min, far less than any patient in our general anesthetic cohort.
Despite the important conclusions made from this study, they must be considered in light of the following limitations. The strength of our conclusions is limited by the retrospective study design which may introduce bias due to data inaccuracies associated with the medical record. The generalizability of the study can also be questioned as the outcomes from two different academic institutions with two different medical records were investigated. Data were collected and recorded differently between the two institutions, and this has the potential to introduce error when data are not recorded in the same fashion. Certain constant elements such as anesthetic time, time in the OR, and general management of emergent cases is likely different between these two institutions which can further skew the data. However, despite these potential differences, one cannot overlook the fact that in the bedside aspiration cohort, almost half of these children did not require operative intervention, thus avoiding general anesthetic. We acknowledge that the documentation of anesthesia time can be variable based on a variety of factors including patient cooperation, intravenous (IV) access, and the presence or absence of complicating medical factors. Finally, the risks of anesthesia in children have not been well established with some studies offering conflicting data (no change in intelligence quotient, for instance). 14 There remains significant value in the present report; however, as it is one of the largest retrospective reviews of pediatric patients who have undergone hip aspiration for suspected septic arthritis to date.
Despite these limitations, we believe important conclusions can be made from this study. Pediatric hip aspiration performed in the OR results in prolonged anesthesia times while synovial fluid is transported and processed. The delay in laboratory results is significantly longer after 5 pm. Aspiration performed at bedside resulted in significantly less anesthesia exposure, and 49% of patients undergoing bedside aspiration avoided general anesthesia altogether.
Footnotes
Author contributions
Z.V.B. performed measurements, statistical analysis, and manuscript preparation. P.P. performed measurements and statistical analysis. M.E.T. performed measurements. H.P. performed cohort identification and study design. W.J.S., A.A.S., A.N.L., B.J.S., and T.A.M. performed study design and manuscript preparation.
Compliance with ethical standards
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The authors have no relevant conflicts of interest to disclose. This study involved data review of human participants and is in compliance with the Helsinki declaration. The study was approved by our Institutional Review Board.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
