Abstract
The use of microsurgery has spread during the last decades, making resolvable many complex defects considered hitherto inapproachable. Although the small vessel diameter in children was initially considered a technical limitation, the increase in microsurgical expertise over the past three decades allowed us to manage many pediatric conditions by means of free tissue transfers. Pediatric microsurgery has been shown to be feasible, gaining a prominent place in the treatment of children affected by limb malformations, tumors, nerve injuries, and post-traumatic defects. The aim of this current concepts review is to describe the more frequent pediatric upper limb conditions in which the use of microsurgical reconstructions should be considered in the range of treatment options.
Introduction
The use of microsurgery has spread during the last decades, making many complex defects, considered hitherto inapproachable, resolvable.
Although the small vessel diameter in children was initially considered a technical limitation, the increase in microsurgical expertise over the past three decades allowed us to manage many pediatric conditions by means of free tissue transfers.
However, pediatric orthopedists have for years looked at microsurgical techniques considering them complex, with uncertain results, high risk of failure, high operating room occupation, and a long learning curve.
One of the causes of this mistrust probably arises from the absence of training in microsurgery for orthopedic residents apart from those passionate for hand or nerve surgeries.
Nevertheless, as in many fields of adult reconstructive surgery, pediatric microsurgery has been shown to be feasible, gaining a prominent place in the treatment of children affected by limb malformations, tumors, nerve injuries, and post-traumatic defects.
The aim of this review is to describe the more frequent pediatric upper limb conditions in which the use of microsurgical reconstructions should be considered in the range of treatment options.
Obstetrical brachial plexus injuries
Since the initial descriptions at the beginning of the past century, the management of obstetrical brachial plexus injuries (OBPI) has significantly changed. The introduction of operative microscopes and surgical instruments dedicated to microsurgery since the 1960s, in addition to the improvement in knowledge about nerve anatomy and pathophysiology, has revolutionized OBPI surgery and outcomes.
The efforts of Seddon, Millesi, and Narakas in approaching adult brachial plexus injuries were followed by Alain Gilbert, who endeavored to apply the concepts known to the reconstruction of the pediatric plexus. 1
The decision whether or not to operate on an obstetrical palsy still remains a challenge even for experienced surgeons; several parameters must be considered, such as the level of palsy, patient’s age, and any comorbidities, especially perinatal brain injuries.
Although the role of a prompt diagnosis and an early approach for total palsy without hand recovery in the first months remains unchanged, the classic dogma of considering the biceps recovery (until the third month of life) as a guide to decide whether surgery is needed or not has been reconsidered for C5C6 and C5C6–C7 palsy.
Currently, for the last two types of paralysis, primary brachial plexus repair should be performed between 3 and 9 months of age, and recent studies appear to show no superior results for reconstructions carried out before or after 6 months of age.2,3
Nevertheless, the well-known age limit of 1 age for performing brachial plexus reconstruction remains unchanged.
Nerve reconstruction continues to be based on the use of autologous nerve grafts to bridge the gap resulting from neuroma resection and on the use of concomitant nerve transfers, especially in cases of gaps that cannot be filled by grafting alone (Figure 1).
Obstetrical brachial plexus Narakas 2 palsy; a neuroma involved upper and medium trunk (a) and it was resected and reconstructed by means of sural nerve graft (b).
Although neurolysis still remains in the armamentarium of many surgeons, it as a single surgical act in OBPI treatment has now been shown to be null and void in terms of improving recovery. 4
An accurate surgical technique is mandatory in terms of intraoperative neurostimulation, evaluation of available roots, and suturing of grafts or nerve transfers, whether performed with micro-sutures or fibrin glue alone, depending on the surgeon’s habits.
During the early 2000s, the role of nerve transfers in pediatric brachial plexus surgery has significantly increased, paving the way for a delayed approach in cases of partial spontaneous recovery, isolated functional deficit, failure of primary repair, or late presentation of the patients. 5 Suprascapular, axillary, musculocutaneous, and radial nerves are the main targets using accessory spinal, intercostal, and branches of the ulnar or median nerves as principal donors of motor fibers.
The key to extending reparative surgery of the obstetrical plexus beyond 1 year of age arises from electromyographic studies that have made it possible to understand that the child’s muscle is rarely completely denervated as in adults. 6
It allows reconstructive microsurgeons to gain time to assess the recovery, both spontaneous and after graft repair, before deciding whether any other procedures are necessary (Figure 2).
Late nerve transfer procedure (accessory spinal to suprascapular) for loss of external rotation in obstetrical brachial plexus sequelae (a, b) and muscle recovery at 2 years of follow-up (c, d).
However, although distal nerve transfers are widening the options for approaching OBPI, these should not diminish the surgeons’ ability to decide when to explore the brachial plexus and reconstruct it with grafts; it would indeed be a great loss if new generations of plexus surgeons lose the ability to carry out an anatomical reconstruction of the plexus, thus becoming only “distal transfer surgeons.”7,8
Congenital pseudarthrosis of the forearm
Over the recent decades, the use of free vascularized fibular grafts (FVFGs) has established itself as the primary treatment modality in pediatric cases with congenital pseudarthrosis of the forearm (CPF) (Figure 3).
A treatment course of a 4-year-old boy with congenital radius pseudarthrosis is presented (a). Following several previous attempts to unite a supposed distal radius fracture at the patient’s home institution, he was referred showing a narrow, dysplastic radius pseudarthrosis with a K-wire in situ (b). Solid union was achieved using a free vascularized fibular transfer (c). No attempt was made to correct the radial length discrepancy, which will be addressed in a future lengthening procedure. Preoperative (d) and postoperative (e) images are shown.
Since its first description by Allieu et al. 9 in 1981, numerous case reports and small case series have been published mentioning exclusively vascularized fibular transfers to achieve bone union in this rare entity. With an incidence of only 2 per 1 million, 10 this pathology can affect the ulna, radius, or even fewer instances in both forearm bones.11–13 It is commonly found in conjunction with neurofibromatosis (NF) type I. 14 As with other congenital pseudarthrosis, such as in the tibia, the main treatment problem remains to achieve a sound bone union in the area of the dysplastic, malformed bone(s). Moreover, surgical treatment provides a stable, growing forearm with a reconstituted ulnocarpal joint compartment and a stable radial head.
While a few reports have attempted to achieve union using non-vascularized bone grafts (NVBG) with internal fixation,15,16 these techniques have almost uniformly provided inferior results in this pathology. In this regard, some authors have stated that bone union is possible in the absence of NF1. 17 However, given the literature of the last century, such successes are rather rare exceptions. As such, CPF cases are unfortunately commonly misdiagnosed as normal ulnar fractures and may undergo a plethora of conservative and/or operative interventions before bone stabilization and union can eventually be achieved. To date, FVFG with or without fibular epiphyseal transfer 18 and, less commonly, the one-bone forearm procedure 2 are preferred by most treating surgeons. The latter may achieve a stable, united forearm at the cost of a loss of forearm rotation. It should be reserved as a salvage procedure for cases with irreducible radial head dislocation or those with an insufficient distal bone stock to adequately secure a graft. In contrast, FVFG can be performed as early as 3 years of age. 19 This can help to avoid potentially irreversible mid-term sequelae pertinent to the disintegration of the dysplastic distal ulna (i.e. bowing, radial head dislocation). It can be performed using a conventional technique or in a double-barrel manner, 20 which is especially attributable to very distal pseudarthrosis to increase bone stock width.
A recent meta-analysis by Sonna et al. systematically reviewed the available literature on this topic. Overall, 55 articles with 94 cases undergoing various techniques were reviewed. Of these, 36 patients underwent primary or secondary FVFGs. Univariate analysis revealed that FVFG had the highest healing rate (100%) compared to the non-operative means (0%) and NVBG (70%). In the presence of NF1, FVFG showed significantly better healing than NVBG. Moreover, when performing a multivariate analysis, the authors found that FVFG healed much better than NVBG with a mean odds ratio (OR) of 25.7 (up to 466 in the most impressive cases). Finally, the proportion of patients who healed after the first intervention was also much higher in the vascularized group (OR = 10.2; 32/35 vs 15/30 in the NVBG).
Another recent large analysis arrived at similar conclusions. 21 Here, 47 studies with 84 cases were evaluated. The authors found that the healing rates of one-bone forearm procedures yielded the best union rate (92%), followed by FVFG (87%). However, it was concluded that, as mentioned before, FVFG should be pursued in the first attempt, and one-bone forearm procedures should be reserved for revision cases. Irrespective of which technique is preferred, most authors would agree that wide bone resection and stable fixation are critical to achieve successful bone union.
In addition to achieving a successful union following FVFG surgery, one must bear in mind that bone union may take quite some time, and several problems may occur. With regard to union time, Mathoulin et al. 22 mentioned consolidation time of a mean of 6 months (3–24 months). This was corroborated by Bauer et al., who found a need for a healing time of even 10 months (3–18 months). Complications may rarely include nonunion at the distal junction, 23 graft fracture, 24 symptomatic hardware (BAE), limited forearm motion, and donor site morbidity. Thus, additional surgery may be necessary over the course of treatment. 25
To maintain a reasonable ulna length during the course of treatment and growth, FVFG has been shown to be successfully lengthened after incorporation using osteodistraction with external fixators. Distraction distances of 2–4.5 cm have been reported for this indication. 13
The donor morbidity rate is not low and has to be examined with the parents: flexor hallucis longus flexion contracture is very common and tibiofibular fusion is indicated in younger children and when the distal fibula remnant is shorter than 8–10 cm to avoid ankle valgus deformity.
Microsurgery in traumatic disorders
Intercalary bone defect
Traumatic segmental diaphyseal bone defects >4–6 cm show a high rate of unsuccessful healing following bone grafting and fixation. 26 This figure might be different in children, probably being inverse to the age of the child. Vascularized bone transplants for reconstruction are a surgical option for the treatment of large bone defects, allowing for a faster and higher rate of consolidation than NVBG. The main alternative to bone flaps is bone transport; no outcome differences exist between both techniques except for very large defects in which FVFG results are superior. 27 Another advantage of the use of an FVFG is the possibility of addressing skin defects by associating a fasciocutaneous flap.
The fibula flap, based on the peroneal vessels, is the most commonly used vascularized bone graft. It has the capacity to enlarge in order to adapt to mechanical solicitations with an approximate rate of 3% per month until 2.5 years afterward 28 (at the pediatric age, the vascularized fibular graft ends can be prolonged with vascularized periosteum, which results in a very fast consolidation).29,30
Alternative bone flaps are the vascularized iliac crest and ribs.
Recalcitrant nonunion
Persistent traumatic nonunion in children is uncommon, occurring in most cases in the context of massive allografts and infections. 31 Provision of osteogenic and angiogenic resources is mandatory in multioperated nonunions to solve this biologically unfavorable situation. Recently described vascularized periosteal grafts (VPGs) in children have shown an enormous and fast-healing capacity, which is the ideal treatment for this problem.32,33
Local pedicled VPGs are used whenever possible. Examples of local pedicled VPG are the following:
Lateral humeral periosteal flap, based on the posterior radial collateral vessel, to treat humeral lateral condyle nonunion;
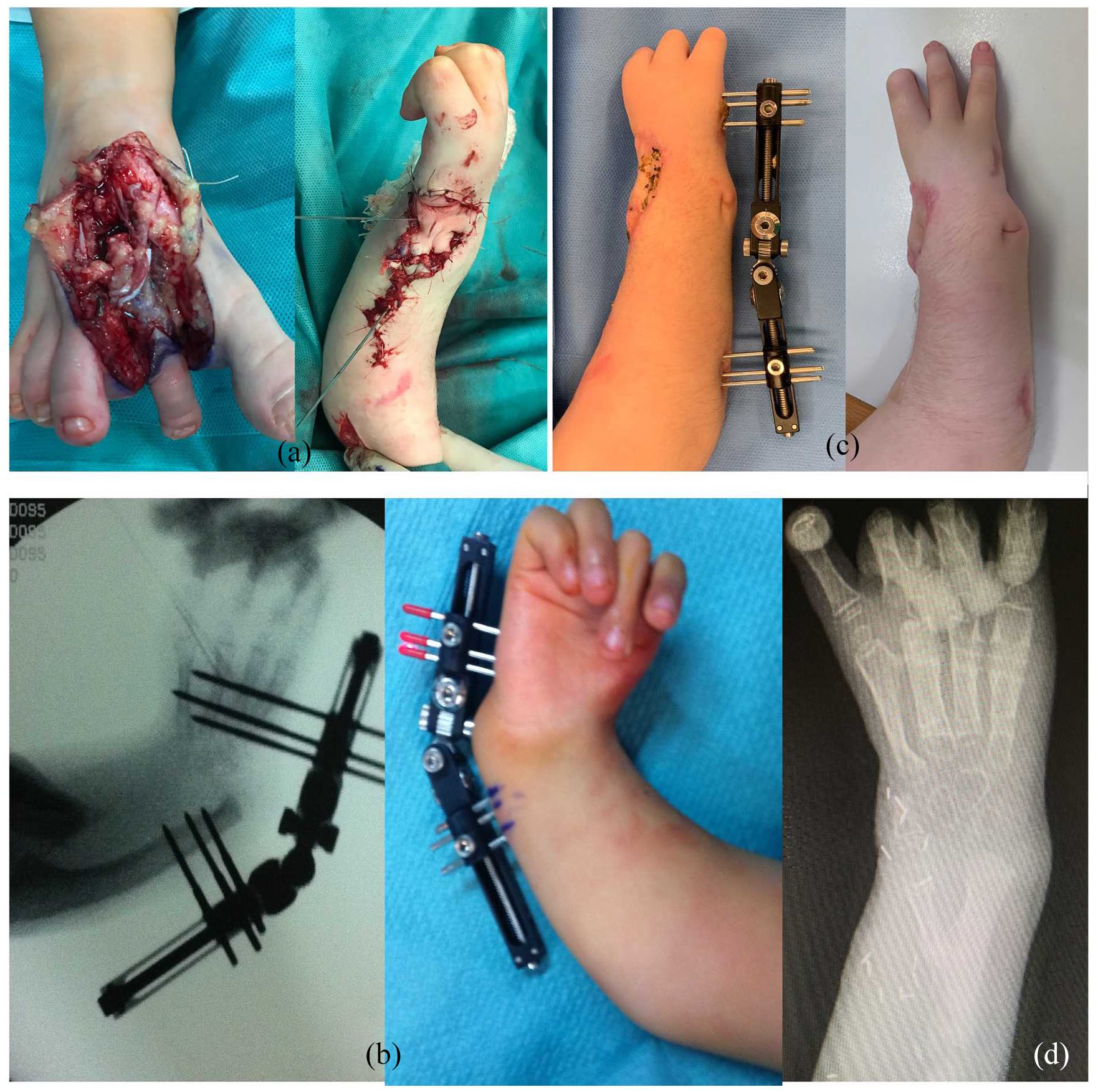
First metacarpal periosteal flap, based on the first dorsal metacarpal artery, for complex scaphoid nonunion (Figure 4);
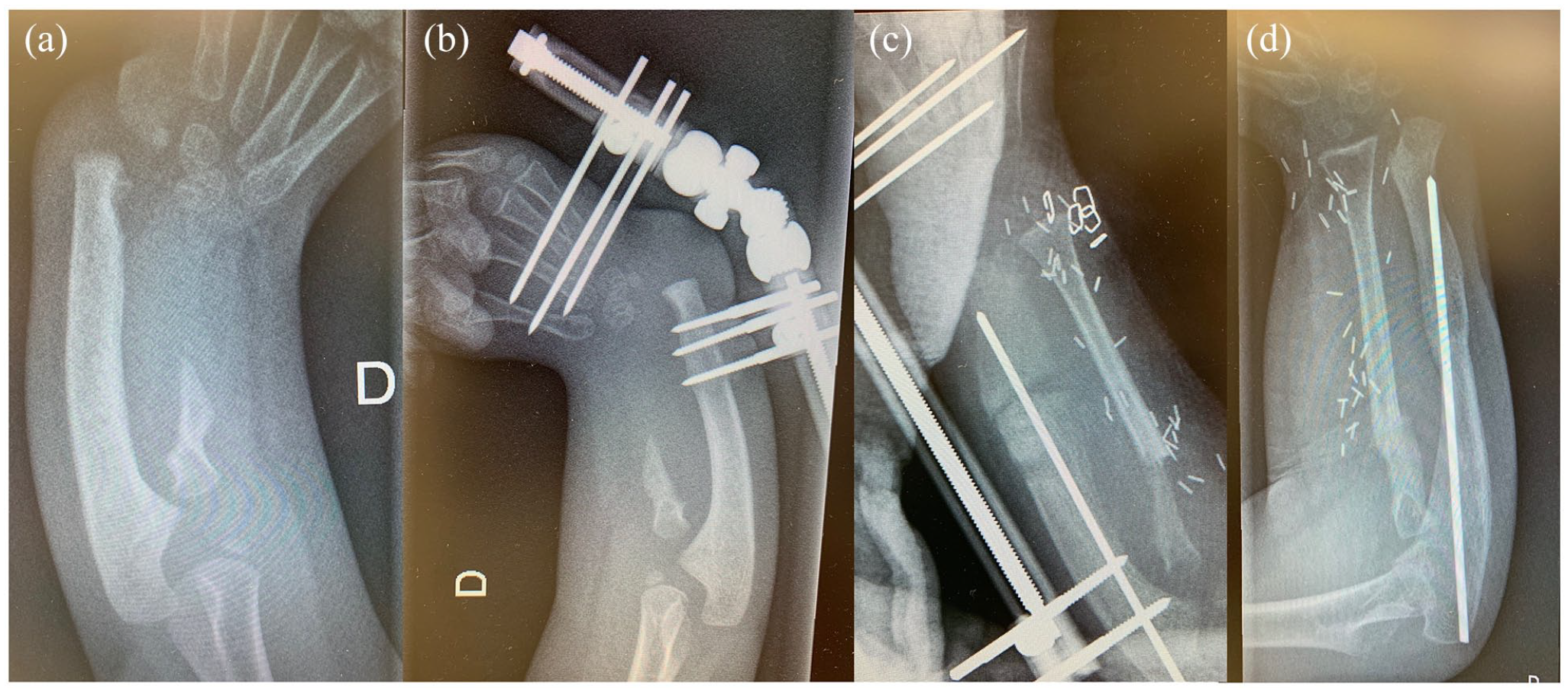
Ulna VPG based on the posterior interosseous vessels to solve radial nonunion (Figure 5) and radial VPG based on the anterior interosseous vessels to treat ulnar nonunions.34–36
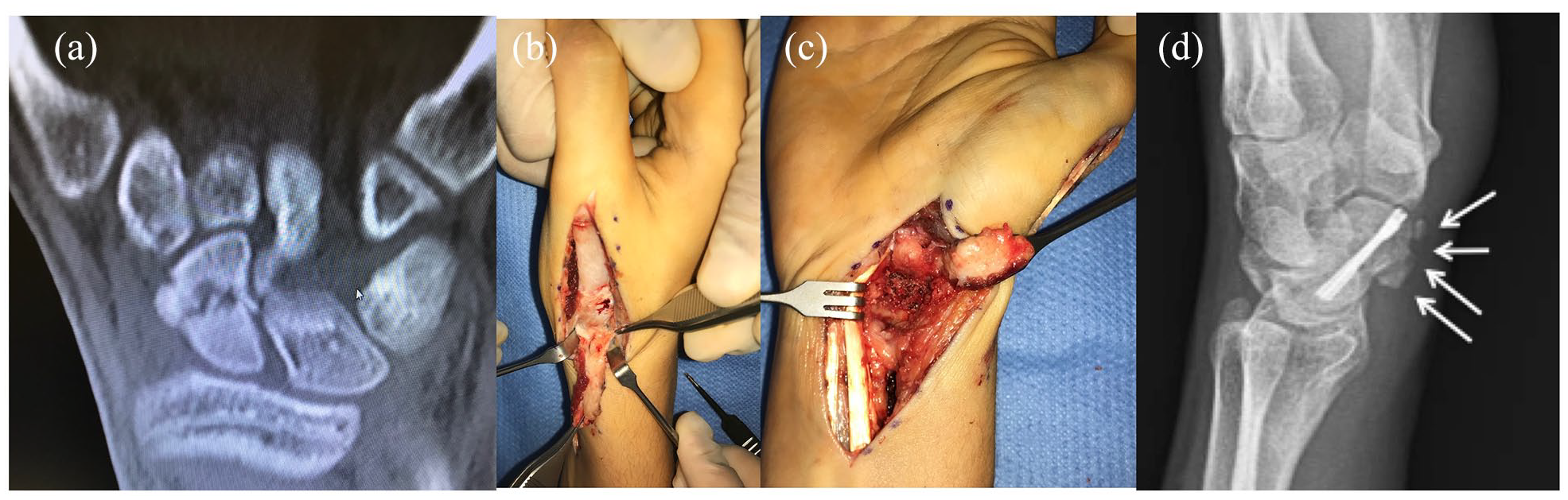
Adolescent scaphoid nonunion (a). Using a dorsal approach, a vascularized first metacarpal periosteal flap was obtained (b). The pseudarthrosis site was debrided, fixed with cannulated screws, and distal radius bone graft was added (c). Finally, the nonunion site was covered with the flap. Union was obtained in 6 weeks (d).
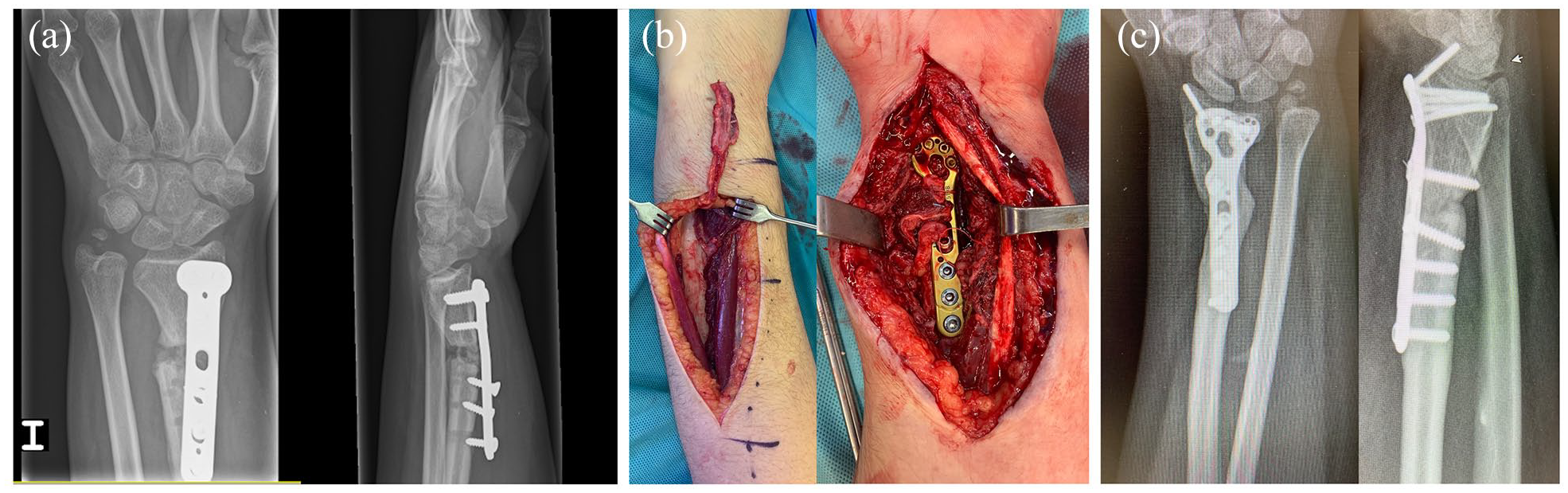
Multioperated distal radius fracture in an adolescent with persistent nonunion (a). After fixation revision, a pedicled vascularized ulnar periosteal graft was used to enhance union (b). Bone healing was obtained 8 weeks after surgery (c).
Free VPGs are used when local options are not available or a larger periosteum is required. The vascularized tibial periostal graft could be considered as the first choice, although the main leg vascular axis is sacrificed (anterior tibial vessels) as it is easier to harvest and provide larger flaps 37 (Figure 6).
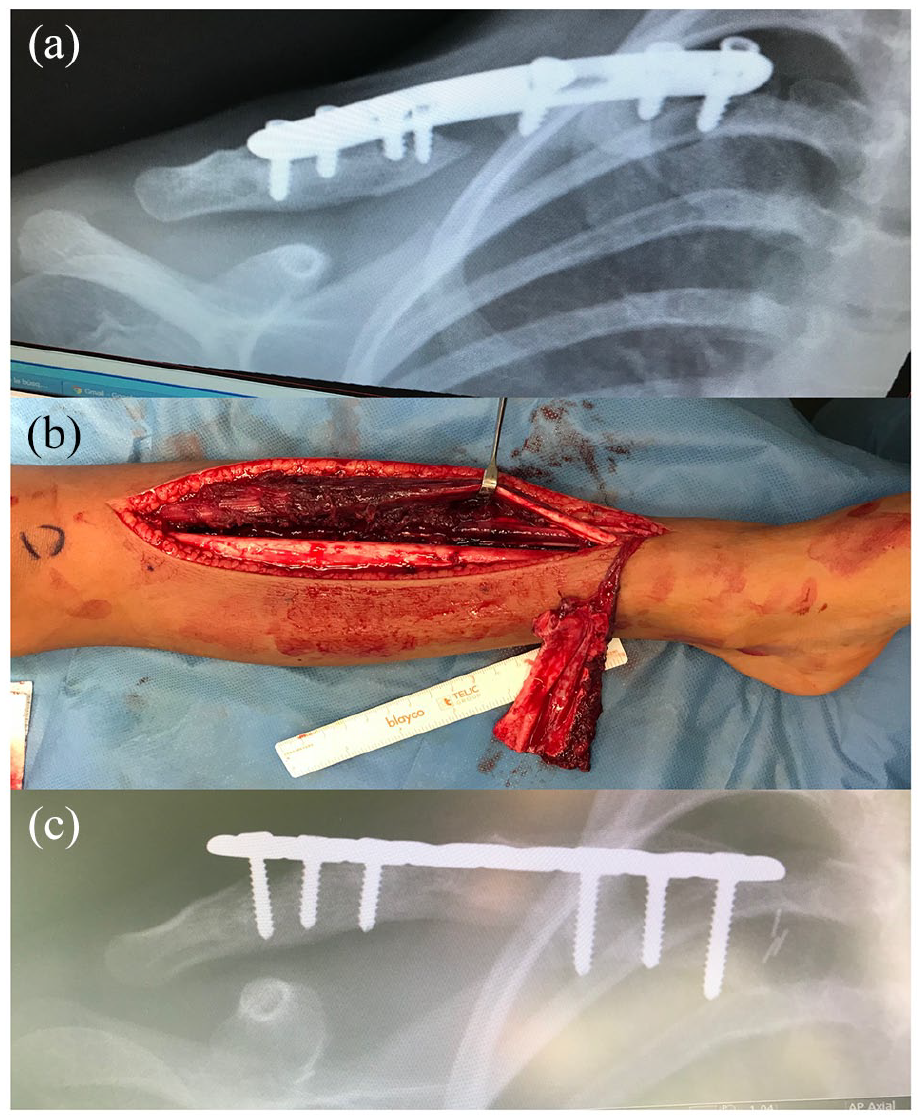
Multioperated clavicle with persistent nonunion and 3 cm bone defect (a). A vascularized tibial periosteal graft (b) without bone grafting was used after fixation revision, resulting in a fast bone consolidation (c).
Other free options are the vascularized fibular periosteal graft and vascularized iliac periosteal graft and medial femoral condyle. 38
VPGs are not combined with bone grafts, except for the first dorsal metacarpal artery flap for scaphoid nonunion; thus, all bones originated from the flap.
Limb amputation
Indications for digit replantation or toe transfer following traumatic amputation in children are similar to those in adults:
Thumb amputation;
Single-digit amputation distal to the PIP (preserved PIP and FDS) 39 (Figure 7);
Multiple digit amputation. 40
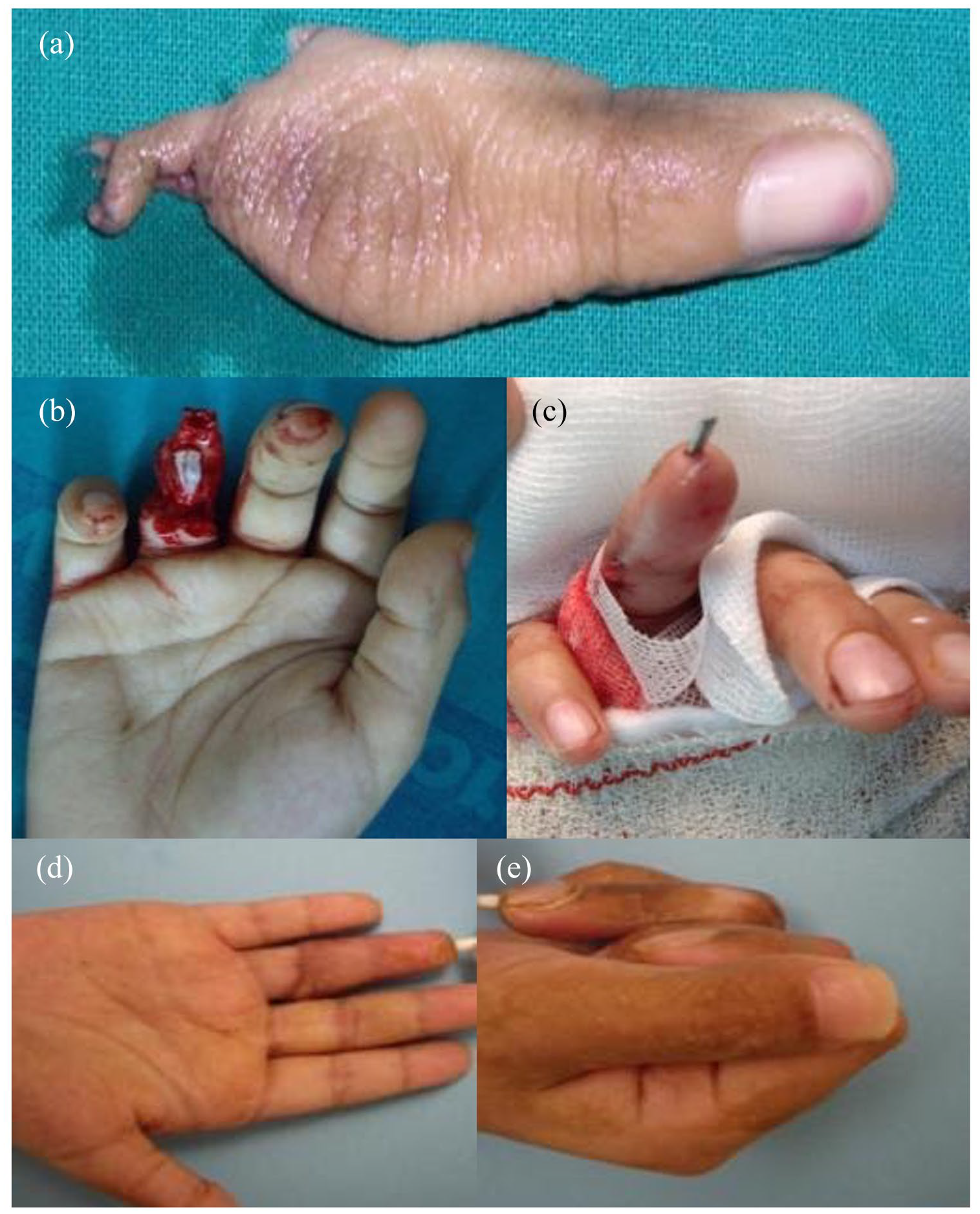
A 10-year-old patient with a ring finger injury (a). PIP and FDS insertion are preserved (b); thus, replantation was performed (c). Full active PIP ROM was obtained (d, e).
Nevertheless, the best results in children allow us to consider any level of amputation as a potential indication to attempt a replantation. 41
Partial hallux or second toe transfer can be used for both finger and thumb reconstruction when the amputated segment is not available or usable 42 (Figure 8).
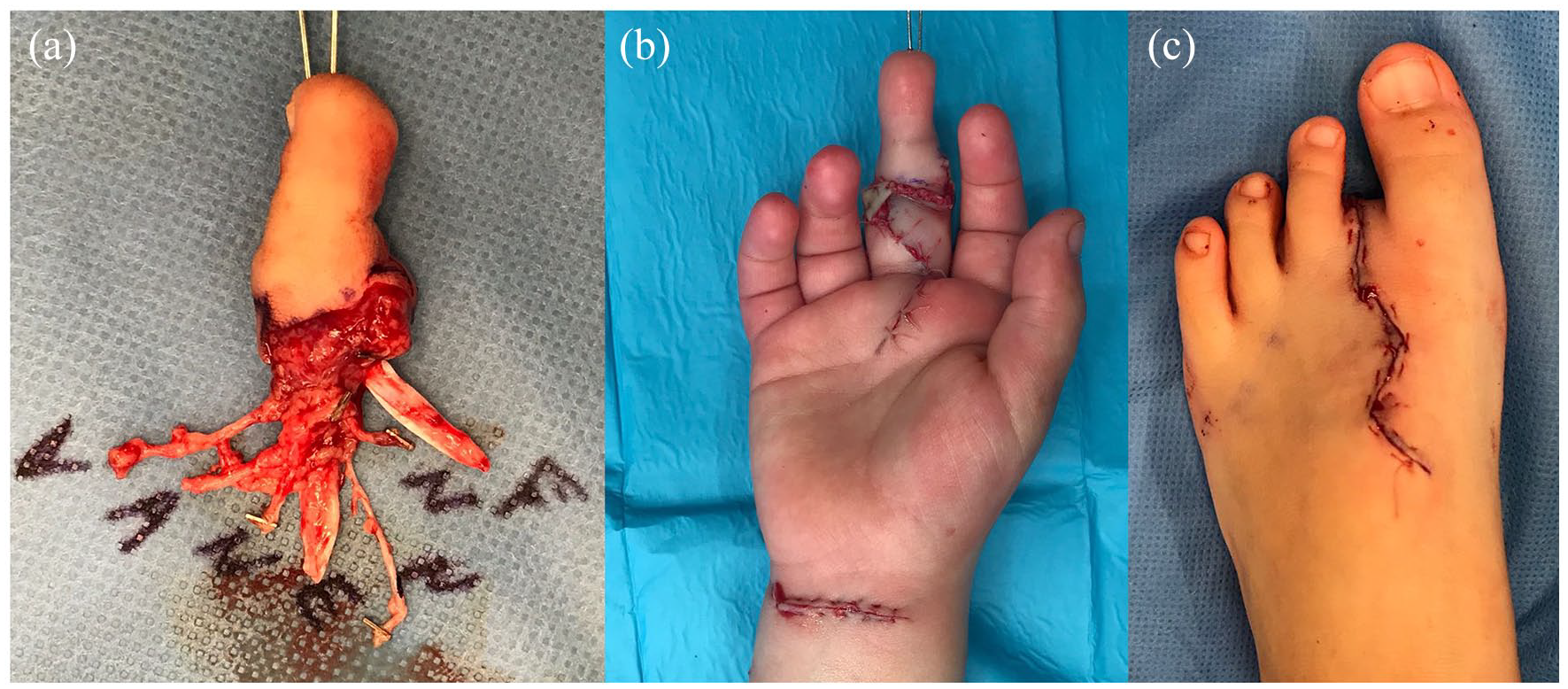
Middle finger amputation in a 4-year-old patient with unavailable amputated segment. (a) An early second toe transfer was performed as PIP and FDS were preserved (b and c).
Major amputations are an indication for replantation in children when the timing and condition of the amputated segment allow for it.43,44
Maximum ischemia times are approximately 12 h of warm and 24 h of cold time for digits, with shorter times tolerated for more proximal amputations. Success rates vary; survival is predicted in part by the mechanism of injury, with sharp cut injuries having better outcomes.
Traumatic soft tissue loss
Indications of vascularized soft tissue transfers (fasciocutaneous or muscular) are similar to those in adults: exposed bone devoid of periosteum, joints, and large multitissutal defects with exposure of neurovascular structures.
Pedicled flaps are used whenever possible; otherwise, a free flap is indicated. Flap selection depends on the size and width of the defect.
The anterolateral thigh flap (ALT flap) is the most commonly used free flap. The rate of success following soft tissue transfer in children is similar to that in adults. 45
Microsurgery in congenital deficiencies
Congenital absent fingers
Congenital absent fingers amenable to vascularized toe transfer are more frequent in the context of symbrachydactyly and amniotic band syndrome. 46
Symbrachydactyly forms with the presence of a thumb and a digit to act as a post and will develop a functional pinch; thus, toe transfers are not generally indicated. Thus, indications for toe transfer in symbrachydactyly can be adactylous and monodactylous forms.
Indications for toe transfer in amniotic band syndrome are similar to symbrachydactyly and traumatic conditions, such as monodactylous and adactylous forms, thumb amputations, and digit amputations. Although toe transfers are a good “theoretical” solution, many parents are reluctant, as donor morbidity can be significant. Long-term problems after toe transfer on the foot must be discussed with families before endeavoring to perform microsurgical toe transfer.
Radial longitudinal deficiency
The management of radial longitudinal deficiency (RLD) remains a challenge because of the high risk of radial deviation recurrence and wrist stiffness. 47
Classical techniques of carpus radialization continue to be adopted by many surgeons, while centralization tends toward progressive abandonment, especially for the risk of distal ulnar physis injury (both for surgical dissection and for transitory fixation with a nail) which might result in a further shortening of the forearm.
Unfortunately, the risk of recurrence of the native deviation is inherent in the initial malformation due to the absence of the radial column of the wrist .
Microsurgical procedures could provide a growing radial support with a potentially lower risk of a stiff wrist.
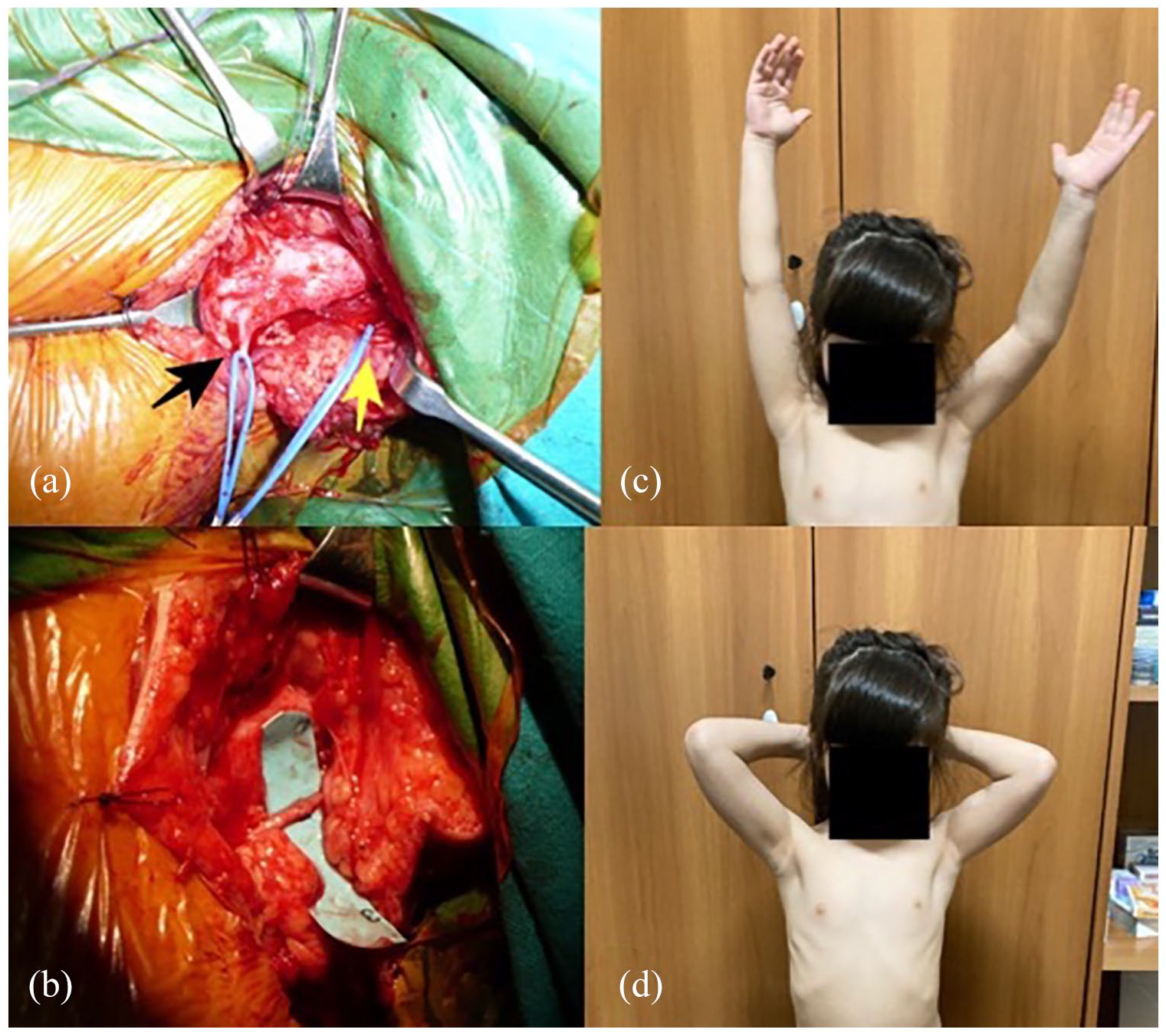
Vilkki 48 and Nayar et al. 49 described a two-stage procedure consisting of the transfer of the second metatarsophalangeal joint with the above-mentioned objectives (Figure 9).
Morphological result of a Vilkki procedure in a Bayne Type IV radial clubhand. The second toe MTF joint is used to provide a growing lateral wrist support. The toe skin is used for lateral wrist coverage and vascular monitoring. (a, b) Radiological images of Bayne Type IV radial clubhand showing progressive soft tissue distraction and once the flap is consolidated (c, d).
After initial soft tissue distraction to position the carpus over the ulnar head, the microsurgical transfer achieves the metatarsophalangeal transfer into a y-shaped position with fixation into the ulna. This procedure resulted in excellent wrist motion and alignment with a growth rate of the two transplanted physes (first phalanx base and metacarpal head) mirroring the ulnar physis. However, after adolescent growth, a growing imbalance occurs, resulting in a mean wrist radial deviation of 30°. Due to the high demands and technical difficulties associated with this procedure, only a few centers worldwide use it routinely.
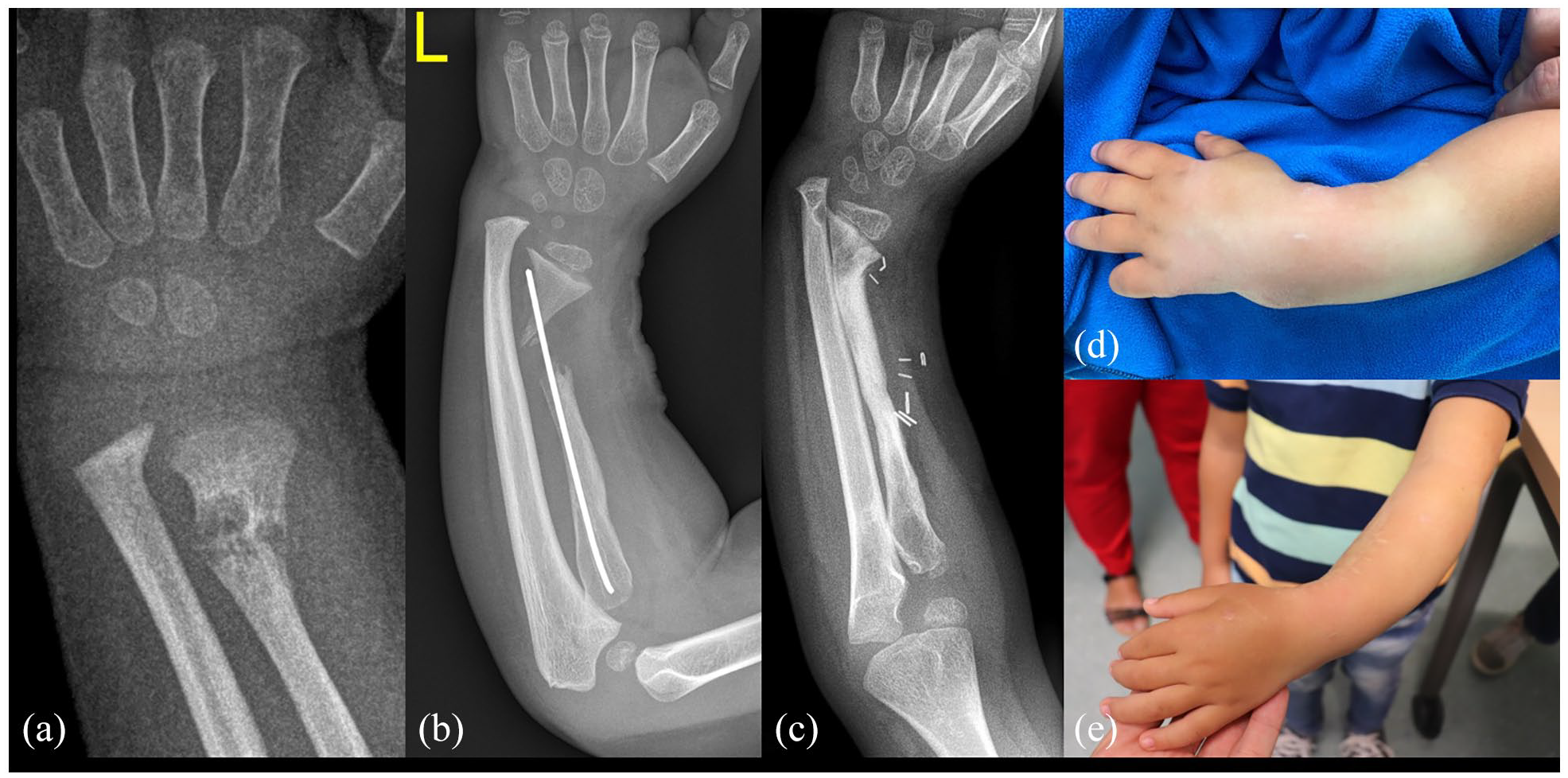
Vascularized fibular epiphyseal transplants are another microsurgical option for reconstructing radial hemimelia. The proximal fibular physis allows for longitudinal growth and three-dimensional reshaping to create a neoradius (Figure 10).
Bayne Type III radial clubhand (a) undergoing two-stage reconstruction (a). Stage I consisted of soft tissue distraction with a monolateral external fixator (b). Stage 2 consisted of a vascularized fibular epiphyseal transfer over the residual radius (c). Fibula epiphyseal remodeling occurred over time (d). Bipolar latissimus dorsi transfer technique: (a) three incisions are used; (b) the LD is isolated on its pedicle; (c) the flap is moved anteriorly through the deltopectoral approach and afterward tunelized toward the cubital fossa incision (d).
Elbow muscle flexor hypoplasia
Elbow muscle flexor hypoplasia may occur in a focal form, frequently associated with shoulder girdle muscle hypoplasia, or in the context of arthrogryposis multiplex congenita. 50 The absence of elbow flexion crease is an obvious sign and allows, in some cases, the differential diagnosis of neonatal brachial plexus palsy. 51
Our preferred reconstructive choice is bipolar latissimus dorsi transfer; 52 another common option in arthorgryposis is triceps to biceps, transferring the long head with the goal to achieve an active elbow flexion preserving active extension. 53
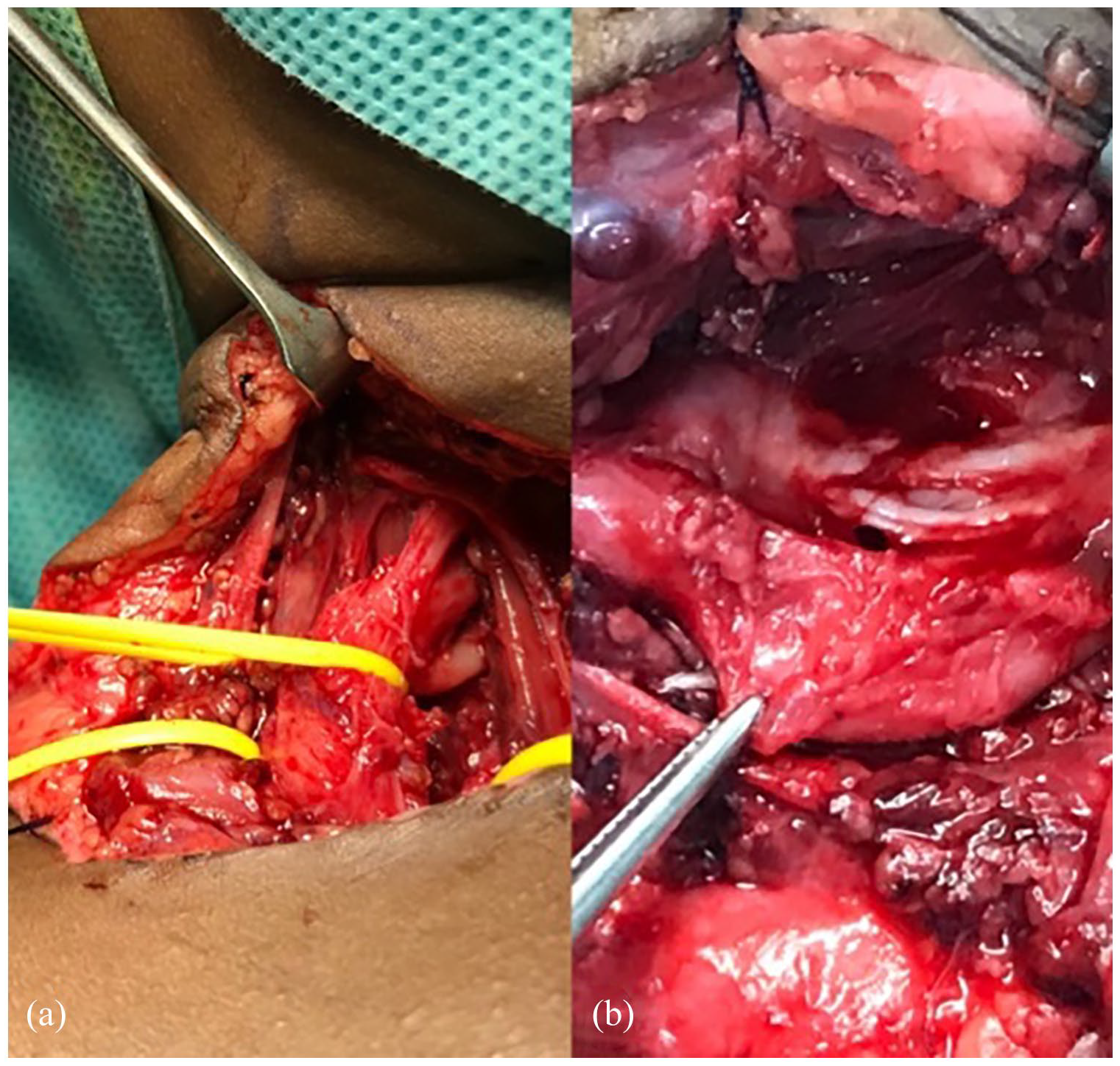
Otherwise, a free gracilis muscle was transplanted (Figure 11). Nerve coaptation is performed on any available and expendable nerve donor (spinal accessory, intercostals, etc.), while donor vessels are the neighboring vessels (suprascapular, acromiothoracic, etc.).
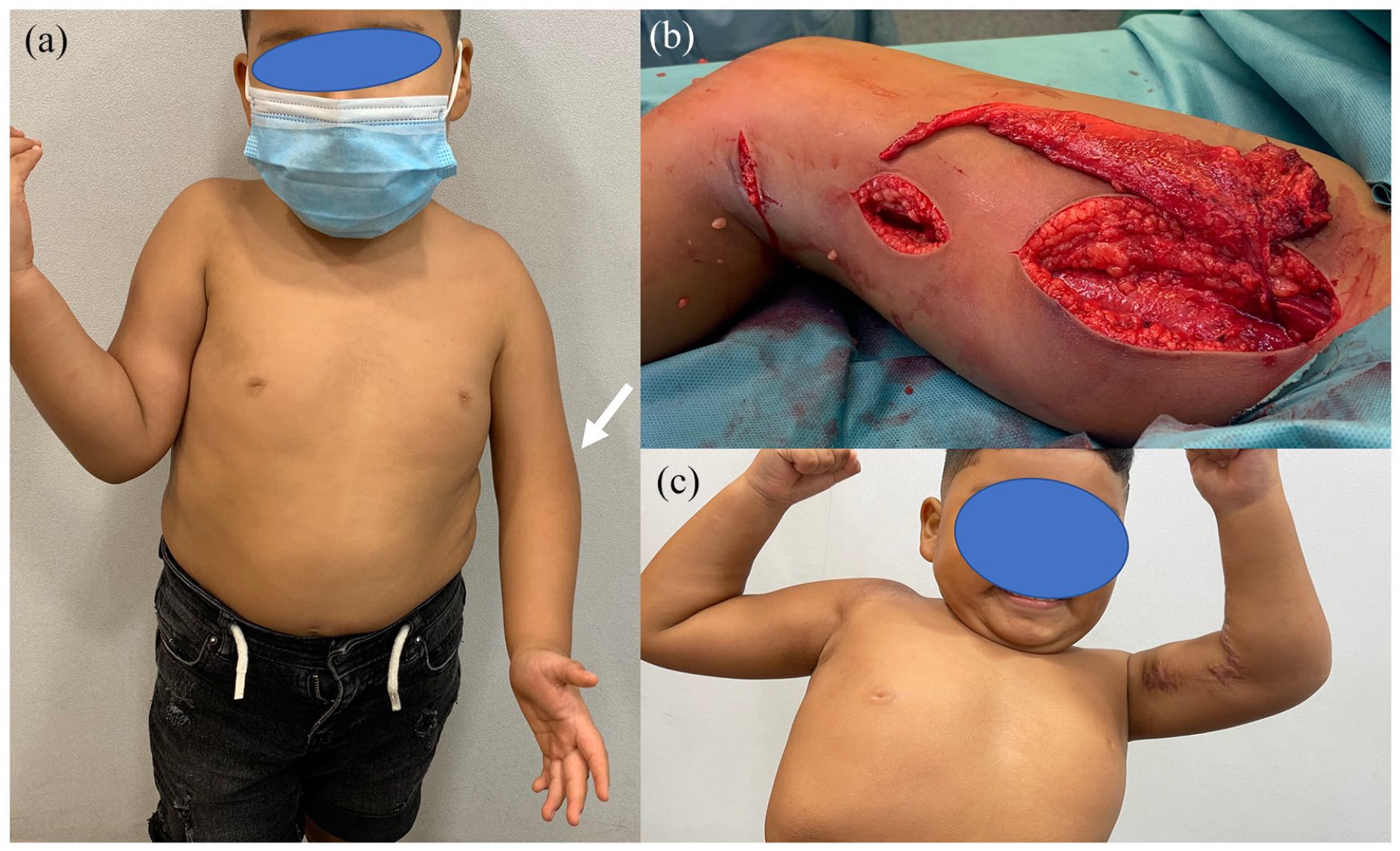
Left elbow flexors hypoplasia as revealed by the absence of elbow flexion crease (arrow) (a) associating deltoid and latissimus dorsi hypoplasia. A reverse gracilis flap was transplanted anasthomosed to the radial vessels and coapted to an ulnar nerve fascicle (b). Twelve months after surgery BMRC, M4 strength was objectivized (c).
Microsurgery in the reconstruction of soft tissue and bone tumors
Microsurgical techniques are an important adjunct in the armamentarium of any reconstructive surgeon when dealing with soft tissue and bone tumors. Microsurgical techniques include all surgical interventions around nerves and blood vessels, requiring the use of micro-instruments, loupe magnification, or microscopy.
Microsurgical techniques are important for resection of the tumor mass as well as for reconstruction after resection. It is important to have good knowledge of the resection and reconstruction techniques.
Soft tissue tumors
Although soft tissue tumors are not rare in children, the majority are benign and rarely require radical resection. As they are a heterogeneous group with different characteristics, it is vital to establish a diagnosis prior to any surgical considerations. Unless the diagnosis is obvious, such as lipoma or pyogenic granuloma, a multidisciplinary discussion needs to establish whether an incisional or excisional biopsy is necessary.
Precise preoperative imaging is necessary prior to surgical decision-making. The X-ray will provide information on whether the bone is abutted or affected. Magnetic resonance imaging (MRI) with contrast will show the extent and especially the involvement of vascular or neural structures. Ultrasound shows the vascularity in detail. Imaging will help plan the surgery in detail. In the case of benign lesions involving vascular and neural structures, the decision is usually to spare them.
The need for microsurgical tissue transfers after resection of soft tissue tumors in children is rare; it is much more frequent to utilize microsurgical techniques when performing an arteriolysis or neurolysis.
There has been an emerging number of dermal replacement matrices, for example, biodegradable temporizing matrix (BTM; PolyNovo) synthetic device used to facilitate the growth of neodermis prior to definitive wound closure, which seems to have some potential to replace the need for free microsurgical tissue transfer with exposed bone and tendon.
Once the decision for removal of complex benign lesions has been established, only experienced surgeons with good knowledge of microsurgical techniques should attempt to remove deep-seated lesions in the extremities. Surgeons should use the same precautions and preparations as when planning a microsurgical tissue transfer (Figure 12).
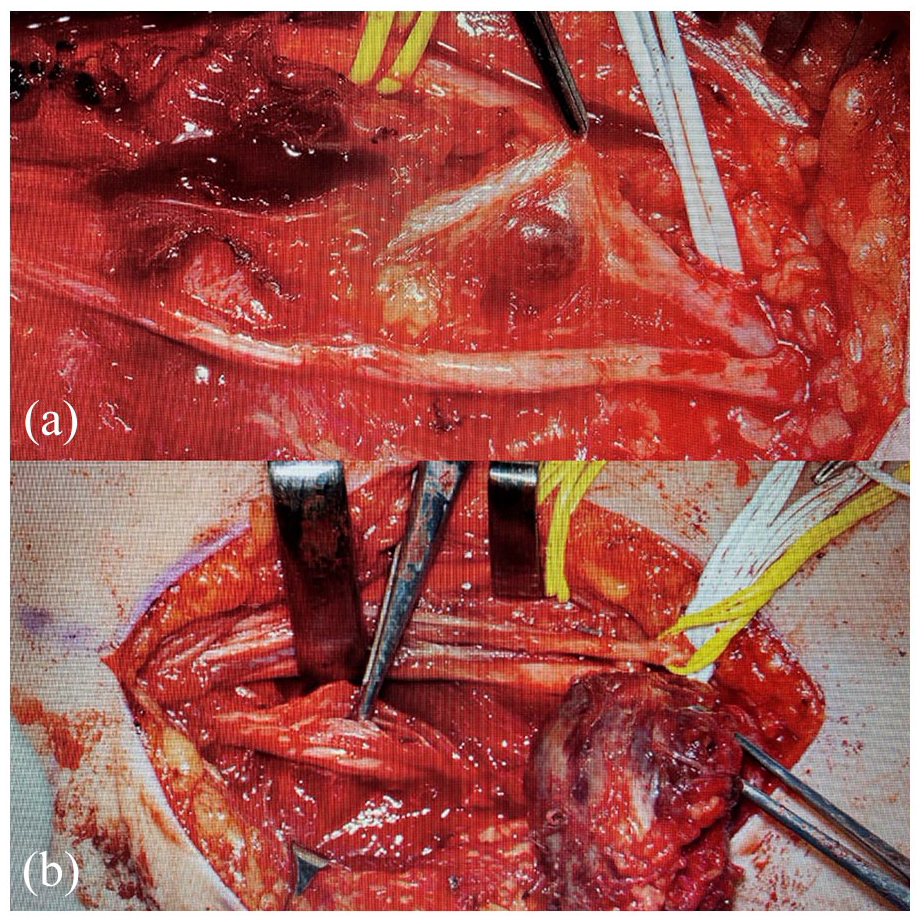
A 9-year-old girl complained about an increasing lesion on the inside of the upper arm (angiomatoid fibrous histiocytoma). The surgery proofed to be challenging as the lesion infiltrated the median, ulnar, and cutaneous nerves and abutted and displaced the radial nerve (a). The procedure had to be performed with adequate microsurgical instrumentation as well as necessary adequate loupe magnification (b).
In the case of benign tumors, a radical and destructive approach is not acceptable; hence, meticulous dissection to maintain the maximum function is paramount. In some cases of significantly increased tumor blood flow, preoperative embolization can prove useful in minimizing intraoperative blood flow. It is important to perform definitive surgical resection either on the same or on the next day; otherwise, the inflammatory response after embolization can increase the difficulty when attempting to remove the lesion.
Bone tumors
Malignant bone tumors comprise 3%–5% of cancers at ages 0–14 years 54 and are the seventh most common group of malignancies in children.
Their age-standardized incidence is approximately 5 per million person-years in the United Kingdom. 55 The majority of these tumors are osteosarcoma or Ewing sarcoma. 56
While significant improvements in survival have been seen in other pediatric malignancies, the treatment and prognosis for pediatric bone tumors have remained unchanged for the past three decades with survival rate at 5 years being approximately 60% in European countries.
When the limbs are affected, in association with chemotherapy protocols, demolitive surgery with wide margins or amputation remains a mainstay treatment.
In the appendicular skeleton, most sarcomas originate in the diaphysis or metaphysis and only later expand into the epiphysis.
While it has been proven that amputation does not improve the survival rate compared to wide oncological excision of the tumor, the quality of life after limb preservation has been shown to be superior to amputation. 57
Benign bone tumors in children, such as chondromas, unicameral bone cysts (UBC), and aneurysmal bone cysts (ABC), may not be life-threatening but can be limb- or function-threatening. When locally aggressive or recurrent, a wide-margin excision and reconstruction is suggested following similar principles as in malignant tumors. Large intercalary diaphyseal or meta-epiphyseal osseous defects are created to obtain adequate oncological margins during tumor resection.
In children, as in adults, diaphyseal defects pose the same reconstructive challenge of overcoming the bone defect and providing structural support for ambulation or large range movements, for example, shoulder joints.
When a loss of physis and epiphysis occurs in a skeletally immature individual, three main issues can present to the reconstructive surgeon: replacement of the osseous defect, restoration of joint function, and, unique to the pediatric population, restoration of longitudinal growth. Failure to achieve these objectives leads to severe deformity and functional impairment, which significantly compromises the quality of life of young patients.
Non-microsurgical techniques employing non-vascularized autologous or allografts have been used to fill these defects; however, microsurgical techniques for the transfer of vascularized bone offer the best results in terms of restoring structural support while providing an osteogenic environment that allows fast bone healing and the capacity to remain viable even in cases of infection, chemotherapy, and radiotherapy.58,59
Maintaining the growth potential is one of the main goals of this surgery and it is a unique demand of the pediatric population.
Non-vascularized epiphyseal transfer can provide a certain amount of growth; however, the best results are in very young children with phalangeal transfer, and the percentage of growth is minimal.
Only vascularized epiphysis can guarantee physiological growth achieving life-lasting biological reconstruction without the need for further surgeries.60–63
Diaphyseal reconstruction
Free vascularized fibular grafts are the gold standard for diaphyseal reconstruction of major long bones. Other donor sites, such as the iliac crest and scapular ridge, are described and used in selected cases. 64
Free vascularized diaphyseal fibular grafts are an excellent match in terms of size for humerus and radius reconstruction. However, despite the ability to undergo hypertrophy and remodeling, fibular grafts alone lack mechanical strength for lower limb reconstruction. Capanna et al. supplemented the vascularized graft using cortical allografts to circumvent the lack of structural support until hypertrophy occurs. This technique combines the structural support of the cortical allograft with the osteogenic potential of the intramedullary free fibular grafts, making the use of these grafts particularly attractive in the reconstruction of bony defects in children. 65
The medial femoral condyle flap is gaining popularity for the reconstruction of smaller osseous defects due to minimal donor site morbidity 66 (Figure 13).
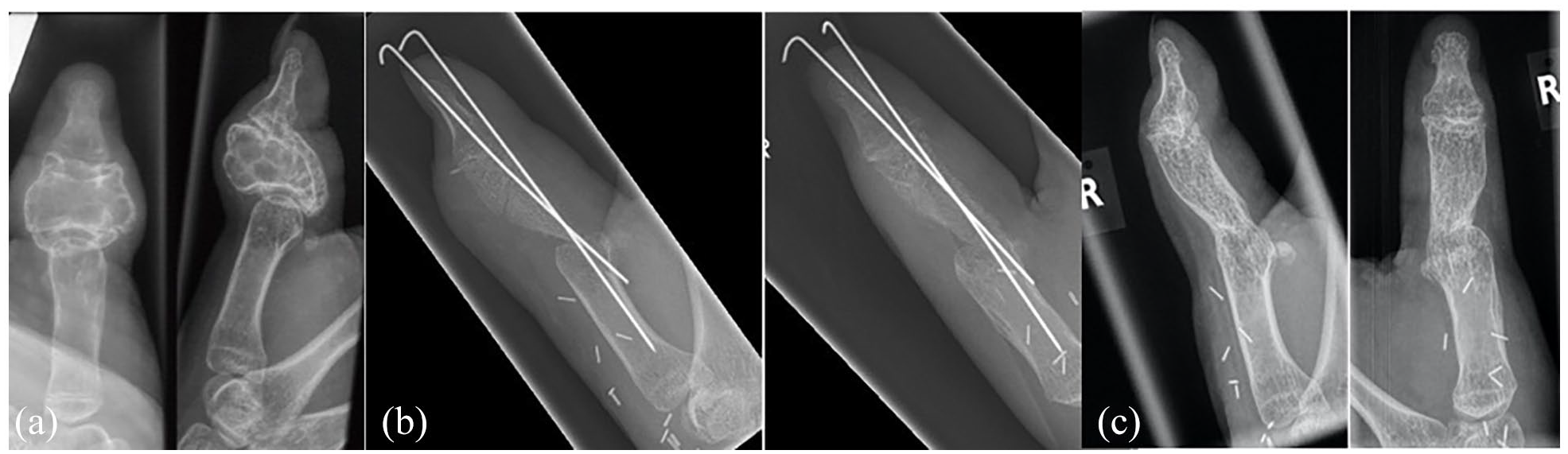
A 12-year-old girl presented with a painful expansile aneurysmatic bone cyst of the proximal phalanx of the right thumb following multiple recurrences after sclerotherapy. Radiographs and CT scan demonstrated a locally aggressive lesion of the proximal phalanx of the thumb with close involvement of the proximal and distal articular surfaces, and complete collapse of bone architecture (a). Salvage-wide local resection and reconstruction with osseous free medial femoral condyle flap based on the descending genicular artery were performed in order to guarantee the lowest chance of recurrence while providing faster bone consolidation with minimal bone fixation and early mobilization (b). Radiographs at 2 years after surgery demonstrating adequate fusion of the MCPJ and remodeling of the graft. There was no evidence of recurrence (c).
Epiphyseal reconstruction
Conventional methods of joint reconstruction, such as customized prosthetic implants and non-vascularized osteoarticular allografts, can achieve good functional outcomes and overcome bone defects and, in fact, are often the treatment of choice in adults, but these procedures cannot provide physiological bone growth. Autologous vascularized epiphyseal fibula transfer is the only procedure capable of simultaneously reconstructing a lost joint, replacing the bone defect, and maintaining the growth potential. Different pedicles have been described to supply proximal fibula epiphysis. The authors preferred the technique based on the anterior tibial artery as described by Innocenti et al. 67 The longer the expected period of time between surgery and the end of growth, the stronger the indication for vascularized epiphyseal transfer (Figure 14).
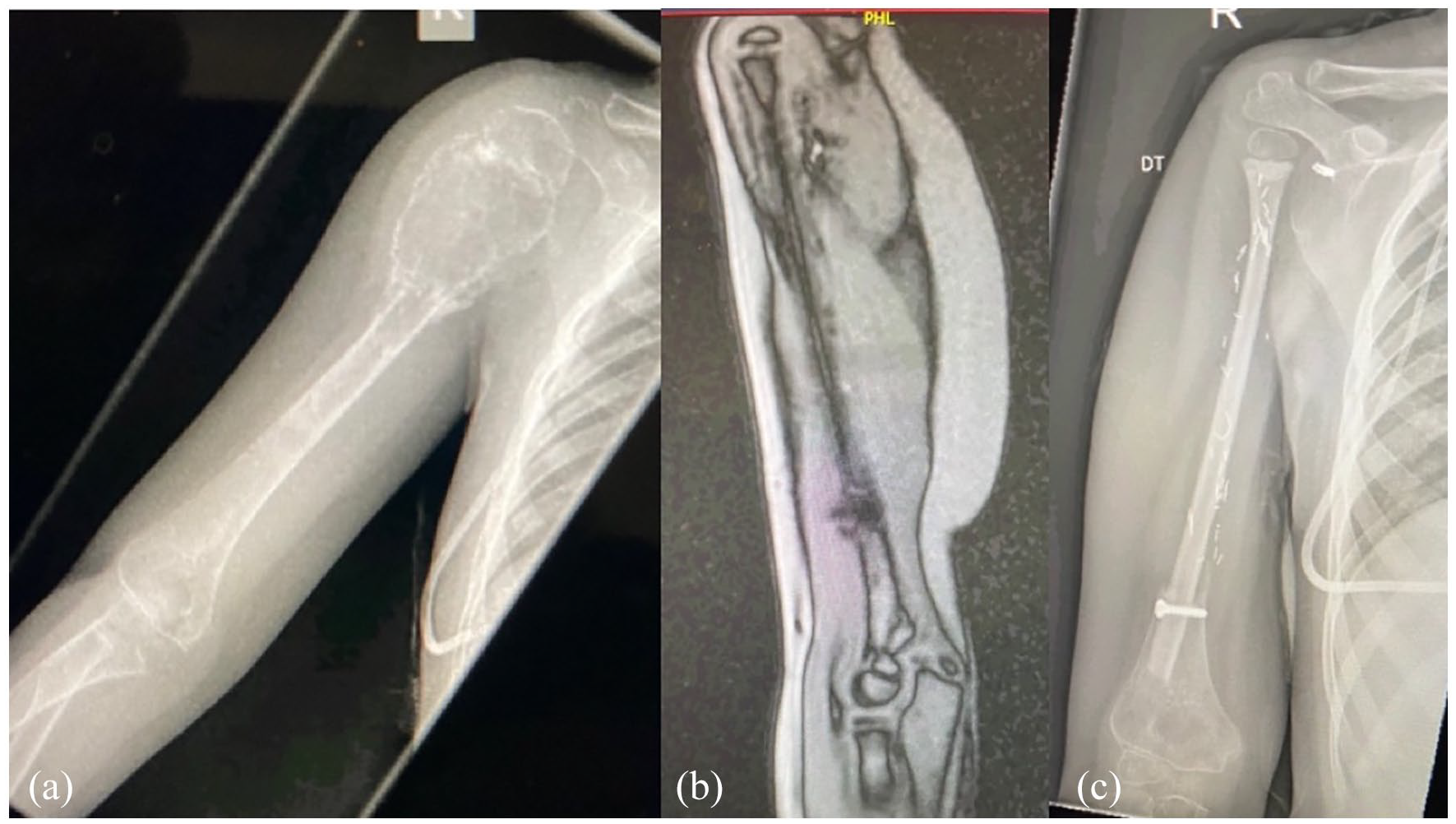
An 8-year-old girl presented with a diagnosis of high-grade telangiectatic osteosarcoma of the right proximal humerus involving the epiphysis (a). At the time of surgery, the entire proximal humerus and two-thirds of the humeral diaphysis were resected for a total length of 15.5 cm. An autologous vascularized epiphyseal fibula transfer was planned; 18 cm of the contralateral proximal fibula was harvested based on the anterior tibial artery (b, c).
Donor site complications: Although knee instability is a feared complication at the donor site, meticulous reconstruction of the lateral collateral ligament proved to be able to prevent this condition in all cases. By contrast, injuries to the small motor branches of the peroneal nerve are virtually unavoidable due to the anatomically intricate configuration of the personal nerve and the anterior tibial artery in this area. Transient foot drop is reported in almost all patients. However, permanent peroneal nerve palsy is less frequent. 68
In conclusion, microsurgical techniques should be part of the armory of pediatric hand and upper limb surgeons, as their application enables the resolution of complex cases that would otherwise be untreatable, as well as the restoration of function lost due to congenital, traumatic, or oncological pathologies.
Footnotes
Author contributions
Nunzio Catena: article design, writing section on obstetrical brachial plexus palsy, and final editing.
Carla Baldrighi and Andrea Jester: writing section on bone and soft tissues tumors and pre-final check.
Sebastian Farr: writing section on congenital pseudarthrosis of the forearm and pre-final check.
Francisco Soldado: writing section on traumatic disorders and pre-final check.
Compliance with ethical standards
Research involving human participants and/or animals (i.e. compliance with Helsinki declaration): nothing to declare.
Institutional Review Board/Ethics committee approval: the nature of the article does not require any approval.
Informed consent: the nature of the article does not require any consent.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research ethics and patient consent
Any consent has been necessary due to the nature of the article.
