Abstract
Cancer nanotheranostics aims at providing alternative approaches to traditional cancer diagnostics and therapies. In this context, plasmonic nanostructures especially gold nanostructures are intensely explored due to their tunable shape, size and surface plasmon resonance (SPR), better photothermal therapy (PTT) and photodynamic therapy (PDT) ability, effective contrast enhancing ability in Magnetic Resonance imaging (MRI) and Computed Tomography (CT) scan. Despite rapid breakthroughs in gold nanostructures based theranostics of cancer, the translation of gold nanostructures from bench side to human applications is still questionable. The major obstacles that have been facing by nanotheranostics are specific targeting, poor resolution and photoinstability during PTT etc. In this regard, various encouraging studies have been carried out recently to overcome few of these obstacles. Use of gold nanocomposites also overcomes the limitations of gold nanostructure probes and emerged as good nanotheranostic probe. Hence, the present article discusses the advances in gold nanostructures based cancer theranostics and mainly emphasizes on the importance of gold nanocomposites which have been designed to decipher the past questions and limitations of in vivo gold nanotheranostics.
Introduction
Despite decades of research and development to regulate tumor growth and progression, cancer remains the second prime cause of deaths globally next to cardiovascular-related diseases. 1 The unconstrained growth and heterogeneity of tumor has made its early stage diagnosis as well as therapy a tough nut for the researchers and medical practitioners. 2 The conventional diagnostic (imaging, marker-based in vitro assays) and therapeutic (surgery, radiotherapy, chemotherapy) strategies fails in the prognosis of tumor tissue with high sensitivity and specificity. 3 For instance, the conventional techniques can detect the tumor only when there are predictable changes in the morphology of tissue and unable to differentiate lesions from malignant tumors. Therefore, by the time the tumor is diagnosed, it would have metastasized to other tissues which reduce the survival rate of the patient. Hence, there is a need for the development of new strategies which can target tumors at their molecular level for the early diagnosis and precise therapy based on the patient's profile.
Theranostics has emerged as an efficient and specific alternative field of medicine by combining real time imaging and tumor therapeutics. With the intervention of nanotechnology in the field of theranostics (Nanotheranostics), various auxiliary strategies of imaging (MRI, photoacoustic (PA) imaging, surface enhanced Raman scattering (SERS) imaging, etc.) and therapy (PTT, PDT, radiation therapy, and chemotherapy) are embraced onto a single platform. Nanotheranostics enhances the specificity and sensitivity of tumor detection at early stage besides providing comprehensive image-guided therapy. It becomes feasible due to the fascinating properties of nanostructures like high surface to volume ratio, flexibility in surface modification, eccentric optical and magnetic properties, high cargo loading capacity, Enhanced Permeation Retention (EPR) effect, etc. It has also led the way for the growth of personalized medicine to bridge nanobiosensing and nanomedicine. 4 The importance of nanotheranostics has tremendously increased in the past few years as nanostructures provide a plethora of approaches for delivering target entities to the target site with high precision. Since then, various organic and inorganic nanostructures such as carbon5,6 liposomes7,8 silica9–11 magnetic12–14 gold15–18 silver19–21 quantum dots22,23 have been employed as theranostic probes for the delivery of drugs, siRNA and fluorescent molecules. Of all the nanostructures used in cancer nanotheranostics, gold nanostructures lead the race. Because their nanotheranostic efficiency is amplified by offering features such as SPR, quantum confinement, short range translational order, formation of local density of states due to the transition of ions between molecules and metallic states, high surface energy, etc. Additionally, these nanostructures also offer strength to other nanostructures such as liposomes. 24 Till date gold nanoformulations such as Aurimmune (NCT00356980, NCT00436410), Auroshell (NCT04240639, NCT02680535), AuroLase (NCT00848042, NCT01679470) and few others are under various stages of clinical trials for the treatment of different cancers. 25 Recently gold nanostructures based theranostic formulation (NCT04240639) has reached the clinical trials phase aiming at prostate cancer. 26 Therefore, this communication briefly discusses the recent developments in gold nanostructures based theranostics to give a holistic view to the readers. In addition, the effect of shape and size of nanostructures on theranostic efficiency is also discussed.
Impact of size and shape of gold nanostructures on theranostic efficiency
Gold nanostructures exhibits excellent optical properties owing to their tunable SPR, plasmon absorption (dissipation and conversion of incident light into heat/vibrational energy) and light scattering ability (Rayleigh scattering or Raman scattering) which makes them a promising candidates in the field of imaging, radiation/laser therapy. Any alteration in the arrangement of atoms and other structural parameters influences these optical properties, thus altering their imaging and ablation therapy capabilities; formation of protein corona and folding of proteins over nanoparticle's surface. 27 Size and shape of nanoparticles greatly impacts the theranostic ability of gold nanostructures by governing their in vivo movement, biostability, circulation time, biodistribution, renal clearance, metabolism, toxicity and catalytic activity. Hence it is important to critically review the effect of size and shape on the fate of nanotheranostic probes which provides a perspective to the researchers for the development of efficient theranostic probes. Hence, this section discusses the impact of size and shape of nanotheranostic agent on various biological parameters here in detail.
Effect of shape
Anisotropic gold nanostructures such as nanostars, nanorods have been demonstrated as better photothermal agents in comparison to spherical nanoparticles due to the exhibition of two different plasmon resonance peaks which can be tweaked near infrared window.28,29 Among the anisotropic nanostructures, nanohexapods were demonstrated as efficient photothermal transducers due to sharp edges followed by nanocages and nanorods. But, nanocages possess higher photothermal conversion efficiency per gold atom than that of nanohexapods. 30 However, few contrasting studies have demonstrated spherical nanoparticles to cause greater cell death than that of nanorods and nanospikes when irradiated with X-ray source. 31 Furthermore, the shape of nanoparticles also influences the osteogenesis of mesenchymal stem cells where sphere and rod shaped nanoparticles significantly up regulated the expression of osteogenic marker genes. 32 The efficiency of theranostic probe also depends on their cellular uptake efficiency which is again determined by the shape of nanoparticle. Few studies reported that spherical shaped nanoparticles exhibited better cellular uptake in comparison to nanorods, spiked and cage shaped nanoparticles.31,33,34 Whilst another study has shown hollow gold nanoshells to display more cellular internalization followed by nanorods and nanocages in HeLa cell lines 35 , furthermore, nanospikes have better internalization capability than that of nanorods 31 . But Xie et al. 36 showed that nanostars were poorly taken up by RAW264.7 cell lines whereas nanorods and nanotriangles caused higher cellular uptake. However, the regular order of cellular internalization with effect of shape changes with the nature of surface functional groups. For example, Cho et al. 37 investigated the effect of shape and surface modification of differently shaped gold nanostructures on the cellular uptake by SK-BR-3 breast cell lines. The group employed nanospheres and nanocages surface modified with PEG, anti-HER2 antibody, poly(allyamine hydrochloride) (PAA) in the study. The cellular uptake studies showed that the cells preferred nanospheres over nanocages when surface is modified with either PEG or anti- HER2 antibody. However, cellular uptake of nanocages was extremely higher upon surface modification with PAA. Carnovale et al. 38 demonstrated that nanoprisms and spherical nanoparticles exhibited higher toxicity than nanorods and nanocages inspite of the lower cellular internalization of nanoprisms. Thus this study reported that cellular toxicity does not linearly depends on the amount of cellular uptake which is consistent with another recent study reported by Navyatha and Nara. 39 During nanoparticle-mediated chemotherapy, the curvature of nanoparticles determines the drug loading capacity or siRNA loading density. Hollow gold nanoshells were found to possess 3 times more capacity to load siRNA compared to nanocages and nanorods. However, release percentage of siRNA under appropriate conditions was higher for nanorods. 35
The main concern about the nanoparticles is their toxicity which depends on the shape, size and surface chemistry of the respective nanoparticles. Wang et al. 40 demonstrated that gold nanorods were more toxic than spherical particles and this toxicity was attributed by CTAB present on the surface of nanorods. Another study investigated the comparative cytotoxicity of nanospheres, nanorods, nanoflowers, nanostars, nanoprisms on HeLa cell lines and showed that nanospheres and nanorods were toxic to cells even at lower concentrations (1, 6, 8 16 μM) after 72 h of treatment. However, nanostars, nanoflowers and nanoprisms were nontoxic at these lower concentrations and showed toxicity at a concentration of 300 μM after 72 h. 41 Circulation of nanoparticles in the blood stream also depends on the shape and curvature of nanoparticles. Hence, choosing the appropriate nanoparticle shape is highly important in designing a theranostic probe. For instance, PEGylated gold nanorods are able to escape macrophage mediated uptake and so had a prolonged circulation period compared to other counterparts i.e., PEGylated spherical nanoparticles. 42 However, Black et al. 43 demonstrated that nanospheres exhibited high cellular uptake, prolonged circulation period compared to nanorods, nanocages, nanodiscs. Though the uptake of nanospheres and nanodiscs was higher, their distribution was restricted to surface of tumors whereas; nanorods and nanocages were able to reach the core of tumors. Besides influencing the therapeutic efficiency and biodistribution of nanoparticles, shape of nanoparticles also determines the imaging ability for their use as contrast agents.
Effect of size
The optical properties and theranostic efficiency of nanoparticles are greatly affected by their size. Many studies have reported the implementation of gold nanostructures as contrast agents in imaging techniques such as PA, CT, MR imaging. Nevertheless, the studies reported so far contradict on the effect of size of nanoparticles to be used as contrast agents. For example, Dou et al. 44 showed that gold nanoparticles of size 34.8 nm i.e., larger size nanoparticles possess high X-ray attenuation property than smaller nanoparticles. Others have shown smaller nanoparticles of size 4 nm, 13 nm as better contrast than larger ones of sizes ∼40 nm, 60 nm.45,46 Few more studies have demonstrated that X-ray attenuation of gold nanoparticles was independent of size of nanoparticle.47,48 The change in size shifts the plasmon peak either bathochromically or hypsochromically which thus affects the photothermal efficiency of nanostructures. As discussed above anisotropic nanostructures own effective photothermal ability however, the effect of size on PTT effect is more prominent in case of anisotropic nanostructures than spherical ones. 49 The photothermal efficiency increases with the decrease in nanoparticle diameter.50,51 PEGylated gold nanoparticles of sizes 12.1 nm and 27.3 nm exhibited stronger radiosensitization effect by inducing both apoptosis and necrosis in comparison to 4.8 nm and 46.6 nm. 52 The osteogenesis of mesenchymal stem cells is not only affected by the shape but also the size of nanoparticles. Zhang et al., 2017 53 demonstrated that spherical nanoparticle of sizes 13 nm and 45 nm promoted the differentiation of stem cells more effectively than 5 nm nanoparticles.
Cellular uptake is another factor that is affected by the size of nanoparticles. Design of a theranostic probe with appropriate size is very crucial because cellular uptake influences the biodistribution and renal clearance. A study reported that nanorods with low aspect ratio had higher cellular uptake in comparison with high aspect ratio 30 and also the orientation of nanorods. Similarly, another study investigated the size effect of spherical particles and nanocages on cellular uptake by SK-BR-3 breast cancer cell lines. The results revealed that spherical particles of size 15 nm have 1.5-2.4 times higher uptake than that of 45 nm particles and 33 nm sized nanocages 37 . Chithrani et al. 33 studied the cellular uptake of various sized (14, 30, 50, 74, 100 nm) spherical nanoparticles by HeLa cell lines and determined that 50 nm sized nanoparticles had prominently higher cellular uptake which was consistent with the study carried out by Arnida et al., 2009 on PC3 cell lines. 54 Previous studies say that larger nanoparticles of size >200 nm rapidly clear by macrophages in the spleen and kupffer cells in the liver whereas smaller nanoparticles generally accumulate at higher concentrations in lungs and brain and lower concentrations in stomach, heart and pancreas.55,56 Similarly, study conducted to explore the effect of size of gold nanoparticles on their biodistribution disclosed that nanoparticles of size <15 nm were accumulated in all the organs including brain, heart, kidneys, liver, spleen, blood, stomach etc. Nanoparticles of size 15-50 nm were able to cross blood brain barrier and nanoparticles of size >200 nm are rapidly cleared by reticuloendothelial system from the body. 57 Moreover, smaller nanoparticles (∼2 nm) were able to pass through the nuclear membrane and accumulate in nucleus whereas nanoparticles of size ∼8 nm accumulate in cytoplasm and nanoparticles >15 nm accumulate on the periphery of cellular membrane. 58 Thus smaller nanoparticles may be applied for treating deeper tumors and larger nanoparticles for superficial tumors.
Besides cellular uptake and biodistribution, cytotoxicity of nanoparticles is a major concern which restricts the human applications. Inspite of many reports on toxicity of gold nanoparticles, the studies reported till so far are largely contradictory. A study conducted by Lee et al., 2016 on human neural precursor cells had determined that 5 nm sized particles were more toxic than 100 nm sized particles. 59 and is in accordance with another study 60 Similarly, 1.4 nm gold nanoparticles were shown to be more toxic to cells in comparison to 15 nm nanoparticles at a concentration 60 folds lower than that of 15 nm particles. 61 Connor et al. 62 demonstrated that nanoparticles of any metal were not inherently toxic to cells but precursors used for the synthesis were responsible for their cytotoxicity.
Inspite of many studies, yet systematic and more controlled studies are required to determine the effect of size and shape on theranostic efficiency of nanostructures and the interaction of nanoparticles and their metabolites with subcellular organelles. Moreover, so far studies reported are controversial studies due to which a precise conclusion could not be drawn on the impact of physicochemical properties on cellular interactions.
Recent advancements in gold nanostructures based theranostics using Photothermal therapy
Despite prompt progressiveness in nanotheranostics still there are various hurdles related to imaging and therapeutic moieties, localization of probes, instant investigation of cellular response etc. that halt the translation of theranostic probes from laboratory to clinical stages. In recent years, different research groups have developed efficient gold nanostructures based theranostic probes with various modifications to overwhelm some of the hurdles faced in the past.
From the point of therapeutics, gold nanostructures offers new paradigm strategies such as PTT and PDT which are non-invasive techniques. The mechanism of PTT behind damaging tumor involves the utilization of localized heat generated by nanostructures upon light irradiation of particular wavelength whereas; PDT utilizes reactive oxygen species released from activated photosensitizer by a particular wavelength light. PDT depends on the oxygen concentration in the tissue and wavelength of light. 63 Hence the application of PDT is limited to surface tumors and could not be applied for larger, deeper and metastasized tumors. However, PTT can be employed for deeper and metastasized tissues without damaging the surrounding healthy cells. Thus gold nanostructures based PTT has become a considerable therapeutic strategy of nanotheranostics now-a-days. In this perspective, some of the recent advancements in gold nanostructures based theranostics using photothermal therapy are discussed ahead.
Most of the theranostic probes comprise different imaging and therapeutic modalities, which delay the monitoring of response to the therapy. To reduce the time between therapy and evaluation of its response via imaging, Wang and his group demonstrated an approach known as self-therapeutic feed-back image-guided therapy. In this study, the group had designed fluorescence based “switch on” gold nanostars’ probe to trace the apoptosis mediated cell damage via caspase-3 activity. Upon light irradiation, caspase-3 enzyme in the cells treated with nanoprobe gets activated and cleaves the peptide conjugated to nanostars, thus switching on the fluorescent molecule indicating the activation of apoptosis. The PTT studies with 2 W/cm2 NIR 808 nm laser for 5 min showed two times higher cell death with nanostars probe in comparison to only nanostars. The fluorescence microscopy and flow cytometry studies demonstrated that the intensity of fluorescence had increased with an increase in the duration of laser irradiation. 64 Thus the study has opened the door for the real time monitoring of effect of therapy on cancer cells.
Despite development of various nanotheranostic probes, the localization of these probes at the tumor site is still a challenge. This challenge is addressed recently by efficiently delivering gold nanoprobes to the tumor site via macrophages and evaluation of theranostic property is depicted in Figure 1. 65 This group evaluated the effectiveness of radioiodine conjugated gold nanoballs (124I-GNBs) labeled macrophages to localize within the tumor. The in vivo PET/CT imaging studies showed that nanoballs labeled macrophages were uniformly distributed throughout the tumor in comparison to only nanoballs. After laser irradiation, the temperature of tumor treated with nanoballs labeled macrophages elevated (55.2°C) more compared to tumor treated with only nanoballs (>40°C). Further studies indicated uniform localization of theranostic nanoballs within the tumor.
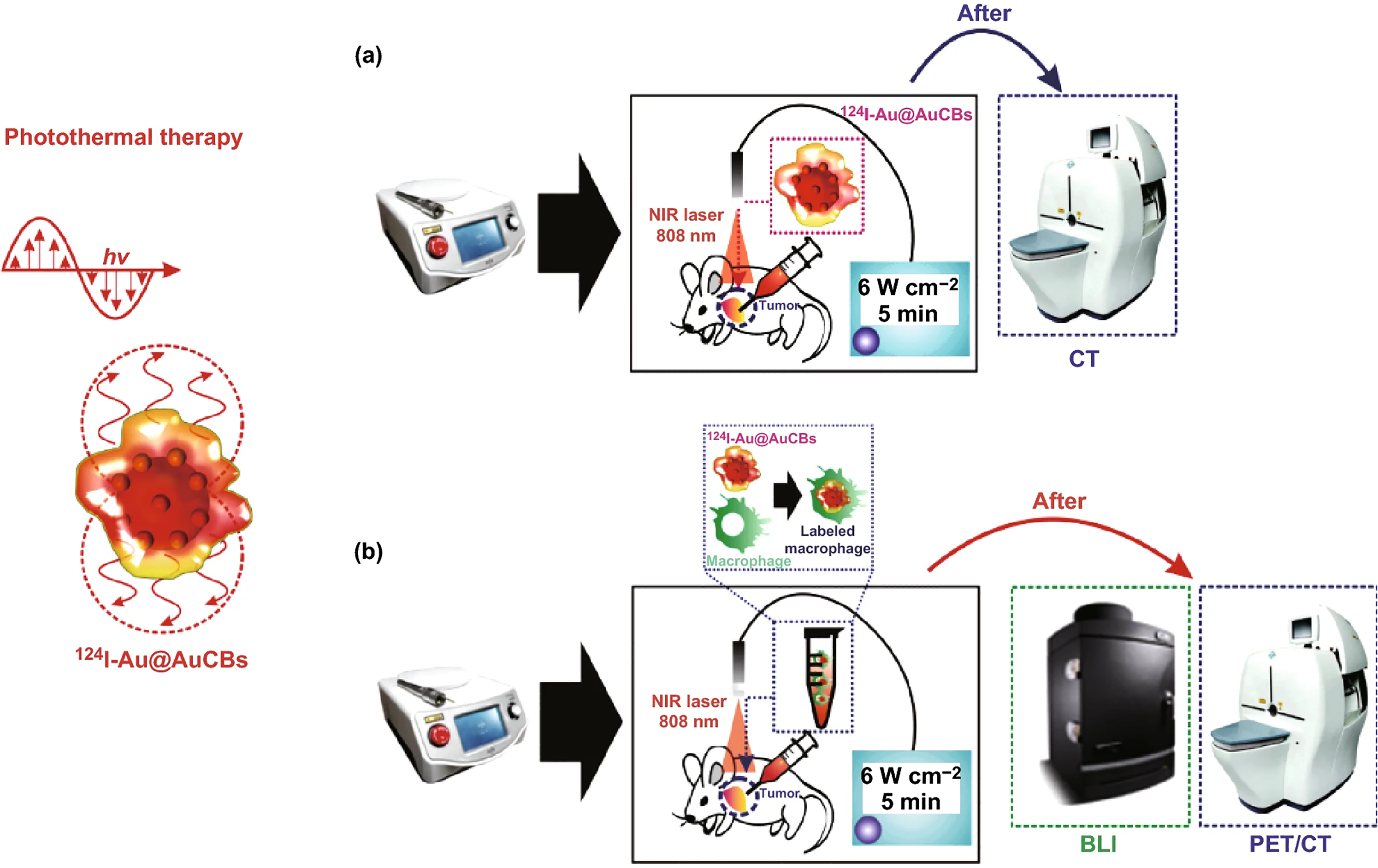
(a) Schematic representation of the evaluation of in vivo photothermal conversion effects of 124I-GNBs. (b) in vivo photothermal therapy using 124I-GNBslabeled macrophages in a colon cancer model, followed by positron emission tomographic imaging of 124I-GNBs labeled macrophage distribution in tumor lesions. Reprinted with permission from reference. 65
Vickers et al. 66 demonstrated that hollow gold nanospheres exhibited better two photon action cross sections and lower photon quantum yield than gold nanorods (GNRs) which attributed to excellent two photon photoluminescence imaging guided (TPP) PTT. Further studies on HeLa cell lines revealed that at a power density of 1.6 W/cm2 and an exposure time of 8 μs, the cell viability had significantly decreased, leading to membrane blebbing. This data showed high theranostic efficiency of gold hollow nanospheres in comparison to GNRs. Similarly, nanobipyramids 67 were demonstrated as promising nanotheranostic probes with increased biocompatibility and enhanced photostability. Another recent advancement is implementation of GNRs in fluorescence endoscopy mediated laser ablation of upper gastrointestinal cancers. 68 Fluorescence endoscopic studies showed that fluorescent dye, Cy5.5 and anti-EGFR antibody fabricated PEGylated GNRs were accumulated in the tumor within an hour and produced a good fluorescence signal in the tumor region. Laser ablation studies demonstrated the efficacy of GNRs in PTT in endoscopic imaging also. Apart from these, gold nanocages were demonstrated as potential nanostructures to conquer multidrug resistance issue by specifically releasing doxorubicin to the tumor using low pH peptide conjugated nanocages. 69
Gold nanocomposites– Better alternative to gold nanostructures as theranostic probes
Inspite of many advancements and achievements, gold nanostructures as theranostic probes are facing issues related to their photostability, in vivo toxicity, poor 3D resolution, off-target effect, etc. Gold nanocomposites have shown a ray of hope to address these issues by synergistically using unique properties of diverse elements. 70 Gold nanocomposites are the alloys of gold nanostructures with other organic/inorganic plasmonic nanoparticles. 71 These nanocomposites include gold-mesoporous silica nanocomposites72,73, gold-iron oxidenanocomposites74,75, gold-other noble metals (silver, platinum, palladium) 76 , gold-polymers (biopolymers, polyethylene glycol (PEG)) 77 , dendrimeric gold nanocomposites etc. The synergistic effect of more than one nanostructure enhances the properties of theranostic probes. For example, the high porosity of silica nanoparticles improves the drug loading capacity, which in turn enhances the therapeutic efficiency of the probe. Similarly, magnetic properties of iron oxide nanoparticles boost up the relaxation rate, which can be employed in MR imaging. Furthermore functionalizing the gold nanostructures with various biological entities such as aptamers, antibodies, ligands to promote specific targeting of theranostic probes and decreases the off target effects of nanoprobes. This section highlights the use of gold nanocomposites in addressing issues associated with gold nanostructures as cancer theranostics probe.
Enhancing in vivo biocompatibility and photostability
The toxicity and stability of gold nanostructures remains a burning issue in the field of nanomedicine. Both in vitro and in vivo studies reported so far on toxicity of gold nanostructures do not present uniformity in their experimental settings, model organisms and hence conclusive findings cannot be drawn. Moreover, the studies conducted on different models have reported the effect and fate of nanoparticles within the models upto six months only and thus this shortage of data on long term effects drive the need for studying long term effect of nanoparticles.78,79 Hence, comprehensive studies shall be conducted on in vivo toxicity assessment of nanostructures to answer the unexamined questions or those left unconcluded so far. Therefore, research is ongoing to decrease the toxicity of nanostructures and increase their stability in in vivo conditions. For example, recently, Rahimi-Moghaddam et al. 80 demonstrated PEGylated curcumin loaded gold nanoparticles as highly stable and non-toxic photothermal agents. The in vitro cytotoxicity studies on C540 (B16/F10) mouse malignant melanoma cell line reported that at 2260 μg/mL concentration, PEGylated nanoparticles showed no cell damaged, whereas, curcumin loaded nanoparticles showed cell death of 83% after 24 h. Similarly, in vivo studies also showed the reduction in tumor volume to ∼100 mm3 in the mice treated with curcumin loaded nanoparticles and subsequent laser exposure, compared to the control group. Ex vivo studies of organs like liver, kidney also showed no decrease in the weight of these organs after the treatment. These results show that functionalization of gold nanoparticles decreases the toxicity in vivo. Some studies demonstrated that nanoparticles of size >8 nm induce potential toxicity 81 and hence use of smaller nanoparticles is required. Furthermore, coating with polymers to increase the biocompatibility also increases the size of nanoparticle which affects their circulation in the blood stream and biodistribution. In this respect, Deng et al., 2015 synthesized gold nanomicelles coated with multi-mercapto terminated comb like amphipathic polymer, poly(ε-caprolactone) (PCL)/ poly (2-hydroxyethyl methacrylate) (PHEMA) (hydrophobic) and poly[2-(2-methoxyethoxy) ethyl methacrylate] (PMEO-2-MA) (hydrophilic) to provide biocompatibility besides restricting the size of GNPs to 6 nm. The synthesized nanomicelles were not toxic to cells in in vitro and in vivo studies. The in vivo studies performed by H&E staining showed that nanomicelles did not induce any obvious lethality or abnormalities even after 20 days of exposure. This was due to decomposition of nanomicelles into 6 nm GNPs within the organs facilitating their easy clearance. 82 Surface functionalization of gold nanostructures with biopolymers enhances the biocompatibility, biostability and circulation period making them a promising candidate in theranostic applications. Though many synthetic polymers have been used to improve biocompatibility, biopolymers such as chitosan83,84, alginate83,85, agarose 86 , glucan/dextran 87 , hyaluronic acid88,89 etc. are receiving much recognition due to their ease of synthesis, reducing property, variation in porosity, extracellular matrix mimetic property. For instance, Li et al., 2019 decorated GNRs with β-glucan polysaccharide and assessed their biocompatibility and PTT efficiency in second NIR window range (1000-1400 nm). In vivo studies carried out on HT-29 cells xenografted mice revealed that the tumor had completely dissolved within 16 days after a single administration of nanocomposites and subsequent laser exposure. Further studies on other visceral organs showed no sign of tissue damage, which was confirmed by H&E staining. 90 Alginate coated curcumin and methotrexate drugs loaded gold nanoparticles were found to be hemocompatible. 91 Similarly, Manivasagan and his group synthesized biocompatible gold nanoparticles by stabilizing with chitosan 92 and fucoidan 93 , to develop photoacoustic imaging mediated chemotherapeutic theranostic probe. Similarly various groups have constructed gold nanostructures based theranostic probes stabilized by biopolymers such as bacterial cellulose 94 , β-cyclodextrin 95 , hyaluronic acid 89 , albumin 96 . Recently, phosphatases sensitive phosphotyrosine (pY) peptide stabilized gold nanoparticles were synthesized to deliver doxorubicin to the site of tumor. In the presence of phosphatases, the peptide gets digested and leads to aggregation of nanoparticles thus releasing the drug. 97
The studies showing gold nanorods, nanoprisms, and other anisotropic nanostructures have been explored much as photothermal agents and gained superior position due to their high longitudinal SPR (LSPR) tuning property. However, nanoparticles upon laser irradiation tend to reshape or aggregate accompanied by a blue-shift causing photoinstability which led to the loss of their optical properties in the therapeutic window and makes them poor contrast agents and PTT probes. Hence, photoinstability is another obstacle faced by gold nanostructures. Gold nanocomposites have proved to maintain the photostability of gold nanostructures and thus the efficiency of PTT. Photostability is defined as the response of nanoparticles to optical power and conserve their physicochemical properties such as stability, shape, size in addition to their optical properties. As per the previous studies, this instability is caused by the presence of semiconductor silver nanoclusters over the surface of anisotropic gold nanostructures. 98 To maintain the photostability of GNRs, Zhao et al. 99 entrapped GNRs within mesoporous silica nanoparticles (MSNPs) (MSN@GNRs) which not only conferred stability but also made them biocompatible. Figure 2 demonstrates the increase in temperature and stability of GNRs upon irradiation with laser. In another study, Yoo et al. 100 reported that gold nanoparticles functionalized with polystyrene and azidopolystyrene blocks were thermally stable with no aggregation even at 200°C for prolonged periods in both solution and solid forms in comparison to naïve gold nanoparticles.
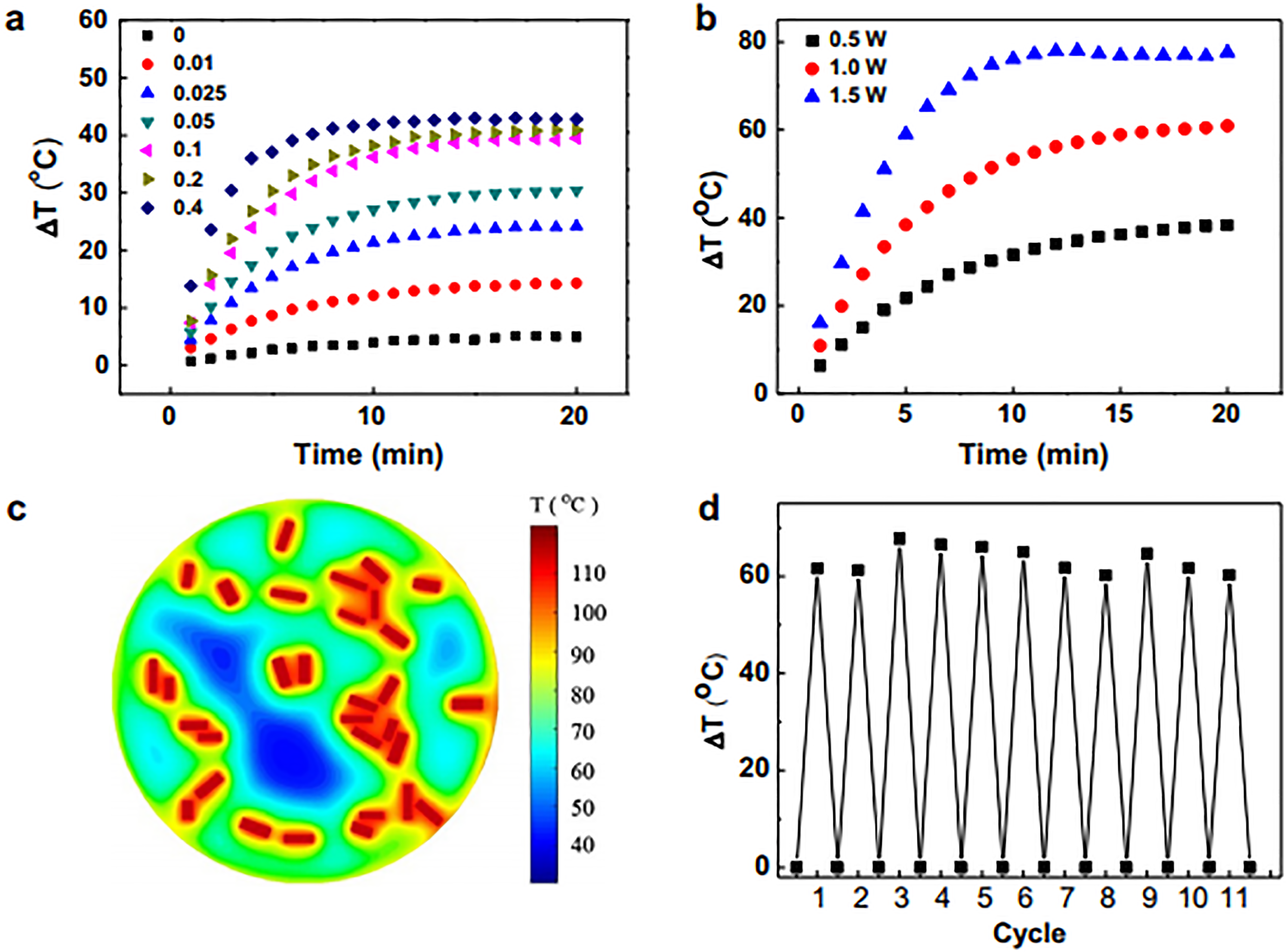
(a) Time dependent temperature changes of MSN@GNRs with different GNRs concentrations. (b) Time dependent temperature changes of MSN@GNRs at different laser power densities. (c) Theoretical simulation of the temperature elevation after NIR irradiation. (d) Temperature elevation record of MSN@GNRs over 11 laser on/off cycles. Reprinted with permission from reference. 99
Specific targeting
One of the major hurdles that has been dragging the translation of gold nanostructures as theranostic probes to clinical trials is their off target effects. Functionalization with specific targeting moieties towards tumor markers enhances the specificity of nanostructures by directly binding with target cells. Besides minimizing the off target effects, target ligand conjugated gold nanocomposites enhances the efficiency of theranostic probes by increasing the localization of these probes at the tumor site. Various methods have been reported to maximize the cell damage by designing specifically targeted and personalized nanomedicines. Some of such recent studies are discussed below in detail.
To reduce the off-target effects, Mangadlao et al. 101 demonstrated that PEGylated gold nanoparticles with surface-bound PSMA-I peptide had specifically targeted PSMA expressing PC3pip cell in vitro as well as in in vivo model. NIR imaging mediated by PC4 photosensitizer, showed faster (2 h) intracellular accumulation of nanoparticles compared to non-targeted nanoparticles (16 h). In vivo photodynamic studies have also demonstrated eight times more damage to PC3 pip cell xenograft by peptide conjugated nanoparticles compared with PC3 flu cell xenograft indicating specific localization of nanoparticles. Aptamers are receiving much interest as targeting moieties because of their high binding potential, ease of synthesis, non-immunogenic, high stability, cost effective etc. 102 . In this perspective, Alibolandi et al. 103 demonstrated the targeted theranostic effect of curcumin loaded PEGylated gold polyamidoamine dendrimer nanohybrids (PEG-AuPAMAM-CUR) conjugated with MUC1 specific aptamer (Apt). CT imaging studies revealed the specific accumulation of aptamer conjugated nanohybrids in the tumor and the results are depicted in Figure 3. Li et al. 104 reported nucleolin targeted gold nanostars as an efficient theranostic probe. The in vitro results demonstrated AS1411 aptamer conjugated nanostars to possess excellent photostability and faster intracellular accumulation. The porous nature of MSNPs enables encapsulation of drugs/fluorescence molecules at high payload and the sustained release of respective molecules into the surrounding environment. Thus amalgamation with MSNPs endows the properties of gold nanoparticles to encapsulate drugs at high payload. For instance, EpCAM aptamer conjugated PEGylated GNPs@MSNPs-5-Fluorouracil nanocomposites are developed to specifically target and damage EpCAM expressing xenograft. Here, GNPs acts as gatekeeper for the release of 5-Fluorouracil at the tumor site under suitable physiological conditions. Both in vitro and in vivo studies analyzed by flow cytometry and CT imaging respectively demonstrated higher cellular internalization of aptamer conjugated nanocomposites in EpCAM overexpressingHepG2 cell lines in comparison to non-expressing CHO cell lines and the non-targeted nanocomposites in both the cell lines. Furthermore, PEGylation enhanced the stability and biocompatibility of nanocomposites in the biological environment as confirmed with toxicitystudies. 105
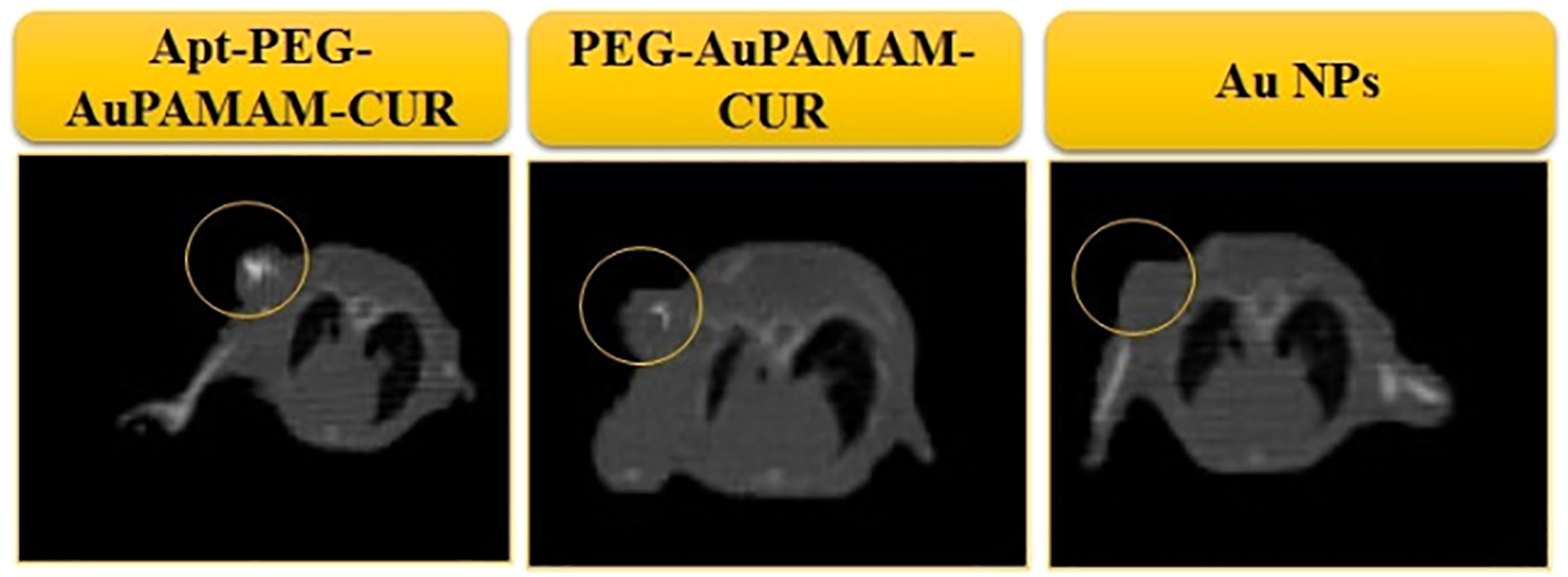
Clinical CT scan imaging of HC26 tumor-bearing mice 12 h post-injection of free gold nanoparticles (AuNPs), PEG-AuPAMAM-CUR and Apt- PEG-AuPAMAM-CUR. Reprinted with permission from reference. 103
Personalized medicine is evolving rapidly to overcome the hurdles (such as variation in the expression levels of specific tumor biomarkers from type of tumor/patient to patient) faced by existing targeted and non-targeted therapeutic strategies. Personalized medicines deliver the required quantity of drug to the site of tumor based on the patient profile (such as nature and level of tumor biomarker, patient health profile) which thus not only enhances the efficiency of drug but also decreases the off-target effects. In one such personalized nanomedicine approach, the tumor targeting peptides can be screened by in vivo phage display technique and conjugated with nanoparticles to promote PTT/chemotherapy. In this context, Qu et al. 106 demonstrated in vivo photoablation of MCF-7 breast tumor by peptide (screened by phage display technique) conjugated gold nanorods. The biodistribution studies showed that the percentage of target peptide conjugated nanorods was 63% after 1 day of administration which is comparatively higher than control peptide conjugated nanorods (i.e., 61% after 5 days). PTT studies were also consistent with the biodistribution studies. AS1411 aptamer with extended sequence conjugated to gold nanoparticles was further used to fabricate DNA template-silver nanoclusters as specifically targeted photoluminescent nanocomposites. The imaging of nucleolin overexpressing cell lines showed better imaging with aptamer conjugated nanocomposites at 50-100 times lower concentrations compared to silver nanoclusters. These nanocomposites were also found to be biocompatible and resistant to nuclease degradation. 107
Enhancing efficiency of PTT and in vivo imaging
Due to its non-invasive nature and utilization of NIR window for damaging cells, PTT and PDT techniques have grown as an alternative to conventional therapies in recent years. However, none of the nanostructures employing PTT technique have approved for human applications to treat cancers till date due to the inability of PTT to completely eradicate deeper tumors and metastatic tumors, less photostability. To overcome these limitations, researchers have been moved towards the synthesis of gold nanocomposites by functionalizing with chemotherapeutic drugs, immunoadjuvants, radiotherapeutic agents etc. Functionalizing gold nanoparticles with high atomic number elements, MR imaging contrast agents enhances the resolution of in vivo imaging which clearly demonstrates the state of tumor before and after PTT. Hence, various approaches to enhance the efficiency of PTT and in vivo imaging are discussed here.
Liu et al. 108 demonstrated the importance of gold-platinum nanodendrites (Au@Pt NDs) as CT imaging guided dual therapeutic (PTT + radiotherapy) agents. The harmonious effect of both radiotherapy and PTT had enhanced the cell death to 68%, whereas; PTT alone showed only 55% cell death. Further, CT imaging showed a linear increase in Hounsefield units with nanocomposite concentration and the results are in shown in Figure 4. Thus, these results prove that high atomic number elements enhance the PTT and radiotherapy efficiency of nanoparticles besides their capability as contrast agents. In another study, GNRs were functionalized with gadolinium oxysulfide to improve biocompatibility, photostability and amplify photoacoustic (PA) and MR imaging attributes. In vitro cytotoxicity and photostability studies on HeLa, Hep2, L02 cell lines demonstrated their non-toxic and photostable characteristics. Further, in vivo MR imaging studies showed great increase in relaxation rate (1/T1) as that of conventional MR contrast agents. Similarly, PA imaging also showed the linear increase in intensity of signal with concentration of nanocomposites. 109 Han et al. 110 developed multilayered gold core/shell nanocomposites (Fe3O4 core-gold nanoshell) to promote multimodal imaging and therapy by encapsulating polypyrrole (Ppy) as photothermal agent. The nanocomposites enhanced the X-ray attenuation intensity and 1/T2 and 1/T1 relaxation rates with increase in iron content, thus improving CT imaging and MR imaging respectively. Furthermore, the synergistic effect of both polypyrrole and gold nanoshell had endowed the photothermal efficiency of nanocomposites under 808 nm NIR laser irradiation for 5 min. Some other studies have shown Fe3O4 functionalized gold nanostructures as better contrast agents for MR imaging.111–113 Liu et al. 114 fabricated gold nanostars with manganese oxide (MnO2) nanosheets to employ them as efficient MR imaging guided PTT agents. Similarly, carbon nanotubes rings fabricated with gold nanoparticles (CNRT@GNPs) enhanced the extinction intensity and SERS signal by 120 and 110 folds than that of carbon nanotubes alone. The enhancement in theranostic property is rendered by the synergistic plasmonic coupling effect of both gold nanoparticles and inner surface of carbon nanotubes ring. 115 Now-a-days transition metal carbides based gold nanocomposites such as titanium carbide@gold (Ti3C2@gold) nanocomposites are gaining much interest due to tuning of their LSPR towards second window of near infrared (NIR-II) region.
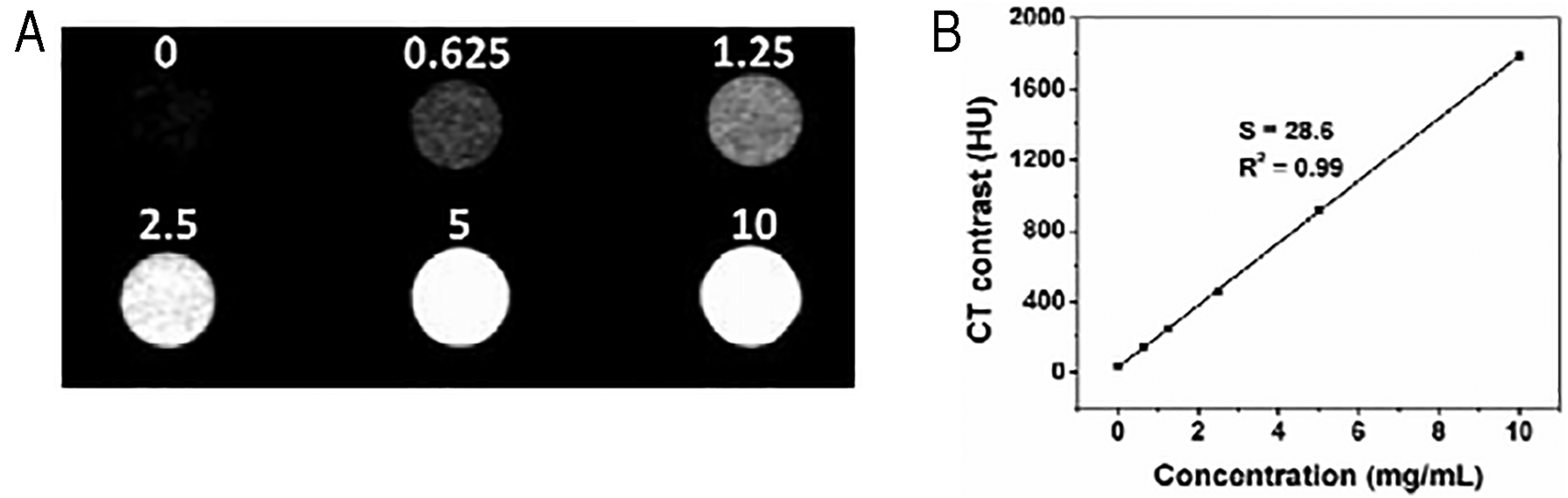
(a) CT images of the Au@Pt NDs aqueous dispersion with different concentrations (mg/mL). (a) CT value (HU) of the Au@Pt ND as a function of the concentration of NDs. Reprinted with permission from reference. 108
These nanocomposites improve PA and CT imaging due to their strong X-ray attenuation ability and optical properties. Furthermore, the strong absorption in NIR-II, improves radiotherapy by inducing tumor hypoxia via mild PTT under laser irradiation. 116 The other nanocomposites that have grabbed the attention of researchers in biomedical applications are graphene-gold nanocomposites due to their biocompatible nature, enhanced PTT effect, etc. In this respect, Chlorin e6 photosensitizer conjugated graphene-gold nanostars based nanohybrids were designed to improve PTT by the synergistic effect of both graphene and gold nanostars in addition to photodynamic therapy (PDT) by chlorine e6. LSPR of nanocomposites was tuned to excite chlorine e6 in such a way that a single wavelength laser can allow both PTT and PDT. This dual ablation therapy had greatly enhanced the tumor damage in comparison to either PTT or PDT which is confirmed by H&E staining of tumor tissue. 117 Gold-silver-carbon quantum dots based nanocomposites have also been proved to be better photothermal agents with high biocompatibility and stability. 118
In summary, fabrication of gold nanostructures with other noble metals besides enhancing the PTT efficiency also increases the photostability of nanostructures which maximizes the cell death. Graphene-gold nanocomposites enhance the biocompatible nature and PTT efficiency of theranostic nanoprobe. Nanoalloying with metals such as iron oxide, manganese oxide endows the resolution of in vivo imaging due to their high conductive nature. All these studies drive nanotheranostic probes to be efficient contrast agents for in vivo imaging and PTT probes.
Conclusion
To date, many gold nanostructures based theranostic probes have been designed and developed for clinical management of cancer. Some of them have reached the clinical trials phase, but the rising statistics of cancer deaths demand the need for improvements in nanostructures based imaging and therapeutics. In this context, this communication summarizes the recent achievements in the development of gold nanostructures based theranostic probes for targeting cancer. The development of new approaches such as self-therapeutic feedback, labeling with macrophages has proven gold nanostructures as efficient theranostic probes. Inspite of these advancements, gold nanostructures still faces challenges of in vivo toxicity, specific targeting, etc. To resolve these issues, gold nanocomposites with other organic and inorganic nanostructures are employed to improve their biocompatibility, imaging properties, cargo loading range, PTT/PDT efficiency, circulation time and biodistribution. These progressive studies establish gold nanostructures and their composites as better nanotheranostic probes to cope up with tumor heterogeneity and its progression.
