Abstract
Along with the increasing requirement for efficient organic conversions using green chemistry, there is a need to develop highly efficient and eco-friendly catalytic reaction systems. Gold nanoparticles (AuNPs)-based nanocatalysts are promising candidates for the reduction of environmental pollutants, such as nitroaromatics and dyes. This study reports on the green synthesis of AuNPs using
Keywords
Introduction
The high impact of nanotechnology on many important areas such as medicine, energy, electronics, transport and space has created a surge in creating greener systems and approaches to solve major health, environmental and energy challenges. Nanotechnology is mainly geared towards the manufacturing of materials that are 10−9 m in size, 1 where at this size they possess unique physical and chemical properties with novel applications. 2 The application of green chemistry principles in nanotechnology for the synthesis of biogenic nanomaterials can help resolve the limitations associated with the conventional methods of synthesis i.e. the chemical and physical approaches. Green synthesis has potential to reduce the use and generation of hazardous substances during the manufacturing and application of nanoparticles (NPs) produced through the conventional methods.3,4 Biogenic NPs are considered to be more safe, eco-friendly, biocompatible and less toxic when compared to the chemically synthesized NPs. 5
Green synthesis of NPs can be accomplished using plant extracts (also called phytosynthesis) and microorganisms. Plant extract-based synthesis has distinctive features such as a higher rate of NPs formation compared to the synthesis rate using microorganisms, no additional reagents are required for synthesis, and the NPs are easy to clean-up, and up-scale.6–8 In addition, plant extracts contain a wide range of biomolecules and metabolites, such as terpenoids, vitamins, polysaccharides, proteins, alkaloids, phenolic compounds, enzymes, etc., 8 which can act as reducing and capping/stabilizing agents in the synthesis of NPs.9–12 Plant extracts-based NPs have a large range of biological properties, such as antibacterial, antifungal, anticancer, anti-biofouling, anti-malarial, anti-parasitic, antioxidant 6,13,14 and catalytic activity.14–17
The use of green-synthesized AuNPs in catalysis, especially for their catalytic reduction of the environmental pollutants, such as p-nitrophenols, are well studied.14–17 These pollutants are by-products in the production of herbicides, pesticides and synthetic dyes, and the US Environmental Protection Agency (EPA) has classified them as priority pollutants due to their toxicity. Green synthesized biogenic AuNPs have successfully reduced these pollutants to p-aminophenols in the presence of sodium borohydride (NaBH4).16,18,19 Due to the high surface area, AuNPs and other NPs show greater catalytic activity at lower concentrations compared to traditional catalysts.20,21 As catalysts, AuNPs are also easy to recover, can be reused several times, and the final product is metal free. 20 In contrast, the conventional environmental remediation approaches for the degradation of nitroaromatic compounds are harmful to the environment.22,23
In this study, Photographs of 
The fruit contains an edible sweet-sour slimy pulp with slimy brown seeds and has a strong, astringent taste. The common medicinal use of sour figs is for purgation,
33
while the dried fruits are processed into jam or added to curry dishes. However, huge amounts of this fruit end up as waste in landfills or are discarded with the dried plants. Therefore, we explored the potential industrial applications for this fruit. So far, and to the best of our knowledge, there is no report on the green synthesis of AuNPs using the
Materials and Methods
Preparation of CeFE
Dried
HPLC analysis of the polyphenols
Phenolic compounds present in CeFE were identified using HPLC following a previously published protocol.34–36 The extract was filtered through a 0.22 µm PTFE filter membrane. The solvents used were water (A) and methanol (B), acidified with 0.1 % formic acid and served as mobile phase at a rate of 0.2 ml/min. Gradient elution was applied as isocratic 5% B from 0 – 3 min, a linear gradient increasing from 5 % - 100 % B from 3 – 43 min, kept at 100 % B from 43 – 61 min, a linear decreasing gradient from 100 % to 5 % B from 62 – 70 min. The temperature was kept at 25°C. The column was combined with a Q-exactive orbitrap mass spectrometer, and the spectra was recorded in positive and negative ion mode, respectively.
Synthesis and optimization of biogenic CeFE-AuNPs
CeFE-AuNPs were synthesized by heating NaAuCl4∙xH2O to a desired temperature for 5 min, and subsequent addition of CeFE in a 9:1 ratio. The formation of the CeFE-AuNPs was confirmed by change in colour from light yellow to ruby-red. Various parameters were tested to obtain the optimum conditions: temperature (25, 37, 50, 80 and 99°C), pH of extract (2.9 – 10), concentrations of gold (0.1 0.25, 0.5 and 1 mM) and CeFE (0.25, 0.5 and 1 g/ml). The reaction was continued for 1 h while shaking in the orbital shaker at 1000 rpm (Eppendorf Thermomixer Comfort, Hamburg, Germany), then cooled to ambient room temperature. The tubes were centrifuged at 14,000 rpm for 15 min at room temperature. The pellets were washed 3 times with deionised water before characterization.
CeFE-AuNPs Characterization
UV-Vis Spectroscopy
UV-Vis spectrophotometer was used to confirm the formation of AuNPs. Exactly 0.03 ml of the CeFE-AuNPs samples were mixed with 0.27 ml of deionized water in a 96-well flat-bottom microplate. The absorbance was read in the wavelength range of 300 - 700 nm using the POLARstar Omega microplate reader (BMG Labtech, Offenburg, Germany).
Size and Hydrodynamic Density
CeFE-AuNPs sample was diluted 10-times to 1 ml. The particle size distribution, polydispersity index (PDI) and zeta potential measurements of the
HRTEM
One drop of the CeFE-AuNPs solution was loaded onto a carbon coated copper grid. The grids were dried for a few minutes under a Xenon lamp. The High Resolution TEM (HRTEM) images were captured using FEI Tecnai G2 20 field-emission gun (FEG) HRTEM operated in bright field mode at an accelerating voltage of 200 kV. The elemental composition of the AuNPs was characterized using EDX liquid nitrogen-cooled lithium-doped Silicon detector. These analyses were performed by the Electron Microscopy Unit, UWC Department of Physics.
FTIR Spectroscopy
The FTIR analysis was performed using the PerkinElmer Spectrum One FTIR spectrophotometer (Waltham, MA, USA) according to the previously reported method. 13 The dried CeFE and CeFE-AuNPs were mixed with the KBr powder and pressed into a round disk. A pure KBr disk was used for background correction.
Total phenolic content analysis
The total phenolic content (TPC) of CeFE was determined using Folin-Ciocalteu reagent (Sigma-Aldrich, USA) as described previously. 37 Total polyphenols were expressed as milligrams Gallic Acid Equivalents per ml of extract (mg GAE/ml).
Catalytic Reduction of 4-Nitrophenol and MB
The reduction of 4-nitrophenol and MB was carried out at room temperature in a 96-well flat-bottom plate as previously described.16,22,38–40 The reaction mixture consisted of 0.14 ml water, and 0.03 ml of 2 mM 4-nitrophenol or MB, 0.1 ml 0.03 M sodium borohydride and 0.03 ml CeFE-AuNPs. The absorption spectra were read in the 200 - 750 nm wavelength range using the POLARstar Omega microplate reader at 1 min intervals from 1 to 10 min, thereafter at 5 min intervals to 60 min.
Determination of 4-nitrophenol reduction to 4-AP by HPLC
The conversion from 4-nitrophenol to 4-AP was determined using reverse phase high performance liquid chromatography (RP-HPLC), as previously described. 16
Results
Characterization of CeFE
Identification of CeFE polyphenols by HPLC analysis.

CeFE-AuNPs synthesis and characterization
CeFE-AuNPs were synthesized with CeFE at different pH conditions (2.5 – 10) by varying one factor at a time, namely: temperature, gold salt and CeFE concentrations. These four parameters are known to strongly affect the hydrodynamic size and shapes of NPs.42,43 The formation of CeFE-AuNPs was confirmed by both changes in colour from light yellow to a ruby-red colour and UV-Vis spectroscopy.
UV-Vis characterization
Temperature and pH have been shown to have great impact on the synthesis of AuNPs.
44
As shown in Figure 2, the reduction of gold ions into AuNPs by CeFE was evidenced by the UV-Vis spectra showing characteristic, well-defined surface plasmon resonance (SPR) band at around 535 - 540 nm. UV-Vis spectra for the synthesis of CeFE-AuNPs at different pH and temperature conditions.
This phenomenon is attributed to the excitation of the SPR of AuNPs. All pH and temperatures tested produced AuNPs. The pH ranging from 2.5 to 8 produced CeFE-AuNPs that were approximately 100 nm in size, as determined by dynamic light scattering (DLS). CeFE at pH 9 and 10 produced redshifted AuNPs, which suggested that larger AuNPs were produced or that aggregation had occurred. DLS analysis of pH 9 and 10 samples revealed that the synthesized AuNPs had a small size range (23.57 – 60.77 nm) and higher PDI range (0.805 – 0.350), thus indicating polydispersity. Coupled with the blue colour of the AuNPs at basic pH, the NPs were considered unstable. This instability could be due to insufficient amounts of CeFE phytochemicals to cap the AuNPs.
These results confirmed the vital role played by pH in controlling the shape and size of the AuNPs. It has been shown that protonation and deprotonation surface reactions are used to obtain local surface charge which depends on particle size and pH. 45 At higher pH, the large number of phenolic functional groups available for gold binding possibly facilitated a higher number of Au+ ions to rapidly nucleate and subsequently form a large number of AuNPs with smaller diameters. It was observed that, if the unknown pH-precipitated material is not removed by centrifugation, this tended to inhibit the formation of AuNPs, Therefore, the impact of the removal of these unknown materials, which could possibly include proteins, amino acids and other phytochemicals that are pH sensitive, in the synthesis of AuNPs is currently unknown and will be further investigated.
Temperature effects were also explored for the synthesis of CeFE-AuNPs (Figure 2). Lower temperatures (25 – 50°C) favoured the formation of CeFE-AuNPs with higher yields (Figure 3(a)) which is represented by the peak intensities and the Amax (Figure 3(b)), the higher the absorbance of the AuNPs the higher the concentration of the AuNPs formed. These AuNPs had a larger hydrodynamic sizes and uniform size distribution (Figure 3(c)), which is in accordance with previous reports.44,46,47 Since the CeFe-AuNPs produced at lower temperatures had similar properties, 25°C was selected as an optimal temperature to fabricate the CeFE-AuNPs. This bodes well for the green chemistry principles, as no external energy was required for the synthesis of CeFE-AuNPs. The optimum pH was pH 8 (Figure 3), because the UV-Vis spectrum was narrow and symmetrical. This suggested that the CeFE-AuNPs were monodispersed. The effects of temperature on the synthesis of CeFE-AuNPs using CeFE at pH 8. a) UV-Vis spectra at different temperatures; b) effect of temperature on the yield (maximum absorbance/Amax and peak intensity) of CeFE-AuNPs; c) effect of temperature on size and PDI, and d) the rate of synthesis of CeFE-AuNPs.
Moreover, increasing temperature was associated with reduced synthesis (yield) of CeFE-AuNPs at pH 8 (Figures 3(a) and (b)), and this was further associated with increases in polydispersity and reduction in the hydrodynamic size of AuNPs (Figure 3(c)). The synthesis of CeFE-AuNPs reached saturation within a minimum time of 8 min, and the synthesis was completed after 15 min (Figure 3(d)).
The results further confirm that temperature is also an important parameter for the synthesis of CeFE-AuNPs. Previous studies have shown that lower temperatures favour the formation of larger AuNPs, and higher temperatures favour the formation of smaller AuNPs.44,48,49 This is attributed to the fact that increasing the reaction temperature led to a rapid reduction of the metal precursors, and the subsequent homogeneous nucleation of gold nuclei, allowing for the formation of smaller spherical AuNPs. Furthermore, it has been well demonstrated that the synthesis of AuNPs using certain plant extracts require heat activation, but only to a certain point.50,51 This is because certain reducing agents in the plant extract gets inactivated at higher temperatures. This phenomenon is also seen by the decrease in absorbance at 100°C and the increase in absorbance or peak intensity at 25 – 50°C.
The concentrations of CeFE and gold solution required for CeFE-AuNPs synthesis were optimized as shown in Supplement Figure S1. The NaAuCl4∙xH2O concentration at 0.1 and 1 mM was either insufficient or too high to synthesize stable AuNPs at 25°C for all CeFE concentrations, respectively. CeFE-AuNPs with desired optical properties were synthesized at 0.25 and 0.5 mM of NaAuCl4∙xH2O at 0.125 – 0.5 g/ml of CeFE. The optimal conditions were thus selected as 0.25 g/ml of CeFE at pH 8 and 0.5 mM NaAuCl4∙xH2O, because the UV-Vis spectrum was narrower, and the SPR was higher than that of the 0.5 mM NaAuCl4∙xH2O and 1 g/ml CeFE. The synthesis was scaled-up using these optimized conditions, and the UV-Vis of the CeFE-AuNPs is shown in Figure 4, with an SPR peak at 535 nm, Amax of 2.1 and a peak intensity of 1.3. UV-Vis spectrum of CeFE-AuNPs synthesized at the optimal conditions. The CeFE-AuNPs were synthesized at 25°C with pH 8 CeFE (0.25 g/ml) and 0.5 mM gold salt.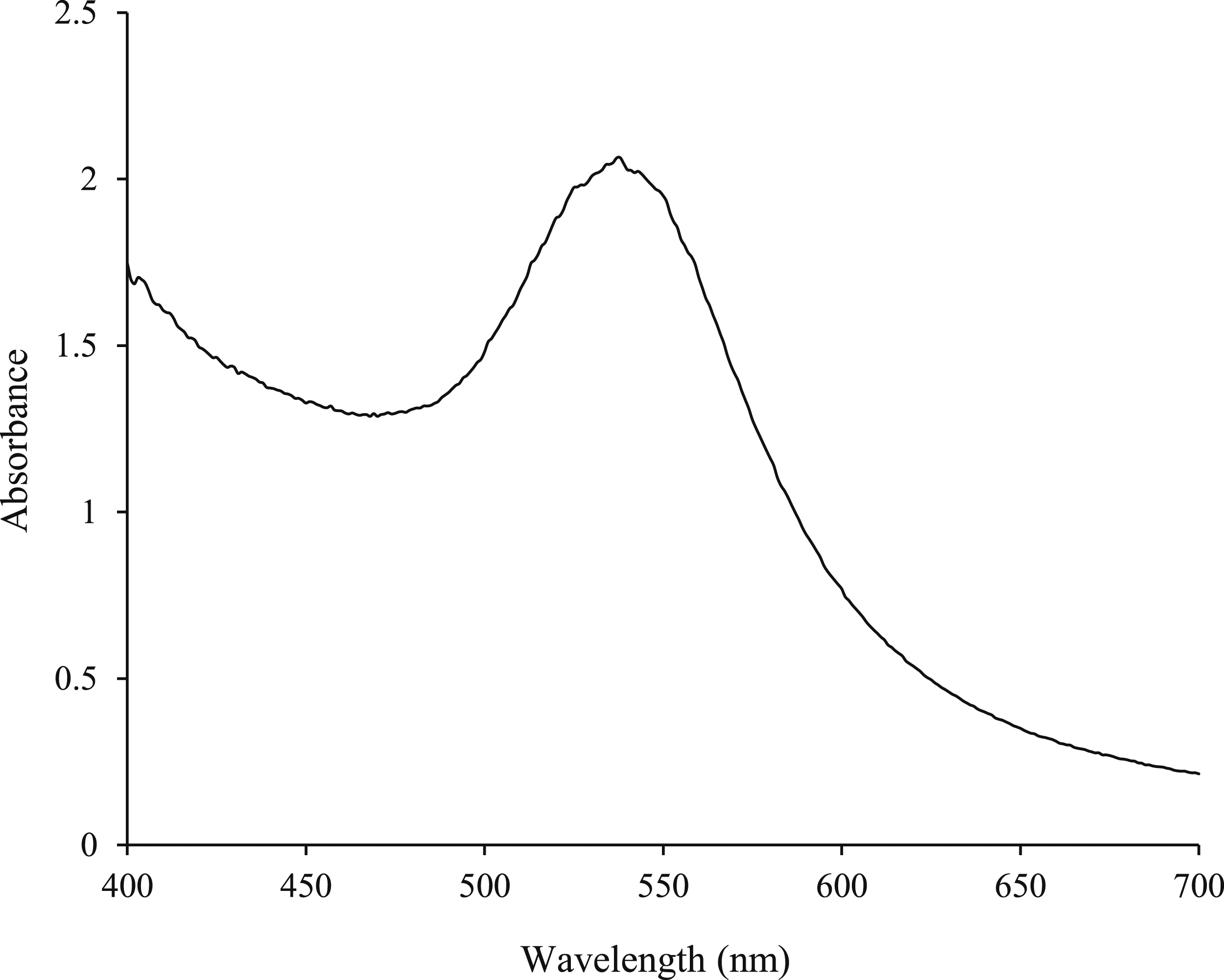
Furthermore, the analysis of the absorption maxima for CeFE-AuNPs at various pH conditions explicitly depicted a gradual shift towards lower wavelengths with an increasing pH from 3 to 7, indicating that the sizes of the particles decreased when pH changed from highly acidic to neutral i.e., from 3 to 7. When the pH was increased from 7 to 10, the absorption maxima shifted towards higher wavelengths, which suggested that the size of the NPs increased when pH is increased from 7 to 9. Taken together, this suggested that the synthesis of smaller CeFE-AuNPs can be synthesized when the pH of the CeFE is around 7. This data is corroborated by Singh & Srivastava. 52
DLS and TEM characterization
DLS and TEM were used to determine the size, shape and morphologies of CeFE-AuNPs synthesised from CeFE produced at pH 8. The DLS showed size distribution in the range of 50 to 200 nm, with an average hydrodynamic size of 108.0 ± 0.2 nm (Figure 5(a)). It is possible that the NPs were synthesized from a combination of different phytochemicals. The TEM images indicated that the CeFE-AuNPs obtained were mostly spherical in shape, with an average core size of 32.96 ± 4.39 nm (Figures 5(b) and (c)). Furthermore, the AuNPs had a “halo” (Figure 5(b): insert), which is indicative of an organic capping layer around the NPs surface.
53
Size distribution of CeFE-AuNPs as measured by DLS (a) and TEM (b). The core size distribution from the TEM micrograph was analysed by ImageJ with Gaussian fitting and represented as a histogram (c).
The mean particle size determined by the TEM analysis was significantly smaller (33 nm) than that measured by the DLS (108 nm). The adsorption of organic stabilizers from the extract on the surface of AuNPs, and the adsorption of water on the stabilized AuNPs could be responsible for this discrepancy, which could have had an effect on the average particle hydrodynamic size obtained by the DLS.54–56 Therefore, CeFE served as a good reducing and a capping agent in the synthesis of AuNPs and yielded monodispersed AuNPs. Since these analyses were performed for the optimized CeFE-AuNPs samples, it is unknown whether AuNPs synthesised at other pH values would give the same results, as different green synthesis methods have been shown to synthesize quasi-spherical AuNPs with an average diameter ranging between 40 and 100 nm. 13
EDX spectrum and SAED pattern
The EDX spectrum showed strong signals for gold atoms, with the strongest signal peak at 2.3 keV,
57
demonstrating that the AuNPs have been formed (Figure 6(a)). The elements such as C, O and Cu were also detected. The C and O could be from the organic compounds present in the extract that played a role in the reduction and stability of the AuNPs. The Cu in the spectrum was from the carbon-coated copper grid used for TEM analysis. As shown in Figure 6(b), SAED pattern analysis indicated the Miller indices at (111), (200), (220), (311), and (222), indicating that the synthesized AuNPs have a phase crystal of face-centred cubic.
58
The formation of AuNPs and their crystal phase was in accordance with the Joint Committee on Powder Diffraction Standards of Au (JCPDS number 04-0784).
58
The EDX spectrum (a) and SAED pattern (b) of the synthesized CeFE-AuNPs. These are representative images of 3 independent experiments done in triplicate.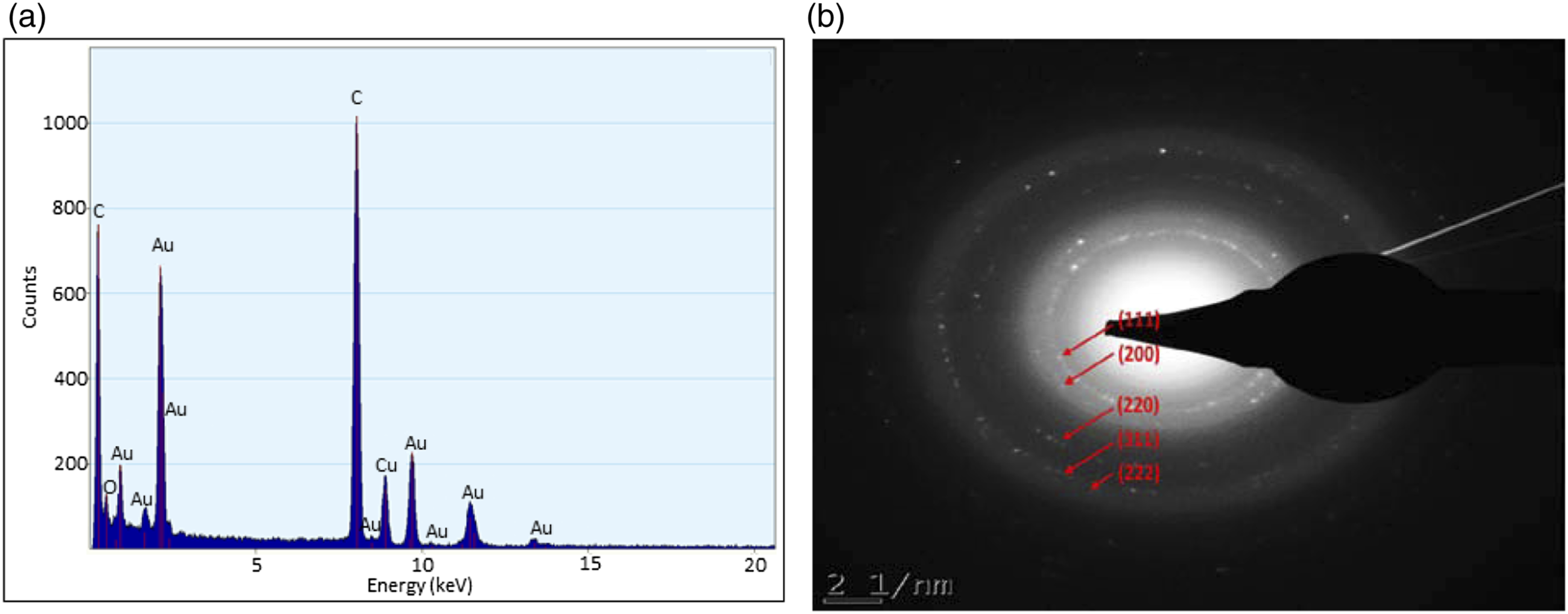
FTIR analysis
The FTIR spectroscopy revealed the surface chemistry of the CeFE-AuNPs compared to CeFE (Figure 7). The band at 3306.15 cm−1 is assigned to the O–H and N–H stretching vibration of carboxylic acid and proteins from the extract. The peak at 1631.99 cm−1 corresponds to the bending vibration of amide I, and this arises due to the carbonyl stretching vibration in the amide linkage of proteins.
59
FTIR analysis of the CeFE and the CeFE-AuNPs.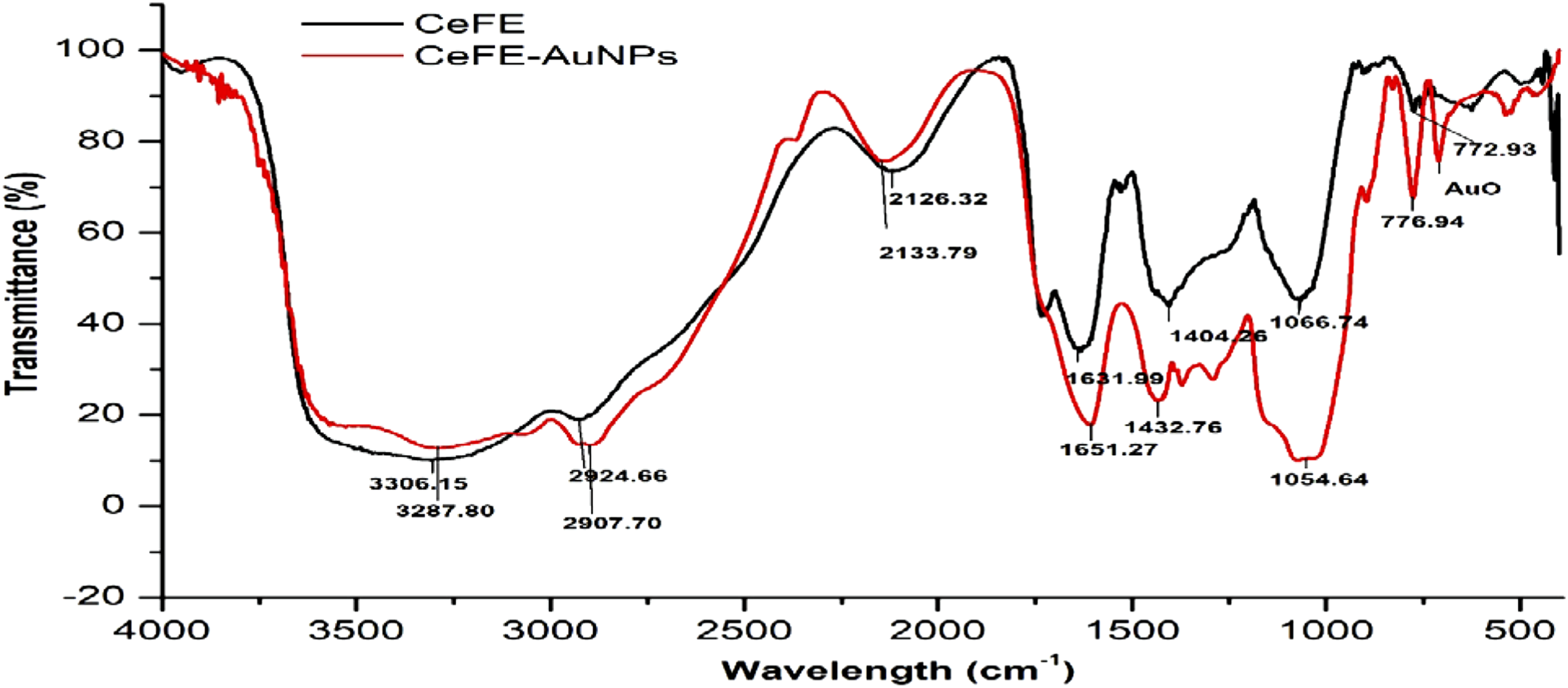
Shifts of the FTIR spectra bands (cm−1) of major peaks of CeFE and CeFE-AuNPs.

Note: The shift values were calculated by subtracting the peak transmittance of the AuNPs from that of the extract.
The spectrum of the CeFE-AuNPs exhibited absorption bands at 3287.8, 2907.6, 1615.3, 1432.8, 1054.6 and 776.9 cm−1. These bands are associated with polyphenols.
22
Comparison of the free CeFE and CeFE-AuNPs polyphenols indicated shifts in the absorption bands of the carbonyl, hydroxyl and aromatic ring groups (Table 2), which further confirmed that the polyphenols are present on the surface of CeFE-AuNPs. Since CeFE has high levels of catechin, epicatechin and EGCG (Table 1) and reducing sugars, these could play a major role in the AuNPs synthesis, as catechin 60,61 and EGCG
62
have been shown to produce AuNPs with diverse shapes. The possible mechanism of formation of CeFE-AuNPs is shown in Figure 8. Green synthesis of AuNPs by CeFE. Chemical structures of abundant compounds in CeFE, which may be responsible for the reduction of Au+ ions and synthesis of CeFE-AuNPs.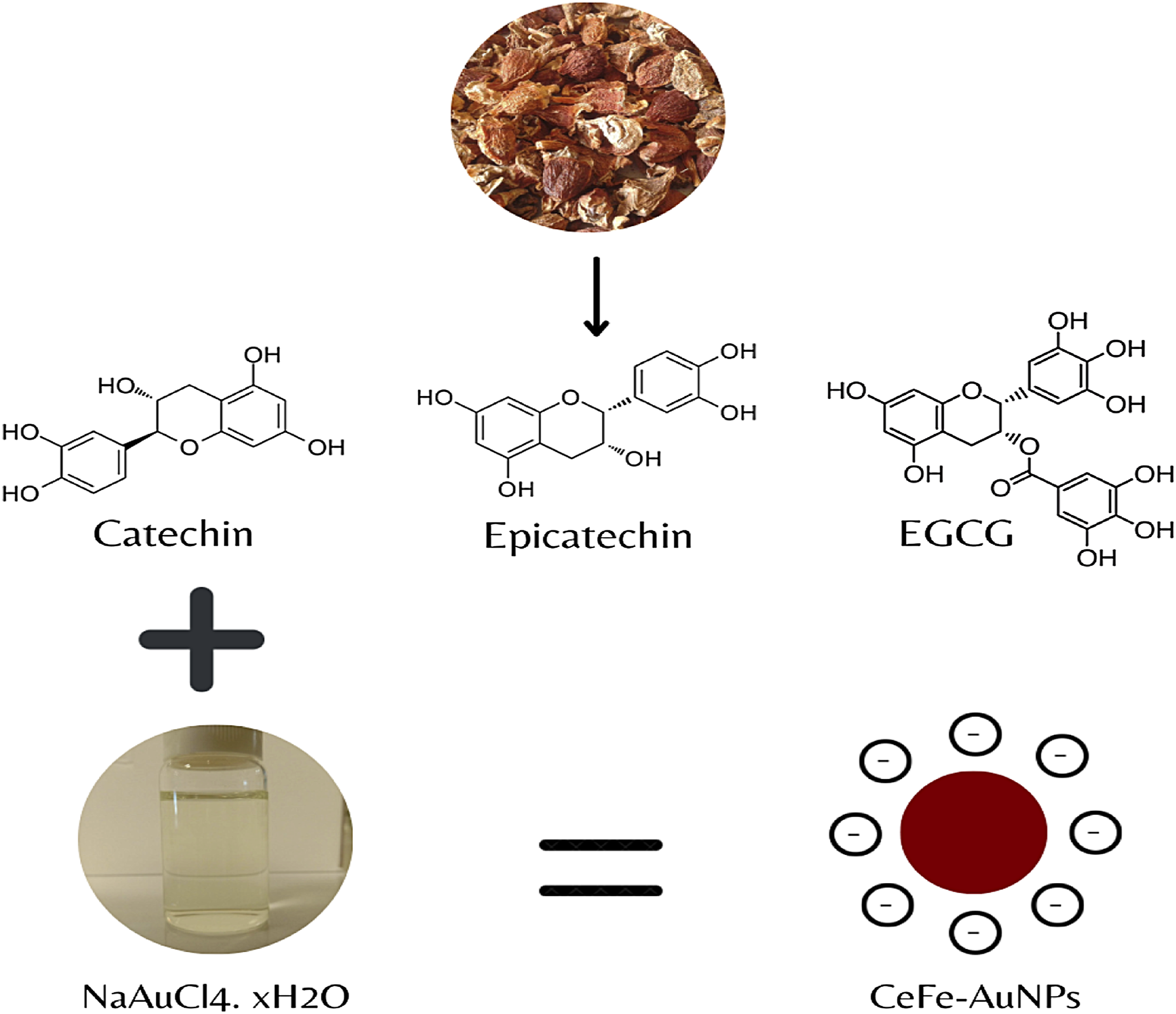
Catalysis
Reduction of 4-nitrophenol
The catalytic activity of green synthesized AuNPs using various plant extracts have been reported.38,63–69 CeFE-AuNPs also displayed catalytic activity in the reduction of 4-nitrophenol to 4-AP (Figures 9(a) and (b)) and MB to leucoMB (Figures 9(c) and (d)) by NaBH4. In this study, CeFE-AuNPs act as a catalyst and overcome the kinetic barrier of hydrogenation and facilitate the electron exchange between the donor and acceptor (Figure 9). Addition of NaBH4 to a light yellow 4-nitrophenol solution resulted in the formation of a bright green solution, which showed an absorption peak at 400 nm, due to the formation of the 4-nitrophenolate anion. The CeFE did not show any catalytic activity when added to the 4-nitrophenol and NABH4 solution (Supplemental Figure S2), only the 4-nitrophenol conversion to 4-nitrophenolate anions occurred but there was no formation of 4AP up to 60 min. In contrast, addition of CeFE-AuNPs resulted in the reduction of 4-nitrophenol to 4-nitrophenolate anion, with the concomitant appearance of a peak at 300 nm because of the 4-AP formation (Figure 9(a)). This reaction was very rapid and completed within 10 min when using as-synthesized CeFE-AuNPs, with a reaction rate of 3.3 x 10−3 s−1 (Figure 9(b)). This rate is equivalent to the catalytic activity of caffeic acid-AuNPs.
16
CeFE-AuNPs completely catalysed the reduction of 0.2 mM 4-nitrophenol to 4-AP in the presence of NABH4, with no residual 4-nitrophenol, as confirmed by HPLC. UV-Vis spectra showing the catalytic reduction of 4-nitrophenol (a) and MB (c) by NaBH4 in the presence of CeFE-AuNPs. Plots of the pseudo-first order reaction kinetics for the reduction of 4-nitrophenol (b) and MB (d) by CeFE-AuNPs in the presence of NaBH4.
Reduction of MB
Addition of CeFE to a light blue MB solution resulted in the formation of a dark blue solution, which was associated with a decrease in the absorption peak at 665 nm. Addition of CeFE-AuNPs resulted in similar characteristics. However, no further reduction of the peak occurred over time, indicating that, in the absence of NaBH4, no activity was possible. Addition of NaBH4 to CeFE+MB resulted in further decrease of the peak at 665 nm. In contrast, addition of CeFE-AuNPs resulted in the reduction of MB to leucoMB over time (Figure 9(c)). This reaction was very rapid and completed within 10 min when using as-synthesized CeFE-AuNPs, with a reaction rate of 1.33 x 10−3 s−1 (Figure 9(d)).
The above reaction kinetics were in good agreement with the pseudo-first order equation: where At is the absorbance at different time points, kapp is the apparent rate constant, and t is the reaction time (in seconds). These results are consistent with previously published studies on the catalytic capacity of biogenic/green synthesized AuNPs.70–72 The possible mechanism of catalytic action of CeFE-AuNPs is depicted in Figure 10. The role of AuNPs in these reactions is still not clear, however, studies have speculated that the NPs as catalysts might possibly overcome the kinetic barrier of hydrogenation and facilitate electron relay from the donor to the acceptor.
73
In this case, the CeFe-AuNPs speed up the degradation of the two dyes by facilitating the electron exchange between NaBH4 and 4-nitrophenol, as well as NaBH4 and MB dye. The reduction of these compounds formed 4-AP and leucoMB, respectively. The catalytic activity and efficiency of AuNPs are dependent on their size, shape, surface composition and pH
74
; and have been reported to have enzyme-like catalytic activities as either antioxidant or pro-oxidant agents.
73
Possible mechanism of catalytic reduction of 4-nitrophenol and MB dye with NaBH4 in the presence of CeFE-AuNPs.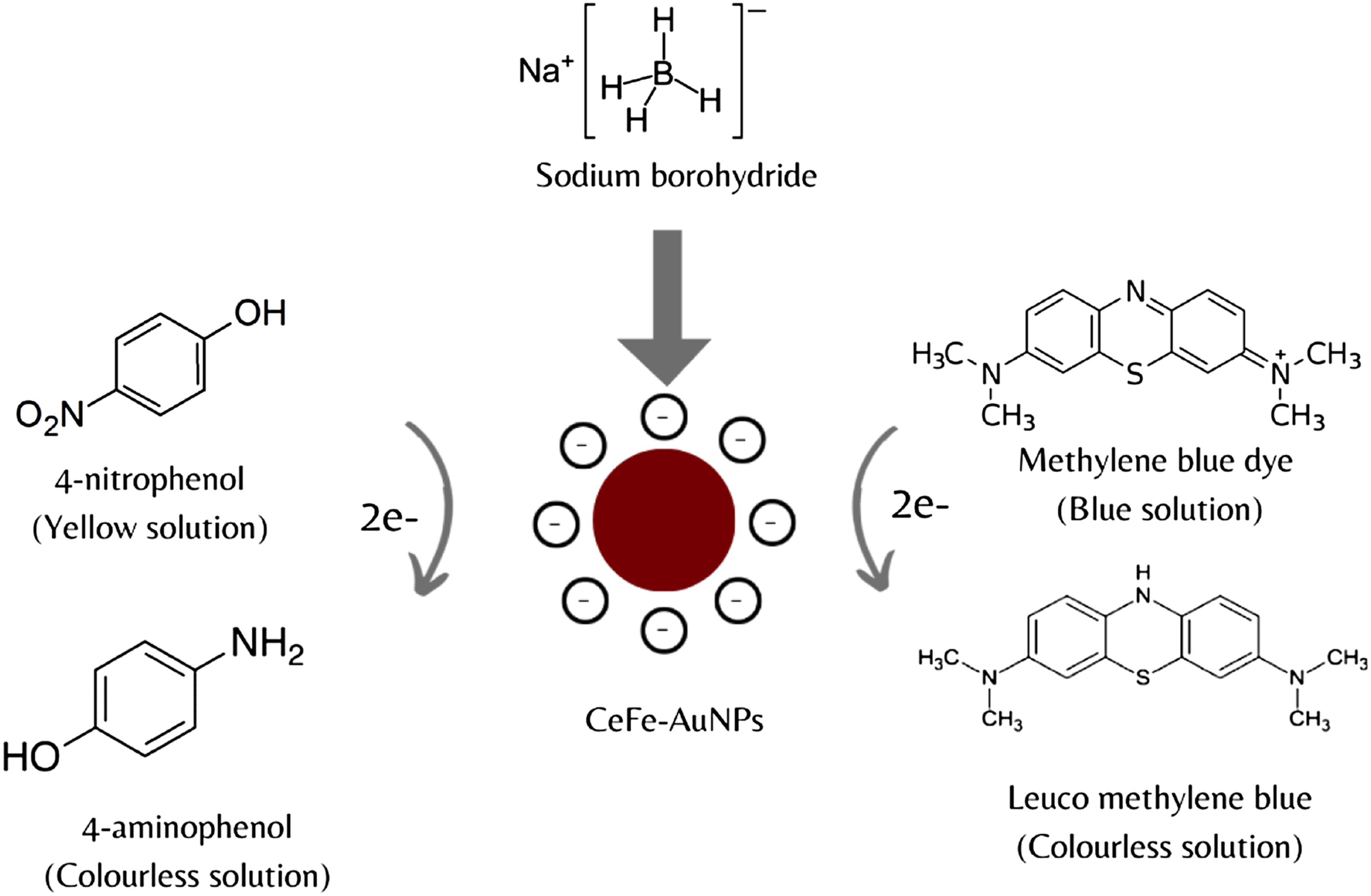
Conclusion
A facile one-step green synthesis protocol that utilizes CeFE as a reducing, capping and/or stabilizing agent to generate highly stable CeFE-AuNPs has been developed. This method produced spherical AuNPs that were characterized using various techniques. These biogenic AuNPs possessed high catalytic activity for the reduction of 4-nitrophenol to 4-AP and MB to leucoMB, respectively, and could be useful in the elimination of these environmental pollutants. If not consumed, this fruit would have ended up as waste, but instead was used to produce AuNPs at low cost that can be used to clean up water pollutants. The synthesised AuNPs can also be retrieved and used in another catalytic reaction, further emphasizing their low cost. The eco-friendliness of these AuNPs will be evaluated in future.
Supplemental Material
Supplemental Material - Catalytic reduction of 4-nitrophenol and methylene blue by biogenic gold nanoparticles synthesized using Carpobrotus edulis fruit (sour fig) extract
Supplemental Material for Catalytic reduction of 4-nitrophenol and methylene blue by biogenic gold nanoparticles synthesized using
Footnotes
Acknowledgements
The authors acknowledge the Department of Science and Innovation/Mintek Nanotechnology Innovation Centre Biolabels Unit for funding this project. The authors thank the UWC Departments of Physics and Chemistry for their assistance with HRTEM and FTIR analyses, respectively. The authors thank Dr. Sarah D’Souza for HPLC analysis of 4-nitrophenol and 4-AP and Mr. Fanie Bezuidenhout at Cape Peninsula University of Technology (CPUT) for TPC and HPLC analysis of CeFE. Nicole Sibuyi is now working at the Health Platform Diagnostic Unit (Advanced Materials Division, Mintek, Johannesburg, South Africa).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article from the Department of Science and Innovation/Mintek Nanotechnology Innovation Centre Biolabels Unit and the University of the Western Cape.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
