Abstract
A gold nanoparticle solution was prepared by adding sodium borohydride (NaBH4), trisodium citrate dihydrate (C6H5Na3O7·2H2O), cetyltrimethyl ammonium bromide (CTAB,(C16H33)N(CH3)3Br), ascorbic acid (C6H8O6), and potassium tetrachloroaurate(III)(KAuCl4) to distilled water and stirring the solution for 15 min. [C60]fullerene nanowhisker-gold nanoparticle composites were synthesized using C60-saturated toluene, the gold nanoparticle solution, and isopropyl alcohol by liquid-liquid interfacial precipitation (LLIP).
The product of the nanocomposites was characterized by X-ray diffraction, scanning electron microscopy, Raman spectroscopy, transmission electron microscopy, and solid-state 13C-nuclear magnetic resonance spectroscopy. The catalytic activity of the [C60]fullerene nanowhisker-gold nanoparticle composites was confirmed in 4-nitrophenol reduction by UV-vis spectroscopy.
Keywords
1. Introduction
The best known fullerene is C60, which consists of carbon bonded into 12 pentagonal and 20 hexagonal rings [1]. Polymerization of [C60]fullerene can occur by a cycloadditive reaction, which forms a four-membered ring between adjacent [C60]fullerenes [2]. A crystalline fibre of this carbon nanomaterial is called a [C60]fullerene nanowhisker. [C60]fullerene nanowhiskers can act as an n-type semiconducting material, and are used in a wide range of applications, including photocatalysis, chemical sensors, solar cells, field-effect transistors, and electronic devices [2,3].
The liquid-liquid interfacial precipitation (LLIP) method relies on diffusion of a poor fullerene solvent, such as isopropyl alcohol, into a saturated toluene solution of fullerenes [4]. [C60]fullerene nanowhiskers are synthesized using the LLIP method [4], which has been widely applied [5-12]. The [C60]fullerene nanowhiskers show good electrical conductivity [13] and have a large surface area [14, 15]. The growth of [C60]fullerene nanowhiskers is affected by temperature, light, ratio of poor solvent to good solvent during LLIP, and concentration of water [16-20]. [C60]Fullerene nanowhiskers are composed of C60 molecules weakly bonded by van der Waals forces [21,22]. After electron beam irradiation, [C60]fullerene nanowhiskers show higher Young's modulus [24] than pristine C60 crystals [22], and stronger thermal stability [23]. When Miyazawa et al. used Raman laser-beam irradiation, [C60]fullerene nanowhiskers became polymerized [21,22]. Rao et al. found that the shift of Ag(2) peak was a good indicator for [C60]fullerene nanowhisker polymerization, as the peak Ag(2) pentagonal pinch mode of C60 shifted downward, from 1469 cm−1 to 1457 cm−1 upon photopolymerization [22,25]. Gold nanoparticles have been used for hydrogenation of aromatic nitro compounds because of their higher catalytic efficiency compared to bulk catalysts, which can be attributed to their large surface-to-volume ratio [26]. The dispersion of gold nanoparticles on nanoporous fullerene nanowhiskers is attractive for catalytic applications [27,28]. The reduction of 4-nitrophenol to 4-aminophenol in the presence of NaBH4 with gold nanoparticles is a commercially important intermediate for the manufacture of analgesic and antipyretic drugs [29-32]. Examples of the use of 4-nitrophenol in classical reaction tests to evaluate catalytic properties of various metal nanoparticles, or in similar kinetic studies with gold nanoparticles dispersed in other conducting matrices, are given in the following literature [33-36]. The present authors have investigated the characterization of gold nanoparticles incorporating [C60]fullerene nanowhiskers and their catalytic activity for 4-nitrophenol reduction in the presence of sodium borohydride (NaBH4), as determined by UV-vis spectrophotometry.
2. Experiment
2.1 Reagents and instruments
Sodium borohydride (NaBH4) was purchased from Kanto Chemical Co., Inc., and trisodium citrate dihydrate (C6H5Na3O7 2H2O), cetyltrimethyl ammonium bromide ((C16H33)N(CH3)3Br), and toluene were obtained from Samchun Chemicals. Ascorbic acid (C6H8O6) and potassium tetrachloroaurate (III) n-hydrate (KAuCl4 nH2O) were supplied by Sigma-Aldrich. [C60]fullerene was supplied by Tokyo Chemical Industry Co., Ltd.
X-ray diffraction (XRD; Bruker, D8 Advance) analysis was used to examine the structure of the nanocomposites at 40 kV and 40 mA. Imaging of the sample surface was performed by scanning electron microscopy (SEM; JEOL Ltd., JSM-6510) at an accelerating voltage of 0.5 to 30 kV. The particle size and morphology of the sample were identified by transmission electron microscopy (TEM; AP Tech, Tecnai G2 F30 S-Twin) at an acceleration voltage of 200 kV. Solid-state 13C-CP/MAS nuclear magnetic resonance spectroscopy (Agilent Technologies Korea, DD2 700) was used for observing the chemical shift of carbon in the [C60]fullerene nanowhisker-gold nanoparticle composites. Raman spectroscopy (Thermo Fisher Scientific, DXR Raman Microscope) was used for observing the polymerization of the composites, and UV-vis spectrophotometry (Shimazu UV-1691 PC) was used to characterize their catalytic activity.
2.2 Synthesis of [C60]fullerene nanowhisker-gold nanoparticle composites
2.2.1 Synthesis of gold nanoparticles
A gold nanoparticle seed solution was prepared by dissolving 2.5×10−4 M potassium tetrachloroaurate (III) (KAuCl4), 2.5×10−4 M trisodium citrate dihydrate (C6H5Na3O7•2H2O), and 6 ml of 0.1M sodium borohydride (NaBH4) in 5 ml of distilled water. A gold nanoparticle growth solution was prepared with 2.5×10−4 M KAuCl4 and 2.5×10−4 M cetyltrimethyl ammonium bromide ((C16H33)N(CH3)3Br) in 20 ml of distilled water. Gold nanoparticles were prepared by mixing 5 ml of seeding solution with 15 ml of growth solution, and then adding 0.1 ml of ascorbic acid (C6H8O6) to the solution and stirring for 15 min.
2.2.2 Synthesis of [C60]fullerene nanowhisker-gold nanoparticle composites
40 mg of [C60]fullerene and 40 ml of toluene were added into a 50 ml Erlenmeyer flask, and then stirred for 15 min and ultrasonicated for 45 min. [C60]fullerene solution was dissolved in toluene and the solution was filtered through filter paper. The [C60]fullerene solution and isopropyl alcohol were placed in the refrigerator for 20 min.
2 ml of [C60]fullerene solution, 1 ml of gold nanoparticle solution, and 15 ml of isopropyl alcohol were placed in a 20 ml vial. The mixture solution was ultrasonicated for 10 min, and refrigerated for 24 h. The cold mixed solution was filtered through filter paper, and then dried to the solid state in an oven at 100°C for 5 h. The amount of gold nanoparticles loaded on the fullerene nanowhiskers was 0.25 mM.
2.2.3 Characterization of [C60]fullerene nanowhisker-gold nanoparticle composites
The XRD pattern of the [C60]fullerene nanowhisker-gold nanoparticle composites was obtained from powder X-ray diffraction with Cu Kα radiation (λ=1.54178 Å). The morphological shape of the nanocomposites was observed by SEM, and TEM was used to observe the specimen size. The composites were characterized using Raman spectroscopy, and analysed by solid-state 13C-CP/MAS nuclear magnetic resonance spectroscopy.
2.2.4 Catalytic activity evaluation through 4-nitrophenol reduction
The absorbance peak in the UV-vis spectrum of 1.5 mg (1.1 mM) 4-nitrophenol at 400 nm was monitored, as it appeared in the presence of 5 mg (13.2 mM) NaBH4 dissolved in 10 ml distilled water. 1 mg [C60]fullerene nanowhisker-gold nanoparticle composites was used as the catalyst for the 4-nitrophenol reduction. The absorbance was monitored at 2 min intervals to confirm the 4-nitrophenol reduction.
3. Results and Discussion
3.1 Characterization of [C60]fullerene nanowhisker-gold nanoparticle composites
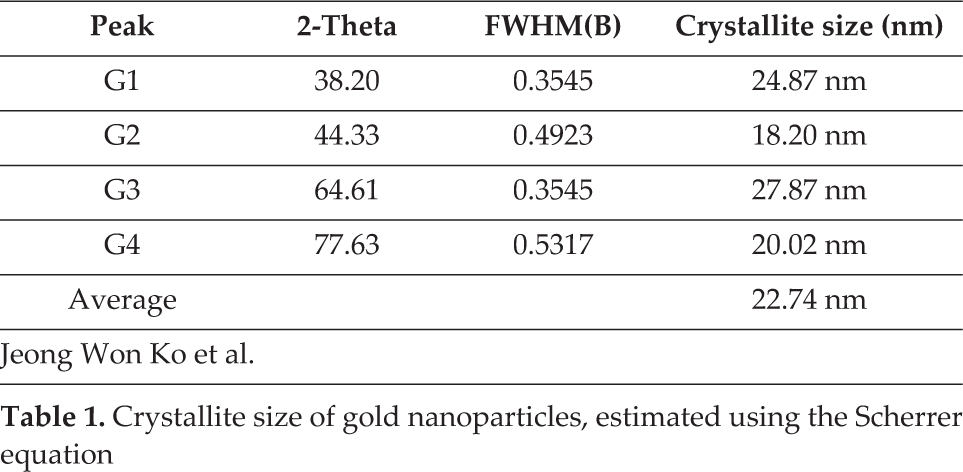
The crystal structure and crystallite size of the [C60]fullerene nanowhisker-gold nanoparticle composites were examined by XRD. Figure 1 shows the XRD patterns of the [C60]fullerene nanowhisker-gold nanoparticle composites. Peaks at 2θ = 10.82°, 17.68°, 20.72°, 27.53°, 30.13°, and 32.14° were due to the [C60]fullerene nanowhiskers, and were assigned to the (111), (220), (222), (420), (422), and (333) planes, respectively. Peaks at 2θ = 38.20°, 44.33°, 64.61°, and 77.63° were due to the gold nanoparticles, and were assigned to the (111), (200), (220), and (311) planes, respectively. The crystallite size of the gold nanoparticles was calculated using the Scherrer equation:

XRD pattern of [C60]fullerene nanowhisker-gold nanoparticle composites

where K is a shape factor taken as 0.9, λ is the wavelength of powder X-ray diffraction with Cu Kα radiation (λ=1.54178 Å), β is the full width at half maximum (FWHM), and 2θ is the angle between the incident and scattered X-rays. The Scherrer equation was used to calculate the crystallite size of the gold nanoparticles, as shown in Table 1. The mean crystallite size of the gold nanoparticles was 22.74 nm. Figure 2 shows the SEM image of the [C60]fullerene nanowhisker-gold nanoparticle composites. The gold nanoparticles agglomerated on the [C60]fullerene nanowhiskers, which are needle-like fibres. The many tiny crystals shown in Figure 2 are gold nanoparticles. The amount of gold nanoparticles loaded on the fullerene nanowhiskers was 0.25 mM. Figure 3 shows the Raman spectra of the nanocomposites. Raman shifts were observed for the peak of the [C60]fullerene nanowhisker-gold nanoparticle composites, which showed squashing mode Hg(1) at 271 cm−1, breathing mode Ag(1) at 494 cm−1, and pentagonal pinch mode Ag(2) at 1460 cm−1.
Crystallite size of gold nanoparticles, estimated using the Scherrer equation

SEM image of [C60]fullerene nanowhisker-gold nanoparticle composites

Raman spectra of [C60]fullerene nanowhisker-gold nanoparticle composites
The Raman spectroscopy of the [C60]fullerene nanowhisker-gold nanoparticle composites used a laser wavelength of 532 nm and a laser power density of 10 mW/mm2.
From the Raman shift data, which showed a blue shift of Ag(2) to 1460 cm−1, we can confirm that the [C60]fullerene nanowhiskers polymerized from [C60]fullerene to form longer needle-like crystals.
Figure 4(a) and 4(b) show TEM images of the [C60]fullerene nanowhisker-gold nanoparticle composites. Heat-treated [C60]fullerene nanowhiskers collapsed and had a porous morphology, as seen from the TEM images shown in Figure 4(b). As can be seen, the gold nanoparticles are located on the surface of the [C60]fullerene nanowhiskers in the composites. The width of the composites is about 200 nm, and the size of the gold nanoparticles is 10–15 nm.

TEM images of (a) unheated and (b) heated [C60]fullerene nanowhisker-gold nanoparticle composites
Figure 5 shows the solid-state 13C-CP/MAS NMR spectrum of the [C60]fullerene nanowhisker-gold nanoparticle composites. The chemical shift observed at 143.60 ppm can be assigned to the

Solid-state 13C-CP/MAS NMR spectrum of [C60]fullerene nanowhisker-gold nanoparticle composites
3.2 Catalytic and kinetic activity of [C60]fullerene nanowhisker-gold nanoparticle composites for 4-nitrophenol reduction
Figure 6 shows the catalytic activity of the [C60]fullerene nanowhisker-gold nanoparticle composites for the 4-nitrophenol reduction using NaBH4. The UV-vis spectrum shows that the peak at 400 nm, related to the formation of 4-nitrophenolate ions under alkaline conditions after the addition of NaBH4, was diminished. A new peak at 300 nm simultaneously appeared due to 4-aminophenol production in the presence of the [C60]fullerene nanowhisker-gold nanoparticle composites. In Figure 6(a), without the composites, the peak due to the 4-nitrophenolate ion remained unaltered for 40 min, showing the inability of NaBH4 to reduce the 4-nitrophenolate ion to 4-aminophenol, even though it is known to be a strong reducing agent. Therefore, the [C60]fullerene nanowhisker-gold nanoparticle composites operated as a catalyst for the 4-nitrophenol, as shown in Figure 6(b). Figure 7 shows the kinetic activity in the reduction of 4-nitrophenol using the composites as a catalyst. In previous studies, the Langmuir-Hinshelwood model has been applied to study the kinetics of 4-nitrophenol reduction [37-40]. The kinetic equation can be written as follows: ln(C/C0)= -κt, where C0 is the initial concentration, C is the concentration at time t, and κ is the rate constant. The reduction of 4-nitrophenol obeyed a pseudo-first-order rate law. The [C60]fullerene nanowhisker-gold nanoparticle composites, as compared to the other system, exhibited similar catalytic efficiency [33,35].

UV-vis spectra of 4-nitrophenol reduction with NaBH4 (a) in the absence of and (b) in the presence of [C60]fullerene nanowhisker-gold nanoparticle composites

Kinetics of reduction of 4-nitrophenol using [C60]fullerene nanowhisker-gold nanoparticle composites
4. Conclusions
[C60]fullerene nanowhisker-gold nanoparticle composites were synthesized from gold nanoparticle solution, [C60]fullerene-saturated toluene, and isopropyl alcohol solution, using the LLIP method. The composites were characterized by XRD, Raman spectroscopy, SEM, TEM, and solid-state 13C-CP/MAS NMR spectroscopy. The reduction of 4-nitropheqnol when applied with NaBH4 resulted in good catalytic activity of the composites using UV-vis spectroscopy. The catalytic activity of the 4-nitrophenol reduction with the [C60]fullerene nanowhisker-gold nanoparticle composites showed pseudo-first-order reaction kinetics.
Footnotes
5. Acknowledgements
This study was supported by research funding from Sahmyook University, South Korea.
