Abstract
High-performance hierarchical cheese-like C-ZSM-5 nanocrystals are successfully prepared by acid–base treatment and are evaluated for oxidative desulfurization of dibenzothiophene. The acid–base treatment can generate a large number of Si-OH bonds and an open-mesoporous structure, which is conducive for the dispersion of TiO2 and also for the transport of dibenzothiophene and its oxidation products simultaneously. The catalytic results indicate that the hierarchical cheese-like C-ZSM-5 nanocrystals are highly active catalysts for the oxidative desulfurization of dibenzothiophene due to open mesoporosity. Complete conversion was obtained within 80 min at 338 K. The excellent performance is due to the large number of active framework sites and open mesopores generated by post-processing.
The cheese-like hierarchical ZSM-5 synthesized via an acid–base treatment method exhibit superior performance in the oxidative desulfurization reaction.

Introduction
Desulfurization of fuels has attracted significant attention in recent years. Environmental regulations in many countries have strict requirements on the sulfur levels of fuels, which requires that refractory sulfur-containing compounds like dibenzothiophene (DBT) in fuels must be highly desulfurized.1–4 However, it is difficult to remove thiophene substances effectively by hydrodesulfurization, which is a commonly used process in industry because of their high stability. Currently, oxidative desulfurization (ODS),5–7 selective adsorption desulfurization, 8 biodesulfurization, 9 and extractive desulfurization 10 have been widely investigated, among which ODS is considered as one of the most promising desulfurization technologies. Recently, many ODS polyphase catalysts have been developed, such as TS-1,11–13 Ti-MIL-101(Cr), 14 Ti/ZSM-5, 15 and Ti/Beta, 16 which have been proved to have good catalytic activity thiophene compounds.
However, due to their pore size, traditional microporous zeolites, such as TS-1 have almost no catalytic reactivity in the ODS of DBT, which has a relatively large molecular size. On the contrary, Ti-modified mesoporous zeolites like Ti-MCM-41 17 have amorphous mesopore walls which greatly reduce their thermal and hydrothermal stability. With the development of hierarchical zeolite materials,18–20 Ti-modified hierarchical zeolite catalysts have been of significant interest in recent research. The hierarchical TS-1 prepared by Du,21,22 can achieve a DBT removal rate of more than 90% within 3 h, while the hierarchical TS-1 synthesized by Lv 23 can achieve almost 100% DBT removal within 1 h. However, the issue is that a considerable amount of framework Ti cannot participate in the catalytic ODS of relatively large molecules such as DBT. What is more, the preparation of hierarchical TS-1 currently requires an expensive template. Leng 24 used tetra-butyl orthotitanate (TBOT) as the precursor of a Ti species and loaded Ti on the mesopores of a hierarchical mordenite to achieve a high ODS activity toward DBT. Among numerous hierarchical zeolite materials, nanometre-hollow and thin-shell zeolites possess the characteristics of layered zeolites, nanocrystalline zeolites, and hierarchical zeolites at the same time. Hence, they have attracted extensive attention from researchers in recent years.25,26
In this paper, hierarchical cheese-like C-ZSM-5 nanocrystalline materials as the catalyst carrier are obtained post treatment. TiO2 was located on the mesopores of C-ZSM-5 using TBOT as the Ti source. In a catalytic evaluation, the hierarchical nanocrystalline catalyst shows high catalytic activity for the ODS of DBT.
Results and discussion
The synthesis of the zeolites was achieved by post-treatment of ZSM-5 nanocrystalline using hydrogen chloride solution and sodium hydroxide solution, successively. The simulation process is shown in Scheme 1. As the distribution of Si and Al atoms in ZSM-5-nano was not uniform, the Al atoms were mainly distributed in the periphery of ZSM-5-nano crystal. As shown in Scheme 1, the Al atoms in the periphery were partially removed by the acid treatment. During the first 20 min of base treatment, the Si atoms in the center were removed first due to the protective effects of the residual peripheral Al atoms, thus forming a closed hollow mesoporous structure. On continuation of the base treatment process to 30 min, when the peripheral Si atoms were also partially corroded, the shell of the sphere becomes damaged, and the cavities open, thus forming cheese-like open mesopores.

Imaginative diagram about the formation process of C-ZSM-5.
N2 adsorption–desorption isotherm
Figure 1 shows the N2 adsorption isotherms of various samples (A: Ti/ZSM-5-0, B: Ti/C-ZSM-5-30, C: Ti/ZSM-5-20). All the samples gave a type-I isotherm at low relative pressure, indicating the existence of micropores. Samples Ti/C-ZSM-5-30 and Ti/ZSM-5-20 gave a type-IV isotherm with a hysteresis loop at high relative pressure, indicating the existence of mesopores. As shown in the mesoporous distribution diagram, the average mesoporous size of C-ZSM-5-30 is larger than that of ZSM-5-20, which is caused by thinning of the shell wall and enlargement of the cavity after desilication.

N2 sorption isotherms and pore size distribution curves of various catalysts with 5wt% Ti content (a: Ti/ZSM-5-0; b: Ti/C-ZSM-5-30; and c: Ti/ZSM-5-20).
The detailed data are listed in Table 1. After the post-treatment process, the Si/Al ratio went down from 94 to 32, the Brunauer–Emmett–Teller (BET) surface area increased (from 340 to 433 m2/g) and the total pore volume was also increased significantly (from 0.37 to 0.99 cm3/g), indicating that removal of silicon and aluminum atoms resulted in the formation of a mesoporous structure. In addition, after introducing Ti, the BET surface area of all three samples slightly decreased, but the total pore volume and the Si/Al ratio did not change.
Textural properties of different samples.

BET: Brunauer–Emmett–Teller.
X-ray diffraction analysis
Figure 2 shows the powder X-ray diffraction (XRD) patterns of three samples (1: ZSM-5-0; 2: ZSM-5-20; 3: C-ZSM-5-30) (A: samples with Ti-modification; B: samples without Ti-modification). As seen in Figure 2(a), the samples after post-treatment still have the framework structure of ZSM-5. But notably, the peak strength decreased following the post-treatment process. As the post-treatment is continued, the thickness of the zeolite shell becomes progressively thicker, and the long-range order of the zeolite crystal decreases slightly. As seen in Figure 2(b), the loading of Ti species did not cause damage to the zeolite framework. The diffraction peak did not deviate horizontally, indicating that the Ti species did not enter the micropores, but were loaded on the outer surface and mesoporous surface of the zeolite. At the same time, it was difficult to find the peak attributed to TiO2 in Figure 2(b), which means that the degree of dispersion of the Ti species should be appreciably high.

XRD patterns of the different catalysts (a: catalysts with 5wt% Ti content; b: catalysts without Ti-modification) (1: ZSM-5-0; 2: ZSM-5-20; 3: C-ZSM-5-30).
SEM and TEM analysis
Scanning electron microscope (SEM) and transmission electron microscope (TEM) images of Ti/ZSM-5-0, Ti/ZSM-5-20, and Ti/C-ZSM-5-30 with a Ti content of 5wt% are shown in Figure 3. It can be seen that the three samples all have a morphology of spherical shape, and most particles are sized around 100 nm (Figure 3(a), 3(c), and 3(e)). The TEM images show that Ti/ZSM-5-0 presents a morphology of nano-solid spheres (Figure 3(b)). Ti/ZSM-5-20, on the contrary, is a medium hollow-shell structure (Figure 3D), with a shell thickness of about 30–50 nm and a mesopore of about 30–60 nm. Ti/C-ZSM-5-30 becomes a cheese-like hollow structure with many holes 10 nm in diameter on the shell wall (Figure 3(f)). Moreover, large TiO2 crystals were not observed in the three samples. This is because, all three samples were nanometre particles with very large surface areas, which is very conducive to the dispersion of Ti species. This result is in good agreement with the XRD results in Figure 2.

SEM and TEM images of the different catalysts with 5wt% Ti content (a, b: Ti/ZSM-5-0; c, d: Ti/ZSM-5-20; e, f: Ti/C-ZSM-5-30).
Figure 4 shows TEM images of the samples without acid treatment before base treatment. Different from the acid–base–treated samples, the morphologies of these samples after different degrees of base-treatment were hollow shells (Figure 4(a)) to open cavities (Figure 4(b) and (c)), and finally fragmented (Figure 4(d)). In this process, no cheese-like morphology, which is similar to the C-ZSM-5 (Figure 3(f)), was observed. This experimental result is consistent with our explanation in Scheme 1.
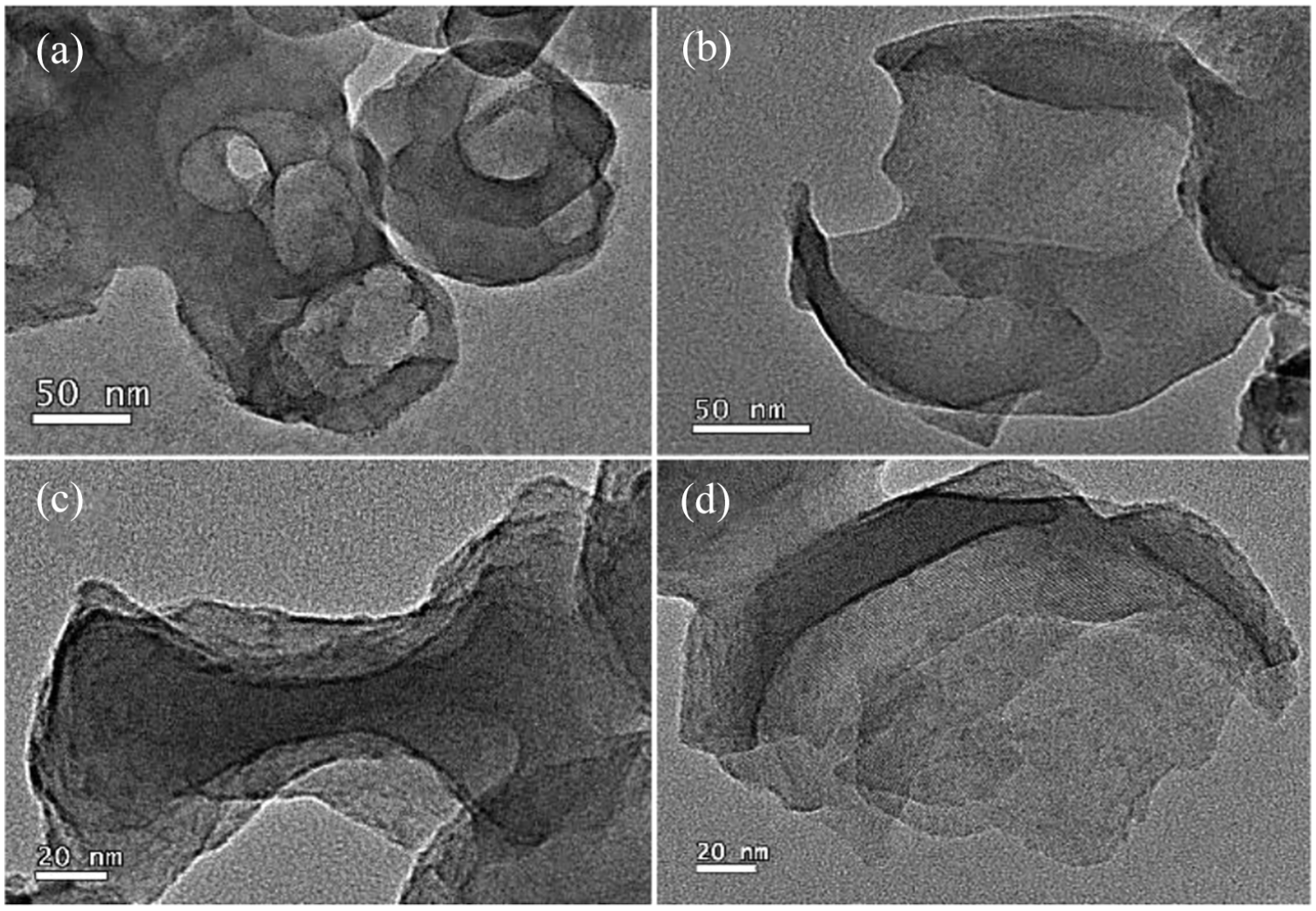
TEM images of ZSM-5 (the original Si/Al ratio was 95) after different base treatment times (a: 20 min; b: 30 min; c: 40 min; d: 50 min) and without acid treatment.
Ultraviolet–visible spectrophotometry analysis
The ultraviolet response is very sensitive to the chemical environment of the Ti element, and is often used to identify the form of the titanium species in titanium-containing catalysts. Generally, the absorption between 210 and 230 nm belongs to the skeleton or independent titanium species (four or five coordination), and the absorption between 260 and 300 nm belongs to non-skeleton titanium (nanoclusters) of six coordination. The absorption at about 330 nm belongs to anatase-phase titanium dioxide.23,27 To detect the coordination of Ti species, the ultraviolet–visible spectrophotometery (UV-vis) spectra of Ti/C-ZSM-5-30 with different Ti contents are given in Figure 5. Obviously, all the curves have an absorption band between 210 and 230 nm which corresponds to isolated tetra-coordinated Ti species. Simultaneously, all samples have an absorption band between 260 and 300 nm, which corresponds to penta- and hexa-coordinated non-framework Ti species. In addition, the sample with a Ti content of 7wt% displayed a weak absorption around 330 nm, indicating the existence of bulky TiO2 crystals. Based on an earlier report, 28 isolated and oligomeric octahedral Ti species are considered to be highly active species in ODS reactions because such species contribute to the formation of numerous of superoxide radicals stabilized at Ti sites. TiO2 of anatase phase does not contribute to ODS, moreover, it is also a waste of Ti resources.

UV-vis spectra of Ti/C-ZSM-5-30 with different Ti content.
Catalytic performance
The ODS reaction of DBT was conducted to evaluate the catalytic performance of the Ti-modified catalysts. After an ODS reaction of 15 min, the DBT conversion rate was analyzed by gas chromatography (GC) (Figure 6). The catalysts were prepared with Ti contents of 1wt%, 3wt%, 5wt%, and 7wt%, respectively. It can be seen that to a certain extent (<5wt%), an increase in the Ti-content had a positive effect on the catalytic ability of the catalysts. However, due to the limited mesoporous capacity of the zeolites, when the Ti content was too high (>7wt%), guest molecules would agglomerate. This leads to the formation of bulky and anatase phase TiO2, which has low ODS activity. Eventually, the catalyst activity was reduced.
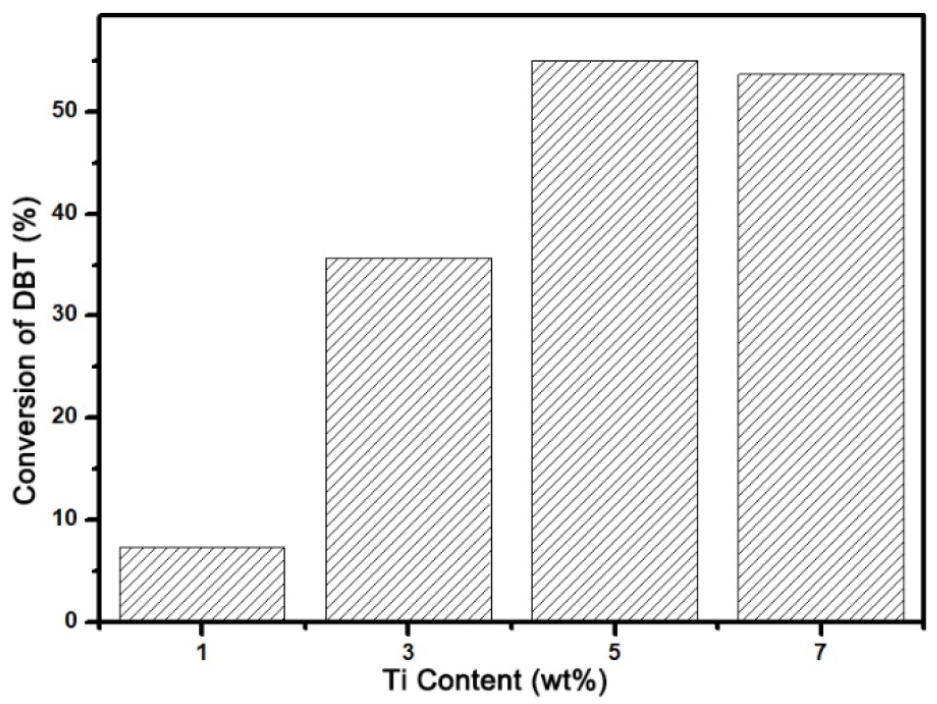
ODS performance of Ti/C-ZSM-5-30 with various Ti content. Reaction conditions: catalyst (50 mg), model oil (10 mL), DBT (1000 ppm), O/S molar ratio (6:1), temperature (338 K).
To determine the optimal dosage of oxidizer, we carried out the experiments with O/S ratios of 2, 4, 6, and 8 (Figure 7). Ti/ZSM-5-30 with a Ti content of 5wt% was used as the catalyst. As can be seen from the figure, when the O/S ratio was 2, the reaction was relatively slow, and only 80% of DBT was oxidized within 120 min. When the O/S ratio was 4, the total conversion time of DBT was 100 min, and when the O/S ratios were 6 and 8, the time was 80 min in both cases. Theoretically, two units of tert-butyl hydroperoxide (TBHP) would be enough to oxidize all of DBT to DBTO. However, in the catalytic reactions, the concentration of O and S is very low (<0.03 mol/L). In this case, the rate of chemical reaction is predictably slow. To avoid the influence of oxidizer concentration, subsequent experiments were carried out with an O/S ratio of 6.
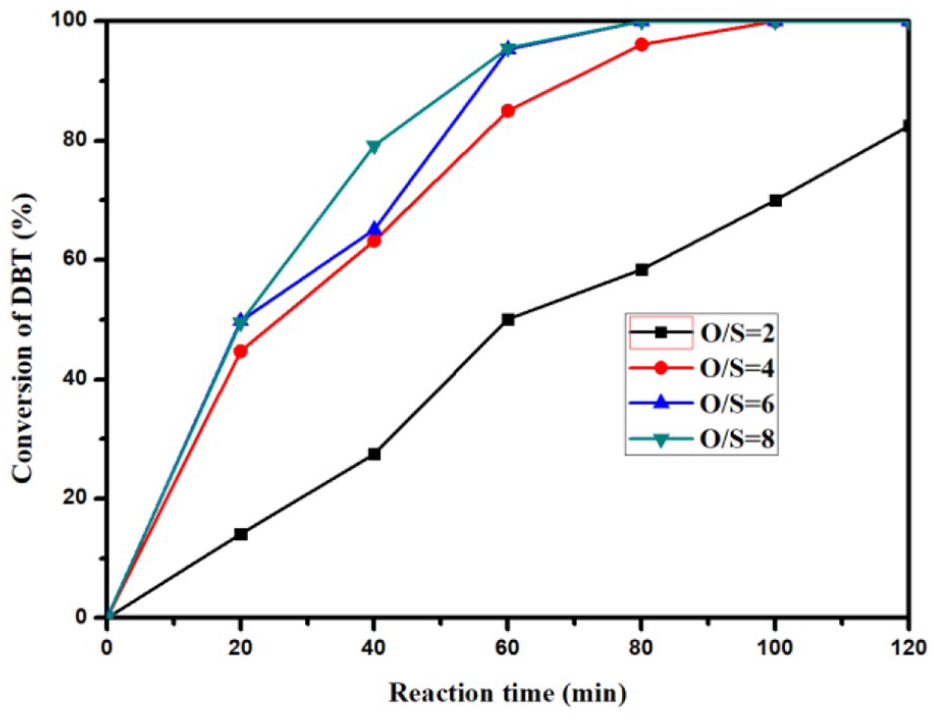
ODS performance of Ti/C-ZSM-5-30 with different O/S ratios. Reaction conditions: catalyst (50 mg), model oil (10 mL), DBT (1000 ppm), Ti content of catalysts (5wt%) and temperature (338 K).
Catalysts with 5wt% Ti content were prepared to research the influence of the pore structure on the ODS reaction of DBT (Figure 8). Although the DBT conversion rate of Ti/ZSM-5-0 was only 11.3% in the first 20 min, it was lower than that of Ti/ZSM-5-20 (41%) and Ti/C-ZSM-5-30 (49.8%). The DBT conversion rate of Ti/ZSM-5-0 reached 90.9%, basically consistent with that of Ti/ZSM-5-20 (95%) after 120 min. However, the DBT conversion rate of Ti/C-ZSM-5-30 reached 100% in 80 min. To explain these catalytic results, a simulative diagram on the ODS process is shown in Scheme 2. Because the molecular size of TBOT is larger than that of the ZSM-5 pore size, the Ti species were mainly located on the external surface and on the mesopores of the zeolites. Therefore, Ti/C-ZSM-5-30 with open mesopores and a larger BET surface area was more conducive to the dispersion of the Ti species. In addition, the abundant open mesopores can facilitate the transfer of reactants and products in ODS reactions. However, in the case of far excessive oxidants, the DBT conversion rate of Ti/ZSM-5-0 and Ti/ZSM-5-20 still did not reach 100%. The main reason may be that the polar products (mainly DBT sulfones) were adsorbed on the surface of the catalysts, such that the active site on the catalysts might be partially covered, and the catalytic activity would gradually decrease. Ti/C-ZSM-5-30 has more abundant and useable mesopores and more active sites. Therefore, Ti/C-ZSM-5-30 had better catalytic performance than Ti/ZSM-5-0 and Ti/ZSM-5-20.
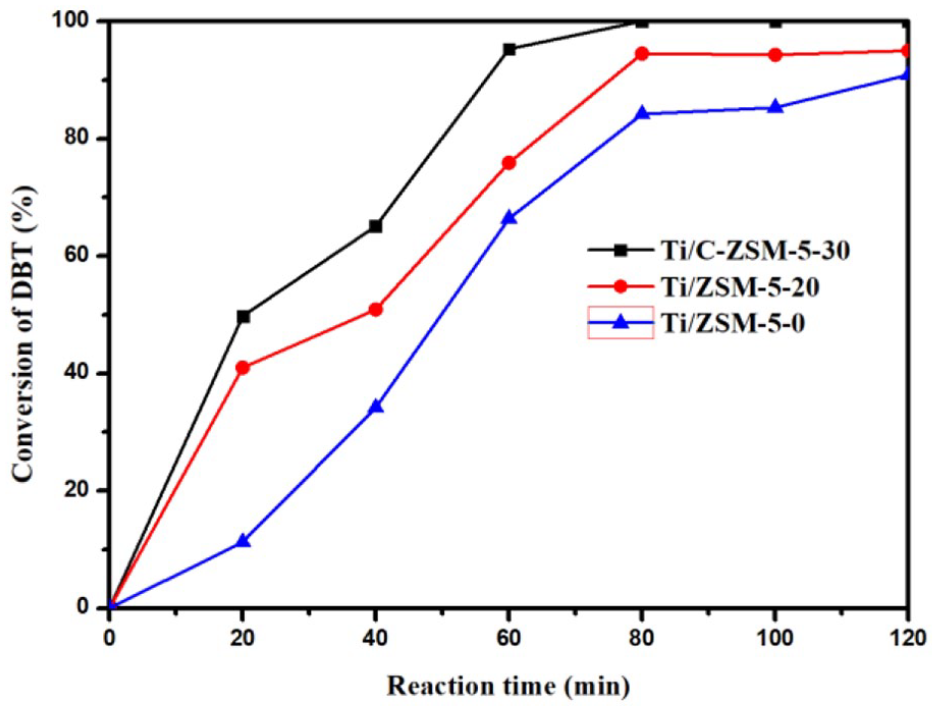
Conversion of DBT over different catalysts with different pore structures; reaction conditions: catalyst (50 mg), model oil (10 mL), DBT (1000 ppm), O/S molar ratio (6:1), Ti content of catalysts (5wt%), and temperature (338 K).
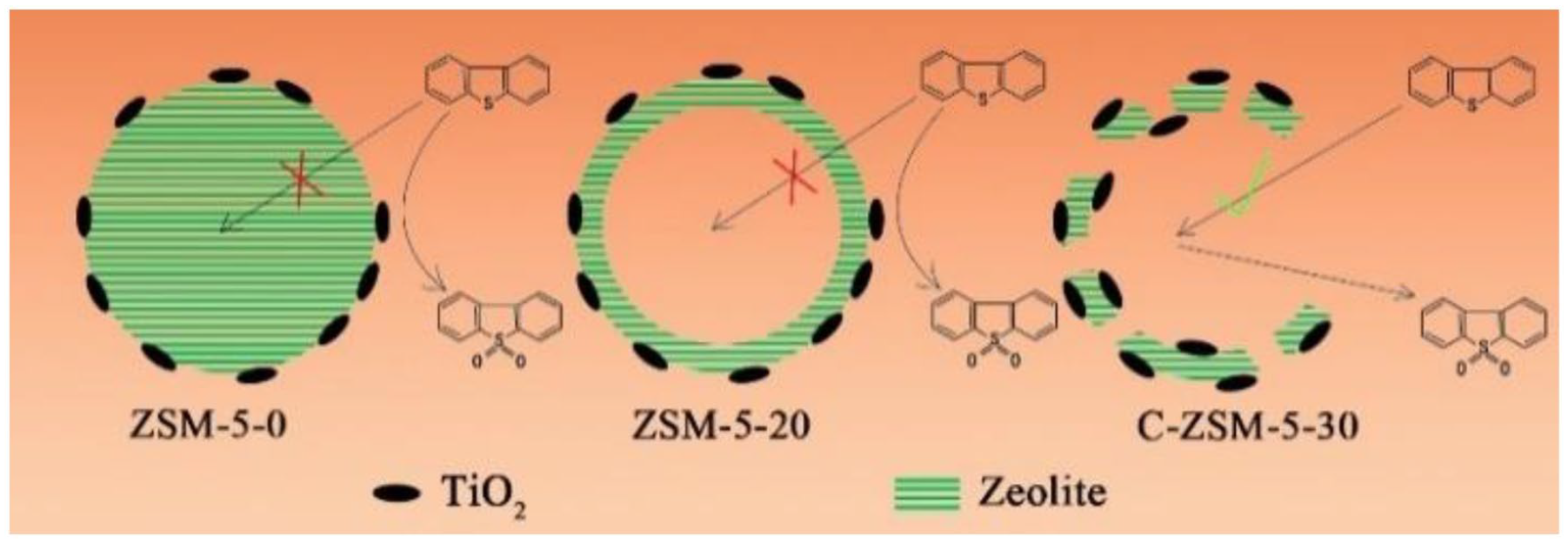
Schematic diagram of the ODS reaction of DBT with different catalysts.
To make a comprehensive comparison, the catalytic activities of several representative Ti-containing porous materials in the ODS reaction of DBT were summarized in Table 2. The turnover frequency (TOF) number of DBT removal over Ti/C-ZSM-30 is 5.7 h−1. Comparatively, the catalyst Ti-HMS, 29 Ti-MCM-41, 30 Ti-MWW, and Ti-MOR 24 gave a TOF number of 0.6 h−1(333 K), 0.7 h−1(343 K), 2.3 h−1(345 K), and 4.3 h−1(333 K), respectively. These results demonstrated that the Ti/C-ZSM-5-30 can be one of highly active ODS catalysts.
Comparison of catalytic activities over some representative Ti-containing porous materials in the ODS reaction of DBT.

ODS: oxidative desulfurization; DBT: dibenzothiophene; TOF: turnover frequency.
TOF = (mole number of converted DBT)/(mole of metal content in catalyst × reaction time (h)).
Reusability
The reusability of the Ti/C-ZSM-5-30 catalyst was tested and the results are shown in Figure 9. After each reaction for 2 hours, the catalyst was separated and calcined at 823 K for 10 h. As can be seen from the figure, after the first regeneration, the catalytic ability of the sample decreased slightly. But after the second and the third regenerations, the catalytic effect of the sample remained almost consistent. A possible reason might be that during the first reaction and regeneration process, some Ti species which were not firmly bound to the framework of the zeolite were removed or destroyed. Thus, the remaining Ti species maintained high activity in the second and third cycles. On the fourth recycle, the catalytic activity remained above 97%, indicating that the catalyst has good regeneration performance. Meanwhile, XRD, SEM, and N2 sorption tests were carried out for all the recycled catalysts. The results showed that there was no difference in morphology and structure between the recycled catalysts and the new catalysts.

Reusability of Ti/C-ZSM-5-30 (5wt% Ti content) in the ODS reaction of DBT. Reaction conditions: catalyst (50 mg), model oil (10 mL), DBT (1000 ppm), O/S molar ratio (6:1), Ti content of catalysts (5wt%), and temperature (338 K).
Conclusion
Hierarchical cheese-like Ti/C-ZSM-5-30 catalysts exhibit excellent ability in the ODS of DBT. In the first 20 min, the catalytic capacity was 4.4 times than that of traditional Ti/ZSM-5-0. DBT was completely removed within the following 60 min. This catalyst has numerous cheese-like mesopores and a large BET surface area, which can significantly improve the TiO2 dispersion, enhance the transfer efficiency of the reactants, and increase the chance of contact between the active sites and reactants. This unique hierarchical nanocrystalline zeolite not only performed well in ODS reactions but also shows potential in other heterogeneous catalytic reactions by loading other active species. Our results on such investigations will be reported in due course.
Experimental
Reagents
Tetrapropylammonium hydroxide (TPAOH, 20% in H2O) was purchased from Aladdin Reagent Co., Shanghai, China. NaOH (A.R.), HNO3 (A.R. 65-68wt%), aluminum isopropoxide (AIP, A.R.), tetraethyl orthosilicate (TEOS, A.R.), TBHP (A.R. 70wt%, in H2O) and TBOT (A.R.) were all purchased from Sinopharm Chemical Reagent Co. (Shanghai, China).
Characterization
Powder XRD patterns were recorded on a Rigaku D/Max-2550 diffractometer at 50 kV and 200 mA with CuKα radiation (λ = 0.15418 nm). Morphological details were examined using a SU8020 SEM and a JEOL JSM-3010 TEM. The specific surface areas were calculated by employing the BET equation. UV-vis spectra were recorded with a Shimazu UV-2550 UV-visible spectrometer. For the catalytic reaction, an Agilent GC6890N gas chromatograph with an FID detector was used to analyze the products.
Synthesis of materials
Nanoscale ZSM-5 was hydrothermally synthesized according to a reported method. 18 The reaction solution consisted of tetrapropylammonium hydroxide (TPAOH), deionized water (H2O), sodium hydroxide (NaOH), aluminum isopropanol (AIP), tetraethyl orthosilicate (TEOS) in a molar ratio of 30:2600:1:1:132, respectively. The reaction mixture was then transferred to a Teflon-lined stainless-steel autoclave that was placed in a pre-heated oven at 373 K for 12 h and then further heated at 453 K for 24 h under static conditions. The as-synthesized zeolites were washed with deionized water, and then dried at 358 K in an oven overnight. The resulting powders were calcined at 823 K for 10 h with a heating rate of 1°C min–1.
ZSM-5 (1 g) was refluxed with a solution of HNO3 (1 M, 40 mL) at 353 K for 0.5 h. The solid sample was then filtered, washed, dried, and calcined at 823 K for 5 h. This sample was named ZSM-5-0.
ZSM-5-0 (1 g) was mixed with a solution of NaOH (0.5 M, 40 mL), the mixture was added to a round-bottomed flask and then transferred into a thermostated water bath at 338 K stirring constantly for 20 or 30 min. After rapid cooling, centrifuging, washing, and drying, hollow thin-shell ZSM-5 was obtained. According to the alkali corrosion time, the samples were named ZSM-5-20 (20 min) and C-ZSM-5-30 (30 min), respectively.
TBOT was dissolved in methanol (0.61 g) and mixed with the zeolites (1 g). The mixture was then placed in an ultrasonic bath for 30 min and then dried in an oven at 353 K overnight. Finally, it was calcined in a muff furnace at 823 K for 6 h to afford catalysts with different Ti contents and named Ti/ZSM-5-0, Ti/ZSM-5-20, and Ti/C-ZSM-5-30, respectively.
Catalytic activity test
An appropriate amount of DBT was dissolved in n-octane to act as model fuel, and the concentration of sulfur in the model fuel was 1000 ppm by weight (ppmw). Next, 50 mg of the catalyst and 10 g of the model fuel were added to a three-neck glass flask. When the reaction temperature reached 338 K, an appropriate amount (O/S = 2, 4, 6, and 8) of TBHP was added to the reactor, and this represented the initial reaction time. Small quantities of samples were taken at specific intervals for GC analysis.
Footnotes
Author contributions
Conceptualization, S.C.Y. and W.Y.; methodology, S.C.Y. and W.Y.; investigation, S.C.Y. and S.H.Z.; writing-original draft preparation, S.C.Y. and W.Y.; writing-review and editing, D.F. and W.C.Y.; supervision, W.Y. and W.C.Y.; funding acquisition, Z.J.Q. and W.C.Y. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Nature Science Foundation of China (grant nos. 61805178 and 61701349), the Shandong Province Natural Science Foundation (grant nos. ZR2017MF040, ZR2018PF016, ZR2017MF042, ZR2017QF012, ZR2020KF017, and ZR2019MD011), and the Weifang Huimin Foundation (grant no. 2019KJHM11).
