Abstract
Background:
Despite widespread use of tumor necrosis factor inhibitors (TNFi) as first-line therapy in rheumatoid arthritis (RA), up to 40% of patients fail initial treatment. Subsequent therapeutic choices remain poorly structured, with limited evidence-based guidance to inform individualized post-TNFi decision-making.
Objective:
To develop evidence-informed, profile-based recommendations to guide treatment selection after inadequate response to a first TNFi in RA, combining evidence and expert consensus.
Design:
Delphi-based consensus study informed by a PRISMA-guided scoping review (ScR) and nominal group methodology.
Methods:
A PRISMA-guided ScR of biologic and targeted synthetic biologic disease-modifying antirheumatic drug (tsDMARDs) after TNFi failure was conducted. Patient profiles were identified by a steering committee, and draft recommendations were evaluated through an anonymized Delphi process. A profile-based decision tree integrated direct and indirect evidence, with evidence strength graded using the Oxford Centre for Evidence-Based Medicine approach.
Results:
The ScR included 43 studies, mostly exploratory analyses of randomized trials. Scenarios included age ⩾65 years; failure of ⩾2 TNFi; monotherapy; rheumatoid factor/anti-citrullinated peptide antibody status; prominent systemic inflammation; interstitial lung disease (ILD); rheumatoid vasculitis; high cardiovascular (CV) risk or prior CV event; venous thromboembolism (VTE) risk; obesity; high infection risk; osteoporosis; nociplastic pain, depression and fatigue; prior solid cancer; hematologic cancer/lymphoproliferative disease; non-melanoma skin cancer; and pregnancy.
Seventeen recommendations were formulated; 15 achieved consensus. Agreed positions included caution with JAK inhibitors (JAKi) in older patients and in those with CV/VTE risk; preference for IL-6 receptor inhibitors or JAKi for monotherapy or prominent systemic inflammation; in RA-ILD, use a b/tsDMARD with a non-TNFi mechanism; rituximab as first choice in rheumatoid vasculitis; abatacept in infection-prone patients; discouraging JAKi in prior malignancy; and TNFi as acceptable during pregnancy. Two statements did not reach consensus: preferential use of non-TNFi in obesity and heightened caution with tofacitinib in osteoporosis or fracture risk.
Conclusion:
This Delphi-validated, profile-based framework provides a practical tool to support evidence-informed clinical decision-making.
Plain language summary
When the first tumour necrosis factor inhibitor (TNFi) does not control rheumatoid arthritis (RA) well enough or causes side effects, the next step is not straightforward. We combined a structured review of published studies with a two-round Delphi exercise in which 30 experienced rheumatologists from Spain and Portugal rated and refined practical, profile-based recommendations. We focused on real-world patient profiles that commonly shape treatment choice: older age; failure of two or more TNFi; need for monotherapy; strong systemic inflammation; interstitial lung disease (ILD); rheumatoid vasculitis; high cardiovascular or venous-thromboembolism risk; obesity; high infection risk; osteoporosis; non-inflammatory pain, depression or fatigue; previous solid or haematological cancer; non-melanoma skin cancer; and pregnancy. The result is a practical, profile-based framework to support shared decision-making after first TNFi failure, aligning drug choice with patient characteristics, safety considerations, and treatment goals to improve outcomes and quality of life.
Introduction
Tumor necrosis factor inhibitor (TNFi) agents are established as first-line biologic therapy for rheumatoid arthritis (RA), supported by more than two decades of clinical use.1,2 Their efficacy and favorable safety are well established, and they have become the most affordable biological agents in this condition. 3
However, up to 40% of patients respond inadequately to an initial TNFi owing to primary inefficacy, secondary loss of response, or treatment-limiting adverse events.4,5 According to the European Alliance of Associations for Rheumatology (EULAR) recommendations for RA management, 6 after an inadequate response to a TNFi, clinicians may either switch to another TNFi (cycling) or change (switching or swapping) to a biologic disease-modifying antirheumatic drug (bDMARD) with a different mechanism of action (MoA) or to a targeted synthetic DMARD (tsDMARD).
Current EULAR guidance does not formalize post-TNFi pathways or recommend a preferred strategy; rather, it emphasizes careful appraisal of contraindications and the risks of subsequent therapies, allowing flexibility in clinical decision-making. In the absence of clear, evidence-based algorithms and reliable biomarkers or validated tools to guide routine selection, clinicians face substantial uncertainty when choosing between cycling and switching, particularly in light of the reason for failure, comorbidities, extra-articular manifestations, prior therapies, and patient preferences. Several publications have sought to address these unresolved questions but have relied primarily on expert opinion rather than on published evidence.7,8
Given these gaps, our aim was to identify the most appropriate post-TNFi strategy across common RA scenarios by conducting a PRISMA-guided scoping review (ScR). Based on this evidence, we developed profile-based recommendations for routine clinical scenarios, which were subsequently Delphi-validated by a larger binational expert panel. This work seeks to reduce ambiguity, answer common clinical questions, and support evidence-informed treatment decisions in routine clinical practice.
Methods
Study design and oversight
A Delphi-based consensus study was conducted, preceded by a ScR and framed by two nominal group meetings, under the supervision of a methodologist (E.L.). The Delphi exercise followed core methodological safeguards for reliability: a prespecified protocol; an expert panel with defined eligibility criteria; anonymous voting; controlled feedback between rounds; and an a priori consensus threshold. The project complied with the principles of the latest version of the Declaration of Helsinki and applicable Good Clinical Practice regulations.
Expert selection and nominal group meetings
A steering committee comprising five RA experts was established. Expert selection criteria included rheumatologists with a special interest in RA, ⩾8 years of clinical experience and/or ⩾5 RA publications, and membership of national or international societies and/or RA-related working groups; geographical representativeness within the Iberian Peninsula was ensured by involving Spanish and Portuguese rheumatologists.
Two nominal group meetings were held. The first, conducted before the ScR, defined the objectives, scope, intended users, key definitions, and document contents; drafted RA patient profiles and treatment strategies to guide selection of bDMARDs/tsDMARDs after TNFi failure (see Supplemental Table 1); and agreed on the inclusion and exclusion criteria for the subsequent ScR. The second, conducted after circulation of the ScR results, refined statements derived from the evidence and expert experience, and formulated recommendations for specific RA subgroups after first-line TNFi therapy, which were prepared for Delphi circulation.
The number of experts involved in the Delphi process was defined a priori to balance diversity of expert opinion, methodological robustness, and feasibility, consistent with methodological standards for Delphi studies in healthcare, where panels of approximately 20–40 experts are considered sufficient to achieve stable consensus while maintaining operational feasibility. A formal power calculation was not applicable to this qualitative, consensus-based design and is acknowledged as a limitation.
Scoping review (evidence base for the Delphi)
The ScR was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) statement. 9 Studies were identified in MEDLINE using disease- and treatment-related MeSH/controlled vocabulary terms and additional keywords; abstracts from the American College of Rheumatology (ACR)/EULAR meetings up to 2023 and relevant guidelines and consensus documents were also screened. The main findings of this literature review will be reported in a separate publication arising from an independent project
Inclusion criteria were adults (⩾18 years) with RA and inadequate response to first-line TNFi; treatment with bDMARDs or tsDMARDs; and outcomes reported within predefined profiles or strategies. There were no restrictions on drug type, dose, route, concomitant medication, or treatment duration. Outcomes included disease activity and function, pain, radiographic progression, serious adverse events, and quality of life.
When published evidence specifically addressing patients with inadequate response to a first TNFi was unavailable for a given clinical scenario, this limitation was explicitly acknowledged. In such cases, indirect evidence derived from broader RA populations was considered and subsequently integrated with mechanistic plausibility and expert judgment within the Delphi process.
Two reviewers independently screened, extracted, and appraised the evidence; disagreements were resolved by a third reviewer. Quality assessment tools included AMSTAR-2 (A Measurement Tool to Assess Systematic Reviews, version 2), 10 the Jadad scale (randomized controlled trials), 11 and the Newcastle–Ottawa Scale (observational studies). 12 Given heterogeneity across populations, outcome measures, and time points, the GRADE framework (Grading of Recommendations Assessment, Development and Evaluation) was adapted to the objectives of this document (see Supplemental Table 2). 13 Evidence tables were produced.
Delphi process
The Delphi was the primary method for recommendation generation and validation. Recommendations derived from the ScR and nominal groups were submitted to an online, anonymized Delphi voting process among 30 rheumatologists from Spain and Portugal with recognized expertise in RA and prior access to the ScR results. Participants rated each statement on a 1–10 Likert scale (1 = totally disagree; 10 = totally agree). The a priori consensus threshold was ⩾70% of ratings ⩾7. Items not meeting consensus were revised based on panel feedback and re-voted in a second round. Controlled feedback between rounds included aggregated statistics (mean, median, and interquartile range) to inform re-voting without revealing individual identities. This design ensured key reliability features of Delphi exercises: anonymity (to minimize dominance bias), iteration with controlled feedback (to foster convergence), and an explicit, prespecified consensus rule.
Decision tree development and grading
Following the Delphi, a profile-based decision tree for treatment selection was created, integrating ScR findings, indirect evidence (e.g., pharmacological mechanisms of action and pathophysiology), and expert judgment. Levels of evidence and grades of recommendation were subsequently assigned by the methodologist according to the Oxford Centre for Evidence-Based Medicine (CEBM) approach. 14 The final document was circulated among experts for review and sign-off.
Results
Results of the literature review and evidence for decision-making across clinical scenarios
The ScR included 43 articles; most were exploratory analyses of randomized controlled trials. Patient subgroups, bDMARDs/tsDMARDs, outcomes, and results varied widely. As the main findings of the ScR will be published separately, only key signals are summarized here.
Across the predefined clinical scenarios, the availability of evidence specifically derived from patients with inadequate response to a first TNF inhibitor was heterogeneous. Direct evidence in this population was prioritized when available; when absent, this limitation is explicitly stated in the sections that follow, and the synthesis relies on indirect evidence from broader RA populations, which subsequently informed the expert consensus process. Evidence relevant to common clinical profiles included:
- RA patients aged ⩾65 years: The ScR found no evidence favoring one bDMARD or tsDMARD over another in elderly patients.15,16 Observational data, regardless of prior TNF inhibitor (TNFi) exposure, suggest that bDMARDs and tsDMARDs remain effective in this population, albeit with higher rates of adverse events.17–20 In this age group, some studies have found that abatacept (ABT) and anti-interleukin-6 receptor (anti-IL-6R) antibodies show higher treatment survival and favorable safety profiles compared with TNFi and JAK inhibitors (JAKi), particularly in those aged ⩾75 years.21–23 In subgroup analyses of the ORAL Surveillance trial (patients with ⩾1 additional cardiovascular (CV) risk factor), major adverse CV events were more frequent with tofacitinib (TOFA) than with TNFi in participants aged ⩾65 years. 24
- Inadequate response to ⩾2 TNFi: The optimal strategy after inadequate response to two or more TNFi remains uncertain. Following failure of a first TNFi, switching to a second TNFi may be effective,25,26 but no head-to-head trials have compared cycling to a third TNFi with switching to a drug with a different MoA. Observational studies suggest limited effectiveness when switching to a third TNFi.27,28 Similarly, a subanalysis of the GO-AFTER trial found that, among patients receiving golimumab (GOL) plus methotrexate (MTX), 24-week outcomes were numerically lower with exposure to ⩾2 prior TNFi, with DAS28-CRP moderate/good responses of 58.4% versus 51.1% versus 58.8% and HAQ-DI improvement ⩾0.25 of 53.3% versus 46.8% versus 41.2% for one versus two versus three prior TNFi, respectively.29,30
Indirect meta-analyses and registry data indicate comparable efficacy among non-TNF agents in patients with inadequate response to TNFi, with no consistent superiority of one agent over another.26,31 Although all therapeutic options are reasonable, both the RADIATE study with tocilizumab (TCZ) and the TARGET trial with sarilumab (SAR) suggest that the clinical response to anti-IL-6R antibodies is independent of the number of TNFi previously used.32,33 Similar findings have been reported with some JAKi.34–36 By contrast, progressively lower responses were observed with increasing numbers of prior TNFi failures with ABT37,38 and rituximab (RTX).39,40
Upadacitinib showed superiority to ABT in a head-to-head trial in patients with prior bDMARD failure. 15
- Monotherapy: The ScR did not identify studies specifically evaluating b/tsDMARD monotherapy in TNFi-inadequate responders. In other RA populations, anti-IL-6R antibodies and JAKi showed good efficacy as monotherapy in randomized trials, whereas some bDMARD showed attenuated monotherapy responses compared with combination therapy.41–44 A systematic literature review (SLR) with meta-analysis in csDMARD-intolerant RA found etanercept (ETN) superior to adalimumab (ADA), certolizumab pegol (CZP), and ABT as monotherapy, 44 suggesting ETN as a reasonable alternative when anti-IL-6R antibodies or JAKi are not suitable.
- Rheumatoid factor (RF) and anti-citrullinated peptide antibody (ACPA) status: In the REFLEX trial, ACR20 responses with RTX versus placebo at week 24 were 54% versus 41% in RF-positive patients and 19% versus 12% in RF-negative patients. 45 In R4RA, RTX and TCZ showed no differences in CDAI-50 responses at week 16, regardless of serostatus (RF-positive, 78% vs 70%; ACPA-positive, 82% vs 77%). 47 Overall, b/tsDMARD efficacy is broadly comparable across serostatus groups. Higher baseline ACPA levels have been associated with better clinical responses to ABT, RTX, and CZP, although the effect is modest and heterogeneous. 46
- Marked systemic inflammatory profile: This profile was defined by the presence of clinically relevant inflammatory anemia, fever or low-grade fever, secondary amyloidosis, or markedly elevated C-reactive protein (CRP). Exploratory analyses in TNFi-inadequate responders and other RA populations suggest that anti-IL-6R antibodies improve these systemic features.47–50 JAKi also reduce CRP, but not beyond the effect seen with anti-IL-6R.15,51,52 CRP reduction alone is not a therapeutic goal, and anti-IL-6R–mediated CRP suppression may overestimate disease control. 53
- Interstitial lung disease (ILD): The ScR did not identify evidence supporting any specific b/tsDMARD for TNFi-inadequate responders. Four comparative studies of biologics in RA-ILD have been published. Regarding efficacy, a systematic review and meta-analysis found no overall change in forced vital capacity (FVC) or diffusing capacity of the lung for carbon monoxide after treatment with ABT, RTX, TCZ, JAKi, or TNFi, indicating overall stabilization of lung function. 54 In subgroup analyses, only RTX improved FVC (mean difference −4.62; p = 0.03). Imaging showed stability or improvement in most patients, with 79.2% non-progression on high-resolution chest CT (HRCT).
Regarding safety, a broad systematic review and meta-analysis found no between-drug differences in incident RA-ILD risk in randomized controlled trials (RCTs) (MTX, TNFi, TCZ, TOFA, baricitinib (BARI)), while observational data suggested lower risk with MTX (odds ratio (OR): 0.49) and with TOFA versus ADA (69% lower risk of ILD; adjusted hazards ratio 0.31).55,56 A multinational registry/claims study reported comparable rates of serious/opportunistic infections and tuberculosis for ABT versus csDMARDs or other b/tsDMARDs after adjustment, 57 and a US Veterans Health Administration trial emulation showed that starting ABT, TCZ, or TOFA yielded a similar 3-year risk of death or respiratory hospitalization to RTX. 58
In addition, accumulating recent evidence suggests that anti-IL-6R and JAKi provide efficacy comparable to RTX and ABT for control of articular disease, stabilization of lung function, attenuation of HRCT progression, and ILD progression-related mortality, without a significant increase in serious respiratory events.59–63
Evidence regarding the safety of TNFi remains conflicting; however, cases of new-onset or exacerbation of ILD have been described in patients receiving TNFi monotherapy or treated for ulcerative colitis or spondyloarthritis, leading some guidelines to advise against or individualize their use.64–67
- Rheumatoid vasculitis (RV): The ScR did not identify studies that specifically assessed the efficacy and safety of b/tsDMARDs in TNFi-inadequate responders with RV. RTX is the most studied bDMARD, with evidence supporting its efficacy for remission induction and glucocorticoid-sparing effects.68–70 In broader RA cohorts, the AIR registry reported remission with RTX in 71% at 6 months and 82% at 12 months, with an acceptable safety profile. 64 ABT, TCZ, and JAKi have emerged as potential alternatives, but evidence remains mainly limited to case reports.71–73 Data on TNFi are less favorable, with 56% achieving complete remission at 6 months, 9% mortality at 3 years, high rates of serious infection, and high relapse rates. 74
- High CV risk or previous CV event: High CV risk is variably defined; a commonly used pragmatic definition is ⩾2 traditional risk factors (smoking, hypertension, hyperlipidemia, diabetes mellitus). Despite improvements over recent decades in CV morbidity and mortality in RA, 75 an excess CV risk persists compared with the general population.76,77 Clinical data from RCTs and observational studies, including biological registries, in mixed RA populations indicate that TNFi, RTX, ABT, and anti-IL-6R are not associated with higher CV risk than csDMARDs and, in some analyses, may reduce risk via rapid improvements in endothelial function and arterial stiffness.78–83 In addition, anti-IL-6R lower CRP, increase apolipoprotein A1 (ApoA1), reduce glycated hemoglobin, and improve insulin resistance.84–86 In some studies, TCZ has also demonstrated a decreased risk of major adverse cardiovascular events (MACE) relative to TNFi and RTX.87–89
In oral surveillance, patients aged ⩾50 years with ⩾1 additional CV risk factor had higher MACE rates with TOFA (0.98) than with TNFi (0.73) 24 ; no excess risk was observed in patients without such risk factors.
- Risk factors for venous thromboembolic events (VTE): VTE risk factors include older age, prior thromboembolic events, pregnancy, smoking, malignancy, obesity, and hormone replacement therapy. 90 In patients with treated RA, the incidence of VTE is approximately 0.7 per 100 patient-years (deep vein thrombosis: 0.6; pulmonary embolism: 0.3). 10 The ScR identified no studies specifically addressing patients with inadequate response to TNFi. Across RA populations, the risk of VTE appears similar between bDMARDs and csDMARDs.84,85 However, signals of increased risk have been observed at higher doses of JAKi, particularly with TOFA and BARI, warranting caution in patients with multiple VTE risk factors.24,91 In the ORAL SURVEILLANCE study, an increased risk of VTE was observed only with TOFA 10 mg compared with TNFi. 24
- Obesity: In TNFi-inadequate responders, two subanalyses of randomized controlled trials showed that short-term (12-week) efficacy of TOFA 15 and filgotinib 92 was not influenced by baseline body mass index (BMI). Evidence regarding the effectiveness of TNFi in obesity is mixed; however, most datasets suggest attenuated treatment responses, with obese patients showing significantly lower odds of achieving remission or a good clinical response compared with non-obese individuals.93–95
Meta-analyses and large registry studies indicate that the odds of achieving a clinical response or remission with ABT and TCZ do not differ significantly between obese and non-obese patients.94,96 For ABT, no conclusive data demonstrate superiority over other biologic or targeted synthetic disease-modifying antirheumatic drugs in patients with obesity. 93 With RTX, available evidence suggests no detrimental impact of obesity, although the number of studies remains limited.94,96
In the RABBIT registry (10,593 patients with RA), obesity negatively affected the real-world effectiveness of cytokine-targeted therapies, but not that of cell-targeted therapies. 97
- High infection risk: Across RA populations, b/tsDMARDs increase the risk of infections, including serious ones.78,98,99 ABT shows a favorable infection safety profile and, in patients with prior infections during TNFi therapy, confers the lowest subsequent infection risk compared with other bDMARDs.100–102 A systematic review also reported lower rates of serious infections with ABT than with TNFi, RTX, or anti-IL-6R. 103 ETN is similarly associated with fewer serious infections than IFX (adjusted HR 0.49, 95% CI 0.29–0.83) and ADA (adjusted HR 0.55, 95% CI 0.44–0.67), and with a reduced risk of tuberculosis reactivation compared with monoclonal TNFi (39 vs 136–144 per 100,000 person-years for IFX/ADA).104,105
- Osteoporosis: The ScR and product information have raised a cautionary signal regarding TOFA use in patients with fracture risk factors. Pharmacovigilance has also indicated a possible osteoporosis risk with TOFA, 106 yet pooled phase I–III and long-term extension data showed a numerically lower fracture risk than placebo and a slightly higher risk than TNFis, with fracture predictors resembling those in the general RA population.107,108 Further evidence is needed to clarify the association between JAKi and osteoporosis or fractures. In TNFi-inadequate responders, TCZ significantly reduced biomarkers of cathepsin K–mediated bone resorption and matrix metalloproteinase-mediated tissue degradation and remodeling at 16 weeks compared with placebo 109 ; post hoc data from TARGET suggest that SAR achieved similar short-term reductions. 110 In a small open-label RCT in ETN-refractory patients, switching to IFX versus continuing ETN yielded similar 16-week improvements in bone biomarkers. 111 In other RA populations, bDMARDs improve bone remodeling biomarkers and bone mineral density, but their effects on fracture incidence remain uncertain. 112
- Nociplastic pain, depression, and fatigue: In the TARGET trial, 19% of patients had baseline non-inflammatory pain (NIP). SAR significantly reduced the proportion with NIP versus placebo at week 12 (24% vs 35%) and week 24 (11% vs 20%). 113 It also improved ACR20/50/70 and low-disease-activity endpoints (SDAI ⩽11, CDAI ⩽10) regardless of probable major depressive disorder, with higher ORs and nominal p < 0.01 in both subgroups (no multiplicity adjustment).
Beyond TARGET, both TCZ and JAKi reduce NIP114–118 and improve mental-health outcomes.119–121 The ScR yielded no evidence specifically addressing fatigue in the target population. In broader RA populations, b/tsDMARDs appear effective and generally safe for fatigue management, with a reported dose–response relationship for anti-IL-6R and JAKi.122,123 As with pain, RA-associated fatigue has multifactorial causes, and IL-6–mediated HPA-axis dysfunction has been proposed as a contributor to chronic fatigue. 118
- Previous solid cancer: Randomized and observational datasets (including registries) indicate that bDMARDs do not increase the incidence of solid cancers compared with the general population or csDMARDs.78,116,124 Real-world registry data indicate that TNFi are the most commonly used bDMARDs after a cancer diagnosis, with no clear evidence of harm. 124 Specifically, in BIOBADASER, the risk of incident cancer did not differ between TNFi and other b/tsDMARDs among patients with prior malignancy. 126 By contrast, in ORAL SURVEILLANCE, overall cancer incidence was higher with TOFA than with TNFi (HR 1.48, 95% CI 1.04–2.09). 24 In this sense, a SLR and meta-analysis reported a higher incidence of malignancy with JAKi compared with TNFi, but not compared with placebo or MTX. 127
- Hematological cancer: lymphoma risk in recent-onset RA is similar to that in historical cohorts, and b/tsDMARDs do not appear to further increase it.128–130 RTX is used to treat lymphoma and is guideline-endorsed over other bDMARDs for patients with prior lymphoproliferative disorders.131,132 The ScR did not identify TNFi-inadequate responder-specific comparative evidence.
- Non-melanoma skin cancer (NMSC): evidence is mixed, although TNFi have been associated with an increased NMSC risk in some studies.124, 133–136 An SLR suggested a potential increase with ABT compared with csDMARDs, but not versus other b/tsDMARDs (lower 95% CI bound near unity). 134 One cohort reported higher NMSC risk with JAKi than with TNFi (HR 1.39, 95% CI 1.01–1.91), particularly after ⩾2 years of JAKi treatment (HR 2.12, 95% CI 1.15–3.89). 136 In ORAL SURVEILLANCE, adjudicated NMSC was more frequent with TOFA at both doses than with TNFi. 24 Data for RTX and anti-IL-6R remain scarce.124–126
- Pregnancy: TNFi, particularly CZP, are broadly considered pregnancy-compatible.137–139 Large national registries including millions of live-born infants show no increased risk of congenital malformations with TNFi.137–140 For TNFi non-responders who become pregnant, alternatives are limited; evidence for RTX, ABT, anti-IL-6R, and JAKis is scarce, and these agents are generally withheld before conception.140–145
Expert recommendations and Delphi results
The steering committee formulated 17 recommendations; 15 reached the predefined consensus threshold, while 2 did not after the second round. These are presented in Table 1, together with the results of the Delphi process. The statements integrate the ScR with expert judgment to provide a profile-based framework for post-TNFi decision-making. Table 2 summarizes the treatment selection decision tree following first-line TNFi therapy in RA.
Main results of the Delphi process.
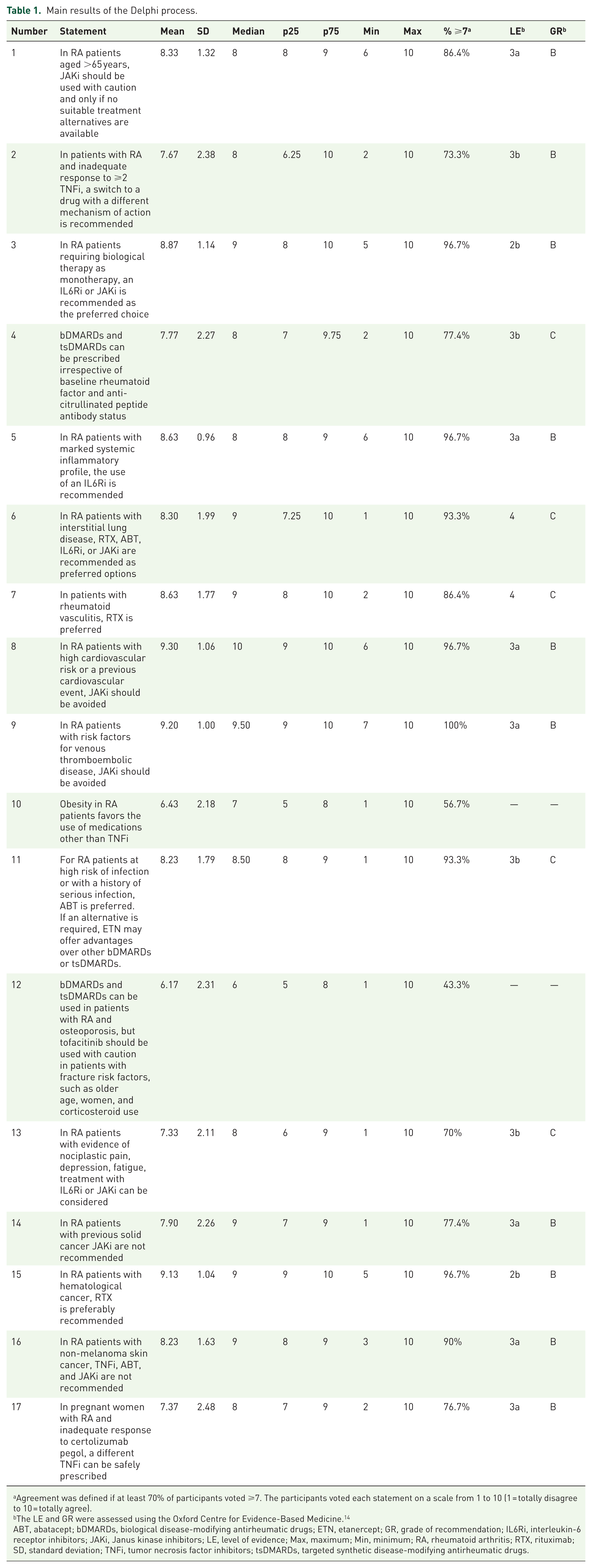
Agreement was defined if at least 70% of participants voted ⩾7. The participants voted each statement on a scale from 1 to 10 (1 = totally disagree to 10 = totally agree).
The LE and GR were assessed using the Oxford Centre for Evidence-Based Medicine. 14
ABT, abatacept; bDMARDs, biological disease-modifying antirheumatic drugs; ETN, etanercept; GR, grade of recommendation; IL6Ri, interleukin-6 receptor inhibitors; JAKi, Janus kinase inhibitors; LE, level of evidence; Max, maximum; Min, minimum; RA, rheumatoid arthritis; RTX, rituximab; SD, standard deviation; TNFi, tumor necrosis factor inhibitors; tsDMARDs, targeted synthetic disease-modifying antirheumatic drugs.
Decision tree for treatment selection following first-line TNFi therapy in RA patients.

 Purple: “Strongly recommended”: robust direct evidence (e.g., randomized controlled trials) in the target RA population or treatment strategy;
Purple: “Strongly recommended”: robust direct evidence (e.g., randomized controlled trials) in the target RA population or treatment strategy;  Dark blue: “Highly recommended”: supported by direct but not robust evidence (e.g., exploratory analyses) and/or by indirect but relevant evidence from different RA populations (e.g., randomized controlled trials or high-quality observational studies, including biologic registries), and/or by a highly plausible mechanism of action;
Dark blue: “Highly recommended”: supported by direct but not robust evidence (e.g., exploratory analyses) and/or by indirect but relevant evidence from different RA populations (e.g., randomized controlled trials or high-quality observational studies, including biologic registries), and/or by a highly plausible mechanism of action;  Light blue: “Recommended”: the available evidence does not meet the threshold for a dark-blue classification;
Light blue: “Recommended”: the available evidence does not meet the threshold for a dark-blue classification;  Gray: “Neutral”: current data are insufficient to support or oppose use; evidence is scarce and no coherent mechanism of action has been demonstrated;
Gray: “Neutral”: current data are insufficient to support or oppose use; evidence is scarce and no coherent mechanism of action has been demonstrated;  Light red: “Use with caution”: direct or indirect evidence points to warning signs suggestive of increased risk; use should be restricted and closely monitored;
Light red: “Use with caution”: direct or indirect evidence points to warning signs suggestive of increased risk; use should be restricted and closely monitored;  Dark red: “Not recommended”: there is relevant and robust evidence, whether direct or indirect, against its use.
Dark red: “Not recommended”: there is relevant and robust evidence, whether direct or indirect, against its use.
ACPA, anti-citrullinated peptide antibody; CV, cardiovascular; CZP, certolizumab pegol; ETN, etanercept; IL6Ri, interleukin-6 receptor inhibitors; JAKi, Janus kinase inhibitors; RA, rheumatoid arthritis; RF, rheumatoid factor; TNFi, tumor necrosis factor inhibitors.
Consensus was achieved in most scenarios. In patients aged ⩾65 years, JAKi should be used with caution and only when no suitable alternatives are available, with ABT regarded as a reasonable option. After inadequate response to ⩾2 TNFi, switching to a drug with a different MoA was preferred over cycling a third TNFi. For biologic or targeted monotherapy, anti-IL-6R or JAKi were favored, with ETN as an alternative when these were unsuitable. Treatment decisions should not rely solely on RF/ACPA status but should consider the overall clinical context.
Anti-IL-6R were preferred in the presence of prominent systemic inflammation. In RA-ILD, ABT, RTX, anti-IL-6R, and JAKi were all considered appropriate, as no specific agent was prioritized based on current evidence. RTX was endorsed as the treatment of choice in RV. In patients with high CV risk or a prior CV event, and in those with VTE risk factors, JAKi should be avoided in favor of agents with a more favorable safety profile. In those at high risk of infection or with a history of serious infection, ABT was preferred, and ETN was considered a suitable alternative. Anti-IL-6R or JAKi could be used when nociplastic pain, depression, or fatigue predominated. JAKi were discouraged in patients with previous solid cancer; RTX was preferred in those with hematological malignancy or lymphoproliferative disease; and TNFi, ABT, and JAKi were not recommended in NMSC. In pregnancy, if response to CZP was inadequate, switching to another pregnancy-compatible TNFi was acceptable, whereas RTX, ABT, anti-IL-6R, and JAKi should be withheld before conception.
Consensus was not reached in two scenarios. The statement that “obesity in RA favors the use of medications other than TNFi” received 56.7% agreement (mean score 6.43). For osteoporosis, the proposal that TOFA should be used with caution in patients with fracture risk factors reached 43.3% agreement (mean 6.17).
Discussion
The therapeutic landscape of RA has evolved substantially in recent years, offering a wide range of targeted treatment options for patients who fail TNF inhibitors. However, selecting the most appropriate second-line therapy remains challenging, as head-to-head trials are scarce and existing guidelines often provide only broad recommendations that do not fully capture the complexity of individual clinical scenarios. 6
This document was conceived as a practical decision-making support tool to assist rheumatologists in choosing the right treatment for the right patient after TNFi failure. By integrating a ScR of the literature with expert opinion through a structured Delphi process, we aimed to provide a clinically oriented, profile-based framework that complements rather than replaces current guidelines. While EULAR recommendations offer overarching treatment strategies, 6 our approach explores specific clinical contexts in greater depth, providing more detailed, scenario-based guidance. In situations where direct evidence in patients failing a first TNFi was unavailable, recommendations were informed by indirect evidence and structured expert consensus.
Overall, consensus was reached in the vast majority of proposed clinical situations, supporting the feasibility of a profile-driven approach in routine care. The final statements reflect a pragmatic balance between efficacy, safety, comorbidities, prior treatment history, and regulatory considerations. The high level of agreement achieved among a large group of rheumatologists from two countries reinforces the external validity of the recommendations. The two areas in which consensus was not achieved (obesity and osteoporosis) highlight ongoing evidence gaps and the need for further research to inform treatment decisions in these settings.
Obesity is highly prevalent among patients with RA and is clinically relevant due to its association with higher disease activity and lower remission rates.146–148 Multiple studies and systematic reviews consistently demonstrate that increased BMI correlates with more severe disease and a reduced likelihood of achieving treatment targets such as low disease activity or remission.146,147 Therefore, obesity should be taken into account when selecting advanced therapies for RA.148,149 Although current evidence tends to favor the use of non-TNFi agents in obese patients, its strength was insufficient to support a formal recommendation against TNFi use, particularly given that certain TNFi, such as IFX and GOL, allow for weight-based dosing.
Regarding osteoporosis, the lack of consensus likely reflects limited awareness among rheumatologists of the warning in the TOFA Summary of Product Characteristics, which advises caution in patients with established fracture risk factors, including older age, female sex, and concomitant glucocorticoid use, irrespective of indication or dose, as fractures have been observed in patients treated with this agent.106–108
The present work specifically focuses on treatment decisions after inadequate response to a first TNFi. This stage precedes the current EULAR definition of difficult-to-treat RA, which requires failure of at least two biologic or tsDMARDs with different mechanisms of action; therefore, management of difficult-to-treat RA was beyond the scope of this manuscript. 150
An important aspect that deserves clarification regarding the available evidence is the safety of TNFi in patients with RA-ILD, which remains a matter of debate. Some studies, such as the retrospective analysis using data from the US Department of Veterans Affairs cohort with up to 3 years of follow-up, found no significant differences in respiratory-related hospitalizations or in overall or respiratory-specific mortality when comparing TNFi with other biologics among patients with RA-ILD. 151 In contrast, a study conducted in an Asian population using the TriNetX database 152 reported a higher risk of mortality and an increased need for mechanical ventilation in patients treated with TNFi compared with ABT. In addition, as previously mentioned, cases of ILD onset or exacerbation have been described in patients receiving TNFi as monotherapy or in those treated for ulcerative colitis or spondyloarthritis. 64 On this basis, and in line with current guidelines,64–67 the expert panel continues to recommend a risk-minimization approach by prioritizing alternative therapies in patients with RA-ILD.
Although until recently RTX and ABT were prioritized64–67,153,154 because they were supported by the largest body of published evidence, accumulating data now suggest that anti-IL-6R agents and JAKi provide efficacy comparable to RTX and ABT for control of articular disease, stabilization of lung function, attenuation of HRCT progression and ILD progression-related mortality, without a significant increase in serious respiratory events.59–63 Therefore, these four options (ABT, RTX, anti-IL-6R, and JAKi) may be used interchangeably in RA-ILD patients requiring treatment intensification. However, this does not imply a systematic contraindication to the use of TNFi in this setting, as there is no solid rationale to support such a position. Indeed, in patients already receiving TNFi with stable lung disease and well-controlled articular symptoms, there is no conclusive evidence to support discontinuation
Although until recently RTX and ABT were prioritized.64–67,153,154 because they were supported by the largest body of published evidence, accumulating data now suggest that anti-IL-6R agents and JAKi provide efficacy comparable to RTX and ABT for control of articular disease, stabilization of lung function, attenuation of HRCT progression, and ILD progression-related mortality, without a significant increase in serious respiratory events.59–63 Therefore, these four options (ABT, RTX, anti-IL-6, and JAKi) may be used interchangeably in RA-ILD patients requiring treatment intensification. However, this does not imply a systematic contraindication to the use of TNFi in this setting, as there is no solid rationale to support such a position. Indeed, in patients already receiving TNFi with stable lung disease and well-controlled articular symptoms, there is no conclusive evidence to support discontinuation.
Regarding predictors of response after TNFi failure, validated tools to guide routine clinical decision-making are currently lacking. However, emerging precision medicine approaches may help refine therapeutic selection. Using machine-learning methods applied to clinical trial data, Rehberg et al. 155 identified a biomarker-based rule combining ACPA positivity and elevated CRP levels to predict response to sarilumab and to discriminate between responses to sarilumab and ADA. Although promising, these results remain exploratory and require prospective real-world validation before clinical implementation, particularly in TNFi-inadequate responder populations.
Finally, it is important to note that, since the development of the present recommendations, two EULAR-endorsed sets of recommendations have been published providing complementary information to that included in our work, although their conclusions were not specifically derived from patients with inadequate response to TNFi.155–158 The EULAR points to consider on the initiation of targeted therapies in patients with inflammatory arthritis and a history of cancer indicate that, when targeted antirheumatic therapy is required in patients with a history of solid cancer, TNFi may be preferred over other options.156,157 In contrast, JAKi and ABT should be used with caution and only in the absence of suitable alternatives. For patients with a history of lymphoma, B-cell depleting therapy may be preferred. 156 The EULAR recommendations for the use of antirheumatic drugs in reproduction, pregnancy, and lactation confirm that all TNFi can be used throughout pregnancy. 158 They also authorize the use of certain non-TNFi bDMARDs (ABT, RTX, TCZ, SAR) when necessary to control maternal disease effectively. Breastfeeding is compatible with the continuation of both TNFi and non-TNFi bDMARDs, but not with JAKi.
The main limitation of this work is the scarcity of robust comparative evidence specifically addressing patients with inadequate response to a first TNF inhibitor across most predefined scenarios. Available data are often heterogeneous or derived from exploratory subgroup analyses, and trials specifically designed for each clinical profile are unlikely. When direct evidence was lacking, this limitation was explicitly acknowledged, and recommendations were developed through structured expert consensus informed by indirect evidence and biological plausibility. In this context, expert consensus represents a pragmatic means of supporting clinical decision-making. Some recommendations also reflect precautionary positions based on safety concerns or regulatory restrictions rather than efficacy data. Accordingly, while the proposed statements offer a structured framework, clinical judgment remains essential, and individualized decisions may justify the cautious use of agents not explicitly endorsed. A formal power calculation was not applicable to this qualitative, consensus-based design and is acknowledged as an additional limitation.
Conclusion
In summary, tailoring treatment after TNFi failure is essential to optimize outcomes in RA. Although high-quality comparative evidence is limited, the recommendations and decision tree proposed in this project guide rational, patient-centered treatment choices by combining the best available evidence with expert consensus. Overall, this approach provides a practical decision-support framework for clinicians, facilitating treatment selection and routine clinical decision-making in everyday patient care.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261425808 – Supplemental material for What to do when the first TNF inhibitor fails in rheumatoid arthritis: stratified expert recommendations from a scoping review and Delphi consensus
Supplemental material, sj-docx-1-tab-10.1177_1759720X261425808 for What to do when the first TNF inhibitor fails in rheumatoid arthritis: stratified expert recommendations from a scoping review and Delphi consensus by Javier Narváez, Rosario García-Vicuña, Jesús Tornero Molina, Susana Romero-Yuste, José A. Pereira da Silva and Estibaliz Loza in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-pdf-2-tab-10.1177_1759720X261425808 – Supplemental material for What to do when the first TNF inhibitor fails in rheumatoid arthritis: stratified expert recommendations from a scoping review and Delphi consensus
Supplemental material, sj-pdf-2-tab-10.1177_1759720X261425808 for What to do when the first TNF inhibitor fails in rheumatoid arthritis: stratified expert recommendations from a scoping review and Delphi consensus by Javier Narváez, Rosario García-Vicuña, Jesús Tornero Molina, Susana Romero-Yuste, José A. Pereira da Silva and Estibaliz Loza in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
