Abstract
Background:
Currently, there is contradictory evidence regarding the best strategy to follow after discontinuation of a first biological agent in patients with rheumatoid arthritis (RA). We aimed to compare the long-term efficacy of switching to a second tumor necrosis factor inhibitor (TNFi) versus biopharmaceuticals with other mechanisms of action (non-TNFi) in patients with RA who previously failed a first TNFi.
Methods:
This prospective cohort study analyzed data from 127 patients who discontinued a previous TNFi between 1999 and 2016. Disease activity was assessed at baseline and at 6, 12, and 24 months (m-6, m-12, m-24) after switching. Primary outcome was the proportion of patients achieving good/moderate EULAR response (E-resp). Factors associated with clinical outcomes were assessed using univariate and multivariate logistic regression models.
Results:
Seventy-seven (61%) patients received a second TNFi and 50 (39%) switched to a non-TNFi. At m-6 and m-12, no differences were observed between groups; nevertheless, at m-24, the proportion of patients with E-resp was higher in the non-TNFi group (49% TNFi group versus 77% non-TNFi group; p = 0.002). In regression models, switching to a non-TNFi was significantly associated with E-resp at m-24 (odds ratio = 3.21; p = 0.01). When assessing the response to the second biological agent based on the reason for discontinuation of the first TNFi, similar results were obtained; at m-24, patients who discontinued the first TNFi due to inefficacy (either primary or secondary) experienced a better E-resp if they had switched to a non-TNFi (primary inefficacy: 52% TNFi group versus 79% non-TNFi group, p = 0.09; secondary inefficacy: 50% versus 76%, p = 0.03).
Conclusion:
In our cohort of RA patients who discontinued a first TNFi, those who switched to a non-TNFi were three times more likely to attain a sustained clinical response, regardless of whether they had discontinued the first biologic due to a primary or secondary inefficacy.
Keywords
Introduction
Biological therapy has dramatically improved the prognoses of patients with rheumatoid arthritis (RA). While tumor necrosis factor inhibitors (TNFis) have routinely been used as first-line biological agents [biological disease-modifying antirheumatic drugs (bDMARDs)], between 30% and 40% of patients have an inadequate response to TNFi either due to inefficacy or intolerance. 1
Currently, there is no clear strategy to follow when a patient fails to respond to the first TNFi, as evidence on the efficacy of trying a second TNFi (cycling) in comparison with using alternative mechanisms of action (e.g. swapping), whether based on randomized controlled trials (RCTs) or epidemiological studies, remains both insufficient and controversial. 2 Because direct clinical comparisons are scarce, researchers have indirectly compared the results of different RCTs using sophisticated statistical analyses. 3 Thus, differences in effectiveness cannot be excluded or concluded with certainty.
On the other hand, two different types of TNFi inefficacy – primary or secondary – have been identified and there are data suggesting differences in treatment response to the second bDMARD depending on the reason for discontinuation of the prior TNFi.4–8 In order to optimize a switching strategy in clinical practice, more information is needed about whether factors such as the reason for switching influence the treatment response to a second bDMARD. By the same token, the impact of the number of previous TNFis has also not yet been established.
To our knowledge, although there are studies evaluating the clinical response after switching to a second biological therapy in patients with RA, most had a short follow-up period.3–14 A recent study assessing this issue had a longer follow-up period after switching. In this report, however, patients who switched due to lack or loss of effectiveness were pooled together. 15 Thus, until now, no evidence has emerged comparing the clinical response between cycling and swapping strategies with a follow-up longer than 1 year after switching and considering both the reason for discontinuation of the first biologic and the distinction between the different types of ineffectiveness. The present study offers the opportunity to clarify which patients will benefit from cycling/swapping strategies in terms of long-term efficacy.
The aim of this study was to compare the long-term efficacy of TNFi versus non-TNFi after discontinuing a previous TNFi in a cohort of patients with RA and to identify potential risk factors affecting the long-term clinical response to a second bDMARD. Secondary objective was to evaluate whether reason for discontinuation of the first biopharmaceutical influences the response to the second biological.
Materials and methods
This prospective observational study included 127 patients with RA, of a total of 215 patients from the Rheumatoid Arthritis Registry at La Paz University Hospital (RA-Paz cohort), who dropped out the first TNFi between January 1999 and March 2016 and who were subsequently treated with a second bDMARD, that is, a TNFi or a biological agent with a different mechanism of action [rituximab (RTX), abatacept (ABA), or tocilizumab (TCZ)]. Before 2010, only TNFi, RTX (from 2007) and ABA (from 2008) could be prescribed in our Unit. All included bDMARD were available from 2010.
The ‘La Paz University Hospital RA’ Registry is a database of all patients who have received or are receiving treatment with a bDMARD and it includes demographic characteristics and clinical information of each visit to the biological therapy unit, which take place by default every 6 months according to the unit’s protocol.
The inclusion criteria for this study were as follows: a diagnosis of RA by a rheumatologist, age >18 years, and discontinuation of a first TNFi. Patients who discontinued the first TNFi due to remission, who had not started a second bDMARD, or without clinical data collected during the second biopharmaceutical were excluded.
There is no consensus on the definition of nonresponse to biological treatments, but generally is categorized as primary or secondary inefficacy based on whether an initial clinical response is observed or not.16–18 Primary inefficacy was defined in our study as patients with a non-EULAR response (delta-DAS28 ⩽ 0.6 or delta-DAS28 < 1.2 with DAS28 > 5.1) during the first 6 months of therapy. Instead, secondary inefficacy was defined as patients with a non-EULAR response at any studied point after a good or moderate EULAR response (delta-DAS > 0.6 and DAS28 ⩽ 5.1) during the first 6 months of therapy. In addition, all patients included had a record in the database by their treating rheumatologist specifying the reason for dropping out the first biological. This was taken into account to ensure that all patients who met the definition criteria were properly classified.
This study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Medical Ethics Committee of La Paz University Hospital (PI nº 1155).
The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement guidelines for reporting observational studies were followed. 19
Data collection and outcome measures
Demographic information was collected before initiating treatment with a second bDMARD. Clinical data were obtained from questionnaires and assessments of disease activity conducted during routine clinical encounters by qualified rheumatologists and with standardized joint evaluation.
Clinical activity was assessed using the Disease Activity Score in 28 joints (DAS28) prior to starting the first TNFi, every 6 months during the first-line bDMARD, before initiating a second bDMARD (baseline visit) and at 6, 12, and 24 months after starting a second-line biological therapy. DAS28 was calculated using erythrocyte sedimentation rate. Neither the clinical disease activity index (CDAI) nor the simplified disease activity index (SDAI) was used since clinical response was measured using the EULAR (European League Against Rheumatism) response criteria at any studied point. Responders were defined as patients with good or moderate response in the clinical visits. All patients needed to have at least the baseline assessment and an additional one (at 6, 12, or 24 months) during the second treatment period.
In order to provide an integrative vision of effectiveness taking into account both treatment response and adherence to therapy, we added the LUNDEX index, which is calculated as the fraction of patients adhering to therapy multiplied by the fraction of patients fulfilling the selected response criterion (EULAR response) at a given time. 20
The treatment response to the second biological therapy was analyzed over 2 years after switching. Patients who discontinued the second bDMARD before their next visit due to a nonclinical response or remission were imputed on the next visit. Thus, clinical data were available for 127 patients at baseline and at 6 months, and for 123 patients at 12 and 24 months.
The reasons for treatment discontinuation of the first TNFi (lack of efficacy, loss of efficacy, adverse event and other causes such as patient’s wishes, pregnancy or loss of follow-up) were collected.
Statistical analysis
Descriptive statistics were reported as median and interquartile ranges (IQRs) for continuous variables and as absolute numbers and relative frequencies for categorical variables. Mann–Whitney U tests and Fisher’s exact tests were used to analyze any statistical differences in baseline characteristics among the groups.
Comparisons of response rates between patients who started a second TNFi and those who switched to a non-TNFi were made at 6, 12, and 24 months after the start of the second bDMARD using Fisher’s exact tests.
Subsequently, a stratified analysis was performed to evaluate clinical response according to three reasons for discontinuing the first biological agent (primary inefficacy, secondary inefficacy, and a third group that included other reasons such as adverse events, patient’s wishes, pregnancy, or loss of follow-up). When stratifying patients according to the cause of withdrawal of the first bDMARD, in order to simplify the reasons for discontinuation, patients were classified taking into account the main reason for stopping second bDMARD, although concomitantly it could exist another associated reason.
Because not all biologics included in the study were available before 2010, we have performed a subanalysis comparing the populations that started the second bDMARDs before and after this period to avoid calendar time bias.
Univariable and multivariable logistic regression models were employed to evaluate the factors associated with being a EULAR responder during the second biological therapy. Age, sex, body mass index (BMI), disease duration, smoking status, rheumatoid factor (RF) and anti-citrullinated protein antibodies (ACPA) status, chosen second-line bDMARD (cycling or swapping), concomitant methotrexate, DAS28 prior to starting the second bDAMRD, and the reason for discontinuing the first TNFi were included as independent variables in the univariable analyses; those variables found to have significant associations in the univariable analysis (p < 0.1) were included as independent variables in the multivariable analyses. In addition, a collinearity analysis was performed to select the final predictor variables for the multivariable analysis.
All analyses were performed using GraphPad Prism 6 (San Diego, CA, USA) and SPSS 21.0 software; significant p values <0.05 and odds ratio (OR) with their confidence intervals (95% CIs) were calculated.
Results
A total of 215 patients with RA who discontinued treatment of their first TNFi between January 1999 and March 2016 were screened. Of these, 62 (29%) were excluded because they did not start treatment with a second biopharmaceutical. The reasons for not starting a second bDMARD included transfer to a different hospital center, loss of follow-up, patient’s wishes, pregnancies, malignancies, severe infections, and death from any cause. Five (2%) additional patients were excluded because they discontinued treatment due to remission. Missing baseline DAS28 values precluded the enrolment of another 21 (10%) patients, leaving 127 (59%) patients for inclusion in the analysis (full analysis population). Of these, 77 (61%) received treatment with a second TNFi and 50 (39%) switched to a non-TNFi. Table S1 specifies what types of TNFi were used as second line of therapy (see in Supplementary Material).
Demographic, clinical, and therapeutic characteristics
Demographic and clinical characteristics of patients at baseline (before starting the second bDMARD) are shown in Table 1. Based on the type of the second bDMARD, patients were divided in two categories: TNFi (77 patients) and non-TNFi [50 patients; 26 (52%) with RTX, 15 (30%) with TCZ, and nine (18%) with ABA]. At baseline, the only two significant differences between the therapy groups were that patients in the non-TNFi group had significantly higher C-reactive protein (CRP) levels and lesser treatment dropouts due to other reasons. The proportion of seropositivity for rheumatoid factor was numerically higher in those switching to non-TNFi, but did not reach statistical significance. In general, patients discontinued the first TNFi mainly due to secondary inefficacy (50%, n = 64) and to a lesser extent due to primary inefficacy (36%, n = 45) or other reasons (14%, n = 18; 15 patients due to adverse events and three due to other causes).
Baseline demographic, clinical and therapeutic patient characteristics.

ACPA, anti-citrullinated protein antibodies; bDMARD, biological disease-modifying antirheumatic drugs; BMI, body mass index; CRP, C-reactive protein; DAS, disease activity score; DMARD, disease-modifying antirheumatic drug; ESR, erythrocyte sedimentation rate; IQR, interquartile range; LFL, leflunomide; MTX, methotrexate; n, number of patients; ns, not statistically significant; PI, primary inefficacy; SD, standard deviation; SI, secondary inefficacy; SZP, salazopyrine; TNFi, tumor necrosis factor inhibitor.
Data are expressed as mean ± SD, positive number/tested number (%), or median with 25–75th percentiles (interquartile range). Statistically significant p values are indicated in bold.
At the start of second bDMARD.
Other causes: patient’s wishes, pregnancy or loss of follow-up.
Patients who experienced not only inefficacy but also other reason of discontinuation of the first TNFi (adverse events, patient’s wishes, pregnancy or loss of follow-up).
A subanalysis was performed comparing baseline characteristics between the three groups established according to the reason for discontinuing the first biological agent. No statistical differences were observed in most characteristics except that those patients who discontinued the first TNFi due to primary inefficacy had a lower DAS28 before starting the first biological therapy (see Table S2 in Supplementary Material).
When comparing the characteristics of each therapy group considering whether the date of starting the second biological was before or after 2010, lower disease activity (by DAS28) upon start of second bDMARD and longer duration of the first TNFi was observed in those patients who started second biologic therapy after or during 2010 (see Table S3 in Supplementary Material).
Clinical response after switching to a second bDMARD
More patients achieved a significant EULAR response in the non-TNFi group at 24 months after switching (49% (37/75) of the TNFi group versus 77% (37/48) of the non-TNFi group, p = 0.002) (see Figure 1). No significant differences were observed between the TNFi and non-TNFi groups with respect to the proportion of patients who achieved a EULAR response (good and moderate) at 6 and 12 months after starting the second biopharmaceutical [at 6 months: 58% (45/77) of the TNFi group versus 66% (33/50) of the non-TNFi group, p = 0.4; at 12 months: 58% (43/74) of the TNFi group versus 67% (33/49) of the non-TNFi group, p = 0.3).

EULAR response of second biological agent in the global cohort during the study period in the two treatment arms (non-TNFi/second TNFi treatment). (a) after 6 months, (b) after 12 months, and (c) after 24 months.
In addition, similar results were observed in a subanalysis evaluating EULAR response 24 months after switching to a second bDMARD in patients who discontinued the first TNFi either due to primary or secondary inefficacy (data shown in Table 2). Although outcome data with a second-line biological therapy in patients who switched due to other causes (intolerance, patient’s wishes, pregnancy, or loss of follow-up) were obtained, the small sample size prevented comparing clinical responses between cycling and swapping strategies.
Proportion of EULAR responders (good and moderate) to a second BT according to reasons for discontinuation of the first TNFi.

BT, biological therapy; EULAR, European League Against Rheumatism; n, number of subjects; ne, nonevaluable, due to the low number of cases; TNFi, tumor necrosis factor inhibitor.
Statistically significant p values are indicated in bold.
When stratifying the response to the second biological agent based on the date of initiation of their second biological therapy, similar results to those of the overall cohort were obtained in both the subpopulation that started second bDMARD before 2010 and in the subpopulation that initiated second bDMARD during or after 2010, with higher response rates in the non-TNFi group at 24 months after switching (see Table S3).
In our study, patients who switched to a non-TNFi had the highest overall LUNDEX values (proportion of patients at each studied point who achieved EULAR response and were still adhered to therapy) in comparison with the group that started a second TNFi at any studied point (6 months: 64% with non-TNFi versus 54% with TNFi; 12 months: 58% with non-TNFi versus 47% with TNFi; 24 months: 44% with non-TNFi versus 35% with TNFi) (see Figure 2).
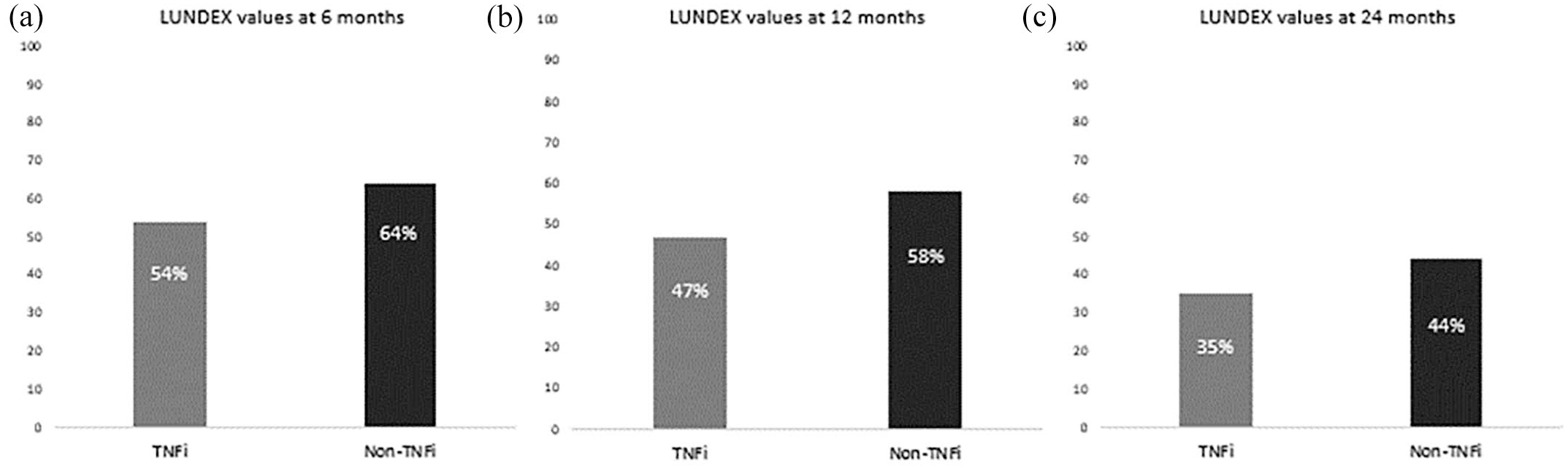
LUNDEX values of second biological agent in the global cohort during the study period in the two treatment arms (non-TNFi/second TNFi treatment).
Predictors of long-term EULAR response to a second biological agent
Univariate analysis showed that longer disease duration before starting the second bDMARD (OR = 1.06, p = 0.02) and receiving a non-TNFi as second biological therapy were associated with a EULAR response 24 months after switching (see Table 3).
Univariable and multivariable logistic regression analyses of predictors for achieving long-term EULAR response (24 months after switching).

ACPA, anti-citrullinated protein antibodies; BMI, body mass index; CI, confidence interval; CRP, C-reactive protein; DAS, disease activity score; EULAR, European League Against Rheumatism; OR, odds ratio; RF, rheumatoid factor; TNFi, tumor necrosis factor inhibitor.
Finally, the multivariate analysis showed that the only factor independently associated with a good EULAR response was receiving a non-TNFi (OR = 3.2, p = 0.007) as a second biological therapy (see Table 3).
Reasons of discontinuation of the second biological agent
Patients withdrew the second biological agent more frequently due to primary inefficacy [19 (34%) primary inefficacy, 13 (24%) secondary inefficacy, 13 (24%) adverse events, six (11%) achieving remission, and four (7%) other reasons]. In the TNFi group, the main reason for treatment discontinuation was primary inefficacy [13 (46%) primary inefficacies, eight (29%) secondary inefficacies, six (21%) adverse events, and one (4%) other reasons]. On the contrary, in the non-TNFi group, six (22%) patients stopped treatment due to secondary inefficacy, five (18%) due to primary inefficacy, seven (26%) due to adverse events, three (11%) due to other causes, and six (22%) because of achieving remission (p = 0.035).
The overall incidence of adverse events was similar between the TNFi and the non-TNFi groups [8% (6/77) in the TNFi versus 14% (7/50) in the non-TNFi group (p = 0.37)].
Discussion
To our knowledge, this is the first observational study assessing, among TNFi-experienced patients, the comparative efficacy of a second TNFi versus an agent with a different mechanism of action 2 years after switching and taking into account whether the first-line bDMARD was discontinued due to a primary or secondary inefficacy. In our study, we observed that after failure to a first TNFi, regardless of the type of inefficacy (primary or secondary), a long-term EULAR response (24 months) was more frequently achieved after using a swapping (using non-TNFi) versus cycling (using a second TNFi) strategy.
The results of this study support the findings of most of the studies that have evaluated the efficacy of a second biological agent.4,8–15 The only head-to-head comparison of these randomized therapies published to date demonstrated greater response rates with RTX, ABA, and TCZ compared with TNFi. 12 Follow-up time, however, did not exceed the 12-month threshold, patients who switched due to intolerance were excluded, and whether the reason for switching influenced treatment response was not evaluated. This last limitation seems important, as other studies have suggested that the reason for discontinuation of a first TNFi could influence the efficacy of a second-line bDMARD.4–8 Due to the controversial evidence about this topic, the findings observed in our study are highly relevant, both in terms of duration of follow-up and demonstrating similar percentages of EULAR response to the second biologic regardless the type of inefficacy experienced with the first TNFi.
On the contrary, in a meta-analysis in which indirect pairwise comparisons were made between four placebo-controlled trials, no differences in the clinical responses of ABA, golimumab, RTX, and TCZ as a second biological therapy were found. 3 Nonetheless, only short-term efficacy was compared (14 weeks), the proportion of patients who switched due to each reason differed between samples, and sensitivity analyses based on this possible confounder were not performed. This last limitation was also observed in most observational studies that found evidence of biologics utilizing other mechanisms of action to be superior to a second TNFi.9–11,13–15 Moreover, mean follow-up remained below 12 months in most of them.
In our study, 6 months after starting the second bDMARD, 69% of patients attained EULAR clinical response and continued biological therapy. This response rate seems remarkably high compared with that reported in a Nordic cohort study, 21 in which 37% of patients remained on treatment and achieved low disease activity (LDA) or remission as measured by DAS28 after 6 months of follow-up. These differences could be explained because therapy response was measured using a different index in both studies. In our study, EULAR response (good and moderate) was the chosen tool to measure therapy response because it takes into account not only the DAS28 value but also the clinical improvement throughout the study. The study from Chatzidionysiou et al. highlights the difficulty of achieving remission or low activity in switchers in their second biological cycle. This should make us ask ourselves whether in patients with a previous failure to bDMARDs, mostly due to inefficacy, such a stringent therapeutical target is feasible.
Focusing on the efficacy of a second TNFi, we are only aware of one report that evaluated clinical response over a 24-month period. 22 In this RCT, the proportion of second TNFi responders who discontinued a previous TNFi due to an insufficient primary response was much lower than that observed in our cohort (52%), with only 15% of patients achieving DAS28 LDA 104 weeks after switching. Again, these differences may be explained by the fact that, in this interventional study, the criteria for primary ineffectiveness and response to treatment differed from ours; thus, the results are not comparable.
In our study, at 24 months after switching, differences supporting the swapping strategy were still evident in the subpopulation that discontinued TNFi treatment due to secondary inefficacy; similar findings were reported in a French meta-analysis of 19 reports, 23 which suggested that a second TNFi appeared to be less effective if the first TNFi was discontinued because of lack/loss of efficacy. Nevertheless, reports included in this meta-analysis pooled together patients who switched because of a primary or secondary inefficacy. On the other hand, there are also observational studies that yielded results opposite to our own, with better response rates to a second TNFi if the reason for switching was a secondary inefficacy.5,6 In these reports, however, comparisons were only assessed at 3 or 6 months after switching.
This study has strengths that are worth highlighting: first, the long-term follow-up during treatment with a second biologic therapy; second, the exclusion of patients with prior exposure to more than one TNFi or in whom the first biological agent was not a TNFi, since their inclusion could contribute to biases to this study; third, the real-world nature of our study makes the results more generalizable; and fourth, as Spanish health care is publicly funded, observer bias is minimized, as physicians can freely choose the bDMARD believed to be of most benefit. Indeed, treatment change decisions are made consensually by all the physicians in the service in a weekly clinical session. Finally, since our biological therapy unit protocol establishes the assessment of activity outcomes within a prespecified time window, and due to the imputation of nonresponders before the next studied point in each visit, incompleteness of data was minimized.
The results of this study should be interpreted in light of some limitations. First, both the observational design of the study and the longer follow-up time confer an unavoidably increased risk of missing data. Second, although multivariate analysis controlled for patient characteristics commonly found in medical record data and known to be prognostic for outcomes in patients with RA, nonrandomization of patients to treatment groups could have resulted in unobserved confounding factors and selection bias. Third, our study applies a longtime interval for patient inclusion, with patients starting second bDMARD when only TNFi was available. As management of RA has changed significantly over the last two decades (due to the use of the ‘treat to target’ strategy and the window of opportunity concept), calendar bias effect cannot be excluded. In order to minimize the impact of this potential confounding factor, stratified analysis by calendar effect was made. This, however, did result in a smaller sample size. Fourth, the limited sample size of the study (due to loss of patients who did not start a second biological agent or in whom clinical measurements were lacking) prevented us to distinguish between clinical responses according to the mechanism of action within the non-TNFi group and precluded comparisons between cycling versus swapping strategies in the subpopulation that discontinued due to causes other than ineffectiveness. Finally, although numerical differences in baseline characteristics could be observed between treatment arms, in small cohorts as ours there is the risk of not drawing statistically powerful conclusions.
Conclusion
In our cohort of RA patients with a previous failure to a first TNFi, response rates to a second biological were favorable, especially when the mechanism of action was changed (swapping strategy). This outperformance of agents with other mechanism of action over a second TNFi remained evident regardless of whether the reason for switching was loss or lack of efficacy.
The patients under a swapping strategy were three times more likely to attain a sustained long-term clinical response after switching a second biological therapy.
Future additional analyses validating these results in a larger cohort may be useful to confirm our findings and to better clarify which second bDMARD will benefit patients the most according their individual characteristics.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X211060910 – Supplemental material for Comparison of long-term efficacy between biological agents following tumor necrosis factor inhibitor failure in patients with rheumatoid arthritis: a prospective cohort study
Supplemental material, sj-docx-1-tab-10.1177_1759720X211060910 for Comparison of long-term efficacy between biological agents following tumor necrosis factor inhibitor failure in patients with rheumatoid arthritis: a prospective cohort study by Patricia Bogas, Chamaida Plasencia-Rodriguez, Victoria Navarro-Compán, Carolina Tornero, Marta Novella-Navarro, Laura Nuño, Ana Martínez-Feito, Borja Hernández-Breijo and Alejandro Balsa in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
We are grateful to the Spanish Society of Rheumatology (SER) English-language editing service, all of the rheumatologists and nurses at the Daycare Department for Biologicals, and the patients who participated in the registry.
Author contributions
PB, CP-R, VN-C, CT, MN-N and AB contributed to conceptualization and methodology. AM-F and BH-B contributed to laboratory assays and material preparation. CT and PB contributed to data collection and literature search. PB, CP-R, VN-C, and AB contributed to analysis and interpretation of data. The first draft of the manuscript was written by PB, CP-R, VN-C, MN-N, and AB and all authors commented on previous versions of the manuscript. All authors have read and agreed to the published version of the manuscript.
Conflict of interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AB has received research fees from AbbVie, Pfizer, Novartis, BMS, Nordic, and Sanofi; consultancy fees from AbbVie, Pfizer, Novartis, BMS, Nordic, Sanofi, Sandoz, and Lilly; and speakers bureau from Pfizer, Novartis, UCB, Nordic, Sanofi, Sandoz, and Lilly. VN-C has received speaker fees and grants from AbbVie, BMS, Lilly, MSD, Novartis, Pfizer, Roche, and UCB. The other authors declare that they have no competing interests.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study object of this article was supported by Sanofi Genzyme (Grant No. CAHMESMMXXX). The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Data availability statement
The data sets generated for this study are available on request to the corresponding author.
Ethics statement and informed consent
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Medical Ethics Committee of La Paz University Hospital (PI nº 1155, June 9, 2011). Written informed consent was obtained from all participants involved in the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
