Abstract
Introduction:
To assess the effect of baricitinib on patient-reported outcomes (PROs) in patients with moderately to severely active rheumatoid arthritis (RA) who had an inadequate response to methotrexate (MTX).
Methods:
This was a 52-week, randomized, double-blind, placebo controlled, phase III study in patients with RA who had an inadequate response to MTX. Patients (n = 290) receiving stable background MTX were randomly assigned (1:1) to receive placebo or baricitinib 4 mg once daily with a primary endpoint at week 12. PROs assessed included Health Assessment Questionnaire-Disability Index (HAQ-DI), Patient’s Global Assessment of Disease Activity, patient’s assessment of pain, Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F), European Quality of Life-5 Dimensions-5 Level index scores and visual analogue scale, and measures collected in electronic patient daily diaries: duration of morning joint stiffness, Worst Tiredness, and Worst Joint Pain. Treatment comparisons were made with logistic regression and analysis of covariance models for categorical and continuous variables, respectively.
Results:
Statistically significant (p ⩽ 0.05) improvements in all PROs were observed in the baricitinib 4 mg group compared to placebo as early as week 1 to week 4; and were sustained to week 24. These improvements were maintained until week 52 for the baricitinib group. A significantly larger proportion of patients met or exceeded the minimum clinically important difference for HAQ-DI (⩾0.22) and FACIT-F (3.56) profiles in the baricitinib group.
Conclusion:
Baricitinib provided significant improvements in PROs compared to placebo to 52 weeks of treatment in patients with RA who had an inadequate response to MTX.
Clinicaltrials.gov identifier: https://clinicaltrials.gov/ct2/show/NCT02265705; NCT02265705; RA-BALANCE. Registered 13 October 2014
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disorder that results in progressive joint damage, joint deformities, functional impairment, progressive disability, and deterioration of health-related quality of life with substantial socioeconomic burden.1–3 The global prevalence of RA was estimated to be 0.24% in 2010. 4 In 2013, the estimated prevalence of RA in China was reported to be 0.42%, affecting more than 5 million individuals. 5
Patient-reported outcomes (PROs) are the patient’s perception of the impact of disease and its treatment without interpretation of the patient’s response by a clinician. There is a growing interest in evaluation of PROs in chronic diseases such as RA,6,7 as PROs are often more meaningful than improvements in underlying disease activity when evaluating therapies.8,9 Recently released SPIRIT PRO extension guidelines indicate that improved clinical trial designs which include PROs can provide high quality data that evaluate patient-centered care. 10 The United States (US) Food and Drug Administration also released guidance on PRO measures to support labeling claims, 11 which resulted in a growing trend to evaluate PROs in clinical trials. Frequently evaluated PRO measures in the field of rheumatology include evaluation of physical function, pain, patient’s global assessment, health-related quality of life (HRQOL), fatigue, morning stiffness, 12 coping, sleep disturbances, and work and social life. 13 Evaluating different PRO measures provides a comprehensive understanding of the health evaluation of patients. The collection and use of PROs is becoming common practice in most western countries; however, utilization in Asia Pacific, Latin American, Middle Eastern, and African countries is not as prevalent14–17 due to factors such as difficulty in identifying appropriate PRO instruments based on cultural background, 17 and lack of knowledge pertaining to the importance of PRO measures. 15 As global trends in healthcare focus on patient-centered medicine, inclusion of PROs is highly warranted in all parts of the world, including Asia.
Baricitinib is a selective inhibitor of the Janus kinase (JAK) family of protein tyrosine kinases with potency and selectivity for JAK1 and JAK2 and less potency for JAK3 or tyrosine kinase 2. 18 Baricitinib19,20 is approved for the treatment of adult patients with moderately to severely active RA, who have had an inadequate response or who are intolerant to one or more tumor necrosis factor antagonist therapies 20 and/or disease-modifying antirheumatic drugs (DMARDs), 19 either as monotherapy or in combination with methotrexate (MTX) 20 or other DMARDs. 19 In the phase III RA-BALANCE clinical study, baricitinib 4 mg once daily was efficacious and safe in patients with moderately to severely active RA who had an inadequate response to MTX therapy. 21 This paper discusses the PROs collected in the RA-BALANCE study.
Materials and methods
Study design
RA-BALANCE was a randomized, multicenter, 52-week, double-blind, placebo controlled, phase III study that evaluated the efficacy of baricitinib compared to placebo in patients with moderately to severely active RA who had an insufficient response to MTX and had never been treated with a biological disease-modifying antirheumatic drug (bDMARD). The primary endpoint of the study was to determine the proportion of patients achieving an American College of Rheumatology criteria of 20% response at week 12 (baricitinib 4 mg versus placebo). More details about the study design, study treatment, and study endpoints of RA-BALANCE have been reported previously. 21
Patients
Major inclusion criteria included adults with moderately to severely active RA (high-sensitivity C-reactive protein of ⩾6 mg/L; ⩾6/68 tender joints; ⩾6/66 swollen joints) who had insufficient response to MTX therapy in the past. The patients received MTX therapy for ⩾12 weeks prior to study entry and were on a stable dose for 8 weeks (7.5 mg to 25 mg/week) prior to study entry. Major exclusion criteria included patients who had received previous biological therapies, a history of recent infection including active tuberculosis, untreated latent tuberculosis, or other serious infections, and immunocompromised patients. Eligible patients who met the criteria for participation in the study were randomly assigned to either baricitinib 4 mg once daily or placebo (1:1) according to a computer-generated random sequence using an interactive web response system.
Ethical considerations
The study was conducted in compliance with international ethical guidelines including the Declaration of Helsinki, Council for International Organizations of Medical Sciences, and Good Clinical Practice. The protocol and its amendments and informed consent form were approved by ethical review boards of 30 study centers in three countries (China, Argentina and Brazil). Written informed consent was obtained from every patient before study initiation. The RA-BALANCE study was registered with the clinicaltrials.gov registry (NCT02265705). The ethics committee approval number for the center of the corresponding author (Peking University People’s Hospital ethics committee) is 2014PHA010-01.
Assessment and schedule of PROs
The PROs presented in the paper include Health Assessment Questionnaire-Disability Index (HAQ-DI), Patient’s Global Assessment of Disease Activity (PtGA), patient’s assessment of pain, Worst Joint Pain, Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F), Worst Tiredness, European Quality of Life-5 Dimensions-5 Level (EQ-5D-5L), and duration of morning joint stiffness (MJS).
The patient’s physical function (disease-associated disability) was measured using the HAQ-DI.22–24 The HAQ-DI includes 24 questions assessing eight categories: dressing/grooming, arising, eating, walking, hygiene, reach, grip, and activities. Scores range from 0 to 3, with lower scores reflecting better physical function (less disability). Change from baseline in HAQ-DI scores and the proportion of patients who met or exceeded the HAQ-DI minimum clinically important difference (MCID) of ⩾0.22 were assessed during the study. The MCID threshold of ⩾0.22 is the commonly used threshold in RA trials for HAQ-DI.25,26
The PtGA measures disease activity while the patient’s assessment of pain measures pain associated with RA. These were assessed using a 0–100 mm visual analogue scale (VAS); higher scores indicate more pain or disease activity. Change from baseline in patient’s assessment of pain and PtGA, and the proportion of patients achieving an improvement of ⩾10 mm in pain score 27 were analyzed.
The FACIT-F scale 28 is a 13-item, symptom-specific questionnaire that specifically assesses the self-reported severity of fatigue and its impact on daily activities and functioning. FACIT-F has a range of 0–52, with higher scores representing less fatigue. The MCID for the FACIT-F score is defined as ⩾3.56 improvement (increase) from baseline. 29 The change from baseline in the FACIT-F score and the proportion of patients who met or exceeded the MCID were analyzed.
The EQ-5D-5L is a measure of general HRQOL. It consists of two components: a descriptive system of the respondent’s health (health index scores) and a rating of their current health state using a 0–100 mm VAS, where higher scores indicate a better health state.30–32 The United Kingdom (UK) and US scoring algorithms provide an index score, using the UK or US population weighting to normalize scores to that population. Change from baseline in index scores (using both the UK and US algorithms), and self-perceived health score were analyzed.
Duration of MJS, Worst Tiredness, and Worst Joint Pain collected through patient daily diaries have been reported previously; 21 PROs assessed at the study visits using an electronic PRO tablet are presented in this paper.
Duration of MJS was recorded as the length of time in minutes that the patient’s MJS lasted. Worst Tiredness and Worst Joint Pain were recorded using numeric rating scales (range 0–10, with 10 being the worst level).
HAQ-DI, patient’s assessment of pain, PtGA, duration of MJS, Worst Tiredness, and Worst Joint Pain were assessed at baseline, weeks 1, 2, 4, and every 4 weeks thereafter to week 32, and then at week 40 and week 52. The FACIT-F and EQ-5D-5L were assessed at baseline and at week 4. These measures followed the same schedules as the PROs after week 4.
Statistical analyses
Health outcome analyses were performed on the modified intent-to-treat (mITT) population, which included all randomly assigned patients who received at least one dose of the study drug. Missing data due to rescue or discontinuation from treatment were imputed using non-responder imputation for categorical variables or modified last observation carried forward for continuous variables. Treatment comparisons (baricitinib 4 mg versus placebo) of continuous PRO variables were made using analysis of covariance with treatment group, country, and baseline joint erosion status as fixed effects and baseline score as a covariate, with change from baseline as the dependent variable, or using a non-parametric method involving the Hodges–Lehmann estimator for duration of MJS. A logistic regression model was used with treatment, country, and baseline joint erosion status as the fixed effect. The Fisher exact test was used for comparison of categorical variables. The proportions and 95% confidence intervals (CIs) were reported for categorical variables and type III sums of squares for the least squares mean (LSM) and 95% CIs were reported for continuous variables. Unless otherwise stated, all analyses were based on a significance level of 0.05 (two-sided), and no multiple comparison adjustment was conducted on the results. All statistical analyses were conducted using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
Patient disposition and baseline characteristics
A total of 290 patients were randomly assigned to baricitinib 4 mg or placebo (145 patients to each group). Of the randomized patients, a total of 269 patients entered the open-label phase, of whom 260 patients (89.7%) completed 52 weeks of the study. A detailed patient disposition is presented in Li et al. 21
The overall mean (standard deviation; SD) age was 49.2 (11.7) years and the mean (SD) body mass index at baseline was 23.6 (5.0) kg/m2. The majority of patients were women (80.3%), Asian (79.7%), and <65 years of age (92.8%). 21 The mean (SD) duration of RA was 9.9 (7.7) years. The majority of patients reported daily usage of corticosteroids (57.9%) with a mean (SD) daily dose of 7.4 (2.9) mg. The mean (SD) weekly dose of MTX was 12.5 (4.9) mg/week. Baseline patient demographics and disease characteristics were similar between the treatment groups. 21 Baseline PRO measures were similar between the groups and indicated a significant disease burden, consistent with baseline disease characteristics (Table 1). 21
Change from baseline at weeks 12, 24, and 52 for patient-reported outcomes.

Data presented at baseline are mean (SD) while data presented at week 12, week 24, and week 52 are LSM (95% CI) change from baseline using mLOCF in the mITT population unless stated otherwise.
Lower scores on HAQ-DI reflect better physical functioning (less disability).
Lower scores on VAS (patient’s assessment of pain and PtGA) indicate less pain/disease activity.
Higher scores on FACIT-F indicate less fatigue.
Higher scores on EQ-5D-5L (index or VAS) indicate a better perception of health.
p⩽0.01, ***p⩽0.001 versus placebo; p-values are based on ANCOVA model for all variables.
ANCOVA, analysis of covariance; CI, confidence interval; EQ-5D-5L, European Quality of Life-5 Dimensions-5 Level; FACIT-F, Functional Assessment of Chronic Illness Therapy-Fatigue; HAQ-DI, Health Assessment Questionnaire-Disability Index; LSM, least squares mean; mITT, modified intent-to-treat; mLOCF, modified last observation carried forward; N, number of mITT patients; PRO, patient-reported outcomes; PtGA, Patient’s Global Assessment of Disease Activity; SD, standard deviation; UK, United Kingdom; US, Unites States of America; VAS, visual analogue scale.
Patient-reported outcomes
HAQ-DI and patient’s global assessment of disease activity
A significantly greater improvement in HAQ-DI 21 and PtGA were observed at weeks 12 and 24 for the baricitinib 4 mg group when compared to the placebo group. A statistically significant improvement from baseline was evident as early as week 1 for PtGA and week 2 for HAQ-DI. The improvements in HAQ-DI and PtGA were maintained from week 24 to week 52 for the baricitinib 4-mg group [Figure 1(a), Figure 2, and Table 1].

(a) Change from baseline in HAQ-DI.

Change from baseline in patient’s global assessment of disease activity.
The proportion of patients who reported scores that met or exceeded the HAQ-DI MCID (⩾0.22) for placebo and baricitinib 4 mg, respectively, at week 12 were 61.4% and 70.3% and at week 24 were 35.2% and 64.8% (p⩽0.001). At week 52, the proportion of patients who reported scores that met or exceeded the HAQ-DI MCID (⩾0.22) for baricitinib 4 mg was 61.4% [Figure 1(b)].
Patient’s assessment of pain and Worst Joint Pain
A significantly greater improvement from baseline in patient’s assessment of pain and Worst Joint Pain was observed at weeks 12 and 24 for the baricitinib 4 mg group when compared to the placebo group. A statistically significant improvement from baseline was evident as early as week 1 for patient’s assessment of pain and week 2 for Worst Joint Pain. The improvements in patient’s assessment of pain and Worst Joint Pain were maintained from week 24 to week 52 for the baricitinib 4 mg group [Figure 3(a) and (b)].

(a) Change from baseline in patient’s assessment of pain.
There was a statistically significant difference between baricitinib 4 mg and placebo groups in the proportion of patients who experienced an improvement in pain of ⩾10 mm at week 12 when patient’s assessment of pain was assessed using VAS. This statistically significant difference was noted as early as week 2 and was maintained to week 24 except for week 8. The improvements with baricitinib were maintained to week 52 [Figure 3(c)].
FACIT-F and Worst Tiredness
A statistically significant improvement in FACIT-F from baseline was observed at weeks 12 and 24 for the baricitinib 4 mg group compared to placebo. A significantly greater improvement was first observed at week 4 for the baricitinib 4 mg group and maintained to week 24. Improvements in FACIT-F were maintained for the baricitinib 4 mg group to weeks 24 and 52 [Figure 4(a)].

(a) Change from baseline in FACIT-F.
A significantly greater percentage of patients reported scores that met or exceeded the MCID for FACIT-F (⩾3.56) at weeks 12 and 24 in the baricitinib 4 mg group compared to the placebo group (64.8% versus 46.2%, p ⩽ 0.01 at week 12 and 64.1% versus 26.2%, p ⩽ 0.001 at week 24). At week 52, the proportion of patients who reported scores that met or exceeded the FACIT-F (⩾3.56) for baricitinib 4 mg was 80.6% [Figure 4(a)].
A statistically significant improvement in Worst Tiredness was observed at weeks 12 and 24 for the baricitinib group compared to the placebo group. The improvement was statistically significant as early as week 2 for Worst Tiredness. The improvement with baricitinib was maintained to week 24 and week 52 [Figure 4(b)].
European Quality of Life-5 Dimensions-5 Level
A significantly greater improvement from baseline health state index scores of EQ-5D-5L (both US and UK) was observed at weeks 12 and 24 for the baricitinib 4 mg group when compared to the placebo group. Compared to placebo, significant improvements from baseline in EQ-5D-5L VAS were observed as early as week 4 for the baricitinib 4 mg group and were maintained to week 24 (except at week 12). The improvements with baricitinib were maintained to week 52 (Table 1).
Duration of morning joint stiffness
A statistically significant improvement in the median duration of MJS was observed at weeks 12 and 24 for the baricitinib 4 mg treatment group compared to the placebo group. The improvement recorded was statistically significant as early as week 1 for the duration of MJS. Improvements were maintained to week 24 and week 52 for the baricitinib 4 mg group (Figure 5).
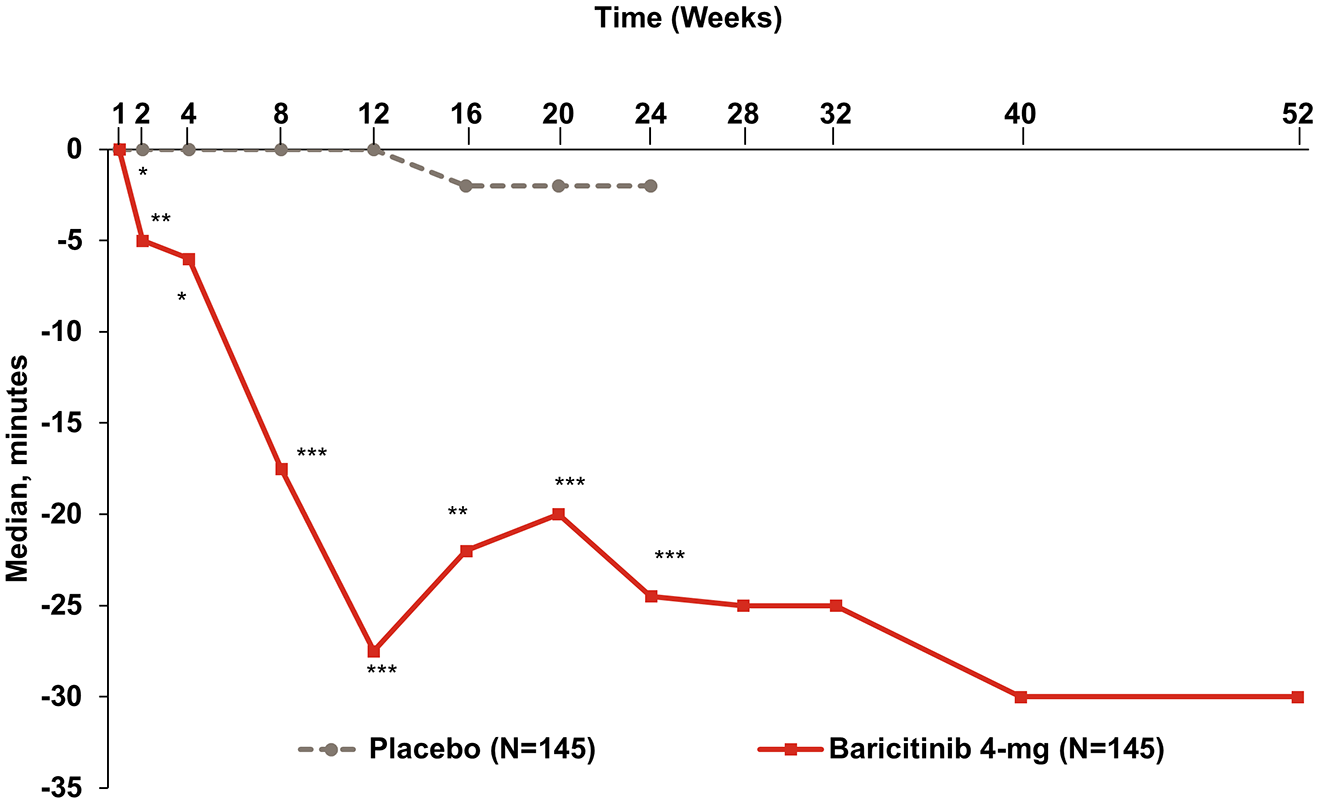
Change from baseline in duration of morning joint stiffness.
Discussion
The RA-BALANCE trial evaluated the efficacy and safety of baricitinib 4 mg once daily in adult patients with moderately to severely active RA who had an inadequate response to MTX and who had never been treated with a bDMARD. 21 The present paper discusses the PROs that evaluated physical function, general health, pain, fatigue, and MJS in the RA-BALANCE trial, which, in conjunction with clinical and radiographic assessments, can serve as important decision making tools in the management of RA.
The present study used well-established PRO measures, some of which are selected from the core set of outcome measures defined by the American College of Rheumatology, the European League Against Rheumatism, and Outcome Measures in Rheumatology33,34 to evaluate patient symptoms thoroughly.
The present analyses show that baricitinib 4 mg produced a significant improvement in PROs consistent with other global phase III studies.35–38 Baricitinib also showed significant improvements in PROs as seen with other JAK inhibitors such as tofacitinib39–42 and peficitinib. 43 The improvements in PROs noted in this study are numerically similar to those reported in the RA-BEAM study at the end of week 12 and week 52. 36 The present analysis showed a numerical improvement in PROs in patients who had an inadequate response to MTX treated with baricitinib (LSM change from baseline scores for HAQ-DI −0.67; patient’s assessment of pain −31.0; PtGA −31.3; and FACIT-F 9.2), compared to those treated with tofacitinib 10 mg twice daily in the ORAL Scan trial 42 after 3 months of treatment (LSM change from baseline scores for HAQ-DI −0.54; patient’s assessment of pain −28.04; PtGA −27.66; and FACIT-F 6.28). The present results (Table 1) also show a numerical improvement when compared to results at the end of 3 and 6 months from the ORAL Sync study in Chinese patients with RA who had inadequate response to conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) or bDMARDs (HAQ-DI, PtGA, patient’s assessment of pain, and FACIT-F scores were −0.48, −24.0, −20.2, and 3.6, respectively, at 3 months and −0.55, −26.1, −24.8, and 4.0, respectively, at 6 months with tofacitinib 10 mg twice daily). 44 The improvements in pain, PtGA, and HAQ-DI (−34.8, -33.4, −0.72, respectively) with baricitinib 4 mg at the end of 24 weeks of treatment were also numerically higher when compared to the results reported in a systematic review that included anti-tumor necrosis factor (−17.9, −19.1, −0.30), abatacept (−23.0, −13.6, −0.21), and tocilizumab (−16.0, −15.1, −0.27) in combination with MTX in patients with RA who had an inadequate response to csDMARDs. 45
The patients recruited in RA-BALANCE had significant disease burden, therefore improvement in PROs in the early stages of treatment was important. Following treatment with baricitinib, improvements in PROs were noted as early as week 1 for patient’s assessment of pain, PtGA, and duration of MJS; week 2 for HAQ-DI, Worst Tiredness, and Worst Joint Pain; and week 4 for FACIT-F and EQ-5D-5L VAS. These improvements were maintained to week 52. These results support the rapid onset of action, as well as the durability of response, of baricitinib 4 mg. This paper demonstrates that the clinical efficacy data for baricitinib seen in the RA-BALANCE study are complemented by corresponding changes in the PRO data. 21
Glucocorticoids are highly beneficial in reducing pain and overcoming the circadian increase of night inflammation due to inadequate cortisol secretion in active disease; 46 however, they do come with a number of long-term side effects. One advantage of the reduction in the duration of MJS with baricitinib as early as week 1 might be in helping wean patients from glucocorticoids at an early stage. In addition, a recent post hoc analysis 47 showed the pain relief effect demonstrated by baricitinib to be independent of the inflammatory process due to antinociceptive effects of JAK1 and JAK2 inhibition. 48 Patients who report marked pain relief are likely to report greater improvements in other PRO measures, as seen in the current analyses.
Current guidelines for the management of RA recommend a treat-to-target approach to achieve higher remission rates and lower disease activity; whereas from the patient’s perspective, improvements in PROs such as fatigue, pain, and quality of life can be more important. In addition, improvements in disease measures have not always had a parallel improvement in PROs, indicating the importance of focusing on PROs along with disease activity measures. Adding PRO assessments to the existing core set of treatment targets helps in addressing the needs of patients.49,50 In the present study, the improvements seen in PRO measures are similar to the improvement observed with efficacy data. 21 As PROs show reliable results in clinical research, it is time to incorporate them into daily clinical practice. The effective and timely use of PROs helps physicians and patients share decision making when evaluating a treatment regimen (determining the treatment plan and duration of treatment), in conjunction with clinical assessment. In a broader perspective, the inclusion of PRO measures in clinical trials helps in the comparison of treatment effects from the patient’s perspective.
There are limitations to be considered in these analyses. Firstly, although we used standard instruments with proved validity and reliability, responses may vary slightly due to ethnic differences in patient populations. Secondly, the results should be interpreted cautiously as the studied population in a controlled environment may be different from the population treated in clinical practice. Thirdly, the use of carrying forward the last observations before rescue or discontinuation as a method of imputation assumes that the PROs do not change over time.
Conclusion
The RA-BALANCE study demonstrated that treatment with baricitinib 4 mg provided a significant improvement compared to placebo in most PROs that evaluated physical function, general health, pain, fatigue, and joint stiffness. Improvements occurred rapidly within 1–4 weeks of initiation of treatment and were maintained to week 52. The efficacy of baricitinib 4 mg as assessed by PROs was shown to have a positive benefit on patients’ quality of life and overall function, suggesting that patients with RA will benefit from treatment with baricitinib 4 mg.
Footnotes
Acknowledgements
The authors would like to thank all study investigators and participants who participated in the study. Eli Lilly and Company contracted Syneos Health (formerly INC Research/inVentiv Health Clinical) for writing and editorial support. The authors would like to thank Pavan Yenduri, Barbara Jackson, and Antonia Baldo (Syneos Health, formerly INC Research/inVentiv Health Clinical) for their assistance in writing, editing, and proofreading the paper, respectively.
Author contributions
Carol L Kannowski and Mengru Liu were involved in interpretation of the data for the work. Hanjun Wu was involved in the conception, design, and drafting of the work and, interpretation of the data for the work. Fei Ji was involved in analysis of the data and interpretation of the data for the work. Lujing Zhan was involved in the conception and design of the work. Zhanguo Li was involved in the design of the work, acquisition of the data for the work and interpretation of the data for the work. All other authors were involved in the acquisition of the data for the work and interpretation of data for the work. All authors were involved in critical revision of the manuscript for important intellectual content, take complete responsibility for the integrity and accuracy of the data, and have given final approval for the version to be published. The present original article has been prepared for Therapeutic Advances in Musculoskeletal Disease and has not been submitted or accepted for publication elsewhere except as a brief abstract in the proceedings of a scientific meeting or symposium.
Conflict of interest statement
Yue Yang, Jianhua Xu, Jian Xu, Xingfu Li, Jiankang Hu, Xiangpei Li, Xiao Zhang, Dongyi He, Zhijun Li, Alberto J Spindler and Zhanguo Li have nothing to disclose. Chunde Bao and Guochun Wang serve as advisory board members for baricitinib and have received consulting and/or speaking fees from Eli Lilly and Company (<$10,000 USD). Cristiano AF Zerbini reports grants from Eli Lilly and Company, Pfizer, Sanofi, and AbbVie outside the submitted work. Carol L Kannowski is a full-time employee of Eli Lilly and Company, Indianapolis, USA; reports personal fees from Eli Lilly and Company outside the submitted work; and is a stockholder in Eli Lilly and Company. The authors Lujing Zhan and Mengru Liu are full-time employees of Lilly Suzhou Pharmaceutical Co. Ltd., Shanghai, China. The authors Hanjun Wu and Fei Ji are full-time employees of Lilly Suzhou Pharmaceutical Co. Ltd., Shanghai, China and own stock in Eli Lilly and Company.
Data sharing and data accessibility
Eli Lilly and Company provides access to all individual participant data collected during the trial, after anonymization, with the exception of pharmacokinetic or genetic data. Data are available to request 6 months after the indication studied has been approved in the US and EU and after primary publication acceptance, whichever is later. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, blank or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at ![]() .
.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Eli Lilly and Company, which was responsible for the design, conduct, and monitoring of the study along with publication of this article.
