Abstract
Background:
Evidence shows that gout attack is closely related to immune and inflammatory levels. However, there are limited evidence-based results for immune-inflammatory indices to predict gout attacks.
Objectives:
This study aimed to explore the predictive value of immune-inflammation index for acute gout.
Design:
Systematic review and meta-analysis.
Data sources and methods:
A systematic search was conducted based on PubMed, Web of Science, Embase, Cochrane, CNKI, and Wanfang. Literature screening and data extraction were performed independently by two investigators, and standardized mean difference (SMD) and 95% confidence interval (95% CI) were used to evaluate the differences in inflammatory indexes between gout patients and individuals in the healthy control group. A random-effects model was used to analyze the predictive value of different inflammatory indexes for acute gout patients. In addition, sensitivity analysis and subgroup analysis were also conducted to explore the sources of heterogeneity.
Results:
A total of 8 articles involving 2655 participants were included in this meta-analysis. The findings showed that both neutrophil-to-lymphocyte ratio (NLR; SMD: 0.75; 95% CI: 0.48–1.03) and platelet-lymphocyte ratio (PLR; SMD: 0.52; 95% CI: 0.24–0.79) showed significant value in predicting acute gout attacks. The sensitivity analysis results showed that both NLR and PLR were significantly stable in predicting acute gout attacks. Meanwhile, subgroup analyses were performed based on region, sample size, and age. The results showed that the predictive value of NLR and PLR for acute gout was significant in all subgroups, and age and sample size were potential sources of heterogeneity in PLR. However, no sources of heterogeneity were found in NLR.
Conclusion:
NLR and PLR are well-performed in predicting acute gout attacks. Given a lack of prospective studies, a limited number of related references, and significant heterogeneity, large-scale prospective studies are needed to verify the relationship between immune-inflammation index and gout attacks.
Introduction
Gouty arthritis (GA) is a metabolic rheumatic disease caused by the deposition of monosodium urate (MSU) crystals within the joints and surrounding tissues. It is generally characterized by recurrent joint redness, swelling, heat, pain, and tissue damage. 1 The burden of gout is increasing globally, with significant differences in incidence and prevalence rates among different genders and age groups. The incidence of gout in men is more than twice that in women, and it increases significantly with age, especially in older men.2,3 The common pathogenesis of GA is hyperuricemia, 4 which leads to the chronic deposition of MSU crystals. These crystals, in turn, activate the innate immune system within and surrounding the affected joint, and cause acute gout through the synergy of multiple inflammatory cells and cytokines.5,6 The clinical course of GA is typically intermittent, characterized by recurrent flares of inflammatory arthritis and asymptomatic intercritical periods. Clinically, drugs such as colchicine, nonsteroidal anti-inflammatory drugs, and glucocorticoids are often used to control acute inflammatory responses, but such drugs can easily cause adverse reactions. 7 The timely diagnosis and treatment of gout is important in clinical practice.
Current methods for predicting GA are less cost-effective and accurate. Hence, serum inflammatory factors such as interleukin 8 (IL-8) are rarely included in routine tests. 8 The commonly used detection indicators, C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR), are at normal levels in some patients with acute GA, 9 while they are elevated in some patients during asymptomatic intercritical periods. Therefore, relying solely on these indicators is insufficient for predicting the severity of gout. Additionally, neutrophils and lymphocytes play important roles in the inflammatory response, but a single-cell indicator is easily affected by dehydration or rehydration, blood sample processing, and blood sample dilution.10–12 Inflammatory indexes, such as neutrophil-to-lymphocyte ratio (NLR), are relatively stable and accurate in predicting diseases. Common inflammatory indexes, such as NLR, platelet-lymphocyte ratio (PLR), lymphocyte-to-monocyte ratio (LMR), and CRP, play an important role in predicting gout. For example, Huang et al. 9 have demonstrated that the levels of NLR and PLR in patients with acute GA are significantly higher than those in the healthy control group. Additionally, Pang et al. 13 have found that the levels of NLR and other indexes significantly decrease in patients with GA after treatment. 13 Therefore, inflammatory indexes have certain predictive value.
Currently, most studies on inflammatory indexes for predicting gout are clinical studies. There is no evidence-based medicine research to provide a theoretical basis for improving clinical evaluation models. Therefore, the purpose of this study is to explore the predictive performance of immune-inflammation index for acute gout, providing theoretical support for further clinical research and the establishment of clinical gout prediction models.
Methods
Search strategy
Relevant studies on immune-inflammation indicators were searched in PubMed, Web of Science, Embase, Cochrane, CNKI, Wanfang, and other databases up to January 2025. The subject words applied were “lymphocytes,” “ratio,” and “gout.” The search strategy was designed by combining subject headings and free words. Details of the search strategy are presented in Table S1.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) the subjects were divided into acute gout group and healthy control group based on the severity of gout attacks, and a precise range of inflammatory indexes was provided in the study; (2) NLR or PLR, odds ratio, relative risk, or hazard ratio for predicting gout attacks, as well as 95% confidence interval (CI) were provided, or these indexes could be calculated from other data in the study. In contrast, conference abstracts, animal experiments, reviews, letters, comments, and studies that did not provide sufficient data were excluded.
Data extraction
The data were independently extracted from all eligible studies by two researchers (Z.G. and M.Z.) based on the inclusion criteria described above. Any disagreements were resolved through discussions with a third person (Y.Y.). The data extracted from the study included author, year of publication, nationality, observation period, sample size, age, body mass index, study type, study indicators, gender, uric acid level, risk of gout attacks, and 95% CI.
Quality assessment
The quality of the included studies was evaluated using the Newcastle-Ottawa Scale (NOS). The NOS scores ranged from 0 to 9. Higher scores indicated a higher quality of a study. The evaluation domains of NOS mainly included the selection of subjects, comparability between groups, and exposure control. Two authors independently assessed the quality of all included studies, and any disagreement was settled by discussion.
Statistical analysis
Standardized mean difference (SMD) and 95% CI were used to evaluate the differences in inflammatory indexes between gout patients and healthy controls. I2 was used to quantify heterogeneity. I2 < 25%, 25% ⩽ I2 < 75%, and I2 ⩾ 75% represented low, moderate, and high heterogeneity, respectively.14,15 If no significant heterogeneity was found, a fixed-effect model would be adopted. Otherwise, a random-effects model was used to pool data. In addition, if there was significant heterogeneity among the studies, subgroup analysis was conducted to explore the sources of heterogeneity. Sensitivity analysis was conducted to test the stability of the overall results. Review Manager V.5.4.1 was used to generate a funnel plot to test potential publication bias. If the funnel plot was symmetric, there was no publication bias. In addition, Egger’s test was also conducted using Stata 15.0 software to determine the presence of significant publication biases in various indicators.16,17 All analyses were performed using Review Manager V.5.4.1 (Revman; the Cochrane Collaboration, London, UK) or STATA V.15.0 (StataCorp LP, College Station, TX, USA).
Results
Characteristics of the results included in the study
We searched 179 potentially relevant articles, of which 152 were excluded. Among them, 82 articles only focused on either inflammatory indexes or acute gout, without exploring the relationship between the two. In addition, 12 conference papers, 3 reviews, and 55 duplicates were removed. The remaining 27 articles were further screened, among which 2 studies were excluded due to inconsistent language and 17 studies were excluded due to no necessary data. Finally, a total of eight articles9,13,18–23 (Figure 1) were included in this meta-analysis. Specifically, a total of 13 comparison groups were extracted. Characteristics of the included studies are listed in Table 1. All gout patients were diagnosed according to the classification criteria for gout established by the 2015 American College of Rheumatology and the European Alliance of Associations for Rheumatology. 24 The NOS scores of the included studies were 7–9 points (Table 2).

Literature search flow.
Baseline characteristics of include studies.
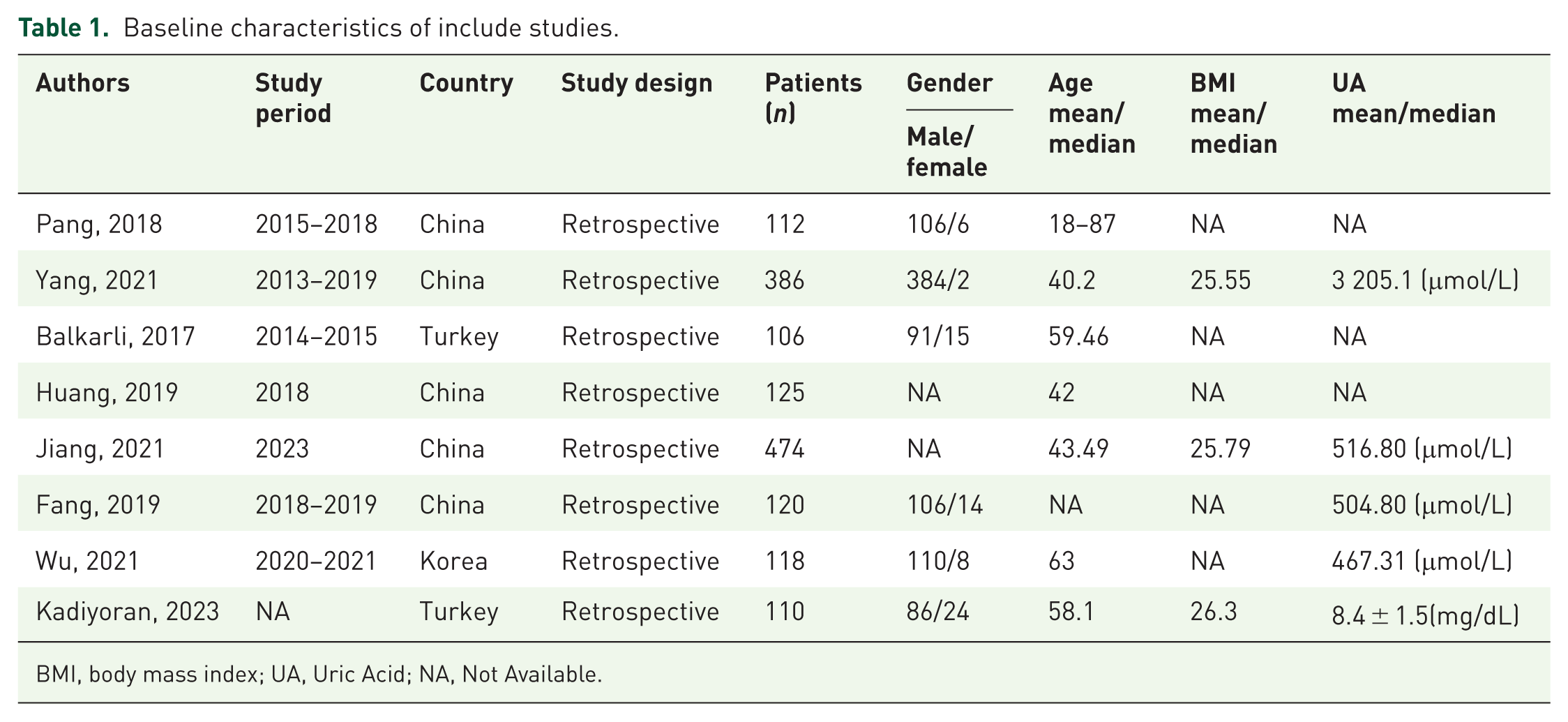
BMI, body mass index; UA, Uric Acid; NA, Not Available.
Quality evaluation of the eligible studies with Newcastle–Ottawa scale.

– Indicates significant of criterion not met.
Indicates criterion met.
Value of NLR in predicting gout attacks
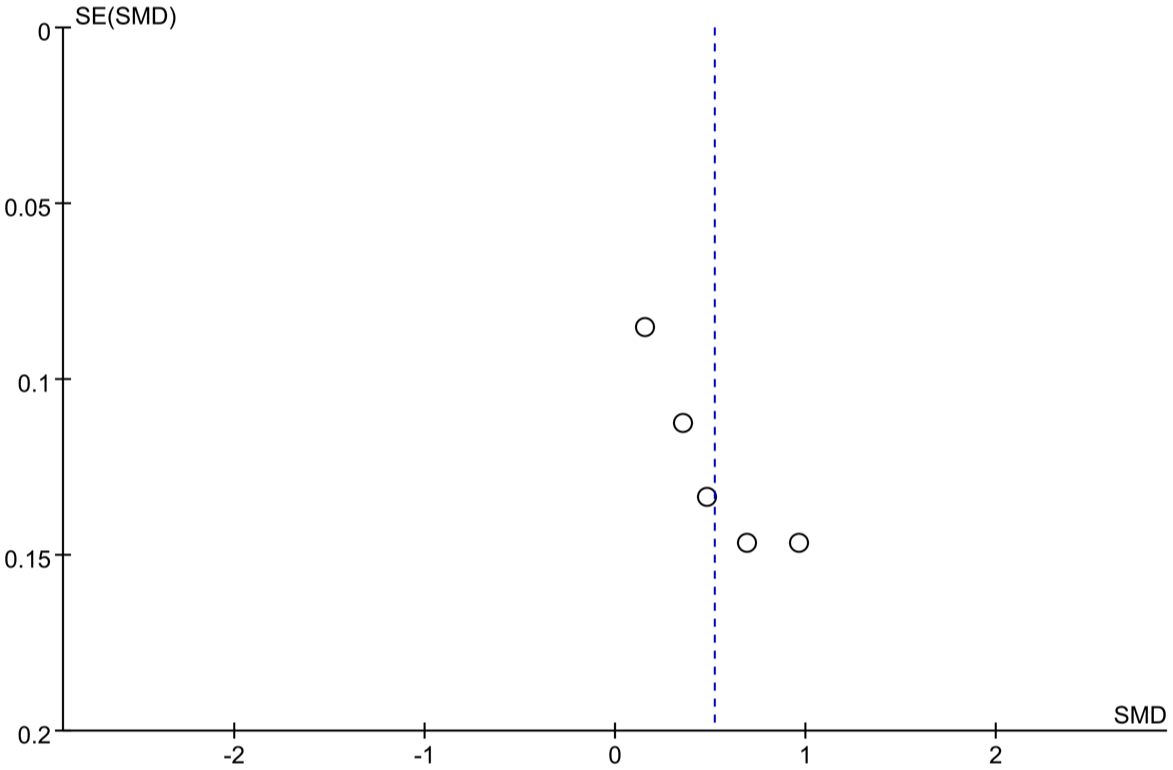
The data related to NLR levels were obtained from eight comparison groups.9,13,18–23 The meta-analysis results showed that the levels of NLR in individuals with acute gout were significantly higher than those in the control group (SMD: 0.75; 95% CI: 0.48–1.03, p < 0.0001, I2 = 90, Figure 2), suggesting that elevated NLR levels had significant value in predicting acute gout attacks. The funnel plot was predominantly symmetric (Figure 3), and the Egger’s test results showed no publication bias (p > 0.275).
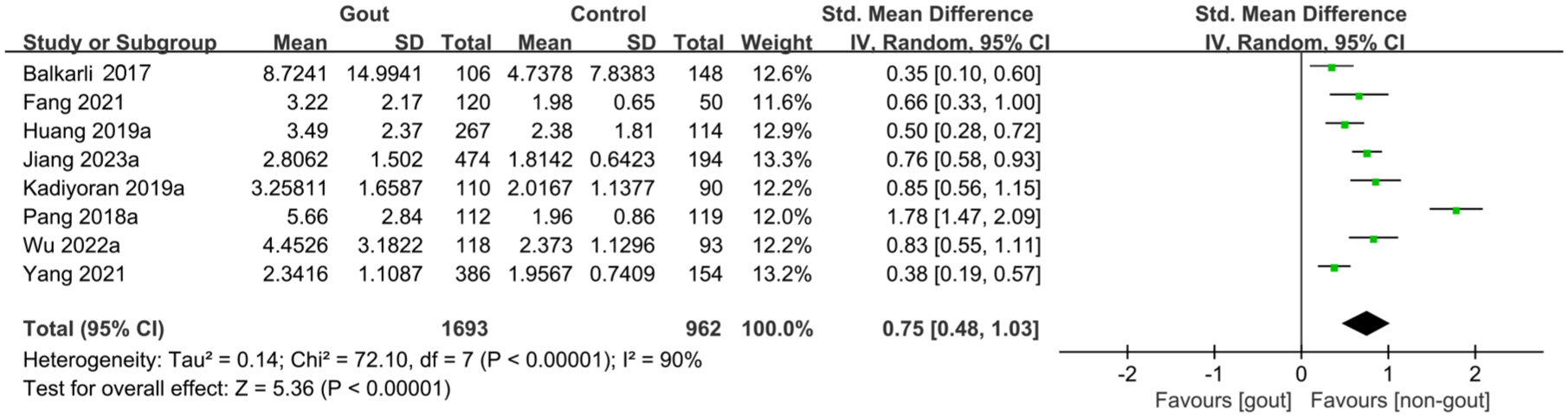
Meta-analysis of the predictive value of NLR.

Funnel plot of the predictive value of NLR in the gout group and the control group.
Value of PLR in predicting gout attacks
The data related to PLR levels were obtained from five comparison groups.9,20–23 The meta-analysis results showed that the levels of PLR in individuals with acute gout were significantly higher than those in the control group (SMD: 0.52; 95% CI: 0.24–0.79, p < 0.0001, I2 = 85%, Figure 4), suggesting that elevated PLR levels had significant value in predicting acute gout attacks. The funnel plot was predominantly symmetric (Figure 5), while the Egger’s test results showed a possibility of publication bias (p < 0.05). This may be attributed to variations in data screening conducted by the investigators and diagnostic criteria.

Meta-analysis of the predictive value of PLR.
Funnel plot of the predictive value of PLR in the gout group and the control group.
Sensitivity analysis
Sensitivity analysis was conducted to observe whether individual studies affected the overall results. The results showed that after excluding any study, both NLR and PLR had significant value in predicting gout attacks, indicating that both indexes were stable predictors (Figures 6 and 7).

Sensitivity analysis of the predictive value of NLR.

Sensitivity analysis of the predictive value of PLR.
Subgroup analysis
Subgroup analyses were performed based on regions (Asia vs Europe), age (older vs younger), and sample size (Table 3). The results showed that NLR and PLR showed significant predictive value across all groups: Asians, Europeans, older adults, younger individuals, and studies with large and small sample sizes. Meanwhile, the analysis results suggested that no sources of heterogeneity were found for NLR. It was speculated that the differences may arise from variations in diagnostic criteria, testing methods, and the observed populations. The results of subgroup analysis suggested that the primary sources of heterogeneity for PLR were age and sample size, with an I2 value of less than 50% in both the older and younger groups.
Subgroup analysis of NLR/PLR in the gout attack group and the control group.

CI, confidence interval; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SMD, standardized mean difference.
Discussion
The meta-analysis results in this study suggested that NLR and PLR could effectively predict acute gout attacks, and both were positively correlated with the risk of gout attacks. Meanwhile, subgroup analysis was performed on the eight included articles. The results showed that the predictive value of NLR and PLR for gout attacks remained stable in all subgroups, further confirming their significant predictive effect. However, no sources of heterogeneity were found for NLR. It was speculated that the heterogeneity may stem from variations in study population selection and data selection. Regarding PLR, we found that its primary sources of heterogeneity were age and sample size, with I2 of less than 50% in younger, older, and large sample size groups. It is worth noting that NLR and PLR are often used as inflammatory markers to predict the prognosis of diseases such as stroke and traumatic brain injury. Gut microbiota affects nerve damage through immune regulation and metabolites (such as Short-Chain Fatty Acids (SCFAs)). NLR and PLR may serve as a potential tool for quantifying inflammation, indirectly associated with peripheral immune activation induced by gut microbiota dysbiosis. 25 Furthermore, elevated inflammation can increase susceptibility to infection following nerve injury. Therefore, NLR and PLR may provide guidance for the use of antibiotics in gout and hyperuricemia-related neurological diseases. 26
In our study, the funnel plot for NLR was symmetric, and the results from the Egger’s test showed no publication bias. The funnel plot for PLR was predominantly symmetric, but the Egger’s test results indicated the possibility of publication bias. This may be attributed to differences in the diagnostic criteria and data processing.
The conclusions of this study are consistent with previous clinical data. Retrospective studies have shown that inflammatory indexes such as NLR and PLR have certain value in predicting gout. For example, Yang et al. 19 reveal that NLR is a predictive factor of the development of gout, and it has important auxiliary diagnostic value for gout and different fractional excretion of uric acid stages. Kadiyoran et al. 23 have found that the values of NLR are higher in patients without gout attacks than in the control group, and gout patients without attacks still have inflammation. Elevated NLR and PLR values during acute attacks indicate aggravated inflammation. LMR, red cell distribution width, and NLR may be potent predictive markers for gout attacks and systemic inflammation.
The inflammatory indicators derived from complete blood count (CBC) are new nonspecific inflammatory indexes, primarily including NLR and PLR. CBC-derived indicators are more stable than lymphocytes, neutrophils, platelets, CRP, ESR, and other inflammatory indexes, and are less susceptible to the influence of blood sample processing, blood sample dilution, dehydration or rehydration, and other factors.10–12 This could enhance the accuracy of disease predictions. CBC has been used to predict various immune diseases, such as rheumatoid arthritis and systemic lupus erythematosus. 27 In the gout pathway, it has been proven to indirectly affect the development of gout. Some studies have found that 28 hyperuricemia causes the deposition of MSU crystals in blood vessels, cartilage, and synovium of joints. Moreover, MSU crystals directly react with monocytes and lymphocytes, increasing monocyte activity and promoting neutrophil chemotaxis and explosive cascade expansion reactions. Subsequently, many recruited and activated neutrophils promote the release of a large number of inflammatory mediators, such as IL-1β and IL-6, ultimately triggering an inflammatory response. Additionally, neutrophils can enhance the interaction between platelets and vascular endothelium, increasing blood viscosity. This may lead to platelet adhesion to endothelial cells and attracts other white blood cells to the site for adhesion, thereby promoting an inflammatory response.28–34
This study found that alterations in the body’s immune inflammatory levels play a crucial role in the occurrence and development of gout. NLR and PLR, as systemic immune inflammatory biomarkers derived from complete blood cell counts, can indirectly reflect the potential pathophysiological link between GA. Gout is attributable to the deposition of urate crystals in the joints and surrounding tissues due to hyperuricemia, which subsequently activates the local innate immune system, promoting neutrophil chemotaxis and infiltration, platelet activation, and the release of inflammatory factors.35–37 This immune-inflammatory process is not limited to the joint area but may also cause changes in the systemic immune status. In this case, changes in NLR and PLR levels may serve as indicators of the degree of inflammatory activation. Simultaneously, immune regulatory factors such as gut microbiota dysbiosis may further influence the occurrence of gout and the risk of complications, such as related neuroinflammation by regulating peripheral immunity. 25 This suggests that NLR and PLR are not only inflammatory markers but may also serve as indirect indicators reflecting immune-metabolic axis dysregulation. The findings of this study provide a theoretical basis for introducing easily accessible inflammatory markers such as NLR and PLR into clinical assessment models of gout, and also provide a reference for further research into the mechanisms linking gout and systemic immune inflammation.
Limitation
This study also has some limitations. First, the majority of studies included in this analysis originated from Asian countries, primarily China, followed by Turkey. Data from African populations were notably absent. Incomplete data may affect the reliability of this study. Second, the included studies mostly employed continuous variables and rarely used categorical variables. Third, the included articles consisted primarily of retrospective studies, with no prospective studies. Fourth, no sources of heterogeneity for NLR were detected in this analysis. Potential sources such as differences in diagnostic criteria, testing methods, and the observed populations may have contributed to heterogeneity. However, this study also has strengths. We conducted an evidence-based medicine study for the first time on the relationship between immune-inflammation index and gout attacks. The results further indicated the correlation of NLR and PLR with gout attacks, and also provided theoretical support for further clinical research and the establishment of clinical gout prediction models. Additionally, clinical doctors should focus on inflammatory indexes such as NLR and PLR in the diagnosis and treatment of gout patients.
Conclusion
In conclusion, the study demonstrates for the first time that NLR and PLR appear to be effective predictive indexes for patients with acute gout. The observed association between lymphocyte-based inflammatory indices and gout attacks should be interpreted cautiously, given the predominance of retrospective studies included and the observed heterogeneity of NLR. Larger, prospective, multiracial studies with standardized staging and treatment are needed to confirm these findings.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251414413 – Supplemental material for Association between immune-inflammation index and the risk of gout attack: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tab-10.1177_1759720X251414413 for Association between immune-inflammation index and the risk of gout attack: a systematic review and meta-analysis by Zhiwei Guan, Minjun Zhang and Ye Yuan in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251414413 – Supplemental material for Association between immune-inflammation index and the risk of gout attack: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tab-10.1177_1759720X251414413 for Association between immune-inflammation index and the risk of gout attack: a systematic review and meta-analysis by Zhiwei Guan, Minjun Zhang and Ye Yuan in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
