Abstract
Left ventricular thrombus is one of the major complications of dilated cardiomyopathy. Although the presence of a cardiac thrombus is a major risk factor for embolization, several probable conditions, the connection of which is not thoroughly studied, such as gout disease and methamphetamine abuse, are also possible causes. We present the case report of a male in his early 40s with a history of alcohol and methamphetamine abuse, gout, and dilated cardiomyopathy, experiencing multiple ischemic attacks, including acute limb ischemia, dysarthria, and renal infarct. Echocardiography revealed a large clot in the left ventricle. The patient received heparin at a rate of 1000 units/hour, followed by warfarin at 10 mg/day, and probenecid at 500 mg/day. The thrombus resolved upon anticoagulant therapy, with improvement in the patient’s symptoms. Left ventricular thrombus formation in dilated cardiomyopathy is influenced by many factors, including drug toxicity (for instance, methamphetamine), uric acid levels, and N-terminal precursor B-type natriuretic peptide. The thrombus can cause cardioembolic attacks in multiple sites, and the choice for emergent therapy is an oral anticoagulant, which may improve the ejection fraction. However, certain aspects related to cardiac thrombosis and embolization risk still warrant more comprehensive studies.
Introduction
Dilated cardiomyopathy (DCM), also known as non-ischemic cardiomyopathy, includes a range of myocardial complications with various etiologies resulting in the dilation of the left ventricle or both ventricles of the heart with systolic dysfunction, usually defined as a left ventricle ejection fraction (LVEF) of less than 40%. Most of the DCM cases are idiopathic, while the remaining can be a consequence of an infiltrative disease, coronary artery disease, alcohol abuse, medications, Takotsubo syndrome, and pregnancy. DCM affects men more than women, and its prevalence in the general population is 40 cases per 100,000. DCM has a high mortality rate and is a leading cause of heart transplantation.1,2
In DCM, left ventricular (LV) thrombus formation occurs owing to stasis, with a reported prevalence of up to 36% for LV thrombus and a risk of 11% for systemic embolization. 3 We report a case of a male with DCM who experienced multiple simultaneous ischemic attacks in the brain, lower limb, and kidney due to a large LV thrombus. The patient had a history of alcohol and drug abuse and uncontrolled gout. This study aimed to discuss the associated risk factors, including drug abuse or high uric acid levels, locations, characteristics of the cardiac thrombus and emboli, and the chosen therapy. We also review the existing literature.
Methods
We reviewed the clinical features of a patient with multiple ischemic attacks and LV thrombus who had DCM, with a history of drug abuse and uncontrolled gout. The study was conducted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. Moreover, the patient provided written informed consent for treatment. We adhered to the CARE reporting guidelines 4 and de-identified all patient details.
We also performed a review of literature that was conducted using PubMed database searches with keywords that included the MeSH terms “Thrombosis” and “Dilated Cardiomyopathy.” The time frame was limited to all English articles from 2000 to the present year. After screening the titles and abstracts of case report studies, we included nine case reports documenting LV thrombus in adults with non-ischemic DCM. The exclusion criteria included venous thrombosis, coagulation disorders, and a history of cardiac procedures (Table 1).
Summary of the left ventricular thrombus in dilated cardiomyopathy cases presented in the literature.
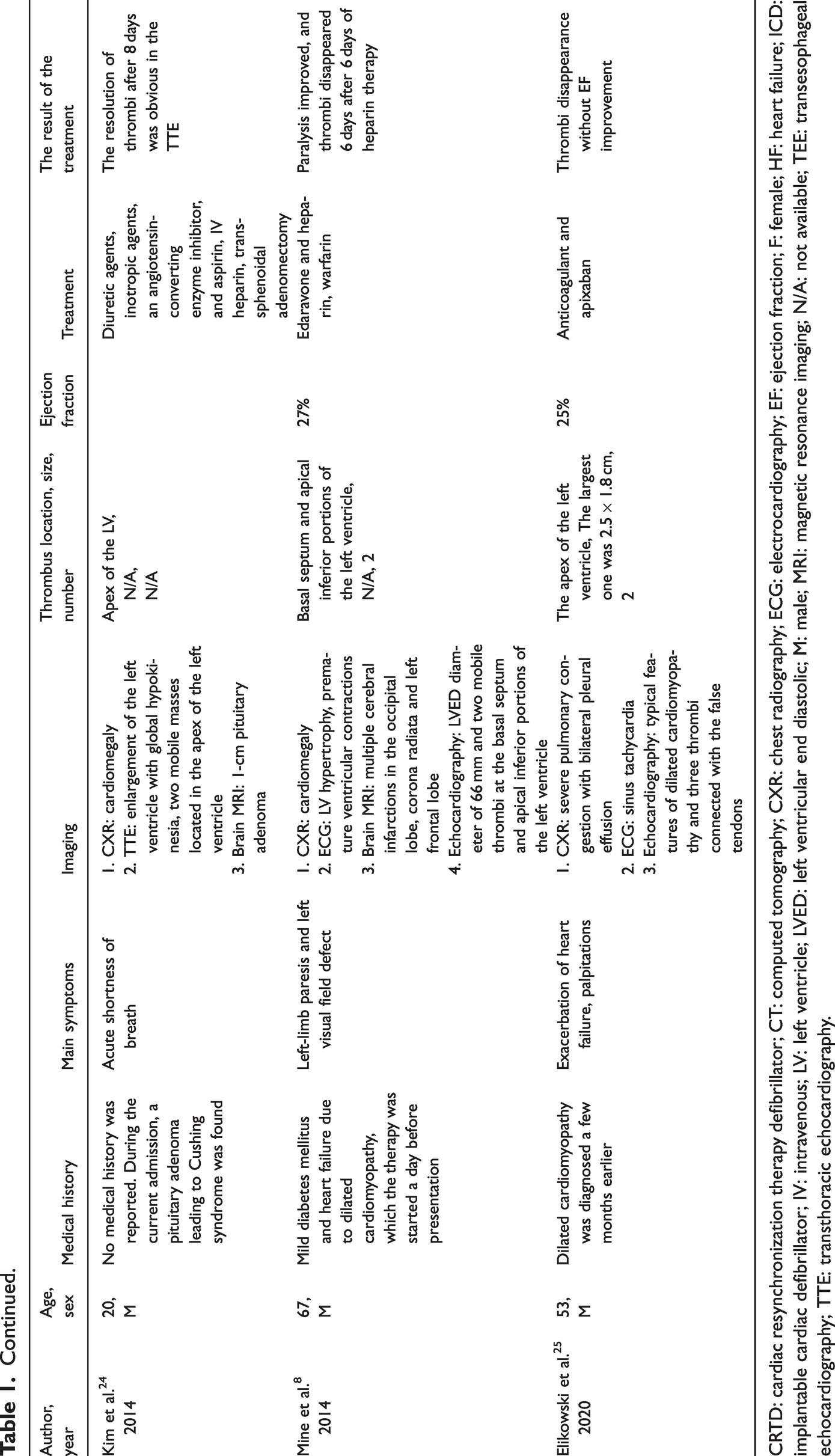
CRTD: cardiac resynchronization therapy defibrillator; CT: computed tomography; CXR: chest radiography; ECG: electrocardiography; EF: ejection fraction; F: female; HF: heart failure; ICD: implantable cardiac defibrillator; IV: intravenous; LV: left ventricle; LVED: left ventricular end diastolic; M: male; MRI: magnetic resonance imaging; N/A: not available; TEE: transesophageal echocardiography; TTE: transthoracic echocardiography.
Case presentation
A Persian male in his early 40s presented with a chief complaint of severe left lower limb pain, which started two hours before the presentation, and was brought into the emergency department by ambulance. The patient reported a self-limited transient episode of dysarthria lasting for 2 hours the night before presentation, and a stage 2A claudication a month ago.
Considering the patient’s medical history, he reported having poorly controlled gout for the past 15 years. The patient was a heavy smoker (50 packs/year), heavy drinker, and methamphetamine user. Additionally, he was hospitalized 6 months ago due to exertional dyspnea. Since that previous admission for exertional dyspnea, the patient has been prescribed sacubitril/valsartan 26/24 mg and furosemide 40 mg twice daily, eplerenone 25 mg as half a tablet, and bisoprolol 2.5 mg daily. He was also taking allopurinol 100 mg each day. Furthermore, he quit smoking, drinking, and using drugs. He also reported a family history of myocardial infarction in his father. The LVEF was reported to be 20%–25% upon admission. Medical documents indicated no signs of LV clots but confirmed a diagnosis of non-ischemic DCM through cardiac magnetic resonance imaging (MRI).
During the physical examination, the patient was found to be afebrile. The heart rate, blood pressure, and jugular venous pressure were 119 bpm, 129/80 mm Hg, and 12 cm H2O, respectively. Grade 2 mitral murmurs and fine crackles were detected at the base of both lungs upon examination. Upon examining the extremities, the affected limb appeared pale, cold, and tender, with a weak pulse, which was recorded as +1. Motor and sensory examinations, as well as reflexes, were normal. The patient’s knee and interphalangeal joints were swollen, tender, erythematous, and warm, exhibiting a decreased range of motion, with many visible dysmorphic tophi (Figure 1(a) and (b)).

(a) Gout arthritis and multiple dysmorphic tophi in the knee and; (b) interphalangeal joints; (c) a large thrombus (10 cm × 7 cm, 25.5 cm2) pointed by the red arrow, is visible attaching to the apex of the left ventricle; (d) a decrease in the size of the left ventricular (LV) thrombus 2 weeks after the initiation of anticoagulant therapy is obvious on the follow-up transthoracic echocardiography. The arrow reveals a clot measuring 7 cm × 3 cm, 17.2 cm2; (e, f) abdominal aorta and lower extremity computed tomography angiogram using maximum intensity projection and volume rendering techniques. Red arrows demonstrate a wedge-shaped defect in the right renal artery, and white arrows point to a significant stenosis of the left common iliac artery and (g) on the 3-month follow-up transthoracic echocardiography, the severe LV enlargement was improved, and the LV thrombus was resolved.
The patient was admitted, and we immediately requested routine laboratory tests, plasma uric acid level, and synovial fluid analysis (Table 2). Electrocardiography revealed incomplete left bundle branch block, left axis deviation, intraventricular conduction delays in the inferior leads, and biatrial abnormalities.
Patient’s laboratory results during hospitalization.

NT-proBNP: N-terminal pro-B-type natriuretic peptide; PMN: polymorphonuclear.
Brain computed tomography (CT) revealed no abnormalities. Left lower limb arterial Doppler ultrasonography was also performed, revealing a monophasic waveform in the lower extremity. This pattern may indicate reduced arterial compliance or increased peripheral resistance, which is often associated with arterial disease or stenosis. For the left lower extremity, iliac artery, and abdominal aorta, CT angiography was requested but postponed due to the patient’s elevated creatinine plasma levels. A transthoracic echocardiography (TTE) indicated very severe LV enlargement (LV end-diastolic volume index: 208 cc/m2) with severe systolic and moderate to severe diastolic dysfunction, LVEF: 5%–10%, global severe hypokinesia, and no LV hypertrophy. A massive (10 cm × 7 cm, 25.5 cm2) protruding LV clot was attached to the apex, anteroapical, and mid-anterior segments of the LV, with a semi-mobile portion and intrinsic liquefaction (high-risk LV thrombus). Severe left atrial enlargement, mild right atrial enlargement (LAVi: 69 cc/m2, RAVi: 34 cc/m3), thickened and tethered mitral valves, and moderate mitral regurgitation were noted (Figure 1(c)). Following the TTE, anticoagulant therapy was initiated; the patient received 1000 units of heparin per hour, followed by warfarin 10 mg daily after 5 days. Additionally, owing to the elevated uric acid levels and severe arthritis, probenecid 500 mg daily was added to allopurinol. A follow-up echocardiography conducted two weeks after the initiation of anticoagulants demonstrated a protruding LV clot (7 cm × 3 cm, 17.2 cm2), which is attached to the apex, anteroapical, and mid-anterior segments of the LV, with a semi-mobile part and multiple mobile particles (Figure 1(d)).
CT angiography was performed 3 days later, following a decrease in the creatinine levels. CT angiography revealed a thrombus involving the left common iliac artery, leading to a cut-off of the origin of the common iliac artery on the left side with good runoff. An extensive wedge-shaped heterogeneous enhancement of the right kidney was also discernible, likely indicating extensive thrombosis resulting in infarction (Figures 1(e) and (f)).
The patient’s leg pain diminished upon treatment. A nephrology consult was requested, and conservative treatment was recommended. Fifteen days later, the patient was discharged once he reached the therapeutic range of warfarin; he was advised to consult a vascular surgeon for iliac stenosis. At the monthly follow-up, after 3 months of warfarin therapy, the clot was resolved, and the N-terminal pro-B-type natriuretic peptide (NT-proBNP) decreased to 950 pg/mL, but the ejection fraction (EF) remained at 10%–15% (Figure 1(g)).
Heart failure treatment and warfarin therapy were recommended to be continued for 6 months. The patient had both lower limb and coronary angiography after discharge. Coronary angiography was normal. Stenting of the left iliac artery was performed, and the patient’s claudication improved completely.
Follow-up
During the monthly visit and evaluation of LV function and thrombosis with cardiac MRI, we noticed that LVEF was 35%, and no further evidence of cardiac thrombosis.
After cessation of addictive drugs and alcohol and the complete elimination of LV thrombosis and upon consultation with a neurologist and vascular surgeon, we decided to discontinue warfarin and evaluate him every month in our clinic. All symptoms improved, and the patient continued with -heart failure treatment.
Discussion
DCM presenting with shortness of breath, cough, fatigue, and swelling in the legs can cause complications, such as heart valve problems, stroke, and arrhythmia. The ischemic stroke in idiopathic DCM is most commonly attributed to cardioembolism; however, other factors, namely hypertension, hyperlipidemia, atrial fibrillation, lower glomerular filtration rate, and intracardiac thrombus can also increase the risk of the ischemic attack in patients with DCM. 5
A previous study introduced high D-dimer levels of more than 444 ng/mL and decreased EF as factors increasing the risk for LV thrombus in patients with DCM. 6 In contrast, the risk factors for systemic embolization of the cardiac thrombus are still inconsistent. Characteristics associated with a higher tendency for embolization include protrusion of the thrombus into the LV cavity, large thrombus size, diffuse LV dilatation, and impaired systolic function. 7 The patient at the time of the presentation had an LVEF as low as 5%–10%, a large protruding LV thrombus attached to the apex, and a severely dilated LV. All these factors together can explain the multiple ischemic attacks our patient experienced.
LV thrombuses can develop at various sites, but it is commonly observed in the apex of the left ventricle, as reported in the literature. The second most common location is the anterior-apical wall. However, thrombi were also observed in the basal 8 and lateral walls of the LV. 9 The biventricular thrombi 10 are usually seen in DCM cases with EF less than 30%. 7
To find the accompanied factors with LV thrombus in adults with non-ischemic dilated cardiomyopathy, among the reported cases in Table 1, 3 of 9 had peripartum cardiomyopathy, described as new onset heart failure occurring during the last month of pregnancy and five months after delivery. Peripartum cardiomyopathy is evaluated with EF less than 45% and a left ventricle end-diastolic diameter of more than 2.7 cm/m.2,9–12 Other factors responsible for DCM and thrombus in the cases included alcohol consumption, amphetamine abuse, and human immunodeficiency virus, of which the first two were present in our case as well. Amphetamine-induced cardiomyopathy, although rare, can cause severe DCM.13,14 Catecholamine-mediated signaling causes the cardiovascular system to react to the stimulants by causing vasoconstriction, vasospasm, tachycardia, and hypertension. Myocardial fibrosis and alterations in myocardial contractility can also be brought on by stimulants, leading to the development of dilated or hypertrophic cardiomyopathy. The core of the treatment of amphetamine-induced cardiomyopathy is the discontinuation of drug usage, with valsartan and sacubitril administration. 15
An interesting point in the medical history of our case was the uncontrolled gout and elevated uric acid levels as high as 9.1 mg/dL. Although the effect of elevated uric acid levels on DCM in adults is still not well elucidated and warrants further research, some studies showed that the elevated uric acid level is a risk factor for ischemic heart disease and can be a marker to evaluate the severity of DCM in children.16,17 It highlights the role of high uric acid levels in endothelial dysfunction, inflammation, and prothrombotic state by increasing factors such as interleukin-6. 18 Krishnan 19 demonstrated that heart failure, subclinical LV systolic dysfunction, and mortality increased in patients with gout. Mouradjian et al. 20 proved in their study that hyperuricemia and gout are independent risk factors for cardiovascular disease and worsening of heart failure. Treatment of these issues with uric acid lowering therapy is recommended for significantly reducing the risk of cardiac disease during gout flare-up. Moreover, Cipolletta et al. 21 reported that high uric acid levels and gout flare were related to venous thromboembolism.
Li X-L et al. 22 and Tigen et al. 23 developed a nomogram to predict the likelihood of ventricular thrombosis in DCM. Their model supports the influence of age, LVEF, uric acid, NT-proBNP, and D-dimer on ventricular thrombosis. Additionally, the increased NT-proBNP levels in our case indicates a poor prognosis for DCM with sinus rhythm.22,23 A review of the literature revealed that LV thrombus in a setting of DCM can present as heart failure exacerbation,9,10,24,25 or ischemic attacks. Additionally, embolisms were reported in the lower limb,7,8,26 brain,8,26,27 kidney, 13 and spleen.11,13 Upon comparing our case to previous studies, it is important to note that simultaneous infarcts have been reported in the left common iliac artery and right renal artery, along with a transient ischemic attack characterized by dysarthria. This underscores the need for prompt initiation of emergent therapy due to the features of the LV clot, such as protrusion and its size, which were more significant than those of the thrombi reported in the literature. Notably, in one of the cases, symptoms began immediately after the administration of vasodilators and diuretics for heart failure treatment. 8 The exact indications for preventing LV thrombus in DCM remain unknown to date. However, Takotsubo syndrome, LV noncompaction, eosinophilic myocarditis, peripartum cardiomyopathy, and cardiac amyloidosis are considered risk factors for LV thrombus, and oral anticoagulants can be prescribed for its management. 8 At present, surgical removal of LV thrombus is not recommended except when anticoagulant therapy is not well tolerated; moreover, a high risk for embolization and occurrence of thromboembolic events exists despite anticoagulation therapy.26,28 The valuable point in the literature review is that after the resolution of thrombi, the EF improved in some cases.13,26,27 The reviewed cases did not mention any recurrence of LV thrombus; however, two studies report a recurrence of LV thrombus or an increase in the size of the thrombus in the patients with inappropriate medical adherence and those with hematologic malignancies, chronic renal failure, and inflammatory disease.27,28
Conclusion
We emphasize the critical impact of LV thrombosis in patients with DCM, leading to multiple ischemic attacks affecting various organs. Our case report underscores the importance of recognizing the associated risk factors for LV thrombus formation and embolic events, characteristics of the thrombus, and potential complications, while also highlighting the need for urgent therapy initiation and the potential for improvement in EF after the resolution of the thrombus. Additionally, the presence of high uric acid levels in our case report warrants for further research to understand better the impact of elevated uric acid levels on DCM and to optimize management strategies for preventing and treating LV thrombus in patients with DCM.
Footnotes
Acknowledgements
We greatly appreciate the officials and staff of the Razi Educational and Remedial Center and ethics committee of Guilan University of Medical Sciences.
Author contributions
Consent to participate
Patient provided written informed consent prior to participating.
Consent for publication
Informed consent for publication was provided by the participant.
Data availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
Ethical approval to report this case was obtained from local ethics committee of Guilan University of Medical Sciences (approval number: IR.GUMS.REC.1402.607).
Funding
The authors received no financial support for the research, authorship, and publication of this article.
Patient perspective
My overall health has improved since starting the treatment, and I have not experienced any further episodes of dysarthria. The claudication has also improved following the stenting procedure.
