Abstract
Background:
Impaired lung function has been noted in individuals with spondyloarthritis (SpA). However, a comprehensive summary of the existing evidence related to these conditions is still lacking.
Objective:
The aim of the study is to explore the association between impaired lung function and the prevalence and incidence of SpA, utilizing data from the UK Biobank.
Methods:
A total of 411,780 participants with complete spirometry data were included in the study. Logistic regression and Cox regression models were employed to investigate the association between impaired lung function and the prevalence/incidence of SpA. In addition, restricted cubic spline (RCS) analysis was applied to evaluate the nonlinear relationships between forced expiratory volume in 1 s (FEV1) and/or forced vital capacity of the predicted value and SpA incidence.
Design:
A cross-sectional study was conducted by using UK Biobank data from 411,780 participants with complete lung function records and HLA-B27 genotype. In addition, a prospective cohort of 409,069 individuals without baseline SpA and lost follow-up was followed longitudinally to assess incident SpA risk.
Result:
The prevalence of SpA was significantly higher in individuals with preserved ratio impaired spirometry (PRISm) compared to those with normal spirometry, as evidenced by adjusted odds ratio (OR; 95% confidence interval (CI)) of 1.538 (1.409–1.680) for SpA. Over a median follow-up period of 14.64 years, 1637 participants (0.40%, 1637/409,069) developed SpA, with PRISm associated with an elevated risk for incident SpA (adjusted hazard ratio (95% CI): 1.249 (1.098–1.420)) when compared to normal spirometry. Furthermore, RCS analysis revealed a nonlinear relationship between FEV1 %pred and the risk of developing SpA (overall p < 0.001 and nonlinear p = 0.011).
Conclusion:
The study unveiled a significant association between impaired lung function and the prevalence of SpA. Individuals with PRISm may have an elevated risk of incident SpA.
Introduction
Spondyloarthritis (SpA) comprises a group of chronic inflammatory rheumatic disorders primarily affecting the axial spine, sacroiliac joints, entheses, and peripheral joints, with an estimated population prevalence of 0.5%–1.4%.1,2 This disease spectrum is clinically classified into two principal phenotypes as axial SpA (axSpA) and peripheral SpA (pSpA) as per the Assessment of Spondyloarthritis International Society (ASAS). 3 The axSpA category is further stratified based on radiographic evidence of sacroiliitis as radiographic axSpA (r-axSpA), also known as ankylosing spondylitis (AS) and non-radiographic axSpA (nr-axSpA). pSpA encompasses several clinical entities, including reactive arthritis (ReA), psoriatic arthritis (PsA), and inflammatory bowel disease (IBD) associated arthritis (IAA), characterized by peripheral manifestations such as arthritis, dactylitis, tenosynovitis, and/or enthesitis. 4 In our study, the term SpA collectively encompasses both axSpA and pSpA. Of axSpA cases, AS represents the dominant and clinically significant subtype, which is characterized by inflammatory back pain, radiographic sacroiliitis, and excess spinal bone formation, affecting ~0.5% of the population worldwide. 5 Individuals with positive HLA-B27 increase the prevalence. 6 nr-axSpA shares similar features with AS and is usually recognized as the early stage of AS. Both of them can cause inflammation and eventual fusion of the spine, particularly the thoracic spine and costovertebral joints. 7
Both axSpA and pSpA may present with extramusculoskeletal manifestations (EMMs), such as anterior uveitis, psoriasis, IBD, and other systemic disorders, highlighting the overlap between axSpA and pSpA.8–10 Current evidence indicates a low prevalence (1.3%) of pulmonary imaging abnormalities in SpA cohorts, predominantly observed among AS patients. 9 Reported rates of pulmonary involvement in AS populations (0%–85%) and PsA cohorts (1.05%–25%) demonstrate substantial heterogeneity.10–13 A large retrospective case-series study revealed that pulmonary involvement occurs in only 1.3% of affected AS individuals.13,14 This significant heterogeneity in pulmonary imaging findings across SpA subtypes raises critical questions: While some patients exhibit no radiological abnormalities, they may already demonstrate pulmonary function impairment. This subclinical dysfunction potentially serves as a more sensitive indicator of early lung involvement in SpA than conventional imaging modalities. Restrictive ventilatory patterns have been documented in AS without radiological abnormalities, while their prevalence across other SpA subtypes remains largely unexplored.13,15
Pulmonary function tests may be associated with the early detection of rheumatoid arthritis, as they can reveal restrictive ventilatory defects and decreased gas exchange, abnormalities that may be detectable even in the absence of clinical symptoms. 16 Preserved Ratio Impaired Spirometry (PRISm) is a distinctive spirometric pattern characterized by a decreased forced expiratory volume in 1 s (FEV1) alongside an intact FEV1/forced vital capacity (FVC) ratio, indicative of lung volume restriction, often linked to conditions such as obesity, chest wall deformities, and interstitial lung diseases. 17 The reported prevalence of PRISm ranges from 17% to 24%, with higher occurrences observed among individuals aged 45–60 years, males, those with elevated body mass index (BMI), and individuals diagnosed with restrictive lung disease. 18 PRISm has also been recognized as the precursor of chronic obstructive pulmonary disease (COPD) in 50% individuals over a 5-year follow-up. 17 In recent years, PRISm has garnered increasing attention due to its clinical significance in evaluating lung function impairment beyond conventional spirometric parameters. 19 This spirometric pattern is not only linked to heightened respiratory symptoms and mortality but also recognized as a risk factor for both adverse cardiovascular and respiratory events. 20 Previous studies had demonstrated a close relationship between PRISm and conditions such as diabetes, metabolic syndrome, and even depression or dementia, underscoring PRISm as a potential public health issue associated not only with respiratory disorders but also with elevated risks of various non-respiratory ailments.21–23 These findings emphasize the crucial significance of early identification and timely management strategies to prevent long-term progression and complications.
Consequently, we aim to investigate whether impaired pulmonary function may serve as an indicator for disease progression in SpA. The utilization of the expansive resources and longitudinal follow-up data of the UK Biobank provides scientific utility for comprehensively elucidating this relationship.24,25 The primary objectives of this study are to examine the association between PRISm and the prevalence of SpA and to investigate its relationship with SpA incidence within a 15-year longitudinal cohort. Second, covariate-adjusted subgroup analyses were conducted to identify heterogeneous associations between PRISm and SpA across diverse populations. Furthermore, restricted cubic spline (RCS) will be utilized to assess potential nonlinear relationships between FEV1 %pred and/or FVC %pred and SpA risk, thus generating significant implications for clinical practice.
Methods
Study design, setting, and participants
Cross-sectional study
The inclusion of participants from the UK Biobank (https://www.ukbiobank.ac.uk/), a comprehensive longitudinal cohort study initiated between 2006 and 2010, was a pivotal aspect of our research. This encompassed a wide demographic, with enrollment of 502,357 individuals aged between 37 and 73 years from various locations across England. Our analysis involved a total of 411,780 subjects who had fully documented lung function data and HLA-B27 testing results for a cross-sectional study.
Longitudinal follow-up
In addition, following exclusion of participants with baseline SpA diagnosis and those lost to follow-up, we conducted a prospective cohort of 409,069 individuals without SpA (Figure 1) to assess incident SpA risk (Figure 1).
Cross-sectional study exclusion criteria
(1) Participants lacking complete lung function data (n = 82,355);
(2) HLA-B27 genotyping results (n = 8222).
Prospective cohort selection
Following these exclusions, we further removed individuals with:
(1) Prevalent SpA at baseline (n = 1666);
(2) Loss to follow-up (n = 1045).
All remaining eligible participants were included in the prospective cohort analysis.

Flowchart of COPD cohort study.
We strictly followed the Strengthening Reporting of Observational Studies in Epidemiology (STROBE) statement. 26
Definition of lung function
During the initial phase of data collection, spirometry was performed by extensively trained personnel using the Vitalograph Pneumotrac 6800 spirometer, Vitalograph (Ireland) Limited, while strictly adhering to standardized procedures. 27 Each participant was tasked with completing 2–3 forced exhalations of at least 6 s’ duration within a 6-min timeframe, with the spirometer being recalibrated before each session. Subsequently, the FEV1 and FVC were computed as a percentage of anticipated values in line with the Global Lung Initiative 2012 standards. 28 Participants were categorized according to the Global Initiative for Chronic Obstructive Lung Disease-2024 Report (GOLD 2024) as COPD: a post-bronchodilator FEV1/FVC ratio less than 0.7 and PRISm: FEV1/FVC ⩾0.7 but FEV1 and/or FVC <80% predicted value after bronchodilation. 29 In addition, participants with spirometry of FEV1/FVC ratios and FEV1 equal to or more than 0.7 and 80% were defined as normal. Qualifying values for further analysis were established as the highest recorded readings if the initial two attempts were within 5% variability; otherwise, a third attempt was made.
Assessment of SpA with ICD-10 codes
The SpA umbrella encompasses axSpA and pSpA. Each subset of the condition was delineated in accordance with the International Classification of Diseases, Tenth Revision (ICD-10), via the assignment of specific codes. For axSpA: (1) r-axSpA (also known as AS): M45; (2) nr-axSpA: M46.8. For pSpA: (1) Arthropathic psoriasis: L40.5; (2) Psoriatic and enteropathic arthropathies: M07; (3) ReA: M02. For extraarticular manifestation: (1) Uveitis: H20; (2) Psoriasis: L40; (3) IBD: K50-K52; For peripheral manifestation: (1) Arthritis: M13; (2) Enthesitis: M46.0, M76, M77; (3) Dactylitis: M79.0, M79.6, M79.8, M79.9.
The diagnostic data for SpA primarily originated from death registries, primary care records, and hospital admissions, supplemented to a lesser extent by self-reported information. If SpA was not documented, the follow-up duration was concluded at the time of death, the date of loss to follow-up, or the data retrieval cut-off date (February 20, 2024).
HLA-B27 genotyping from whole-exome data
HLA genotypes were obtained from the UK Biobank’s imputed dataset. Per UK Biobank recommendations: Individuals were classified as carrying an allele if posterior probability values (PPV) between 0.7 and 1; and subjects with PPV between 1.5 and 2.0 for a single allele at any locus were considered as two copies of the allele. Participants were excluded if they had: (1) Two nonzero alleles with either/both PPV <0.7, or (2) A single nonzero allele with PPV between 1.0 and 1.5 at any of the eight HLA loci. 30
Variables
A comprehensive set of potential risk factors associated with lung function or SpA was compiled as covariates at baseline, sourced directly from the UK Biobank, encompassing sociodemographic factors (age, sex, ethnicity, BMI, Townsend deprivation index (TDI), family income), lifestyle components (smoking status, alcohol consumption, and level of physical activity (PA)), comorbidities (asthma, hypertension, coronary heart disease (CHD), chronic kidney disease (CKD), and diabetes mellitus (DM), cancer), treatments (NSAIDs and biological disease-modifying antirheumatic drugs), and HLA-B27 genotype.
Statistical analysis
For all statistical analyses, continuous variables were depicted as means (SD) or median (IQR) through Student’s t tests or Mann–Whitney–Wilcoxon tests, while categorical variables were represented as frequencies using Chi-square tests or Fisher’s exact test. A multivariate logistic regression analysis was executed to ascertain the prevalence of SpA across various lung function levels, generating odds ratios (ORs) and 95% confidence intervals (CIs) to delineate the strength of the associations. The link between baseline impaired lung function status and the incidence of SpA was evaluated via Cox proportional hazards models, presenting hazard ratios (HRs) and 95% CIs. The potential linear and nonlinear relationships between FEV1 or FVC of predicted values and the prevalence and risk of SpA were investigated using RCS regression with four knots within the R software platform.
Our analytical strategy involved the development of five progressive models. Commencing with Model 1 as the foundation, no adjustments for confounders were made. Subsequent models integrated adjustments for age, sex, and ethnicity. Model 3 included BMI, family income, TDI, alcohol consumption, smoking status, and activity levels based on model 2. Model 4 further refined the analysis by accommodating a range of comorbidities, such as asthma, hypertension, CHD, CKD, DM, and cancer. Model 5 was an extension based on HLA-B27 genotype and medications from Model 4.
IBM SPSS Statistics (IBM Corporation, version 26.0) and R software (version 4.2.2) were employed for the statistical analyses, GraphPad Prism (GraphPad Software, Inc., version 9.0) for forest plot creation, and the RSpiro software package and rcssci package for FEV1% and FVC calculations and RCS regression, respectively. Statistical significance was attributed to p-values below 0.05, in adherence to stringent hypothesis testing criteria safeguarding the statistical integrity of the results.
Results
Baseline characteristics of study population based on SpA diagnosis
The demographic and baseline characteristics of the cross-sectional study encompassing SpA are succinctly detailed in Table 1. Within the cohort of 411,780 individuals with pulmonary function data and full HLA-B27 genotype sourced from the UK Biobank database, participants diagnosed with SpA exhibited notably lower FEV1 Pred% and FVC Pred% compared to those without SpA (89.35 (76.53–100.21) vs 94.03 (82.42–104.24), and 91.97 (80.88–102.71) vs 96.60 (86.53–106.71), respectively). Furthermore, individuals with SpA demonstrated a 10.60% decrease in normal pulmonary function (66.55% (2200/3306) vs 77.17% (315,223/408,474)), an 8.85% increased prevalence of PRISm ((23.20% (767/3306) vs 14.35% (58,624/408,474)).
Baseline characteristics of the study based on SpA diagnosis.
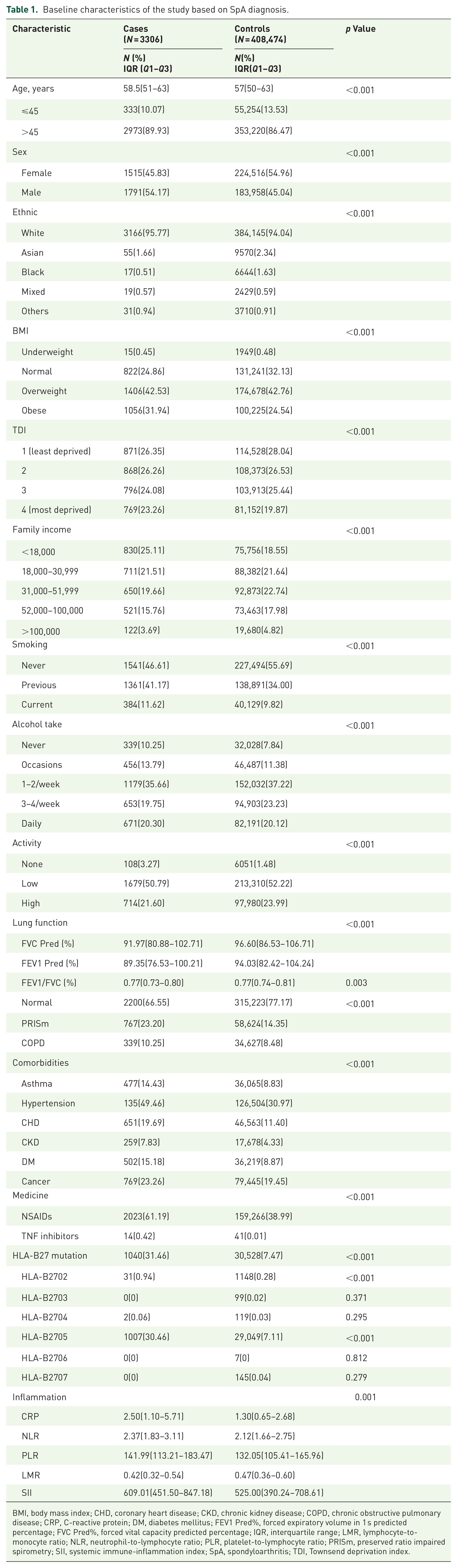
BMI, body mass index; CHD, coronary heart disease; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; DM, diabetes mellitus; FEV1 Pred%, forced expiratory volume in 1 s predicted percentage; FVC Pred%, forced vital capacity predicted percentage; IQR, interquartile range; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; PRISm, preserved ratio impaired spirometry; SII, systemic immune-inflammation index; SpA, spondyloarthritis; TDI, Townsend deprivation index.
Among 3306 SpA patients, axSpA predominated (54.45%, 1800/3306), while pSpA accounted for 47.52% (1571/3306). Within these subtypes, AS constituted the majority (97.00%, 1746/1800) in the axSpA cohort, and PsA was the primary manifestation (81.29%, 1277/1571) in the pSpA group, with both ReA and IAA at 14.58% (229/1571) each. Comprehensive details of EMMs and peripheral joint involvement across SpA subtypes are presented in Supplemental Table 1.
HLA-B27 prevalence was 7.67% (31,568/411,780) in the overall cohort. Among SpA patients, positivity rates were substantially higher at 31.46% (1040/3306). Notably, axSpA demonstrated threefold greater HLA-B27 prevalence than pSpA, with AS showing the highest frequency at 46.85% (818/1746 AS patients; Supplemental Table 2).
Cross-sectional analysis of pulmonary function and SpA association
A significant protective role of lung function (FEV1 Pred% and FVC Pred%) for SpA was demonstrated in the fully adjusted models (Model 5). The ORs (95% CI, p value) for SpA in relation to FEV1 Pred% were 0.787 (0.717–0.863, p < 0.001), 0.780 (0.708–0. 860, p < 0.001), and 0.636 (0.571–0.708, p < 0.001) in quartiles 2–4, as compared with the lowest quartile. Furthermore, the ORs (95% CI, p value) for SpA in relation to FVC Pred% were 0.728 (0.663–0.799, p < 0.001), 0.732 (0.664–0.806, p < 0.001), and 0.643 (0.579–0.715, p < 0.001) in quartiles 2–4 compared with the lowest quartile (Table 2). In addition, individuals with PRISm exhibited a 53.8% increased prevalence of SpA (OR = 1.538, 95% CI = 1.409–1.680) compared to those with normal lung function.
Association between lung function and prevalence of SpA.

Calculated by means of a Logistic regression analysis with no adjustment.
Adjusted by age, sex, and ethnicity.
Adjusted for model 2 plus economic status (BMI, Townsend score, and family income) and lifestyle (alcohol intake, smoking status, and amount of activities).
Adjusted for model 3 plus underlying diseases (asthma, hypertension, CHD, CKD, DM, and cancer).
Adjusted for model 4 plus HLA-B27 genotype and medicine.
BMI, body mass index; CHD, coronary heart disease; 95% CI, 95% confidence interval; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; FEV1 Pred%, forced expiratory volume in 1 s predicted percentage; OR, odds ratio; PRISm, preserved ratio impaired spirometry; SpA, spondyloarthritis.
In subtype-specific analyses comparing axial axSpA versus pSpA, and their predominant manifestations AS and PsA, impaired pulmonary function demonstrated significantly stronger associations with axSpA (OR (95% CI), 2.022 (1.804–2.266)) and its cardinal subtype AS (OR (95% CI), 2.056 (1.832–2.308); p < 0.001). While the PRISm subgroup within PsA (OR (95% CI), 1.165 (1.006–1.350)) maintained a measurable association with lung impairment, the overall correlation between pulmonary dysfunction and pSpA incidence did not reach statistical significance (OR (95% CI), 1.115 (0.974–1.276) p = 0.114; Supplemental Tables 3–6). Further, it was observed that the HLA-B27 mutation emerged as a more critical factor in AS than in SpA. This aligns with established epidemiological patterns, where HLA-B27 demonstrates its highest prevalence in AS among SpA subtypes (Supplemental Figure 1).
Longitudinal association between baseline lung function and incident SpA
A cross-sectional investigation identified a close relationship between impaired lung function and the prevalence of SpA. Following the removal of SpA individuals at baseline (n = 1666) and those lost to follow-up (n = 1054), further inquiry was conducted to ascertain whether impaired lung function correlated with the development of SpA (Figure 1). Over a median follow-up period of 14.64 years, elevated lung function was associated with a reduced SpA risk, as evidenced by HRs (95% CI, p value) of 0.871 (0.763–0.993, p < 0.001), 0.855 (0.746–0.981, p < 0.001), and 0.782 (0.676–0.905, p < 0.001) for FVC %pred in quartiles 2–4, relative to the lowest quartile. Similar trends were identified for FEV1 %pred, while the highest quartile showed a significant p value. Subsequently, in the fully adjusted model (Model 5), the impaired lung function group exhibited a higher risk of SpA compared to the control group, demonstrating a 24.9% increased risk of SpA (HR = 1.249, 95% CI 1.098–1.420) (Supplemental Table 7). Refer to Supplemental Tables 8–11 for the association between lung function and the prevalence of axSpA, pSpA, AS, and PsA. Similar to the cross-sectional study, impaired pulmonary function demonstrated significantly stronger associations with SpA risk in axSpA, especially in the AS subtype.
Furthermore, the Kaplan–Meier curve illustrated an elevated risk of incident SpA in the PRISm cohort compared to the control cohort based on sex and age (Figure 2). Stratification analyses were conducted across various subgroups encompassing age, sex, race, BMI, lifestyle, and comorbidities (Figure 3). In the PRISm population, we further discovered that the HR for the HLA-B27 mutation, particularly for the B*27:05 allele, was 5.472 (95% CI 4.503–6.651; Supplemental Figure 2).
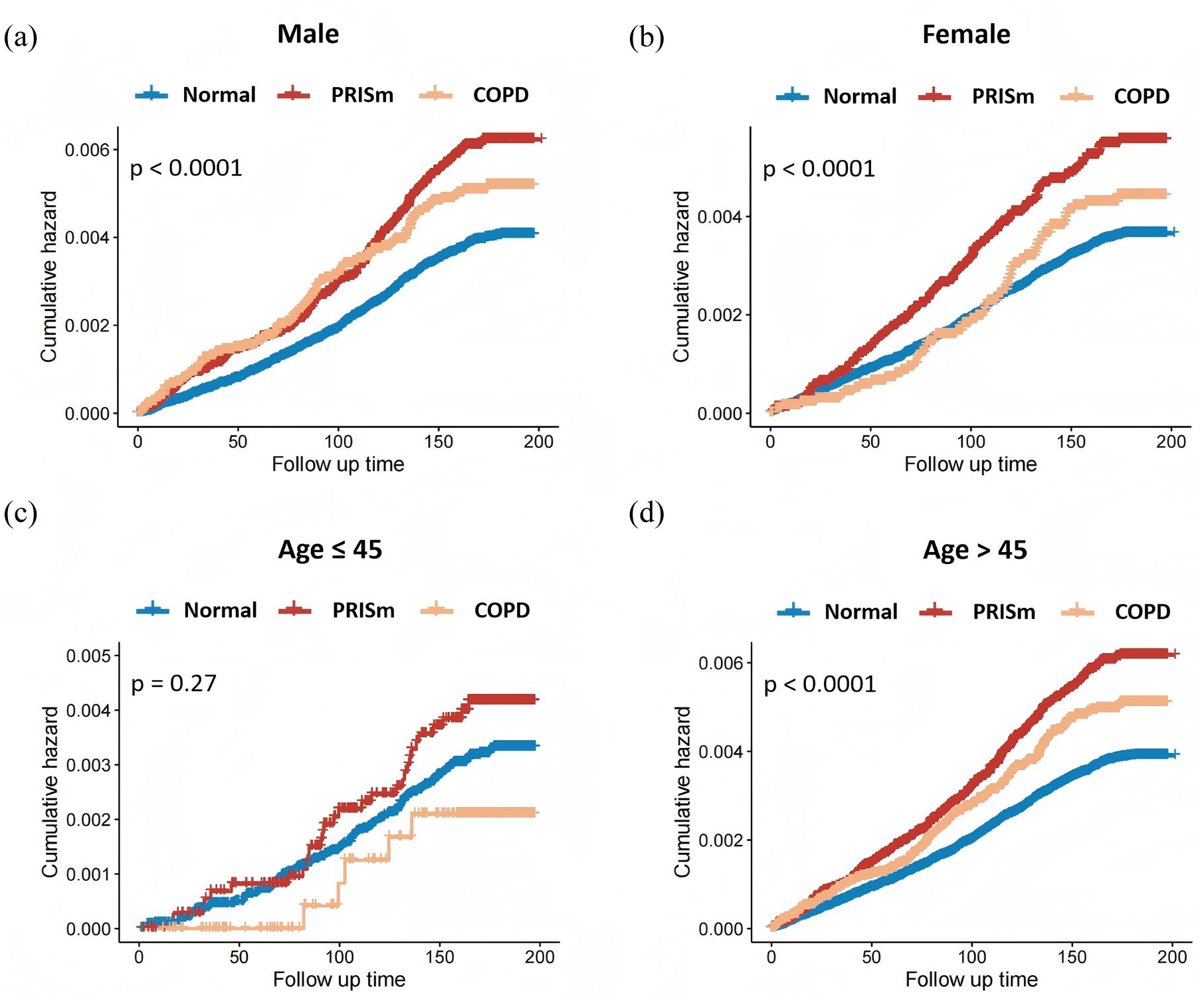
The Kaplan–Meier analysis of the risk of SpA with respect to various lung function levels stratified by age and sex. The Kaplan–Meier curve illustrated an elevated risk of incident SpA in the PRISm and COPD cohort compared to the control group based on sex ((a) and (b), p < 0.001 for both male and female) and age ((c) and (d), p = 0.27 for age <45 years old and p < 0.001 for age at and >45 years old).

Subgroup of Forest plots of the associations between PRISm and SpA incidence.
The detection of nonlinear/linear relationships between lung function indicators and risk of SpA
Upon categorizing lung function into quartiles, we have observed that elevated lung function was associated with a reduced SpA prevalence and incidence. Since FEV1 %pred and FVC %pred as the indicator role of PRISm, we subsequently used the RCS method to explore the relationship between the two indicators and the prevalence/incidence of SpA. The RCS curve was plotted with four knots at the 5th, 35th, 65th, and 95th percentiles of FEV1 and FCV levels. Our study found that a nonlinear dose-response relationship between FEV1 %pred and the prevalence of SpA (nonlinear p = 0.012; Supplemental Figure 3) and the incidence of SpA (overall p < 0.001 and nonlinear p = 0.011), while a negative and linear associations between FVC %pred and SpA risk (overall p < 0.001 and nonlinear p = 0.294; Figure 4).

Association between FVC %pred and/or FEV1 %pred and the incidence of SpA.
Discussion
Our cross-sectional study, utilizing a large sample from the UK Biobank, identified an association between lower lung function (FEV1 %pred and FVC %pred) and a higher prevalence of SpA, particularly in the AS subtype within SpA. Specifically, we observed that compared to individuals with normal lung function, those exhibiting PRISm had a 58.8% increased prevalence of SpA. Furthermore, the presence of the HLA-B27 positive mutation intensified these outcomes. Subsequently, we designed a prospective study to evaluate the association between lung function and the risk of SpA incidence (HR (95% CI): 1.249 (1.098–1.420)). In our fully adjusted model, the group with impaired lung function exhibited a substantially higher incidence of SpA compared to the control group. RCS analysis revealed a nonlinear relationship between FEV1 %pred and/or FVC %pred and the risk of developing SpA (overall p < 0.001 and nonlinear p = 0.011).
Baseline data from the cross-sectional analysis indicate that individuals with SpA were more inclined to be of older age, male gender, obese, have lower income levels indicating poor economic status, exhibit a higher rate of current smokers, lower amounts of activities and crucially, have a greater prevalence of comorbidities, with the HLA-B27 mutation emerging as a pivotal factor in the SpA status. Inflammation markers were more elevated in the SpA group than in the control population as well (all p < 0.001).
Impaired pulmonary function demonstrates established links to systemic autoimmune disorders.31,32 While PRISm often presents with lung abnormalities, its transition to COPD creates a precursor state relevant to SpA risk through shared inflammatory pathways.20,33,34 Our data confirm elevated inflammation markers in SpA patients versus controls, aligning with reports that pulmonary impairment elevates circulating inflammatory mediators that drive musculoskeletal pathology.35–37 TNF-α/IL-17-driven inflammation—central to SpA pathogenesis—simultaneously promotes pulmonary parenchymal stiffness via fibroblast activation and aberrant extracellular matrix remodeling.38–41 Our stratified analysis confirmed smoking’s association with elevated SpA risk—likely mediated through inflammation—this relationship operated primarily via pulmonary dysfunction. Smoking-induced mucosal barrier disruption triggers autoantibody production pre-SpA onset, amplifying inflammatory cascades that mechanistically link to disease pathogenesis.42,43
In addition, our study confirms prior reports of nonlinear associations between lung function and adverse outcomes, demonstrating this pattern specifically for FEV %pred in relation to SpA risk, while revealing a linear relationship for FVC% pred.44,45 Notably, our analysis revealed a stronger association between impaired pulmonary function and axSpA, particularly AS, compared to pSpA. This differential risk profile may stem from the characteristic spinal kyphosis in advanced AS. Progressive spinal curvature exacerbates thoracic cage restriction, mechanically compromising lung expansion capacity through reduced chest wall compliance, diaphragmatic compression, and altered pulmonary parenchymal mechanics. This explains why 18%–57% of AS patients develop restrictive ventilatory dysfunction correlating with kyphosis severity. 15
Crucially, our stratified analyses indicate that these lung function-SpA relationships are modulated by PA levels—a finding with significant clinical implications given that most patients with pulmonary impairment experience functional limitations in daily activities. 46 PA not only counteracts these limitations but also actively protects against AS progression by improving symptoms and reducing disease activity through mechanisms like manual soft tissue mobilization and spinal mobility exercises.47–50 While our subgroup analysis supports advocating PA as standard care throughout AS management, optimal exercise dosing requires further investigation. 51 This reinforces the clinical significance of PRISm as a biomarker for identifying at-risk individuals during modifiable pre-symptomatic phases, thereby potentially preventing progression and disability.
Patients with immune-mediated rheumatic diseases (including SpA) face nutritional depletion as well, similarly observed in COPD.52–54 Reduced protein, calcium, and vitamin D intake exacerbates muscle atrophy and osteoporosis, compounding functional limitations from pulmonary impairment.55,56
Emerging research indicates that obstructive lung diseases may be significant pulmonary manifestations of rheumatic diseases, while the risk of SpA is more associated with a restrictive spirometry pattern, such as PRISm, and absence of increased risk in COPD. 57 This differential susceptibility might explain: (1) Disease stage relevance: PRISm represents an early inflammatory phase characterized by mild restrictive ventilatory impairment (FVC <80% predicted), often preceding overt COPD. At this juncture, patients are frequently asymptomatic yet already exhibit elevated SpA susceptibility. While clinical practice typically identifies SpA symptom onset before age 45, UK Biobank data revealed a median diagnosis age of 57 years (IQR: 50–63). This discrepancy likely reflects well-documented diagnostic delays in real-world settings, where the mean interval from symptom onset to diagnosis spans 8–10 years. Our findings underscore PRISm as a critical preclinical window for early SpA detection and intervention, potentially mitigating disease progression and socioeconomic burdens. (2) Distinct pulmonary phenotypes: COPD manifests as obstructive ventilatory dysfunction (post-bronchodilator FEV1/FVC <0.7), where concurrent FVC and FEV1 reductions may mask diagnostic thresholds. PRISm primarily reflects restrictive patterns (isolated FVC reduction), mechanistically linked to SpA via shared inflammatory pathways. (3) Age distribution alignment: SpA predominantly affects younger adults (typically <45 years), coinciding with the PRISm demographic where 20%–50% may progress to COPD later in life. In contrast, COPD peaks in older populations (>60 years) with lower SpA incidence, diminishing its predictive utility for early SpA detection.
The etiology of SpA remains unclear, yet it is known to harbor a significant genetic component, primarily associated with HLA-B27. 58 Over the past few years, our comprehension of genetic susceptibility to SpA, particularly AS, has greatly improved due to findings from comprehensive genome-wide association studies based on single-nucleotide polymorphism arrays. Our analysis indicates that the HLA-B27 mutation, especially the HLA-B*27:02 and HLA-B*27:05 alleles, significantly increases the prevalence and incidence of SpA, with a particularly strong predisposition to AS. HLA-B*27:05 is frequently associated with AS, particularly in Caucasian and American Indian populations, which are also the predominant demographics in the UK Biobank, while HLA-B*27:02 is more prevalent among Mediterranean populations. 59 The mechanism underlying the increased susceptibility to SpA due to HLA-B27 is believed to involve the presentation of an “arthritogenic peptide” to CD8+ T cells, leading to chronic inflammation. Additionally, HLA-B27-induced immunological changes result in the early and sustained expression of CD4+ T cells, such as T-helper 17 cells. 41
For the overall UK Biobank, broad SpA, axSpA, AS, and pSpA were 7.67% (31,568/411,780), 31.46% (1040/3306), 45.72% (823/1800), 46.85% (818/1746), and 15.79% (248/1571), respectively. The overall population rate (7.67%) aligns with published estimates in the UKB study. 5 Despite widely recognition of over 95% of HLA-B27 positive in AS, International Genetics of Ankylosing Spondylitis Consortium (IGAS) 60 reported that HLA-B27 was only accounting for only 20.1% of AS, a proportion lower than that observed in our findings. We assume the lower HLA-B27 positivity was probably due to the incomplete subtype coverage in the UK Biobank, which only tests for six common HLA-B27 subtypes (e.g., B2702–B2707). This approach probably underestimates true positivity rates, given the existence of more than 160 HLA-B27 subtypes globally. 61
Our study is strengthened by a large sample size and rigorous methodology; however, several limitations must be acknowledged. First, the cross-sectional design of the spirometry data may fail to adequately capture the evolving changes in lung function over time. Second, although we adjusted for numerous confounding variables, there remains the possibility of unmeasured confounding factors, including medication data, such as dosage, duration, adherence, and non-captured therapies (e.g., biologics), which may introduce residual bias. Undiagnosed respiratory comorbidities, environmental exposures may as potential contributors to these associations as well. Third, the predominantly Caucasian demographic of the UK Biobank cohort may limit the generalizability of our findings, highlighting the need for research involving more diverse racial groups. Fourth, we recognize that the UK Biobank’s inherent paucity of structured radiological records limits large-scale quantitative analysis of imaging biomarkers (e.g., sacroiliitis grade, syndesmophyte burden). This constraint precluded subgroup stratification by radiographic severity and may introduce unmeasured confounding in pulmonary-SpA associations. Fifth, the limitation in diagnostic validation methodology resulting from the UK Biobank’s absence of secondary confirmation data existed. While our case definition relied on a single ICD-10 code entry and self-reported diagnoses—consistent with established epidemiological practices for large-scale cohorts—we recognize that this approach warrants further refinement to enhance diagnostic precision. Sixth, the existence of a causal relationship remains uncertain. Future research should focus on longitudinal assessments of lung function to clarify the relationship between respiratory health and the risk of SpA.
Conclusion
In summary, our study has identified a significant link between impaired lung function and a heightened risk of SpA. Incorporating routine lung function tests into standard clinical practice could be a valuable strategy for the early identification of high-risk individuals. This methodology not only aids in early detection but also deepens our comprehension of the pathogenesis of SpA, thereby paving the way for the creation of more targeted and effective treatment approaches to improve patient outcomes. Nonetheless, further studies are warranted to ascertain the causality of this relationship.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251371112 – Supplemental material for Association between impaired lung function and risk of spondyloarthritis: a cross-sectional study in the UK Biobank
Supplemental material, sj-docx-1-tab-10.1177_1759720X251371112 for Association between impaired lung function and risk of spondyloarthritis: a cross-sectional study in the UK Biobank by Ying Zhu, Zijian Kang, Chen Zhu, Dajiang Du, Jianzheng Zhang and Qiang Tong in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251371112 – Supplemental material for Association between impaired lung function and risk of spondyloarthritis: a cross-sectional study in the UK Biobank
Supplemental material, sj-docx-2-tab-10.1177_1759720X251371112 for Association between impaired lung function and risk of spondyloarthritis: a cross-sectional study in the UK Biobank by Ying Zhu, Zijian Kang, Chen Zhu, Dajiang Du, Jianzheng Zhang and Qiang Tong in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
Our thanks to all the study participants and the anonymous editors and reviewers for their constructive feedback, which has significantly enhanced the quality of this publication.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
