Abstract
Background:
Axial spondyloarthritis (axSpA) is a chronic inflammatory disease primarily affecting the spine and sacroiliac joints, potentially leading to pain, stiffness and disability. Despite diagnostic advances, delays persist. Orthopaedic physicians, often the first specialists consulted for back pain, play a crucial role in early detection and referral to rheumatologists.
Objectives:
To evaluate orthopaedic physicians’ awareness and management of axSpA and factors that might affect appropriate management.
Design:
An online nationwide survey was conducted among orthopaedic physicians in Germany to assess their axSpA knowledge, diagnostic and therapeutic practices and referral behaviours.
Methods:
Descriptive statistics were used to summarize participant characteristics and management practices. Machine learning and subgroup analyses identified factors influencing clinical practices.
Results:
Among 103 orthopaedic physicians (mean age 49.2 ± 11.4 years; 46.6% female), 48.5% practised in both conservative and surgical settings, and 20% held additional qualifications in orthopaedic rheumatology. While 92% regularly treated chronic back pain, only 11.0% estimated the prevalence of inflammatory back pain (IBP) at ⩾5%. Human leucocyte antigen (HLA)-B27 testing was always used by 17.5%, magnetic resonance imaging (MRI) by 14.6% and IBP classification criteria by 22.5%. Nonsteroidal anti-inflammatory drugs were the most common treatment (77%), while 72.1% never used biological disease-modifying antirheumatic drugs. Digital health applications were rarely recommended (14.6%). The majority of physicians (63%) directly referred suspected axSpA cases to rheumatologists, 29% collaborated with rheumatologists and 12% managed cases independently. Referral barriers included long waiting times (66%) and limited appointment availability (33%), while timely appointments (59%) and better referral knowledge (58%) facilitated referrals. Higher self-reported axSpA knowledge was associated with attending ⩾2 rheumatology seminars, conservative orthopaedics settings, regular HLA-B27 testing and familiarity with axSpA MRI results.
Conclusion:
This study reveals substantial opportunities to improve axSpA awareness, management and referrals among orthopaedic physicians. Targeted education and streamlined referral systems, including easier access to rheumatology appointments, could enable earlier diagnosis and better axSpA management.
Plain language summary
This study shows there are many ways to improve how orthopaedic doctors identify, treat, and refer patients with axSpA. Providing better education and making it easier to refer patients to rheumatologists could help diagnose axSpA earlier and manage it more effectively.
Keywords
Introduction
Axial spondyloarthritis (axSpA) is a chronic inflammatory disease primarily affecting the axial skeleton, notably the spine and sacroiliac joints, leading to persistent pain, stiffness and functional impairment. 1 Beyond its axial manifestations, axSpA can present with peripheral manifestations such as arthritis, dactylitis and enthesitis. Moreover, extra-musculoskeletal manifestations, including inflammatory bowel disease, uveitis and psoriasis, significantly impact patients’ quality of life and complicate disease management. If left untreated, axSpA may progress to irreversible structural damage and disability, emphasizing the necessity of early diagnosis and timely intervention to prevent disease progression. 2
Despite significant advancements in magnetic resonance imaging (MRI) for early detection of axial inflammation and the introduction of classification criteria for non-radiographic axSpA, diagnostic delays remain challenging. 3 Educational programmes and referral strategies mainly targeted at primary care physicians have significantly reduced the time from symptom onset to diagnosis over the past two decades.3–6
In spite of these advances, recent data reveal an average diagnostic delay of 5.7 years in Germany and 7.4 years globally, posing substantial risks to long-term patient outcomes.4,7
This persistent delay in diagnosis underscores the need for enhanced healthcare provider education and optimized referral pathways to facilitate earlier recognition and management of axSpA. A major contributor to these diagnostic delays is the frequent misinterpretation of axSpA symptoms, especially in early disease stages.7–9 Chronic back pain, a hallmark of early axSpA, typically affects the sacroiliac joints or spine and may persist for weeks or months. However, degenerative spinal conditions such as osteochondrosis or disc degeneration also cause pain in these regions. This frequently leads to misinterpretation. Physicians unfamiliar with the distinct features of inflammatory back pain (IBP) and axSpA-specific clinical signs may misclassify symptoms as mechanical or degenerative in origin, leading to delayed care.10,11
Studies from the USA, UK and South Korea highlight insufficient awareness of axSpA diagnostic criteria among non-rheumatologic physicians,7,12,13 particularly concerning the distinction between inflammatory and mechanical back pain.14–16 Given their frequent contact with patients experiencing back pain, orthopaedic physicians are pivotal in the early detection and referral of axSpA cases. Because different levels of axSpA awareness and diagnostic competence in orthopaedic physicians could potentially influence both their referral behaviour and use of diagnostic tools, we designed an exploratory study to systematically evaluate orthopaedic physicians’ knowledge, diagnostic practices and clinical management of patients with potential axSpA. We hypothesized that there would be substantial variance in the management of potential axSpA patients among orthopaedic physicians and that provider-level factors could influence axSpA management practices.
This study employed an interview-based approach using a questionnaire developed by experts in rheumatology and orthopaedics, informed by the existing literature. It aimed to identify structural and knowledge-based barriers and facilitators influencing referrals to rheumatologists. Furthermore, the study assessed the utilization and perception of digital health tools, including digital health applications and telemedicine, as potential enablers to enhance axSpA care. To uncover hidden patterns and associations in our dataset that traditional methods might miss, we applied automated machine learning (AutoML), a powerful tool from recent advances in artificial intelligence, providing deeper insights into underlying patterns.17–19
Methods
Design
An anonymous, cross-sectional online survey was conducted among orthopaedic specialists in Germany between March 16, 2023, and March 1, 2024, using a convenience sampling approach. The survey was open and voluntary, and participation was anonymous. Eligible participants were orthopaedic physicians currently living in Germany, proficient in German, and with sufficient digital literacy to complete an online questionnaire. The reporting of this study conforms to the Consensus Based Checklist for Reporting of Survey Studies (CHERRIES) statement. 20
Based on existing literature and clinical expertise, a comprehensive 30-item questionnaire was developed and refined through cognitive debriefing with 12 orthopaedic physicians from diverse demographics and clinical settings. The ‘think aloud’ technique and predefined as well as ad hoc questions ensured that all items were clear, appropriate and relevant. This iterative process enhanced the questionnaire’s clarity and content validity.
Designed by an interdisciplinary team of rheumatology and orthopaedic experts, with input from experienced healthcare researchers (M.O., A.F.), the final web-based survey covered topics such as physicians’ demographics, professional settings, educational practices and resources, patient population characteristics, awareness and knowledge of axSpA symptoms, management strategies for suspected axSpA, referral behaviours, barriers and facilitators to referral and the use and perception of digital health tools (translated questionnaire available in Supplemental Material 1).
Depending on the item, response formats included Likert scales, numeric rating scales, multiple-choice formats and free-text fields. Frequency was assessed using 5-point Likert scales (‘never’ to ‘always’), while attitudes and preferences were measured on agreement scales (‘strongly disagree’ to ‘strongly agree’). Multiple-choice questions permitted single or multiple responses as appropriate.
Orthopaedic physicians in private practices and hospitals across Germany were invited to participate anonymously through survey links disseminated via newsletters and websites of major orthopaedic associations. All participants provided informed consent through an introductory text that explained the study’s purpose, the investigators involved, the estimated time required and the procedures for data handling. The survey introduction provided complete information on the study’s purpose, estimated time required, data handling procedures and contact details for the research team. Data collection and storage adhered to institutional and legal standards for anonymity and data protection. A detailed overview of the survey methodology and implementation is available in Supplemental Table 1, which presents the CHERRIES checklist for reporting of survey studies.
Statistical analysis
No formal sample size calculation was conducted due to the exploratory nature of this study; all survey responses were included in the analysis. Descriptive statistics summarized the data, with means and standard deviations calculated for continuous variables and percentages for categorical data. Data analysis and visualization were performed using GraphPad PRISM (version 10.0, GraphPad Software, LLC, San Diego, CA, USA) and R software (R Core Team, Vienna, Austria) with packages such as ggplot2, dplyr and tidyr. In addition to descriptive analyses and machine learning (ML) models, we performed supplementary subgroup analyses to further explore key clinical and demographic differences. Mann–Whitney U tests were used for continuous or ordinal outcomes, and Chi-square tests were applied for categorical comparisons. These analyses focused on the primary outcome variables (axSpA knowledge, use of IBP classification criteria, MRI use and digital health application use), stratified by age group, practice setting and clinical specialization. Statistical significance was defined as p < 0.05. Figures were generated in R using the ggplot2 and ggpubr packages, with significance levels visualized using standard notation (*, **, ***).
ML models were employed to identify factors influencing diagnostic practices and referral behaviour, with performance metrics focusing on geographic location and professional specialization.
Exploratory data analysis, model validation and building and feature impact
DataRobot’s AutoML platform was used for exploratory data analysis (EDA), model building and feature evaluation. EDA provided key descriptive statistics and identified data quality issues, ensuring a robust foundation for modelling. Various supervised learning algorithms, including ensemble methods, were tested. The AutoML platform enabled systematic model development with stratified sampling, fivefold cross-validation and an independent holdout set. Model performance was validated using cross-validation and holdout sets, using the area under the curve (AUC) as the primary metric. For binary classification (Target 1), the Light Gradient Boosting model, based on ElasticNet predictions, achieved a cross-validation AUC of 0.77 and LogLoss of 0.79. Regression models showed moderate to strong performance: Target 2 (regularized logistic regression) had a root mean square error of 1.56; Target 3 (gradient boosted trees) explained 49.9% of variance (R2 = 0.50). In multiclass classification (Targets 4–6), AUCs varied, with Target 5 performing best (AUC = 0.81), while Target 4 showed limited performance (AUC = 0.46).
Feature impact analysis quantified how individual variables influenced model predictions, offering insights into critical factors affecting diagnostic and referral practices. The results can be interpreted through partial dependence, focusing on the specific model target and the feature under analysis. For clarity in binary features, ‘yes’ is represented by ‘1’ and ‘no’ by ‘0’. Further methodological details are available in the Supplemental Material 2 and previous publications.17,18
Results
Sociodemographic data and practice setting
A total of 103 orthopaedic physicians participated in the survey, with a mean age of 49.2 years (±11.4). Of these, 46.6% were female and 53.4% male (Table 1). Notably, 41.7% had over 20 years of experience as board-certified orthopaedic specialists. Regarding practice setting, 36.9% practiced in a conservative (non-surgical) setting, while 14.6% were involved in surgical orthopaedics, with almost half (48.5%) practising in both fields. Twenty percent (21/103) reported having an additional qualification in orthopaedic rheumatology. The majority (54.4%) worked in private practices, 36.9% in hospitals and 8.7% divided their time between both settings. Geographically, 49.5% were based in urban areas (population ⩾100,000) and 50.5% in rural regions. Younger physicians (⩽40 years) were more frequently located in urban settings (60% vs 40% in rural areas). ML analysis indicated that physicians working in both clinics and private practices (AUC: 0.84; Relative Feature Impact: 1.00) and those specializing in conservative orthopaedics (AUC: 0.77; Relative Feature Impact: 0.64) were predominantly located in urban areas.
Sociodemographic characteristics and clinical background of respondents.

SD, standard deviation.
Incidence and management of back pain in axSpA
The survey results highlighted diverse approaches to managing chronic back pain and axSpA. Specifically, 34% of respondents treated more than 30 back pain patients per week, 35% managed 11–30 patients and 26% saw 1–10 patients weekly. Only 8% reported not treating any back pain patients (Figure 1(a)). Despite this, the reported prevalence of IBP – a key feature of axSpA – was relatively low: 15% of practitioners reported no IBP cases, while 37% estimated the prevalence to be less than 1% and only 1% of respondents indicated that IBP affected more than 10% of their patients (Figure 1(b)).

Estimated incidence of back pain and axSpA management practices. Self-reported data from orthopaedic physicians (n = 103) by questionnaire. (a) Estimated frequency of back pain patients per week. (b) Estimated proportion of IBP cases. (c) Referral patterns for management of suspected axSpA. (d) Diagnostic modalities used for suspected axSpA. (e) Use of IBP classification criteria and proportion of axSpA cases treated by respondents. (f) Therapies used by respondents treating axSpA independently (n = 61). (g) Use of prescription-based digital health applications. Pre-defined response options were provided for categorical responses.
Management strategies varied, with 63% of respondents referring axSpA patients directly to rheumatologists, 29% managing them collaboratively with rheumatologists and 12% opting for independent diagnosis and treatment (Figure 1(c)).
The diagnostic strategies applied by orthopaedic physicians for axSpA included in the category ‘always used’ were as follows: medical history (85.4%), laboratory values (53.4%) and human leucocyte antigen (HLA)-B27 testing (17.5%; Figure 1(d)). MRI was reported as ‘always used’ by a mere 14.6%, although 29.1% reported that it was ‘almost always’ used. X-rays were indicated as ‘almost always’ by 33.0%. Formal IBP classification criteria (e.g., Calin, Berlin, Assessment of SpondyloArthritis International Society (ASAS)) were used by only 22.5% (Figure 1(e)).
Regarding treatment, 38.4% of respondents indicated utilizing both pharmacological and non-pharmacological therapies for axSpA, while 61.4% stated that they do not treat the condition themselves (Figure 1(e)). Nonsteroidal anti-inflammatory drugs (NSAIDs) were the most prevalent therapy, with 42.6% reporting their ‘always’ use and 34.4% indicating their ‘almost always’ use (Figure 1(f)). Physiotherapy was similarly widely reported as ‘always used’ by 36.1% and ‘almost always used’ by 45.9%. In contrast, other options were rarely employed, particularly systemic glucocorticoids (3.3% ‘almost always’), conventional synthetic and targeted synthetic disease-modifying antirheumatic drugs (csDMARDs; 59% ‘never’) and biological disease-modifying antirheumatic drugs (bDMARDs; 72.1% ‘never’). Surgery was rarely performed (49.2% ‘never’, 36.1% ‘almost never’). Additional therapies, provided as free-text responses, included psychological therapy, rehabilitation exercise programmes, functional training, physiotherapy and sports therapy counselling.
When orthopaedic physicians were asked about the utilization of digital health applications, 1.0% reported using them for personal purposes, 13.6% for direct patient care and 14.6% recommended them to their patients (Figure 1(g)).
Barriers, facilitators and referral dynamics for axSpA
Key barriers to referring patients with suspected axSpA to a rheumatologist included long waiting times for rheumatology appointments (66%), limited appointment availability (33%), lack of clarity on referral pathways (24%) and long travel distances to rheumatologists (21%; Figure 2(a)). Free-text responses highlighted issues such as delays in securing rheumatology appointments, even for cases that were thoroughly pre-diagnosed by orthopaedic physicians or referred by general practitioners. The most frequently cited facilitators were timely appointments (59%), enhanced knowledge of referral processes (58%), strengthened collaboration with rheumatologists (56%) and higher expertise in recognizing axSpA (43%), with the latter two being less prominent in rural areas according to ML analysis. Telemedicine was selected as a facilitator by only 20% of respondents (Figure 2(b)). ML analysis showed that those working in private practice reported being more familiar with referral pathways (Model AUC in Cross Validation: 0.84; Relative Feature Impact: 0.37) and reported stronger collaboration with rheumatologists (Model AUC in Cross Validation: 0.84; Relative Feature Impact: 0.36).
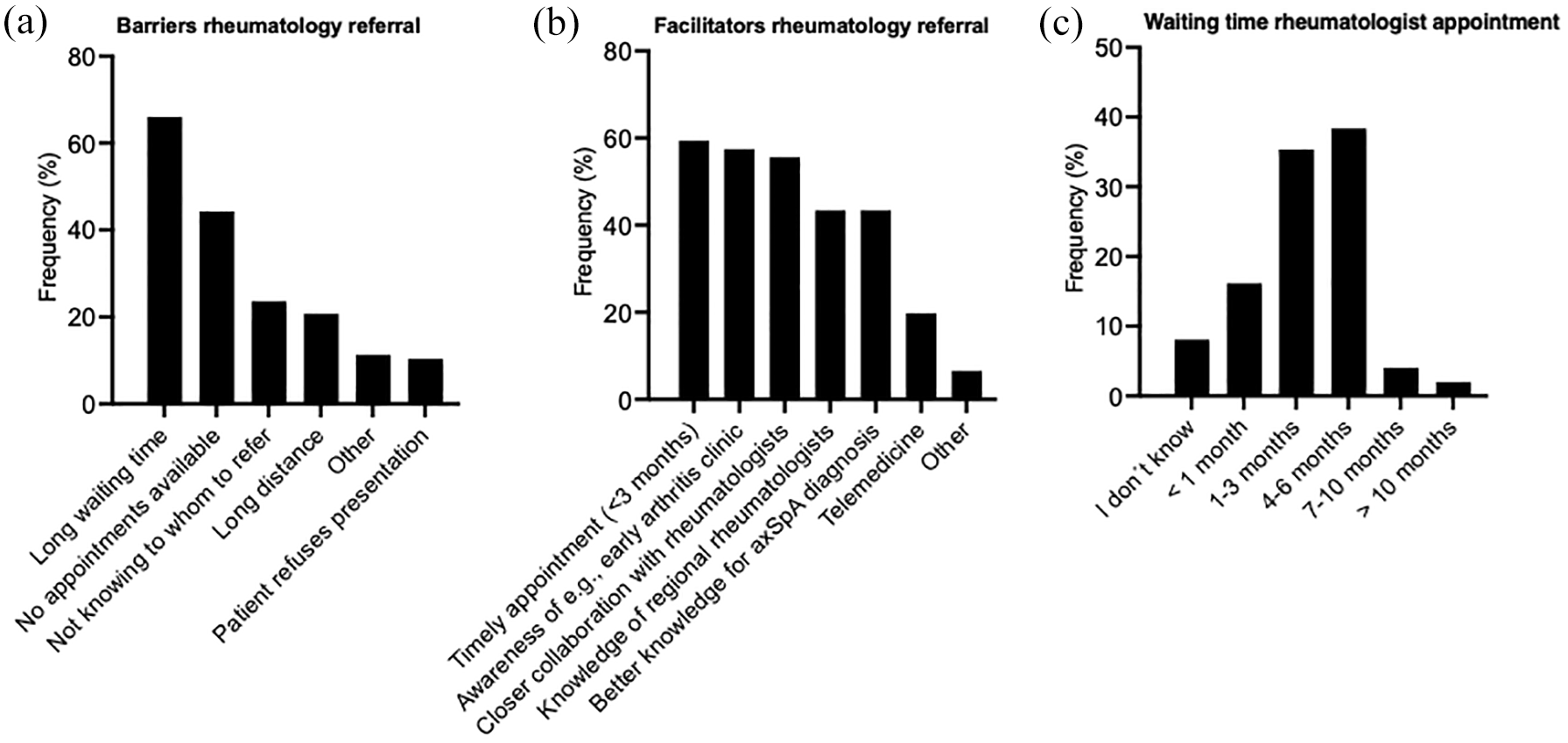
Barriers, facilitators and waiting times for referral of patients with suspected axSpA to rheumatologists. Data are derived from questionnaire responses by orthopaedic practitioners (n = 103). (a) Reported barriers to referral of patients with suspected axSpA to rheumatologists. (b) Factors identified as facilitators for improving referral processes and care of patients with suspected axSpA. (c) Average waiting times for patients to secure appointments with rheumatologists.
Knowledge, learning resources and axSpA-related educational events
Respondents rated their knowledge of IBP as an average of 5.9 (±2.1) and their knowledge of axSpA as 6.1 (±2.1) on a scale from 0 to 10, with scores of 5/10 and 7/10 being the most common for both conditions (Figure 3(a)). ML analysis revealed a strong association between higher self-rated axSpA knowledge and physicians working in both clinics and private practices. Regarding educational resources, the most utilized were in-person congresses (67%), scientific journals (56%), textbooks (55%) and online training (45%), while less commonly used resources included flyers (6%), apps (3%) and other options (2%). Only 2% reported not using any educational resources.
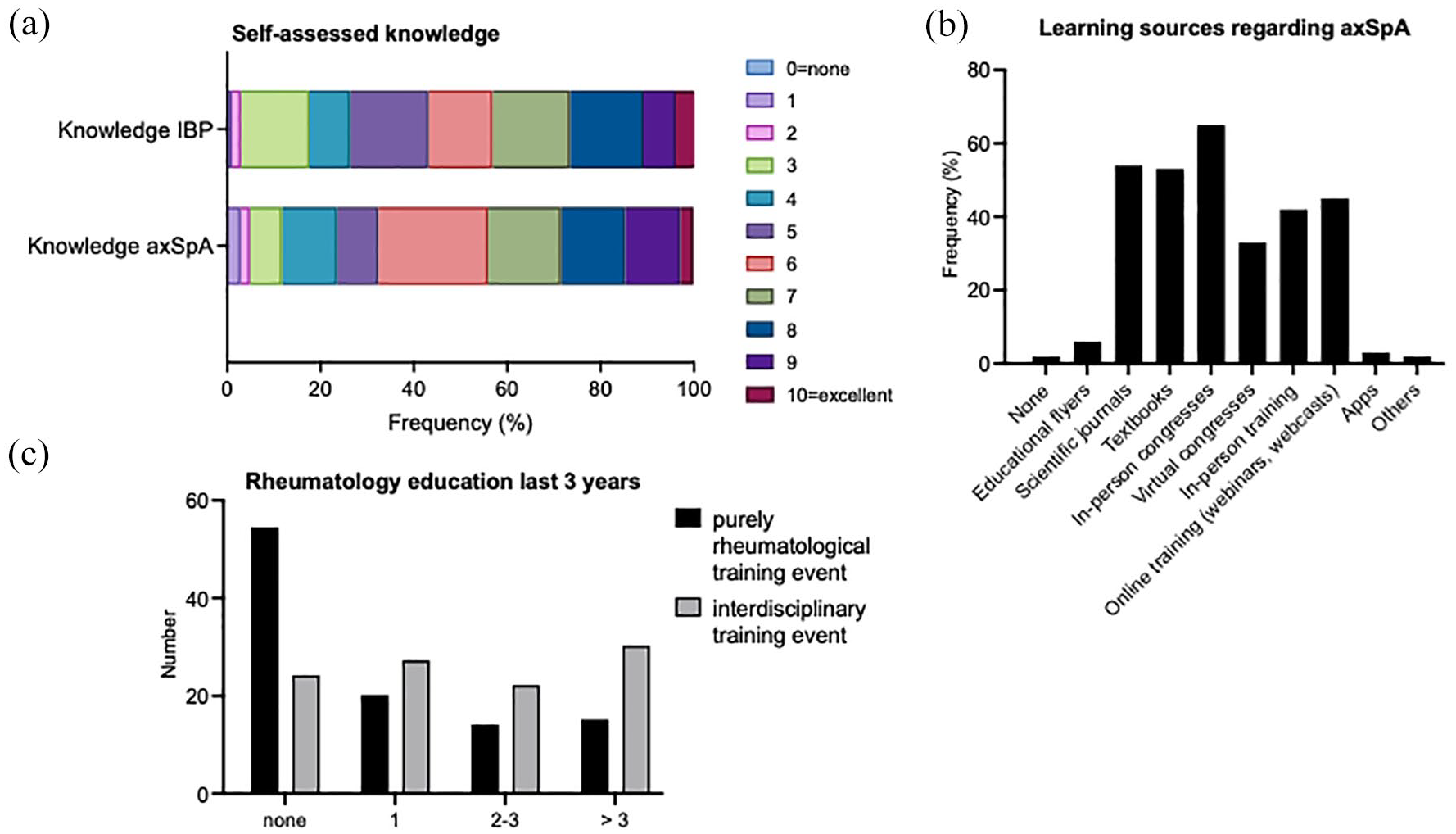
Knowledge, learning resources and rheumatology-related educational events related to axSpA. (a) Respondents’ self-assessed knowledge of inflammatory back pain and axSpA. (b) Resources used to learn about axSpA (others included interdisciplinary rheumatology case discussions, UpToDate (UpToDate, Inc., Waltham, MA, USA)). (c) Attendance at rheumatology-related educational events in the last 3 years.
More than half (52%) of the respondents reported not attending any rheumatology training seminars. In the past 3 years, 19% attended one seminar, 14% attended two to three and 15% attended more than three. Higher self-reported knowledge of IBP and axSpA was associated with attending ⩾2 rheumatology training seminars (Feature Impact: 0.84 for IBP and 1.00 for axSpA), specializing in conservative orthopaedics (Feature Impact: 0.53 for axSpA), and regular use of HLA-B27 testing (Feature Impact: 0.69 for IBP and 0.10 for axSpA) and rating the relevance of active sacroiliitis on MRI as very high (10/10; Feature Impact: 0.57 for IBP and 0.04 for axSpA). Self-rated knowledge levels were also higher among physicians born before 1966 for IBP and before 1972 for axSpA.
Subgroup analyses of the impact of provider-level factors on patient management
To further explore differences across demographic and clinical subgroups, we performed additional subgroup analyses of the primary outcomes. Age-based comparisons showed higher axSpA knowledge scores among participants aged ⩽40 years (Supplemental Figure 1(A)), as well as more frequent use of IBP classification criteria (Supplemental Figure 3(B)), while MRI use did not differ significantly by age (Supplemental Figure 3(C)). Use of digital health applications for personal purposes was markedly higher in the younger group (Supplemental Figure 3(D)).
With respect to practice location, no significant differences were observed in axSpA knowledge (Supplemental Figure 3(E)), use of IBP criteria (Supplemental Figure 3(F)) or MRI use (Supplemental Figure 3(G)), although digital app use showed a non-significant trend towards greater uptake in urban settings (Supplemental Figure 3(H)).
Regarding clinical specialization, axSpA knowledge did not differ between physicians focused on conservative care and those also performing surgery (Supplemental Figure 3(I)). However, IBP classification criteria (Supplemental Figure 3(J)) and MRI (Supplemental Figure 3(K)) were used more frequently by those with surgical or mixed-care roles. No significant difference in digital health application use was observed by specialization (Supplemental Figure 3(L)).
A strong positive correlation was observed between self-reported knowledge of IBP and the use of classification criteria (ρ = 0.85). Moderate correlations were also observed between knowledge and frequency of IBP diagnosis (ρ = 0.41), as well as between use of criteria and frequency of diagnosis (ρ = 0.32), supporting the influence of familiarity with classification tools on diagnostic outcomes (see Supplemental Figure 1(M)–(O)).
Discussion
This study underscores significant opportunities to improve awareness, diagnostic practices and referral pathways for axSpA among orthopaedic physicians in Germany. Despite advancements, the diagnosis of axSpA remains substantially delayed – averaging up to 5.7 years – which aligns with global trends, particularly in healthcare settings lacking direct rheumatological involvement.21–23 Our findings offer critical insights into axSpA management by orthopaedic specialists and highlight key areas for targeted educational outreach.
A key strength of our study is the use of an integrated approach to assess awareness and referral behaviour, including AutoML to detect complex patterns beyond conventional analysis. The goal of this exploratory study was not outcome prediction but rather pattern discovery beyond traditional methods; hence, these metrics should be interpreted cautiously. In particular, smaller sample size likely contributed to reduced holdout performance, highlighting the need for larger datasets and prospective validation to confirm real-world applicability.
The survey of 103 orthopaedic physicians revealed a diverse demographic, geographic distribution and professional focus, providing a representative overview of the current landscape in Germany. Younger orthopaedic surgeons, those practising in both hospital and private settings and those specializing in conservative orthopaedics were predominantly located in urban areas, reflecting established healthcare access trends.
The results of this survey confirmed our hypothesis of variability in orthopaedic specialists’ management of potential axSpA patients as well as the impact of provider-level factors on management practices. Although most respondents reported managing chronic back pain, the recognition of IBP – a hallmark of axSpA – was notably limited. The majority estimated its prevalence to be less than 1% among their back pain patients, contrasting with previous reports indicating a prevalence of 7.7% using ASAS criteria, 13.5% with Calin criteria and 15.4% with Berlin criteria. 24 This discrepancy may be attributable to referral biases related to a lower tendency of general practitioners to refer suspected axSpA cases to orthopaedic physicians, resulting in an underrepresentation of IBP in orthopaedic practices. However, it may also reflect a true underestimation of IBP among patients with back pain, perhaps due to diagnostic uncertainty, overlap with mechanical back pain symptoms or limited familiarity with classification criteria.
Diagnostic practices indicated potential gaps. The relatively low utilization of MRI and HLA-B27 testing – both essential for axSpA diagnosis – and a partial reliance on rather outdated methods like scintigraphy instead of the standardized ASAS recommendations may contribute to diagnostic delays. Additionally, while bDMARDs were rated as highly relevant for axSpA management, csDMARDs were also rated highly despite their limited efficacy in this condition, contrary to ASAS-EULAR recommendations. 25 This mirrors findings from other studies highlighting diagnostic challenges and conservative treatment approaches for axSpA in non-rheumatology specialities26,27 and emphasizes the need for improved education on diagnostic criteria and evidence-based treatments.
AutoML analysis revealed that private practitioners were more likely to have established referral networks with rheumatologists and stronger interdisciplinary collaboration (Relative Feature Impact: 0.37 for knowledge of referral pathways, 0.36 for collaboration). This suggests that orthopaedic physicians in private practice have developed networks that hospital-based physicians without rheumatology departments may lack. Closer collaboration with rheumatologists in the vicinity may have the potential to improve patient care.
Physicians aged 40 and under reported lower levels of axSpA knowledge, potentially due to limited time to gain experience or shifts in medical curricula with reduced emphasis on rheumatological education.28–30 As reported in the literature, attendance at two or more rheumatology-specific training sessions was associated with improved self-reported axSpA knowledge, highlighting the importance of targeted education in improving diagnostic accuracy. 31 Gender differences also emerged: male physicians preferred in-person educational events and direct referrals to rheumatologists, as identified by AutoML (Regularized Logistic Regression (L2); Model AUC: 0.65; Feature Impact: 0.25), aligning with prior studies on gender-based differences in medical education and referral practice.32–34 This highlights the need for diverse, customizable educational offerings to accommodate varying preferences.
To further contextualize observed disparities, we performed subgroup analyses of key outcomes across physician demographics and clinical roles (Supplemental Figure 3(A)–(L)). In addition to descriptive and model-based analyses, subgroup analyses provided a more nuanced view of the data. Trends across age, experience, practice setting and specialization revealed interesting exploratory findings that complement the main results and may help tailor future educational and organizational strategies. While digital health application use was more common in urban settings and among younger physicians, these trends did not reach statistical significance, perhaps due to the sample size. Similarly, differences in axSpA knowledge and diagnostic tool use by experience level and practice location suggest potential structural and experiential influences, although statistical significance was not observed.
Barriers to rheumatology referrals were prominent, with long waiting times (66%) and limited appointment availability (33%) frequently reported, particularly in rural areas. Facilitators included timely appointments (59%) and closer collaboration with rheumatologists (58%). Enhanced interdisciplinary collaboration, identified as a key facilitator by 56% of respondents, was associated with greater diagnostic and referral confidence, reinforcing its importance for early axSpA detection and management. 35 AutoML analysis further revealed that orthopaedic physicians with higher referral confidence had established referral pathways (Partial Dependence: ‘0’=0.57/‘1’=0.65), highlighting the need for streamlined referral processes.
A positive association was found between higher levels of self-reported knowledge of axSpA and attendance at rheumatology-specific training, routine HLA-B27 testing and familiarity with MRI findings of axSpA. These findings provide strong support for the impact of education on improved axSpA management. It is therefore noteworthy that 52% of respondents had not attended rheumatology training in the past 3 years, indicating a need for enhanced continuing education. Furthermore, digital tools, health applications and telemedicine were used infrequently, with only 14.6% of respondents recommending them to patients. The broader integration of digital tools into clinical practice holds significant potential to enhance diagnostic accuracy and patient monitoring, particularly in underserved regions.
Taken together, the data suggest an opportunity to enhance axSpA awareness among orthopaedic physicians in Germany. Our findings are consistent with and expand upon previous international research. Mathieson et al. 36 and Yong et al. 37 reported limited awareness of IBP, non-radiographic axSpA and extra-articular features among secondary care specialists – including orthopaedic, ophthalmology, emergency, gastroenterology and dermatology physicians – as well as among chiropractors and osteopaths in the UK. These findings align with our observations, including lower self-assessed IBP knowledge and limited recent participation in rheumatology-specific education, which point to some training needs. In addition, our survey showed a greater reliance on radiography over MRI for evaluation of back pain, despite current guideline recommendations favouring MRI for early axSpA detection. Similarly, Magrey et al. 38 identified diagnostic delays driven in part by underuse of imaging and suboptimal referral processes. In agreement with our study, these authors also emphasized the importance of timely referral to rheumatologists and concluded that greater awareness among primary care providers could improve early diagnosis and patient outcomes. These shared findings underscore the broader need for targeted cross-specialty training, better access to diagnostic tools and more efficient referral pathways. Focused educational initiatives for orthopaedic physicians – such as incorporating axSpA modules into orthopaedic training and offering regular rheumatology workshops – could significantly improve early identification and referral of axSpA patients.
Limitations of this study include the self-selected sample, which may have resulted in underrepresentation of certain subgroups and may have also introduced other forms of bias, such as selection bias towards physicians with a greater interest or knowledge of axSpA. In addition, the study relied on self-reported data without external validation of actual axSpA knowledge and diagnostic accuracy. Subgroup analyses were exploratory and unadjusted for multiple testing. They should therefore be considered hypothesis-generating and interpreted with caution.
Despite these limitations, the findings provide valuable insights into the role of orthopaedic physicians as initial points of contact for undiagnosed axSpA patients. The identified factors inform strategies to optimize targeted education, raise awareness and improve diagnostic referral practices, ultimately reducing diagnostic delays and enhancing patient outcomes.
Conclusion
This study highlights critical gaps in the recognition and management of axSpA among orthopaedic physicians, with limited use of classification criteria and advanced diagnostics. AutoML analyses identified specific subgroups – such as those working across sectors or attending rheumatology training – as more likely to apply evidence-based strategies. To reduce diagnostic delays, targeted Continuing Medical Education programmes tailored to practice settings and clinical roles are needed. Clear referral pathways and stronger interdisciplinary collaboration should be established, especially in rural areas.
Moreover, the underuse of digital tools suggests untapped potential for scalable solutions. Institutional and political stakeholders are urged to invest in structured educational initiatives and incentivize integrated care models to improve early diagnosis and patient outcomes in axSpA.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251360162 – Supplemental material for Orthopaedic physicians’ awareness, diagnostic challenges and referral barriers in axial spondyloarthritis: insights from a nationwide survey in Germany
Supplemental material, sj-docx-1-tab-10.1177_1759720X251360162 for Orthopaedic physicians’ awareness, diagnostic challenges and referral barriers in axial spondyloarthritis: insights from a nationwide survey in Germany by Fabian Proft, Angela Patricia Moissl, Natalia Kirsten, Alexander Pfeil, David Simon, Igor Bibi, Victor Olsavszky, Anastasia Fleyder, Marina Otten, Milena Pachowsky and Jan Leipe in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251360162 – Supplemental material for Orthopaedic physicians’ awareness, diagnostic challenges and referral barriers in axial spondyloarthritis: insights from a nationwide survey in Germany
Supplemental material, sj-docx-2-tab-10.1177_1759720X251360162 for Orthopaedic physicians’ awareness, diagnostic challenges and referral barriers in axial spondyloarthritis: insights from a nationwide survey in Germany by Fabian Proft, Angela Patricia Moissl, Natalia Kirsten, Alexander Pfeil, David Simon, Igor Bibi, Victor Olsavszky, Anastasia Fleyder, Marina Otten, Milena Pachowsky and Jan Leipe in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-3-tab-10.1177_1759720X251360162 – Supplemental material for Orthopaedic physicians’ awareness, diagnostic challenges and referral barriers in axial spondyloarthritis: insights from a nationwide survey in Germany
Supplemental material, sj-docx-3-tab-10.1177_1759720X251360162 for Orthopaedic physicians’ awareness, diagnostic challenges and referral barriers in axial spondyloarthritis: insights from a nationwide survey in Germany by Fabian Proft, Angela Patricia Moissl, Natalia Kirsten, Alexander Pfeil, David Simon, Igor Bibi, Victor Olsavszky, Anastasia Fleyder, Marina Otten, Milena Pachowsky and Jan Leipe in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
We thank all participants for their valuable contribution and participation in the survey.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
