Abstract
Background:
Non-pharmacological interventions (NPIs) are recommended in current international clinical practice guidelines for the management of axial spondyloarthritis (axSpA) as a complementary therapy.
Objectives:
To assess the comparative impact of different NPIs on the clinical outcomes of patients with axSpA.
Design:
Network meta-analysis and traditional meta-analysis of randomized controlled trials (RCTs).
Data sources and methods:
We searched systematically PubMed, EMBASE, the Cochrane Library, Web of Science, and Chinese Biological Medical Database up to May 2023, and included these RCTs of patients with axSpA receiving the managements of NPIs. The risk of bias of individual RCT was assessed through a modified version of the Cochrane Risk of Bias Tool. Bayesian random-effects network meta-analysis and traditional meta-analysis were performed to calculate the mean difference.
Results:
A total of 51 RCTs involving 3457 patients with axSpA were included in this study. Both supervised combined exercises and neuromuscular training showed a significant reduction in disease activity (Bath Ankylosing Spondylitis Disease Activity Index (BASDAI): 1.13 lower, 1.17 lower, respectively), physical function (Bath Ankylosing Spondylitis Functional Index (BASFI): 1.0 lower, 0.88 lower, respectively), and spinal mobility (Bath Ankylosing Spondylitis Metrology Index (BASMI): 0.7 lower, 1.35 lower, respectively), compared to standard care. Both supervised combined exercises and neuromuscular training also presented more significant improvement than conventional exercises in disease activity (BASDAI: 0.91 lower, 0.95 lower, respectively), physical function (BASFI: 0.67 lower, 0.56 lower, respectively), and spinal mobility (BASMI: 0.18 lower, 0.82 lower, respectively). A traditional meta-analysis demonstrated that supervised combined exercise and neuromuscular training could significantly reduce BASDAI, BASFI, and BASMI scores when compared to unsupervised exercise or standard care. Aerobic exercise, supervised combined exercise, and conventional exercise could significantly reduce pain scores, compared to standard care.
Conclusion:
Compared to either standard care or conventional exercises, supervised combined exercises and neuromuscular training were more beneficial for ameliorating disease activity, physical function, and spinal mobility in patients with axSpA. The choice of exercise modalities may depend on patient values and preferences.
Trial registration:
PROSPRO: CRD42021251219.
Introduction
Axial spondyloarthritis (axSpA) is a chronic inflammatory disease that predominantly involves the spine and the sacroiliac joints, seriously impairing physical function and reducing patients’ quality of life. 1 For patients with axSpA, the treatments are expected to alleviate symptoms, improve spinal flexibility, ameliorate physical function, and maintain the ability to live and work. The primary treatment for axSpA is pharmacotherapy, including non-steroidal anti-inflammatory drugs, synthetic targeted drugs such as JAK inhibitors or biological synthetic disease-modifying anti-rheumatic drugs, which can provide clinically important improvement via alleviating inflammation and symptoms.2,3 However, some patients with axSpA do not respond well to pharmacological treatments or may experience relapse after drug tapering, and concerns have been raised about the safety of these pharmacotherapies, particularly risks of gastrointestinal bleeding, malignancy, and infection. 4 Therefore, it is very important to explore the complementary and alternative treatments of axSpA to assist pharmacotherapy.
Believed to be safe with a few side effects, non-pharmacological interventions (NPIs), especially physical exercise, have been recommended in some clinical practice guidelines as a co-intervention in combination with pharmacological interventions to treat patients with axSpA throughout the disease course. 5 Some guidelines suggested that exercise for patients with axSpA appeared to be more effective than for other types of arthritis, solidifying its role as a cornerstone of treatment. 6 The ASAS quality standard, developed to enhance healthcare quality for patients with axSpA, proposed two specific quality indicators focused on NPIs: one aimed at exercise and another at education and self-management, which highlighted the importance of these therapies. 7
A systematic literature review that informed the 2022 update of the ASAS/EULAR recommendations for the management of axSpA suggested that education and exercise were effective interventions for axSpA. 7 Recent evidence has assessed the paradoxical effects of different types of exercises in patients with axSpA but lacked specific information to guide exercise strategies in clinical practice. Most studies have focused on mobility exercise, with limited emphasis on other aspects of exercise program design such as neuromuscular training, balance, or cardiorespiratory exercise. This gap persists despite the recognition that axSpA may impair cardiorespiratory function, muscle strength, and balance.
Network meta-analysis allows a unified coherent analysis of all randomized controlled trials (RCTs) comparing different NPI modalities head-to-head or with a control intervention, while fully preserving randomization. Not all NPIs are effective for axSpA, highlighting the importance of investigating the effectiveness of different NPIs. Therefore, we aimed to compare different NPI modalities recommended in axSpA clinical guidelines for improving clinical outcomes in patients with axSpA through Bayesian random-effects network meta-analysis and traditional meta-analysis.
Methods
This study protocol has been registered in the International Prospective Register of Systematic Reviews (PROSPRO: CRD42021251219). This meta-analysis was developed based on guidance from the Preferred Reporting Items for Systematic Reviews and Meta-Analysis Protocols (PRISMA-P) statement. 8
Data sources
Systematic literature searches were performed in PubMed, EMBASE, CENTRAL, and Chinese Biomedical Database from inception up to May 2023. A detailed description of the search strategy is shown in Appendix S1. References cited in the relevant articles were tracked for potential and eligible studies. Due to many possible NPI modalities, each modality might have some synonyms. If only a specific search term was used, there was a risk of missing some NPIs. Therefore, any RCT reporting axSpA was initially searched, and the potential and eligible NPIs were screened and identified from those results.
Eligibility criteria
To be eligible, a study had to meet the following PIOS (patient, intervention, outcome, and study design) criteria:
Type of study design: RCTs published in peer-reviewed journals.
Type of patients: Patients (⩾18 years old) with a diagnosis of axSpA confirmed by a physician, based on the 1984 modified New York criteria and/or ASAS classification criteria for axSpA. 9
Type of interventions: NPIs included different exercise modalities, physiotherapy, and no intervention. The definition of each intervention is shown in Appendix S2.
Outcomes: Reporting at least one clinical outcome of axSpA, such as Bath Ankylosing Spondylitis Disease Activity Index (BASDAI), Bath Ankylosing Spondylitis Functional Index (BASFI), Bath Ankylosing Spondylitis Metrology Index (BASMI), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP).
Study selection
ENDNOTE 20 was used to manage the records. To ensure high inter-rater reliability among the raters, pilot-testing eligibility criteria for citations and full-text articles and screening were conducted. The titles and abstracts of all the retrieved bibliographic records were independently screened by pairs of raters (L.H. and X.L., X.J. and Y.X.) based on the eligibility criteria. Any disagreement was resolved by a third rater.
Data extraction
Data extraction was completed by independent pairs of raters after pilot-testing of the data extraction form. Any disagreement was resolved by discussing, or by consulting a third rater if necessary. NPIs were classified into the following broad categories based on suggestions of a rheumatologist and a rehabilitation physiatrist: supervised combined exercise, aerobic exercise, high-intensity interval exercise, neuromuscular training, aquatic exercise, conventional exercise, physiotherapy, cognitive therapy and education, and standard care.
Risk of bias of individual study
We assessed the risk of bias in each included study based on Cochrane’s “Risk of Bias” tool. Two raters independently examined seven specific domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other potential sources of bias.
Data synthesis and statistical analysis
Across all outcomes, Bayesian random-effects network meta-analysis was conducted to calculate pooled estimates and 95% confidence intervals (CI) using the “netmeta” package in the statistical software R (Version 4.3.1). Effect estimates were reported as mean differences (MD) for continuous outcomes. Studies reporting continuous outcomes with the average effect estimate but not reporting the associated measure of variance (e.g., standard deviation) were included in this analysis, with standard errors imputed when feasible.
For the direct comparison, we also performed the traditional meta-analysis using the STATA/MP 17.0 (College Station, TX, USA). For the continuous variables, the meta-analysis was performed on the changes from baseline, and the MD was used to pool the continuous variable. If studies documented the outcome as medians and ranges, the MD was estimated according to the methods described by Hozo et al. 10 p < 0.05 was considered statistically different.
Results
Selection and characteristics of included trials
The PRISMA flow diagram for the selection of RCTs in axSpA is illustrated in Figure 1. From the 6646 citations initially identified in the systematic literature searches, 51 RCTs met our inclusion criteria and were finally included in this study. The basic characteristics of the included studies were summarized in Appendix S4. A total of 3457 patients with axSpA were included in this meta-analysis. Most of the included studies consisted of two arms and were published between 2003 and 2023 in 15 countries from Asia, Europe, and South America. The median duration of the eligible studies was 12 weeks (ranging from 8 days to 48 weeks), and the sample sizes ranged from 19 to 756 patients. In this meta-analysis, network plots for BASDAI, BASFI, BASMI, ESR, and CRP are shown in Figure 2.
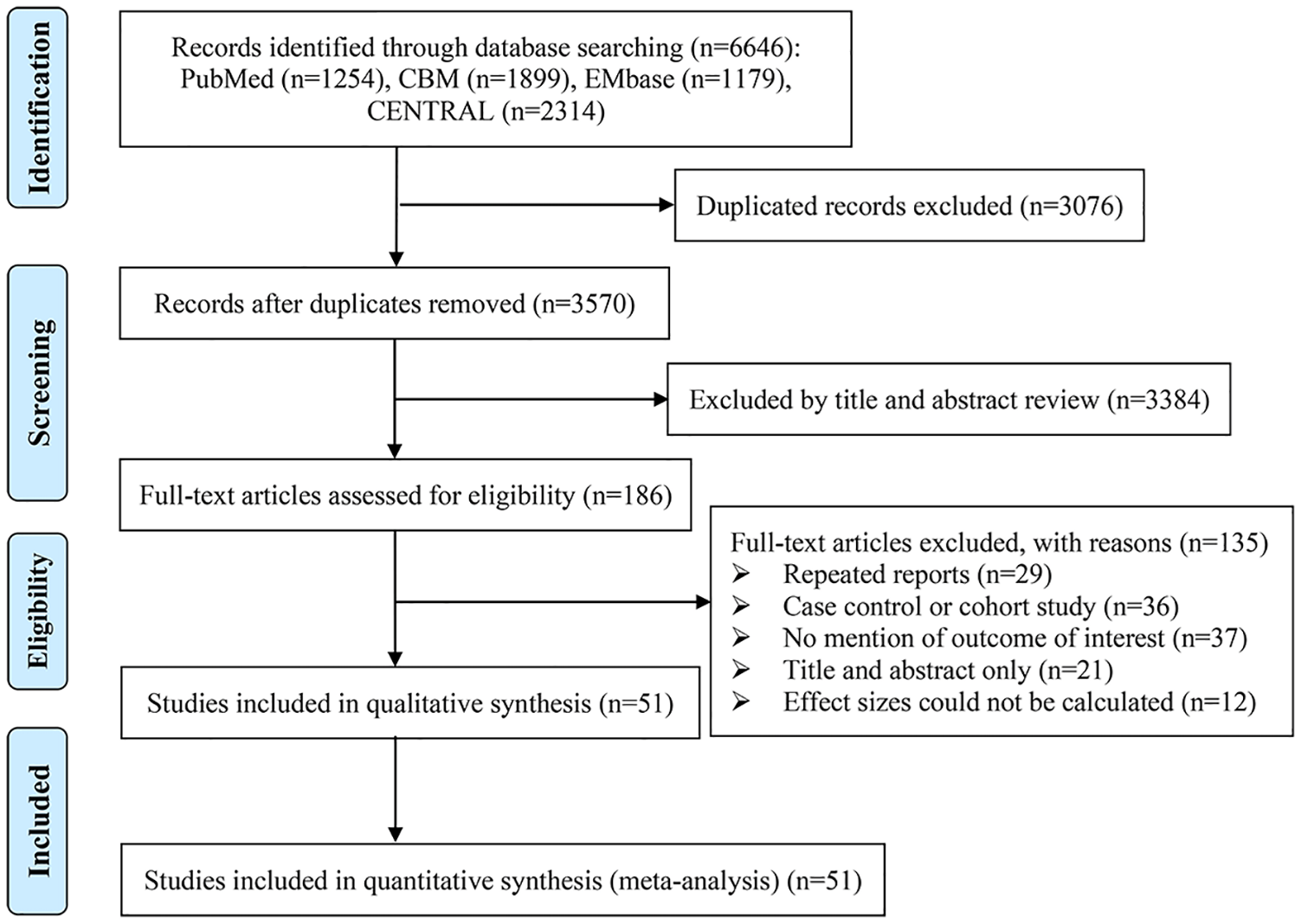
PRISMA flow diagram.

Network plot. (a) BASDAI. (b) BASFI. (c) BASMI. (d) CRP. (e) ESR.
Risk of bias assessments
The results of the risk of bias assessment were demonstrated in Appendix S3. The generation of the allocation sequence was adequate in the majority of studies (n = 44, 86.3%). Only 41.2% of studies reported their approaches to allocation concealment. Eleven studies masked outcome assessors to intervention allocation. About one-third of studies had a high risk of bias for the blinding method, which might be due to blinding shortcomings caused by the nature of the intervention. The potential risk of bias likely to be introduced attrition bias and reporting bias was low in most studies (86.3% and 72.5%, respectively). No studies were supported by commercial funding.
Disease activity
Forty-eight studies reported disease activity reflected by BASDAI. The results of network meta-analysis demonstrated that compared to standard care, supervised combined exercise (−1.13, 95% CI: −1.72 to −0.55), high-intensity interval exercise (−1.37, 95% CI: −2.50 to −0.25), and neuromuscular training (−1.17, 95% CI: −1.71 to −0.63) showed significant decrease in BASDAI; compared to conventional exercise, supervised combined exercise (−0.91, 95% CI: −1.50 to −0.32), and neuromuscular training (−0.95, 95% CI: −1.49 to −0.40) showed significant decrease in BASDAI (Table 1). However, there was no significant difference between the efficacy of the other NPI modalities in reducing BASDAI.
Results of network meta-analysis.
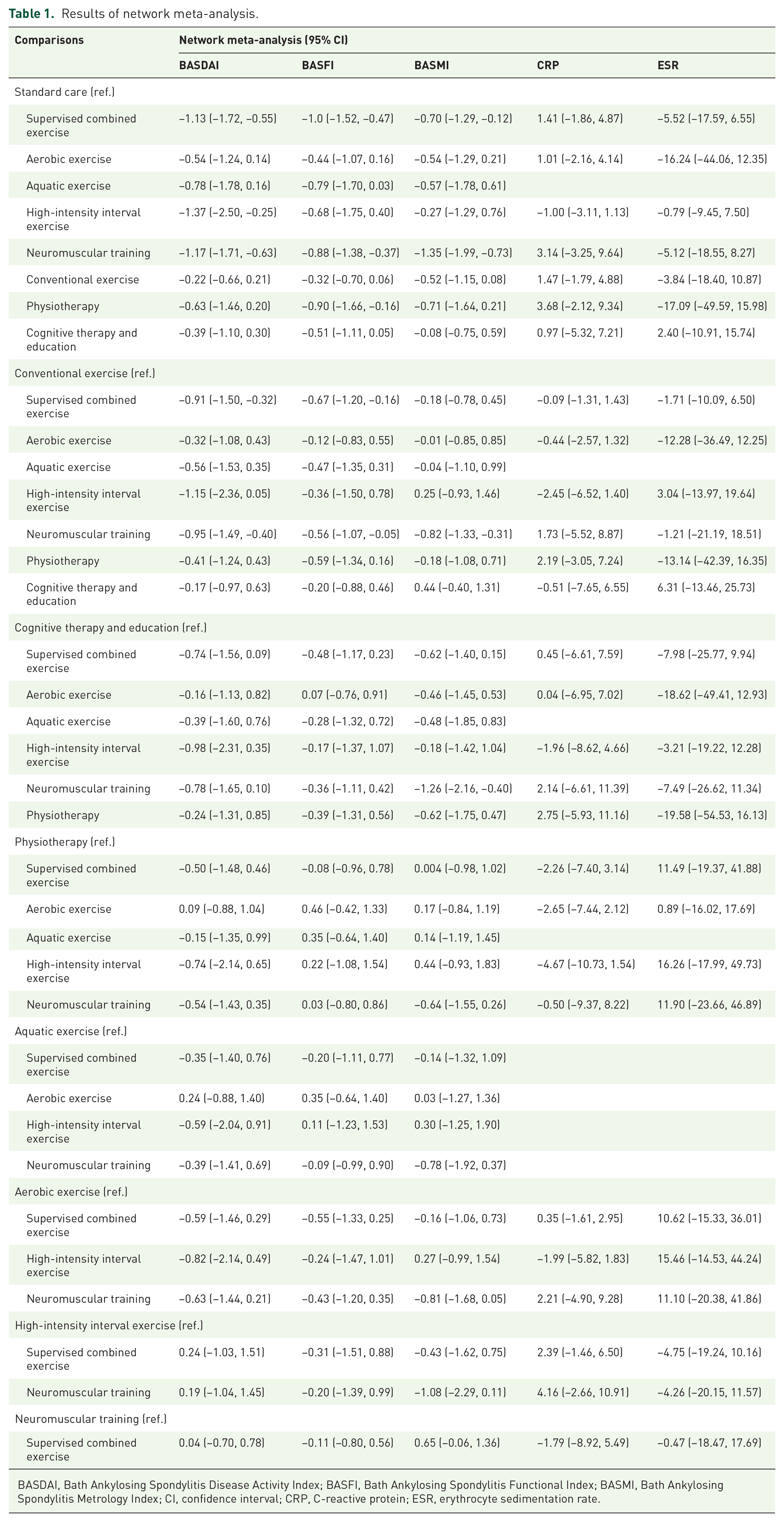
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate.
The results of traditional meta-analysis demonstrated that supervised combined exercise could significantly reduce BASDAI score, whether compared to unsupervised exercise (WMD = −0.62, 95% CI: −0.96 to −0.28, p < 0.001) or standard care (WMD = −0.49, 95% CI: −0.73 to −0.25, p < 0.001; Supplemental Appendix Figure 1). Similarly, neuromuscular training could significantly reduce BASDAI score, whether compared to conventional exercise (WMD = −0.78, 95% CI: −1.03 to −0.53, p < 0.001) or standard care (WMD = −0.75, 95% CI: −1.02 to −0.49, p < 0.001; Supplemental Appendix Figure 2). Compared to standard care, high-intensity interval exercise (WMD = −0.78, 95% CI: −1.15 to −0.40, p < 0.001; Supplemental Appendix Figure 3), aquatic exercise (WMD = −1.40, 95% CI: −2.20 to −0.59, p = 0.001; Supplemental Appendix Figure 4), and physiotherapy (WMD = −0.38, 95% CI: −0.58 to −0.17, p < 0.001; Supplemental Appendix Figure 5) could significantly reduce BASDAI score. However, aerobic exercise did not significantly reduce BASDAI score, whether compared to conventional exercise (WMD = −0.26, 95% CI: −0.62 to 0.11, p = 0.116) or standard care (WMD = −0.23, 95% CI: −0.50 to 0.04, p = 0.095; Supplemental Appendix Figure 6).
Twenty-two studies reported pain scores. The results of traditional meta-analysis demonstrated that compared to standard care, aerobic exercise (WMD = −0.57, 95% CI: −1.08 to −0.06, p = 0.029), supervised combined exercise (WMD = −1.07, 95% CI: −1.53 to −0.61, p < 0.001), and conventional exercise (WMD = −0.37, 95% CI: −0.71 to −0.02, p = 0.039) could significantly reduce pain score. However, compared to standard care, neuromuscular training (WMD = −0.33, 95% CI: −0.82 to 0.15, p = 0.176), aquatic exercise (WMD = −0.47, 95% CI: −1.35 to 0.41, p = 0.294), and physiotherapy (WMD = −0.09, 95% CI: −0.34 to 0.17, p = 0.503) did not significantly reduce pain score.
Physical function
Forty-eight studies reported physical function reflected by BASFI. The results of the network meta-analysis suggested that compared to standard care, supervised combined exercise (−1.00, 95% CI: −1.52 to −0.47), and neuromuscular training (−0.88, 95% CI: −1.38 to −0.37) showed significant decrease in BASFI; compared to conventional exercise, supervised combined exercise (−0.67, 95% CI: −1.20 to −0.16), and neuromuscular training (−0.56, 95% CI: −1.07 to −0.05) showed significant decrease in BASFI (Table 1). However, there was no significant difference between the efficacy of the other NPI modalities in reducing BASFI.
The results of traditional meta-analysis demonstrated that supervised combined exercise could significantly reduce BASFI score, whether compared to unsupervised exercise (weighted mean difference (WMD) = −0.62, 95% CI: −0.88 to −0.25, p < 0.001) or standard care (WMD = −0.36, 95% CI: −0.59 to −0.12, p = 0.003; Supplemental Appendix Figure 7). Similarly, neuromuscular training could significantly reduce BASFI score, whether compared to conventional exercise (WMD = −0.58, 95% CI: −0.84 to −0.32, p < 0.001) or standard care (WMD = −0.42, 95% CI: −0.68 to −0.16, p = 0.002; Supplemental Appendix Figure 8). Compared to standard care, aquatic exercise (WMD = −0.97, 95% CI: −1.52 to −0.42, p = 0.001; Supplemental Appendix Figure 9) and physiotherapy (WMD = −0.69, 95% CI: −1.21 to −0.18, p = 0.009; Supplemental Appendix Figure 10) could significantly reduce BASFI score. However, aerobic exercise did not significantly reduce BASFI score, whether compared to conventional exercise (WMD = −0.15, 95% CI: −0.51 to 0.21, p = 0.412) or standard care (WMD = −0.21, 95% CI: −0.48 to 0.06, p = 0.121; Supplemental Appendix Figure 11). High-intensity interval exercise was not statistically different in reducing BASFI score, compared to standard care (WMD = −0.36, 95% CI: −0.72 to 0.00, p = 0.052; Supplemental Appendix Figure 12).
Spinal mobility
Thirty-three studies reported spinal mobility reflected by BASMI. The results of the network meta-analysis suggested that compared to standard care, supervised combined exercise (−0.70, 95% CI: −1.29 to −0.12), and neuromuscular training (−1.35, 95% CI: −1.99 to −0.73) showed significant decrease in BASMI; compared to conventional exercise, neuromuscular training (−0.82, 95% CI: −1.33 to −0.31) showed significant decrease in BASMI (Table 1). However, there was no significant difference between the efficacy of the other NPI modalities in reducing BASMI.
The results of traditional meta-analysis demonstrated that supervised combined exercise could significantly reduce BASMI score, whether compared to unsupervised exercise (WMD = −0.52, 95% CI: −1.00 to −0.03, p = 0.036) or standard care (WMD = −0.27, 95% CI: −0.50 to −0.03, p = 0.026; Supplemental Appendix Figure 13). Similarly, neuromuscular training could significantly reduce BASMI score, whether compared to conventional exercise (WMD = −1.39, 95% CI: −1.67 to −1.11, p < 0.001) or standard care (WMD = −0.87, 95% CI: −1.29 to −0.44, p < 0.001; Supplemental Appendix Figure 14). Compared to standard care, aerobic exercise (WMD = −0.47, 95% CI: −0.79 to −0.16, p = 0.003; Supplemental Appendix Figure 15) and physiotherapy (WMD = −0.29, 95% CI: −0.54 to −0.05, p = 0.019; Supplemental Appendix Figure 16) could significantly reduce BASMI score. Aquatic exercise was not statistically different in reducing BASMI score, compared to conventional exercise (WMD = 0.04, 95% CI: −0.36 to 0.45, p = 0.843; Supplemental Appendix Figure 17). High-intensity interval exercise was not statistically different in reducing BASMI score, compared to standard care (WMD = −0.21, 95% CI: −0.57 to 0.15, p = 0.259; Supplemental Appendix Figure 18).
Inflammatory markers
CRP and ESR were reported in 12 and 10 studies, respectively. However, there was no significant effectiveness of exercise, physiotherapy, cognitive therapy, and education in reducing serum CRP and ESR levels (Table 1).
Discussion
As a chronic inflammatory disease characterized by pain and structural and functional impairments, axSpA requires treatments focused on suppressing inflammation, decreasing disease activity, and ameliorating physical function. The optimal management strategy combines pharmacologic treatment with non-pharmacologic interventions recommended by ASAS/EULAR guidelines. “Exercise as medicine” has been coined for some time with benefits for ameliorating pain, function, and quality of life being reported as an important therapeutic strategy for patients with axSpA. Therefore, which exercise modality is the most appropriate for axSpA needs to be further explored.
BASDAI is a comprehensive self-administered instrument for assessing disease activity in patients with axSpA, which includes five major symptoms: fatigue, back pain, joint pain/swelling, enthesitis, and morning stiffness. 11 These symptoms have a direct impact on patients’ feeling well and the capability to live a full life. Therefore, BASDAI is one of the core indicators for assessing clinical outcomes in patients with axSpA. Neuromuscular training, such as Pilates, Yoga, and Tai Chi, could significantly improve disease activity in patients with axSpA, and Pilates was superior to the home exercise in decreasing BASDAI score. 12 Our study corroborated these findings. Studies suggested that neuromuscular training could reduce stress by downregulation of the hypothalamic–pituitary–adrenal axis, potentially lowering inflammation and disease activity. 13 Another finding was that aerobic exercise showed no additional benefit in reducing BASDAI score, compared to standard care and conventional exercise. The same result was indicated in another systematic review. 14 This may be because aerobic exercise primarily benefits cardiorespiratory fitness and the anti-oxidant system rather than directly targeting disease activity. 15 The efficacy of high-intensity exercise in reducing disease activity in patients with axSpA was suggested in RCTs, and then further supported by our meta-analysis.16,17
Despite the positive trend toward exercise interventions in patients with axSpA, most patients failed to meet exercise recommendations, and their physical activity levels appeared to be lower than those in the general population. 18 In addition, adherence to exercise appeared to decrease with increasing disease duration. Studies suggested that 25%−54.7% of patients with axSpA exercised frequently, and among those who did, walking, stretching, and swimming were most commonly reported.19,20 Therefore, supervised combined exercise represents a key factor in the management of patients with axSpA, as it may improve the efficacy of exercise programs, and lead to a rapid and more sustained relief of axSpA symptoms. Studies have indicated that supervised exercise could allow patients to maintain an optimal frequency and duration of therapy, in contrast to home-based or unsupervised exercise programs or usual care, which did not achieve the same benefits. 15 In our study, supervised combined exercise could reduce disease activity, whether compared to standard care or conventional exercise.
Physical function, compared to disease activity, appears to be a relatively neglected target for intervention in the clinic for patients with axSpA, despite the fact that limitation in physical function is a major underlying factor contributing to the costs and disability associated with the disease.21,22 Thus, based on the aim of following a treat-to-treat treatment strategy, ameliorating physical function needs to be put back in focus. Our meta-analysis demonstrated that supervised combined exercise and neuromuscular training could reduce functional limitations and improve spinal mobility, whether compared to standard care or conventional exercise. These findings reaffirmed the critical role of supervised regular exercise in optimizing therapeutic outcomes for patients with axSpA. Studies have demonstrated that the spine and limbs were stretched, and the core muscle strength could be enhanced when the patients performed neuromuscular training.15,23 Besides, neuromuscular training could promote muscle relaxation and reduce fatigue in patients with axSpA. 24 Thus, this exercise could relieve muscle spasms, alleviate stiffness in the spine, and improve the flexibility of the body. The increase in flexibility is considered to lead to a reduction in BASFI score by enabling the patients to perform better during movements such as unassisted sock-wearing, reaching upward, and bending forward that provided a comprehensive improvement in functional capacity. 25
We also compared the BASMI score to explore the effect of various exercise modes on spinal mobility. Our meta-analysis demonstrated that compared to standard care, neuromuscular training, supervised combined exercise, and aerobic exercise could significantly reduce BASMI score, but there was no statistical difference as compared to conventional exercise except for neuromuscular training. Neuromuscular training has a significant impact on BASMI, likely due to the specific components that focus on core strength, balance, and dynamic stability. These components are crucial for improving flexibility, coordination, and functional joint stability in patients with axSpA. Other studies have suggested that practicing neuromuscular training not only strengthened muscles but also improved posture, reduced stress, and enhanced overall well-being, which might collectively contribute to better physical outcomes as measured by the BASMI. 26 However, high-intensity interval exercise and aquatic exercise demonstrated limited effectiveness in improving BASMI in patients with axSpA. High-intensity interval training, being quite vigorous, may not be appropriate for many patients with axSpA. While it primarily benefits cardiovascular fitness and muscle strength, it may not adequately address spinal mobility and flexibility—the core domains measured by BASMI. Similarly, although aquatic exercise can reduce joint stress, it may fail to significantly improve spinal mobility if the exercise duration is insufficient or movement variety is restricted.
Another finding in our meta-analysis was that Stanger bath therapy in physiotherapy could significantly reduce disease activity, and ameliorate physical function and spinal mobility, which might be attributed to the therapy itself. The thermal effect and buoyancy of the water might be real facets of Stanger bath therapy that produced many physiologic effects, such as muscle relaxation and enhanced joint mobility. 27 However, further study is needed to confirm its efficacy.
Our study showed the efficacy of supervised exercise in reducing BASDAI, BASFI, and BASMI scores, whether compared to unsupervised exercise or standard care, highlighting the importance of adherence. There were many factors affecting exercise adherence, such as socioeconomic factors, demographic characteristics, lifestyle, disease condition, and poor perceptions of the benefits of exercise.28 –30 In addition, contextual factors, such as communication with the clinicians and feeling taken care of by clinicians, have also been suggested to significantly affect exercise adherence.31,32 Therefore, we could increase the motivation to participate in exercise through booster sessions, reminders, behavioral change, and improving mindsets about exercise, thereby improving adherence to exercise. Our previous study has preliminarily explored the adherence and efficacy of a wearable technology-assisted exercise program in ankylosing spondylitis. 33 The results demonstrated that this wearable technology-assisted exercise program could improve the clinical outcomes of patients with axSpA who had no regular exercise, with good adherence and a favorable safety profile.
Exercise is recommended for the treatment of axSpA in clinical guidelines; however, not all exercises are beneficial for patients with axSpA, and choosing an appropriate exercise mode is crucial for optimizing patient outcomes. 34 Therefore, network meta-analysis was employed to investigate which mode was beneficial for patients by comparing different NPI modes. Our analysis demonstrated that supervised combined exercises and neuromuscular training were more effective in improving key outcomes, such as BASDAI and BASFI, whether compared to unsupervised exercise or standard care. Furthermore, we provided specific quantitative estimates of treatment effects, which enhanced the evidence base and facilitated more informed clinical decision-making for healthcare providers. However, there were several study limitations to consider. First, the number of studies reporting ASDAS was limited in this study, with only 7 out of 51 studies reporting it. Consequently, BASDAI was used to reflect disease activity in patients with axSpA. This may limit the ability to accurately capture true inflammatory disease activity. Second, some studies documented the outcome as medians and ranges; therefore, the MD ± SD were estimated according to the methods described by Hozo et al., which may not be fully aligned with the actual data.
Conclusion
Compared to either standard care or conventional exercises, supervised combined exercises and neuromuscular training were more beneficial for ameliorating disease activity, physical function, and spinal mobility in patients with axSpA. Therefore, supervised combined exercises and neuromuscular training should be available as complementary and alternative treatment strategies in the management of patients with axSpA. In addition, this suggests that future research should focus on the individualized application of various NPIs and further explore their impact on long-term clinical outcomes.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251329696 – Supplemental material for Non-pharmacological interventions for patients with axial spondyloarthritis: a meta-analysis
Supplemental material, sj-docx-1-tab-10.1177_1759720X251329696 for Non-pharmacological interventions for patients with axial spondyloarthritis: a meta-analysis by Lidong Hu, Xingkang Liu, Xiaojian Ji, Yiwen Wang, Jiaxin Zhang, Lulu Zeng, Yangqin Xun, Jianglin Zhang, Jian Zhu and Feng Huang in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251329696 – Supplemental material for Non-pharmacological interventions for patients with axial spondyloarthritis: a meta-analysis
Supplemental material, sj-docx-2-tab-10.1177_1759720X251329696 for Non-pharmacological interventions for patients with axial spondyloarthritis: a meta-analysis by Lidong Hu, Xingkang Liu, Xiaojian Ji, Yiwen Wang, Jiaxin Zhang, Lulu Zeng, Yangqin Xun, Jianglin Zhang, Jian Zhu and Feng Huang in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
