Abstract
Background:
Hypertension (HTN) is the most important modifiable risk factor for the development of cardiovascular events (CVEs). Patients with axSpA are also associated with an increased risk of future CVE.
Objectives:
To ascertain whether baseline early-stage HTN is a predictor of future CVE in addition to inflammation in patients with axial spondyloarthritis (axSpA).
Design:
A retrospective cohort study.
Methods:
Patients with axSpA were recruited from 2001 to 2017. Patients with at least 2 years of follow-up and without prior CVE were divided into three groups according to the calculated mean blood pressure (BP) over the first 2-year follow-up period (adjusted mean BP) (⩾140/90, 130–139/80–89, and <130/80 mm Hg). They were followed from baseline until the end of 2020 or the occurrence of a first CVE. Multivariate Cox regression analyses adjusting for baseline and time-varying variables were used to assess the relationship between mean BP and CVE.
Results:
Out of the 437 patients fulfilling the inclusion criteria, 49 (11.2%) and 132 (30.2%) had an adjusted mean BP ⩾ 140/90 and 130–139/80–89 mm Hg, respectively, and 256 (58.6%) were pre-HTN. After a median follow-up of 12 (7–18) years, 56 (12.8%) CVEs were documented. The incidence rates were 21.4, 14.2, and 5.9 per 1000 patient-years for the three groups, respectively. Baseline adjusted mean BP of 130–139/80–89 mm Hg was independently associated with the occurrence of CVE after adjusting for the baseline covariates as well as time-varying high inflammatory burden.
Conclusion:
Baseline-defined early-stage HTN carries excessive risk of developing CVE which may be due to untreated inflammatory burden. Early antihypertensive therapy should target this BP level to minimize their future risk of CVE.

Introduction
It is well established that patients with axial spondyloarthritis (axSpA) have an increased cardiovascular (CV) risk compared with the general population.1,2 Various conventional CV risk prediction algorithms underestimated CV risk in rheumatoid arthritis (RA) and psoriatic arthritis (PsA), most likely because the inflammatory component was not considered.3–6 The European Alliance of Associations for Rheumatology (EULAR) recommended an intrinsic multiplication factor of 1.5 for patients with RA. 7 However, the modified CV risk scores still underestimated CV risk in axSpA patients.8,9 Therefore, there is an increasing recognition that inflammation plays an important role in driving the development of CV events (CVE) in addition to traditional CV risk factors in inflammatory arthritis.9,10
As one of the most important modifiable risk factors for the development of CVE,11,12 hypertension (HTN) can be detected in up to 40% of patients with axSpA.13,14 Higher systolic blood pressure (SBP) is consistently associated with an increased risk of cardiovascular disease (CVD). 15 A meta-analysis including 61 prospective studies reported that each 20 mm Hg increase in SBP (or each 10 mm Hg increase in diastolic blood pressure (DBP)) was associated with a twofold increased risk of death from CVD. 16 Therefore, the American College of Cardiology/American Heart Association (ACC/AHA) recommended a lower threshold of SBP and DBP for arterial HTN diagnosis in adults in 2017. 17 Accordingly, blood pressure (BP) of 130–139/80–89 mm Hg is classified as early-stage HTN and BP ⩾140/90 mm Hg is classified as stage 2 HTN. With this new definition, the prevalence of HTN increased by 15.5%–49.6%.18,19 However, there were controversies regarding its clinical application.20–23
Of note, a more recent meta-analysis of randomized clinical trials (RCTs) revealed that lowering BP in patients with baseline SBP less than 140 mmHg was not beneficial in reducing mortality and CVD. 24 As our previous study showed that baseline inflammatory burden (baseline disease duration and delay of diagnosis) is associated with incident HTN in patients with axSpA, 25 it is possible that the baseline state of early-stage HTN (130–139/80–89 mm Hg) may carry excessive risk of future CVE. A recent study has confirmed that patients with systemic lupus erythematosus (SLE) with a baseline-defined mean BP of 130–139/80–89 mm Hg under this new definition had a significantly higher incidence of atherosclerotic vascular events compared with the normotensives. However, the association between baseline-defined early-stage HTN with future CVE in addition to time-dependent inflammation remains unknown.
Given the relationship between untreated inflammatory burden and future incident HTN in our previous study, we hypothesize that baseline-defined early-stage HTN is associated with excess CVE in patients with axSpA. The present study was designed to address whether a BP of 130–139/80–89 mm Hg at baseline is a predictor of future CVE after controlling baseline traditional CV risk factors and time-dependent inflammation.
Methods
Study design and ethics
This is a retrospective cohort study to address the relationship between early-stage HTN and CVE in axSpA. Data were retrieved from case notes and the city-wide electronic health record (EHR) database. Briefly, patients who were diagnosed with axial spondylarthritis and fulfilled the 2009 ASAS classification criteria for axSpA, 26 and aged between 18 and 60 years were recruited from the rheumatology clinic of a regional hospital in Hong Kong from January 2001 to December 2017. The reason we established the upper age limit cutoff is based on a meta-analysis that reported a higher age-specific hazard ratio (HR) for CV death within this age group compared to older individuals in the general population. 16 All patients should have at least 2 years of follow-up after recruitment, and at least one clinic visit afterward. Patients with an established CVE or with missing BP measurements during the first 2 years were excluded. The baseline year was defined as the beginning of the 3rd year time point after recruitment. Patients who lost to the follow-up were also excluded. For patients diagnosed before 2001, the baseline year was 2003; for those who were diagnosed after 2001, the baseline year was 2 years after diagnosis. All individuals were followed from the baseline year until the occurrence of the first CVE or the end of 2020.
The study was approved by The Joint Chinese University of Hong Kong—New Territories East Cluster Clinical Research Ethics Committee (No. 2020. 519). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 27 The study was conducted in compliance with the Declaration of Helsinki and the International Council for Harmonisation guideline for Good Clinical Practice. Written informed consents were waived.
BP categories
Recorded BP (SBP/DBP) (measured in the sitting position with an aneroid sphygmomanometer after at least 5 min rest) at each clinic visit and the antihypertensive medication history were retrieved from the city-wide EHR. To adjust for the fluctuation of BP over time, an “adjusted mean BP” was defined at baseline and calculated from BP measurements over a period of 2 years prior to the baseline visit for all enrolled axSpA patients in this study. The calculation has been validated and elaborated previously.28,29 The formula was:
In short, xi and xi − 1 represented the SBP or DBP level at visit i and i − 1, and ti represented the time interval between visit i − 1 and i. By incorporating the ti in its calculation, the adjusted mean BP considered the length of time that SBP and DBP were presumed to have remained at a particular level.
Patients were then categorized into three different groups according to their baseline adjusted mean BP: Group 1: adjusted mean BP <130/80 mm Hg (normotensives); Group 2: adjusted mean BP 130–139/80–89 mm Hg (early-stage HTN); and Group 3: adjusted mean BP ⩾140/90 mm Hg (HTN).
BP measurement at each visit
BP was measured by a nurse or trained healthcare assistant in the non-dominant arm using a digital automatic BP monitor (Colin 8800 NIBP monitor [Colin Medical Instruments Corp., USA] with a cuff of the appropriate size, which was subsequently replaced by the A&D Medical TM-2657-P [A&D Company, Limited, Japan]) following standard recommended procedures. Measurement was initiated after a 5-min rest. If the BP reading was suboptimal, an extra measurement would be subsequently implemented. If both BP readings are suboptimal, clinicians may measure extra BP readings for the patients and select the most appropriate BP reading based on clinical judgment.
Clinical data
Each patient was followed at 6–12 months intervals and clinical data were regularly updated. Inflammatory markers including erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) were measured. Disease activity and function were assessed using the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) 30 and Bath Ankylosing Spondylitis Functional Index (BASFI), 31 respectively. At baseline, demographics (age, sex, smoking, and drinking history), clinical variables (disease duration, disease activity, inflammatory markers), traditional CV risk factors, and medication history were retrieved from case notes and the EHR.
Time-varying inflammatory burden (BASDAI, ESR, and CRP) and drug history (disease-modifying anti-rheumatic drugs (DMARDs) including the use of biologic DMARDs (bDMARDs), conventional synthetic DMARDs (csDMARDs), nonsteroidal anti-inflammatory drugs (NSAIDs)) were also retrieved from the EHR at a yearly interval starting from baseline visit till the end of the study.
Endpoint
The study endpoint was defined as the first occurrence of a CVE, including angina, myocardial infarction (MI), ischemic heart disease (IHD), ischemic and hemorrhagic stroke, acute and chronic heart failure (HF), transient ischemic attack (TIA), percutaneous transluminal coronary angioplasty or carotid endarterectomy, and CVE-related deaths. These data were retrieved from the EHR.
Statistical analysis
Variables were presented as counts (percentages), mean ± SD or for normal distributed data, or median with interquartile range for non-normally distributed data. The baseline demographic and clinical characteristics of the three groups of patients with different adjusted mean BP were compared using one-way ANOVA analysis for continuous variables and χ2 tests for categorical variables. When a significant difference (p < 0.05) was observed, the Scheffe test was used for post hoc comparison at a statistically significant adjusted level of p < 0.0083. CVE-free survival for the three groups was assessed using the Kaplan–Meier survival analysis. The survival distribution was compared using the log-rank test. Univariate and multivariable Cox proportional hazards regressions were used to investigate the association between the development of a first CVE and the potential risk factors (baseline characteristics listed in Table 1 with p < 0.05) after adjusting for the baseline CV risk scores (which were used to account for traditional CV risk factors). Baseline statistical analysis was performed using SPSS V.26.0 (IBM, USA) for Windows. Inflammatory markers, BASDAI, and drug use were also analyzed as time-dependent predictors (being updated at each visit) using time-varying Cox proportional hazard regression analyses (R version 4.2, available at https://www.r-project.org/, in the package named “survival” and “survminer” with the counting process method). p < 0.05 was considered significant.
Baseline characteristics of different hypertension categories.
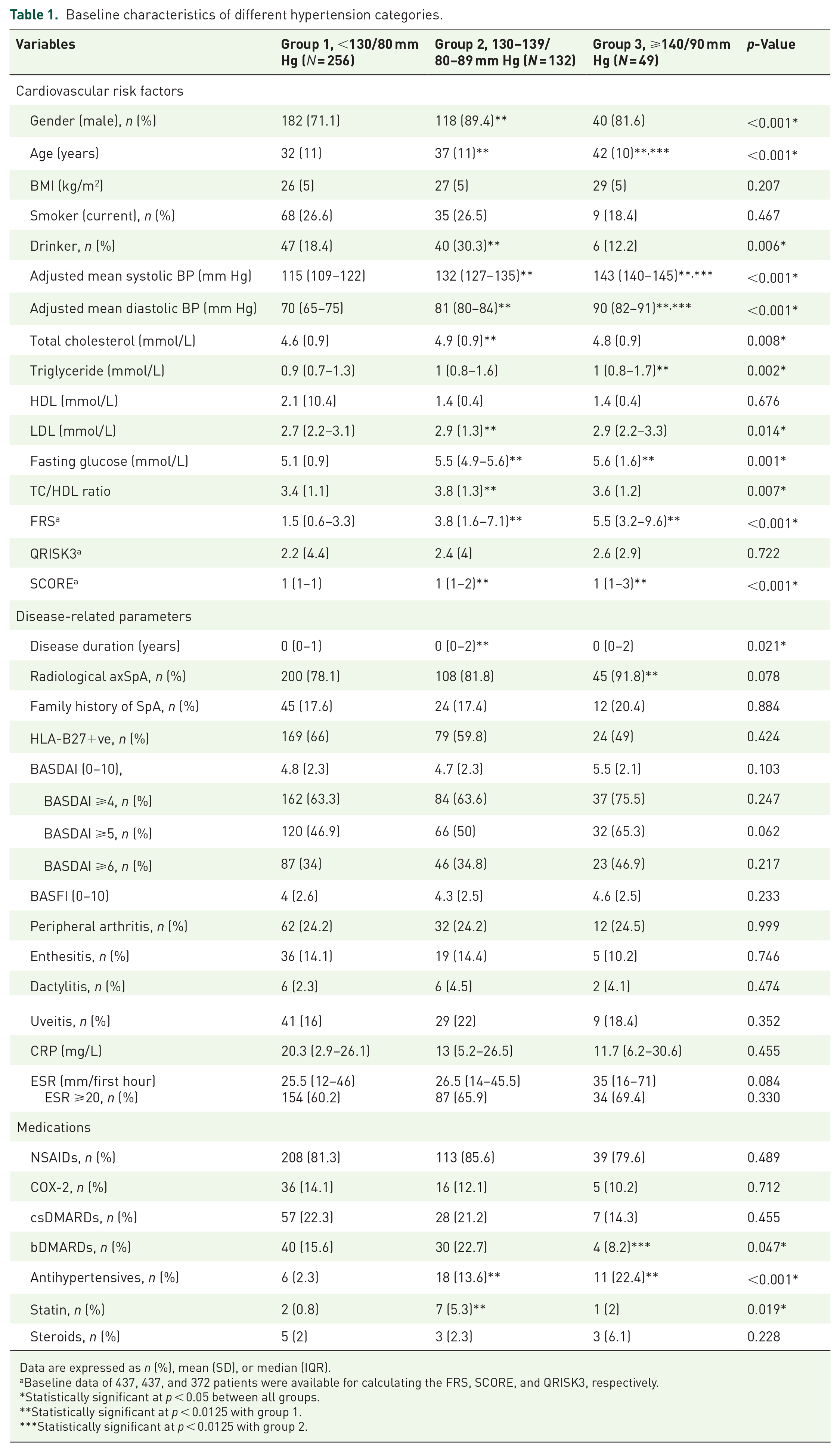
Data are expressed as n (%), mean (SD), or median (IQR).
Baseline data of 437, 437, and 372 patients were available for calculating the FRS, SCORE, and QRISK3, respectively.
Statistically significant at p < 0.05 between all groups.
Statistically significant at p < 0.0125 with group 1.
Statistically significant at p < 0.0125 with group 2.
Results
Baseline characteristics in different HTN category groups
Of the 437 patients included, 256 (58.6%) were normotensive (group 1), 132 patients (30.2%) had baseline-defined early-stage HTN (group 2), and 49 patients (11.2%) had baseline-defined HTN (group 3) at baseline. The baseline characteristics of different BP groups are shown in Table 1. Compared to the normotensive patients (group 1), patients with HTN (groups 2 and 3) were older and had higher sugar and lipid levels. Compared to group 1, longer disease duration and a higher prevalence of radiographic sacroiliitis were observed in groups 2 and 3, respectively. Fewer patients in group 3 (8.2%) used bDMARDs compared with group 2 (22.7%) (p = 0.047).
Association of baseline adjusted mean BP and future CVE
After a median follow-up of 12 (7–18) years (a total of 5494 patient-years), 56 (12.8%) CVEs were documented, including 7 (1.6%) CVE-related deaths. The incidence rate of new-onset CVEs is 10.2 cases per 1000 person-years. Recorded CVEs were IHD (n = 18), CHF (n = 12), ischemic stroke (n = 11), acute MI (n = 7) (including MI-related revascularization procedures (n = 3)), unstable angina (n = 3), TIA (n = 3), and hemorrhagic stroke (n = 2). The overall incidence of CVEs was 13 (26.5%), 24 (18.1%), and 19 (7.4%) and the incidence rates were 21.4, 14.2, and 5.9 per 1000 patient-years in group 3, group 2, and group 1, respectively. The Kaplan–Meier curves for the CVE-free survival (censored at 18 years) of the three groups are shown in Figure 1, demonstrating significant differences between the three groups (p < 0.001) using the log-rank test. Post hoc analysis showed that significant differences were present between group 1 and group 2 (p = 0.005) and between group 1 and group 3 (p < 0.001). There was no difference between groups 2 and 3 (p = 0.188).

Kaplan–Meier analysis for CVE-free cumulative survival among the three patient groups (green line: ⩾140/90 mm Hg, red line: 130–139/80–89 mm Hg, blue line < 130/80 mm Hg). The differences were statistically significant among all groups (p = 0.003* between group 1 and group 2, log-rank χ2 (1 df) = 9.075; p < 0.001* between group 1 and group 3, log-rank χ2 (1 df) = 17.326; p < 0.001* among three groups, log-rank χ2 (1 df) = 18.048). There is no difference between groups 2 and 3 (p = 0.143, log-rank χ2 = 2.151).
The univariate Cox regression model revealed that both HTN groups (groups 2 and 3) had a significantly increased risk of developing a first CVE (Table 2). In the multivariable Cox regression models, patients in group 2 (HR: 2.09–2.35) and group 3 (HR: 3.17–3.80) remained at higher risk of developing CVE after adjusting for traditional CV risk scores, inflammation, and treatment used at baseline (Table 2).
Univariate and multivariate Cox regression analyses for baseline predictors of CVE.
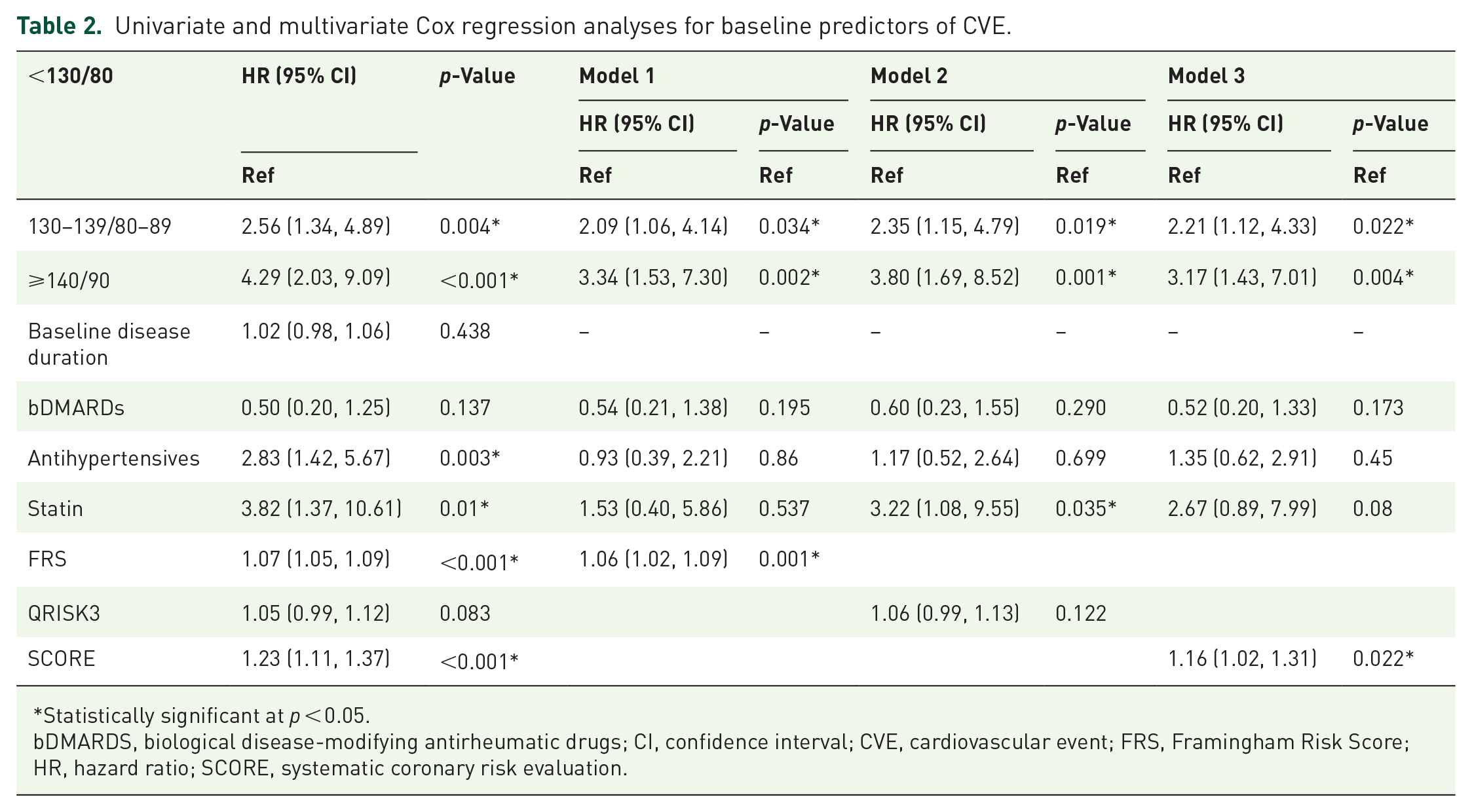
Statistically significant at p < 0.05.
bDMARDS, biological disease-modifying antirheumatic drugs; CI, confidence interval; CVE, cardiovascular event; FRS, Framingham Risk Score; HR, hazard ratio; SCORE, systematic coronary risk evaluation.
Predictors for CVE in time-dependent analysis
In the time-dependent univariable Cox proportional hazard regression model (Table 3), groups 2 and 3 had a significantly increased CVE risk with HRs of 2.59 and 4.21, respectively. Higher inflammatory burden as reflected by ESR ⩾20 (p = 0.039, HR: 2.17, 95% confidence interval (CI): 1.04, 4.35) was associated with an increased risk of developing CVE.
Univariate analysis with time-dependent Cox proportional hazard regression for BP and CVE.

Variables are time-varying unless otherwise specified.
Variables with a p-value of <0.05 are included in the multivariable analysis.
Statistically significant at p < 0.05.
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; bDMARDS, Biological Disease-Modifying Antirheumatic Drugs; BP, blood pressure; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; CVE, cardiovascular event; ESR, erythrocyte sedimentation rate; HR, hazard ratio; NSAIDs, nonsteroidal anti-inflammatory drugs.
Using time-dependent multivariable Cox regression analysis, group 2 (HR: 1.94, 95% CI: 1.00, 3.75, p = 0.048 in model 1 and HR: 2.81, 95% CI: 1.13, 7.01, p = 0.026 in model 2) and group 3 (HR: 2.19, 95% CI: 1.01, 4.75, p = 0.046 in model 1 and HR: 4.34, 95% CI: 1.62, 11.61, p = 0.004 in model 2) remained at higher risk of developing CVE after adjusting for age, sex, and high inflammatory burden (Figure 2(a) and (b)). Moreover, high ESR level (ESR ⩾20) was also the independent predictor of future CVE (HR: 2.52, 95% CI: 1.04, 6.12, p = 0.041) (Figure 2(a) and (b)).

Forest plot illustrating the multivariate time-varying Cox regression for the prediction for CVE with (Figure 2b) and without (Figure 2a) adjusting for inflammatory markers.
Subgroup analysis
In subgroup analysis, we stratify patients into different age group to ascertain the impact of early-stage HTN on CVE in different age groups. Group 2 (HR: 2.13, 95% CI: 1.10, 4.14, p = 0.026 in model 1 and HR: 3.07, 95% CI: 1.22, 7.70, p = 0.017 in model 2) and group 3 (HR: 2.29, 95% CI: 1.05, 4.97, p = 0.037 in model 1 and HR: 4.14, 95% CI: 1.54, 11.16, p = 0.005 in model 2) was independently associated with future CVE in the time-dependent multivariable analysis after adjustment for high inflammatory burden, age groups, and sex (Figure 3(a) and (b)). Patients who were aged 40–60 years (HR: 5.38–10.39) at presentation and high ESR level (HR: 2.44, 95% CI: 1.16, 5.10, p = 0.018) were also independent predictors for future CVE (Figure 3(a) and (b)).

Forest plot illustrating the multivariate time-varying Cox regression for the prediction for CVE stratified by age groups with (Figure 3b) and without (Figure 3a) adjusting for inflammatory markers.
Discussion
Our study found that patients with baseline-defined early-stage HTN (group 2, adjusted mean BP 130–139/80–89 mm Hg) had a persistently increased risk of future CVE after adjusting for multiple baseline traditional risk factors and medications and time-dependent high inflammatory burden. As our previous findings have confirmed that baseline inflammatory burden (baseline disease duration and delay of diagnosis) is associated with incident HTN in patients with axSpA, 25 suggesting a potentially significant impact of untreated inflammatory burden at baseline, not time-varying inflammatory burden, on the increased BP. The increased BP may proceed to the state of early-stage HTN and lead to future CVE, which may be underestimated in clinical practice.
With the new definition of HTN, it is notable that axSpA patients with baseline-defined early-stage HTN and HTN had a higher prevalence of traditional and axSpA-related CV risk factors. When inflammation that changes over time are taken into consideration in the time-dependent Cox-regression analysis, baseline-defined early-stage HTN remains as significance predictor for future CVE. This finding suggests that in patients with chronic inflammatory arthritis such as axSpA, while inflammation is conformed to be associated with a relative increase in CV risk, there is an excessive risk of future CVE dependent on the state of early-stage HTN which may due to the effect of untreated inflammatory burden. Using the old definition of HTN (BP ⩾140/90), it has also been reported that inflammatory burden (longer disease duration and higher disease activity) in axSpA was associated with a higher prevalence of HTN.32,33 Therefore, our findings together with previous evidence may provide more insights into how inflammatory burden in axSpA patients interacts with increased BP. Future studies are needed to address the association between untreated disease duration, delay of diagnosis, and the state of early-stage HTN in axSpA.
A meta-analysis suggested that higher SBP or DBP was associated with higher CVE-related death rates at younger age group (40–69 years) compared to older age group in the general population. 16 As the majority of our axSpA patients in the clinic belong to this age group, subgroup analysis in our study found that baseline-defined early-stage HTN also has a higher impact in these middle-aged patients and conferred approximately twofold to threefold risk of future CVE. However, it would be better to confirm these findings in larger scale, prospective cohort studies before people can consider applying a lower threshold, such as the 130–139/80–89 mm, in routine management of our axSpA patients.
A study from the American National Survey reported an age-standardized prevalence of 45.4% and an increase of over 20% in the total number of individuals with HTN with the 2017 definition. 34 Similarly, the prevalence of HTN in our study was 12.2% according to the previous definition and increased dramatically to 43% using the new threshold. A latest meta-analysis including prospective cohorts also revealed optimal control of early-stage HTN could reduce the risk of CVE for more than 10%. 35 HTN is the most common comorbidity (pooled prevalence 23%), in patients with axSpA using the old definition. 14 This new definition inevitably increases the prevalence of HTN in patients with axSpA. Whether treatment of the state of early-stage HTN could potentially reduce the excessive CV risk in the future would definitely need to be addressed in future studies.
The strength of our study includes the long-term follow-up (a median of 12 years) to investigate the effect of early-stage HTN on CVE in axSpA with time-varying adjustment of high inflammatory burden and traditional risk factors. Moreover, adjusted mean BP over the first 2 years prior to baseline was utilized instead of the traditional approach of averaging BP of ⩾2 readings in two separate occasions, 36 as the cumulative HTN exposure over time was more reliable than BP at single time point. 28 However, there are several limitations to the study. First, missing data are inevitable due to the retrospective design. Body mass index was not included in the analyses as body height was not routinely recorded in the EHR. Second, the relatively small sample size may limit the power of time-dependent analysis of multiple covariates and the interaction between inflammation and traditional CV risk factors. Third, causality cannot be definitively delineated in a longitudinal study, our results should be interpreted with caution, and further larger prospective study is needed to clarify whether early-stage HTN should be the new target of BP management in patients with axSpA. Also, given the limited number of females 98/437 (22.4%), there may be uncertainty regarding the effect of gender. In addition, patients were diagnosed exclusively as AS prior to 2009, as the 2009 ASAS classification criteria were implemented that year. Subsequently, 84/437 (19.2%) patients met the ASAS classification criteria for non-radiographic axial spondyloarthritis (nr-axSpA). Whether the results from this study are applicable to patients with nr-axSpA will need to be confirmed by future studies. Lastly, whether ASDAS may be a better predictor of CVE in these patients with or without early-stage HTN deserves further study.
Conclusion
In patients with axSpA, the state of early-stage HTN carries excessive risk of future CVE, which may be due to untreated inflammatory burden. Antihypertensive therapy targeting BP < 130/80 should be initialed in these patients to minimize their future risk of CVE.
Footnotes
Acknowledgements
We would like to show our gratitude to all medical staffs and research assistants. Furthermore, we would like to thank all the supports from family members.
Declarations
Supplemental material
Supplemental material for this article is available online.

