Abstract
Background:
Axial spondyloarthritis (axSpA) patients are at higher risk of cardiovascular (CV) disease (CVD) than the general population, partly due to consequences of inflammation or its treatment. But relationship between inflammation in axSpA and cardiovascular events (CVE) is unknown.
Objectives:
To examine whether inflammatory burden over time can predict CVE independent of baseline CV risk factors in axSpA patients.
Design:
A cohort analysis was performed in patients who had been recruited since January 2001. The primary outcome was a first CVE occurring between January 2001 and December 2020.
Methods:
Three CVD risk scores were computed at baseline. The performance of the original and modified (*1.5 multiplication factor) CV risk algorithms were assessed. Time-varying Cox proportional hazard models and Kaplan–Meier survival analysis were used to assess whether inflammatory burden (Bath ankylosing spondylitis disease activity index [BASDAI] and inflammatory markers), nonsteroidal anti-inflammatory drugs (NSAIDs) and disease modifying antirheumatic drugs (DMARDs) can predict the development of first CVE.
Results:
463 patients (35 [26–45] years, male: 360 [77.8%]) were recruited. After a median follow-up of 12 (7–19) years, 61 patients (13.2%) experienced a first CVE. Traditional/modified CV risk scores underestimated CV risk. Erythrocyte sedimentation rate (ESR) ⩾ 20 mm/h was associated with a significantly higher risk of CVE during follow-up (HR: 2.07, 95%CI [1.10, 3.98], p = 0.008). Active disease as indicated by a rising BASDAI also showed positive trend towards a higher risk of developing CVE over time. After adjusting for CV risk scores in the multivariable models, high ESR level (ESR ⩾ 20 mm/h) over time remained significantly associated with a higher risk of developing CV events.
Conclusion:
Increased inflammatory burden as reflected by elevated ESR levels (ESR ⩾ 20) was associated with increased risk of CVE, while the use of NSAIDs and DMARDs were not.
Introduction
Axial spondyloarthritis (axSpA) patients are at higher risk of cardiovascular (CV) disease (CVD) than the general population, partly due to shared risk factors, consequences of inflammation or its treatment. The presence of CVD was associated with an increased risk of death in patients with radiographic axSpA (r-axSpA), which is also known as ankylosing spondylitis (AS).1,2 A recent meta-analysis of 12 longitudinal studies showed a significant increase in the risk of myocardial infraction (MI) (RR = 1.44; 95% CI 1.25–1.67) and stroke (RR = 1.37; 95% CI = 1.08–1.73) in patients with AS compared with controls. 3 Compared with patients with rheumatoid arthritis (RA) and psoriatic arthritis (PsA), studies have reported a higher 4 or similar incidence of CVD in axSpA patients, 5 suggesting that inflammation, rather than a particular disease, drives the increased risk of CVD.
The increased CV risk in axSpA may be partially explained by an increased prevalence of traditional CV risk factors. 6 CV risk scores have been developed for the general population, including the Framingham Risk Score (FRS), 7 Systematic Coronary Risk Evaluation (SCORE), 8 and QRISK3. 9 Nonetheless, underestimated CVD risk in RA and PsA were observed as the inflammatory component was not considered except QRISK3.10–13 The European Alliance of Associations for Rheumatology (EULAR) recommends a 1.5 multiplication factor only in RA patients. 14 However, adaptations of the CV risk algorithm did not improve discriminative ability and calibration among PsA patients. 15 All CV risk scores also underestimated the risk of subclinical atherosclerosis in PsA 16 and the EULAR-recommended modification improves the sensitivity of FRS and Atherosclerotic Coronary Artery Disease (ASCVD) only to a moderate level. 17 A small study in AS(n = 133) showed that all the CV risk algorithms included exhibite a poor discriminative ability, except for Reynold’s Risk Score (RRS) and SCORE. 18 Whether adaptation of CV risk algorithms according to EULAR recommendations could provide a significant improvement in discriminative ability for axSpA patients remained uncertain.
Increased subclinical atherosclerosis by high-resolution carotid ultrasound has been demonstrated in AS. 19 Meta-regression analysis revealed that AS patients with active inflammation (as reflected by the Bath AS Disease Activity Index [BASDAI] and C-reactive protein [CRP] level) could significantly impact on the carotid intima-media thickness (IMT). 19 Baseline CRP and AS disease activity score (ASDAS) were significantly associated with increased arterial stiffness 5 years later, supporting the notion that disease activity was linked to future risk of CVD in AS. 20 Whether increased inflammatory burden over time (as reflected by the time-varying BASDAI and/or inflammatory markers) and the use of nonsteroidal anti-inflammatory drugs (NSAIDs) and disease modifying anti-rheumatic drugs (DMARDs) can predict future CV events (CVE) in axSpA deserve more detailed study.
We hypothesize that baseline CV risk scores underestimate CV risk, and inflammatory burden over time could predict CVE in axSpA patients independent of traditional CV risk factors. In this study, we aimed to (1) compare the observed incidence of CVE with that predicted by the CV risk scores to determine the applicability of thes scores in this patient population, and (2) investigate the effect of inflammatory burden over time (as reflected by the time-varying BASDAI and inflammatory markers) on CVD risk in patients with axial SpA patients after adjusting for baseline traditional CV risk factors and drug use (time-varying NSAIDs and DMARDs).
Methods
Study design and patients
A retrospective cohort analysis was performed in axSpA patients (age > 18) fulfilling the 2009 Assessment in Ankylosing Spondylitis (ASAS) classification criteria, 21 who were consecutively recruited and being followed at the rheumatology clinic of the Prince of Wales Hospital from 2001 to 2019. All patients were de-identified. Patients with established CVE (angina, MI, ischemic heart disease [IHD], ischemic and hemorrhagic stroke, acute and chronic heart failure [HF], transient ischemic attack [TIA], surgery including percutaneous transluminal coronary angioplasty or carotid endarterectomy) at baseline were excluded. Follow-up began with the baseline clinic visit and continued until the occurrence of CVE, end of the study (December 2020), or loss to follow-up. For patients who were diagnosed before 2001 (n = 142), the baseline visit was defined as the first clinic visit in the year 2001; for patients diagnosed from 2001 onwards (n = 321), the baseline visit was defined as the first clinic visit. Symptom duration was defined as the duration between symptom onset and the baseline visit. Disease duration was defined as the duration between diagnosis and the baseline visit.
The study was approved by the Ethics Committee (EC) of the Chinese University of Hong Kong (CUHK) and Hospital Authority (HA) ethics committee (no. 2020. 519). The study was conducted in compliance with the Declaration of Helsinki and ICH guideline for Good Clinical Practice (GCP) and conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 22 Written informed consents were waived.
Clinical assessment
Patients were seen during scheduled visits every 6–12 months. During these visits, inflammatory markers were measured including erythrocyte sedimentation rate (ESR) and CRP. Disease activity and function was assessed using the BASDAI 23 and Bath AS Disease Functional Index (BASFI), 24 respectively. Baseline clinical and demographic data, drug history, and traditional CV risk factors were retrieved from case notes and the citywide electronic medical record system (Clinical Management System [CMS]). CV risk factors included age, weight, blood pressure (BP), the history of hypertension (HT), diabetes mellitus (DM), dyslipidemia, and drinking and smoking habits. Time-varying drug history (disease-modifying antirheumatic drugs [DMARDs] including biologic DMARDs [bDMARDs], conventional synthetic DMARDs [csDMARDs], nonsteroidal anti-inflammatory drugs [NSAIDs] and lipid-lowering drugs) and inflammatory burden (BASDAI and inflammatory markers) were also retrieved from CMS at a yearly interval starting from baseline visit till the end of study.
CV risk score assessment
Three CV risk scores were assessed at baseline. FRS was calculated according to the Framingham Heart Study. 7 SCORE Risk Calculator V.1.0 8 developed by the European Society of Cardiology was utilized to calculate SCORE. As no specific charts developed for Asian and the Chinese has a lower CVD risk in general, the European low-risk chart was adopted. 25 QRISK3 9 was computed using the QRISK3-2018 risk calculator with and without assuming CV risk similar to patients with RA (QRISK3*RA). Modified FRS (mFRS) and SCORE (mSCORE) were calculated by applying a multiplication factor of 1.5. Patients with preset cut-off values of FRS/mFRS > 20%, QRISK3/QRISK3*RA > 20%, and SCORE/mSCORE > 5% were regarded as high CV risk groups. Predicted risk for first CVE in patients with a follow-up time <10 years were adjusted proportionally.
Endpoints
The primary outcome was first CVE abovementioned during follow-up, and death due to CVD. The assignment of cause of death was based on case notes or CMS records. For patients who died during or shortly after a hospital stay for a documented condition, the cause of death was considered ascertained. The secondary outcome was the performance of three original and adapted CV risk algorithms.
Statistical analysis
Descriptive statistics were expressed as number with percentage, mean ± SD for normally distributed data, and median with interquartile range (IQR) for nonnormally distributed data. Hosmer–Lemeshow (HL) test was used for testing the goodness of fit between the observed and predicted CVE stratified in deciles and quartiles as appropirate. Differences between the two groups with or without CVE at baseline were assessed using Student’s t-test, Mann–Whitney U test and χ2 test where appropriate. Univariable and multivariable Cox proportional hazard regressions were used to investigate the association between the time-varying inflammatory burden (inflammatory makers [CRP and ESR] and disease activity [BASDAI]), medications (NSAIDs and DMARDs) and the development of a first CVE after adjusting for the baseline CV risk scores. Inflammatory marker was added as a continuous variable (ESR/CRP) and as a dichotomous variable: pro-inflammatory response (yes/no). The cut-off value for the presence of pro-inflammatory response was defined as CRP > 3 mg/L which was suggested to be associated with elevated CV risk in the general population. 26 The cut-off for ESR was ⩾20 mm/h. Inflammatory markers were measured regularly (every 6–12 months) and a yearly interval was maintained for the time-dependent analyses. Patients with high inflammatory burden (CRP > 3 mg/L or ESR ⩾ 20 mm/h or BASDAI ⩾ 4) was compared with patients with low inflammatory burden (CRP ⩽ 3 mg/l or ESR < 20 mm/h or BASDAI < 4) at each interval. In addition, drug use was treated as a time-varying exposure, non-users of NSAIDs and DMARDs were compared with NSAIDs and DMARDs users for each interval of follow-up. Different kinds of NSAIDs and bDMARDs use were conflated respectively to form a mutually exclusive group of either user or nonuser. Intervals with missing data were dropped from the analysis. Time-varying variables with a p ⩽ 0.06 in the univariable analysis were included in the mutivariate models. Time-varying Cox proportional hazard regression analyses were performed using R version 4.0 (https://www.r-project.org/) in the package named ‘survival’ and ‘survminer’ with counting process method. The effect of high versus low inflammatory burden during the follow-up intervals on CVE-free survival was assessed using the Kaplan–Meier survival analysis. The survival distribution was compared using log-rank test. All baseline statistical analyses were performed using SPSS V.26.0 for Windows. A p ⩽ 0.05 was considered statistially significant.
Results
Baseline characteristics
Four hundred and sixty-three patients were recruited (median age of 35 [26–45] years, 360 were male [77.8%]). Three hundred and seventy-six (81.2%) r-axSpA and 87 (18.8%) nr-axSpA patients were included. Other baseline characteristics were shown in Table 1. One hundred and forty-two (30.7%), 18 (3.9%), 5 (1.1%) patients had one, two, three or more traditional CV risk factors, respectively (Table 1). Baseline data of 463, 463, and 397 patients were available for calculating the FRS, SCORE, and QRISK3, respectively. Sixteen out of 463 (3.5 %), 13/463 (2.8%), and 5/397 (1.1%) patients were classified as having high CV risk according to the FRS, SCORE, and QRISK3 respectively.
Baseline characteristics of entire cohort, the subgroups of patients who did or did not develop cardiovascular events during subsequent follow-up.

Data are expressed as n (%) or mean (SD) or median (IQR).
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Disease Functional Index; bDMARDS, biological disease-modifying antirheumatic drugs; BMI, body mass index; COX-2, cyclooxygenase-2 inhibitor; CRP, C-reactive protein; csDMARDs, conventional synthetic Disease-Modifying Antirheumatic Drugs; CVE +ve, patients who developed cardiovascular events during subsequent follow-up; CVE −ve, patients who did not develop cardiovascular events during subsequent follow-up; ESR, erythrocyte sedimentation rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; MTX, methotrexate; NSAIDs, nonsteroidal anti-inflammatory drugs; SCORE, Systematic Coronary Risk Evaluation; SSZ, sulfasalazine; HLA-B27, human leukocyte antigen B27; TC, total cholesterol . QRISK3 means the third version of QEISK.
Baseline data of 463, 463, and 397 patients were available for calculating the FRS, SCORE, and QRISK3, respectively.
Statistically significant at p ⩽ 0.05.
Drug-use over time
At baseline, 377 (81.4%), 76 (16.4%), 30 (6.5%), and 77 (16.6%) patients were NSAIDs, SSZ, MTX, and bDMARDs users (Supplementary Figure 1). A further 23 (total 400, 86.4%), 86 (total 162, 35%), 31 (total 61, 13.2%), and 99 (total 176, 38%) patients were identified as using NSAIDs, SSZ, MTX, and bDMARDs during the follow-up assessments. The mean duration of NSAIDs, SSZ, MTX, and bDMARDs exposure during the period of this study, estimated from the number of study assessments where these drugs’ use was reported, was 6.2 ± 0.2 years, 4 ± 0.3 years, 1 ± 0.1 years, and 3.9 ± 0.3 years, respectively.
CV events during follow-up and the performance of the CV risk scores
A total of 5,501 patient-years of follow-up were available for analysis and 301 (65%) patients were being followed for ⩾10 years. After a median follow-up of 12 (7–19) years, 12 (2.6%) patients died. The cause of death was adjudicated to be due to CVD in 5 (1.1%) patients. Sixty-one patients (13.2%) developed a first nonfatal CVE (1.1 events per 100 patient-years), which included 18 (29.5%) IHD, 8 (13.1%) MI, 3 (4.9%) TIA, 14 (23%) stroke, 15 (24.6%) CHF, and 3 (4.9%) percutaneous transluminal coronary angioplasty for obstructive IHD.
Among the 61 patients who developed first CVE (CVE +ve group), 5 (8.2%), 7 (11.5%), and 1 (1.8%) patient were identified as high CV risk at baseline using FRS, SCORE, and QRISK3 (Figure 1). The proportion increased to 9 (14.8%), 11 (18%), and 1 (1.9%) using the mFRS, QRISK3*RA, and mSCORE, respectively. Overall, a discrepancy between predicted risk and observed CVEs was found. All risk scores underestimated CV risk and the HL test demonstrated poor model fit (p < 0.001, p < 0.001, and p = 0.039, respectively). FRS and QRISK3 underestimated CV risk in the middle and top deciles, but overestimated CV risk in the lower deciles. SCORE underestimated CV risk in the middle quartiles and overestimated CV risk in the top and lowest quartiles (Figure 2); the model fit of modified CV risk scores did not demonstrate significant improvement (HL test: p < 0.001, p < 0.001, and p = 0.033, respectively).

Cardiovascular risk scores in patients who did (CVE +ve) or did not (CVE –ve) develop cardiovascular events. Left: Original risk scores. Right: Modified risk scores using 1.5 multiplication factor recommended by EULAR (mFRS and mSCORE) or assuming patients’ CV risk was similar to RA patients (QRISK3*RA).

Hosmer–Lemeshow test for the goodness of fit for the observed and expected risk of cardiovascular event (CVE) according to the original and modified cardiovascular risk scores. Left: Original risk scores. Right: Modified risk scores using 1.5 multiplication factor recommended by EULAR (mFRS and mSCORE) or assuming patients’ CV risk was similar to RA patients (QRISK3*RA).
Baseline characteristics comparison between CVE +ve and CVE –ve group
The CVE +ve group were older, with more traditional CV risk factors and higher CV risk scores; the use of statins, antihypertensives, and aspirin were also more common. Higher inflammatory burden (as reflected by longer disease duration, higher BASDAI, CRP, and ESR) and worse function (higher BASFI) were observed in the CVE +ve group (Table 1).
Time-varying inflammatory burden and treatment predicting CV events
The time-varying univariable cox regression model revealed that a higher ESR (⩾20 mm/h) level was associated with a significantly higher risk of developing CVE during follow-up [HR: 2.07, 95% CI (1.10, 3.98), p = 0.008] (Table 2). In addition, active disease as indicated by a rising BASDAI or a high BASDAI (⩾4) also showed positive trend towards a higher risk of first CVE [HR: 1.17, 95% CI (1.00, 1.36), p = 0.052 and HR: 2.18, 95% CI (1.00, 4.32), p = 0.051, respectively]. ESR and CRP levels, baseline disease duration all types of NSAIDs, bDMARDs and csDMARDs were not associated with increased risk of CVE.
Univariable analysis with time-dependent Cox proportional hazard regression for the predictors of cardiovascular events.

BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; bDMARDS, biological disease-modifying antirheumatic drugs; BLDD, baseline disease duration; CI, confidence interval; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; ESR, erythrocyte sedimentation rate; FRS, Framingham Risk Score; HR, hazard ratio; NSAIDs, nonsteroidal anti-inflammatory drugs; SCORE, Systematic Coronary Risk Evaluation. QRISK3 means the third version of QEISK.
Variables are time-varying unless otherwise specified.
Parameters with a p < 0.06 are included in the multivariable analysis.
Statistically significant at p < 0.05.
Kaplan–Meier survival analysis was performed to illustrate the effect of high versus low inflammatory burden on time to first CVD event. A significant difference in the CV event-free survival between patients with ESR ⩾ 20 mm/h and ESR < 20 mm/h was demonstrated in Figure 3(a). A trend suggestive of lower CV event-free survival in patients with active disease (BASDAI ⩾ 4) versus low disease activity (BASDAI < 4, p = 0.05; Figure 3(b)) was also observed.
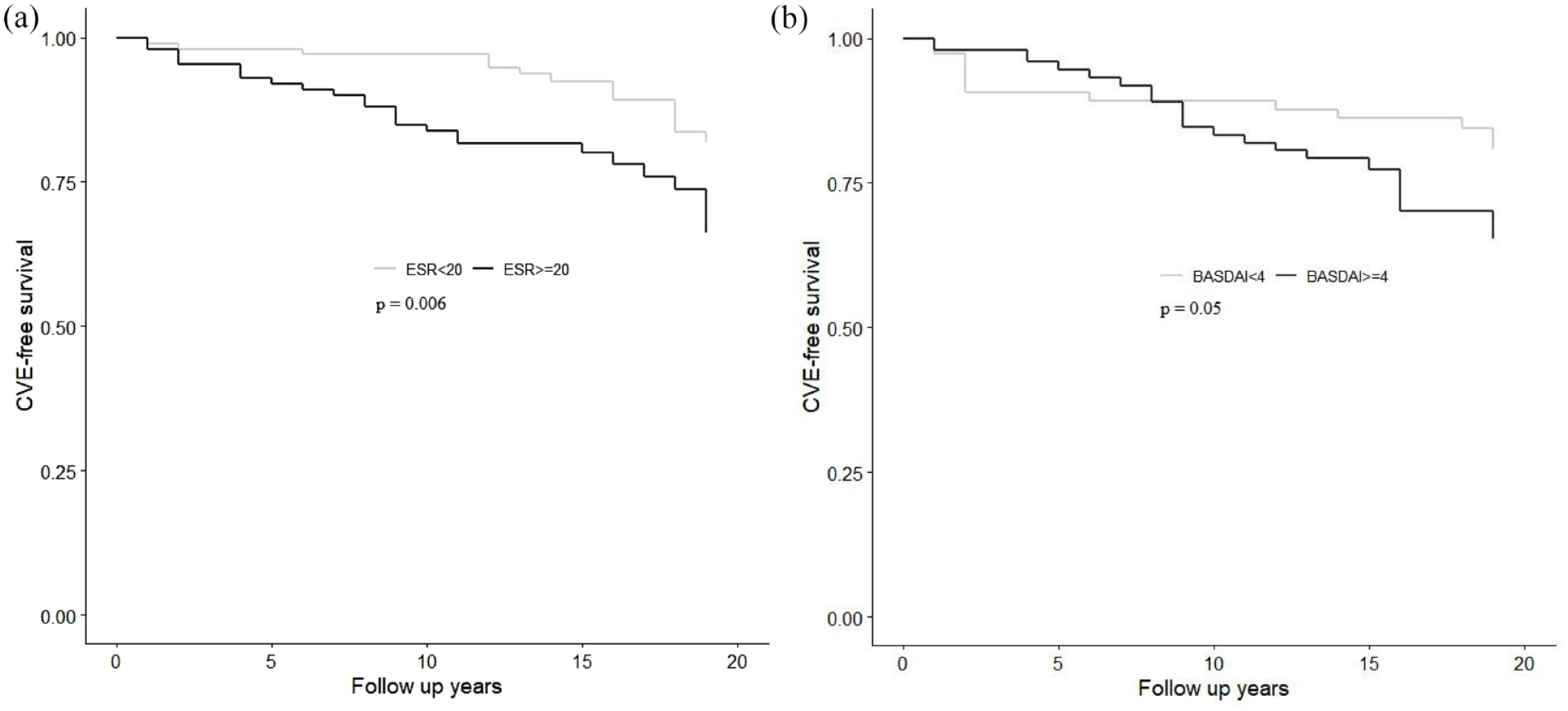
(a) Kaplan–Meier survival analysis between time-varying erythrocyte sedimentation rate (ESR) level and cardiovascular event (CVE). p = 0.006 for difference in CVE-free survival rate between patients with ESR ⩾ 20 mm/h and ESR < 20 mm/h during their follow-up intervals using log-rank test. (b) Kaplan–Meier survival analysis between time-varying Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) level and cardiovascular event (CVE). p = 0.05 for difference in CVE-free survival rate between patients with BASDAI ⩾ 4 and BASDAI < 4 during their follow-up intervals using log-rank test.
After adjusting for baseline disease duration and the traditional CV risk scores (FRS, QRISK3, and SCORE) in the multivariable models (Table 3), the association between time-varying high ESR level (ESR ⩾ 20 mm/h) remained significant, with a HR ranging from 2.74 to 2.95 (Table 3). The association between BASDAI and BASDAI ⩾ 4 with CVE was no longer significant after adjusting for covariates. No significant interactions were found between age and ESR ⩾ 20 mm/h (data not shown).
Multivariable analysis with time-dependent Cox proportional hazard regression for the predictors of cardiovascular events.
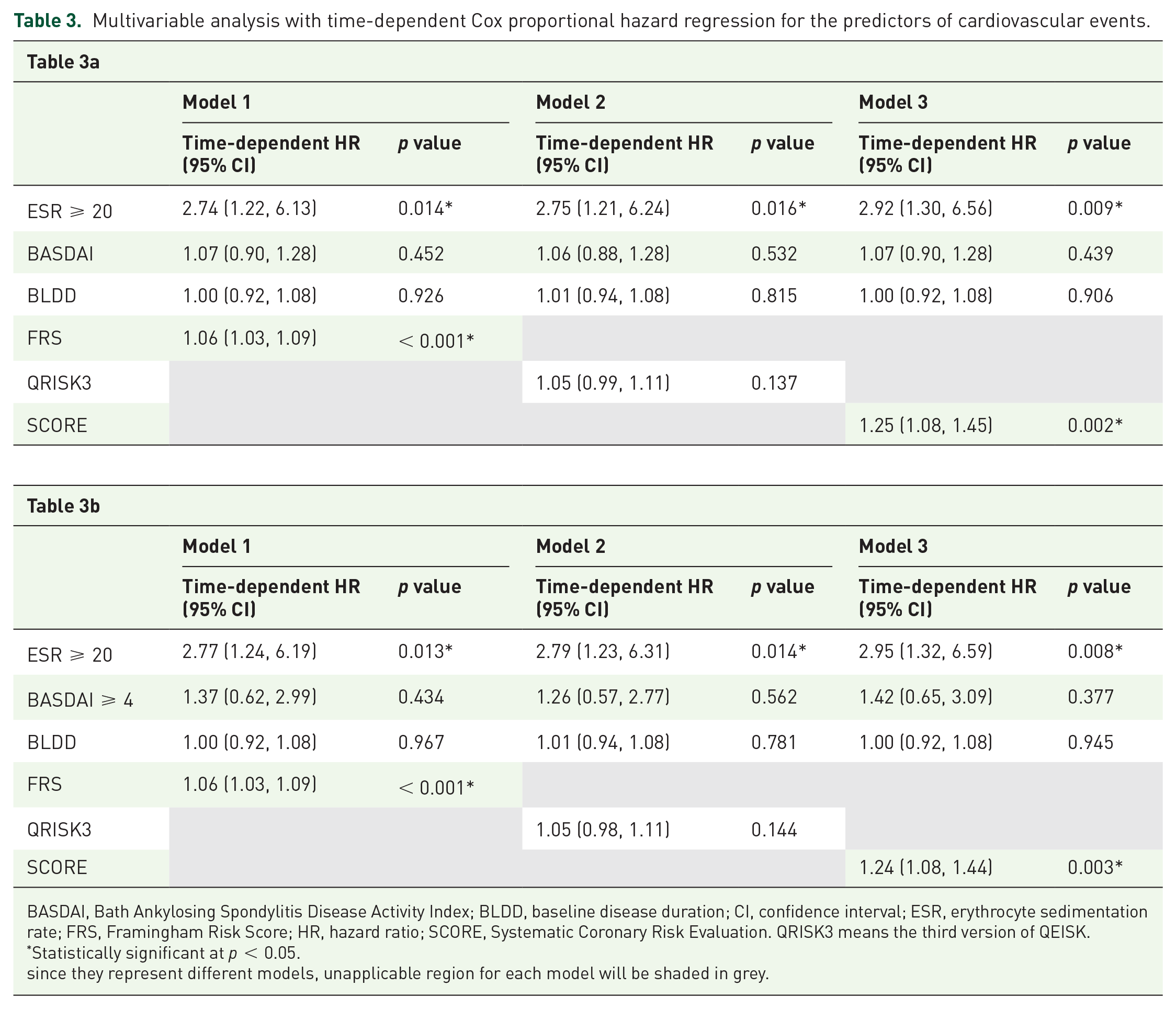
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BLDD, baseline disease duration; CI, confidence interval; ESR, erythrocyte sedimentation rate; FRS, Framingham Risk Score; HR, hazard ratio; SCORE, Systematic Coronary Risk Evaluation. QRISK3 means the third version of QEISK.
Statistically significant at p < 0.05.
since they represent different models, unapplicable region for each model will be shaded in grey.
Discussion
In our long-term follow-up study, we compared the observed incidence of CVEs with the traditional risk scores in mostly (321/463, 69%) newly diagnosed axSpA patients, and found that the risk scores underestimated the true CV risk. Adapting with multiplication factors for these CV risk algorithms has not yielded precise CVD risk estimation, confirming the finding by Navarini et al. 18 This discrepancy between the moderate discriminating abilities of the three scores (as continuous variables) and poor calibrating abilities of the cut-off values could be explained by the threshold selection (Supplementary Table 2). The cut-off values with best accuracy (highest Youden index) were much lower in the original FRS, QRISK3, and SCORE compared with the preset ones. We also tested whether the performance of the modified scores is improved by adopting the EULAR modification. Although the sensitivity of the various CV risk scores increased from 1.8–12.3% to 1.8–14.8%, a significant proportion of patients with CVE were still misclassified as having ‘low risk’ (Figure 1). A possible explanation for the underestimation of the CV risk is chronic inflammation in patients with AS. Chronic systemic inflammation can lead to endothelial dysfunction and accelerated atherosclerosis which cannot be accounted for by these scoring systems. Therefore, we hypothesize that the excess CV risk in axSpA was attributed to inflammatory burden (measured by disease activity and inflammatory markers). This is the first study to demonstrate that chronic inflammatory burden, as reflected by the increasing time-varying ESR level, can predict CV events in axSpA patients independent of baseline traditional CV risk.
Inflammation accelerated subclinical atherosclerosis in AS had been reported. 19 This study further demonstrates that active disease in axSpA is associated with an increased risk of developing CVE. Patients who were able to achieve and maintain low inflammatory burden with ESR < 20 mm/h over time appeared to have a significantly lower risk of CVD than patients with more active disease regardless of baseline traditional CV risk factors. Several other studies have reported similar results in RA and PsA. In RA, bouts of uncontrolled high disease activity were associated with a higher risk of CVD, 27 and reduced time-averaged disease activity was linked to fewer CVEs. 28 Remission in RA, defined as Clinical Disease Activity Index (CDAI) ⩽ 2.8, was found to significantly reduce CV risk markers, supporting remission as a management target. 29 In PsA, we have recently demonstrated that baseline and time-varying higher inflammatory burden was associated with an increased risk of CV events.30,31 Overall, these data suggest that inflammatory arthritis patients who can achieve and maintain remission or low disease activity during follow-up may be less likely to develop bouts of uncontrolled, sustained high systemic inflammation needed to drive the atherosclerotic process.
Previous study reported that baseline CRP and ASDAS predicted future increased arterial stiffness. 20 In contrast, we did not find any association between CRP level (baseline or time-varying) and future CVE. It is well known that CRP levels are not elevated in the majority of axSpA patients. 32 Even though baseline high-sensitivity C-reactive protein (hs-CRP) can independently predict future CVE in the general population, whether it can also predict CVE in axSpA patients deserves further studies.33,34 On the other hand, high ESR level (⩾20 mm/h) was associated with almost three folds increased risk of developing CV events in our study, concurred with the findings from the general population. 35 Elevations in ESR reflect disease states that involve increased plasma protein/fibrinogen levels such as autoimmune conditions or cardiovascular disease. Furthermore, CRP has a shorter half-life (4–7 hours), while ESR level can take weeks to completely normalize after a bout of inflammation. 36 Therefore, ESR may be a better marker for monitoring or following the course of inflammation over time. ESR were also reported to be a predictor for the presence of carotid plaques in patients with AS without CVD, 37 suggesting that ESR may be a better marker of ‘low-grade’ inflammation responsible for accelerating atherosclerosis in axSpA. In terms of disease activity marker, time-varying BASDAI (or BASDAI ⩾ 4) lost statistical significance after adjusting for the time-varying ESR level and the CV risk scores. Unfortunately, patient global assessment was not routinely performed in our cohort and therefore ASDAS cannot be computed. Whether ASDAS may be a better disease activity marker associated with CV event would need to be addressed in future study.
Long-term or a high-dose NSAIDs exposure are associated with increased risk of CV events, especially among patients with CV risk factors or CVD. 38 Variation in CV risk among different NSAIDs have been reported.39,40 Nonetheless, these effects may be different in axSpA patients who have increased inflammatory burden. While CV toxicity was observed with diclofenac, but not with Naprosyn use in a recent spondylarthritis cohort, 41 data from population-based study suggest that NSAID use may be cardioprotective, and frequent use of NSAID (medication possession rate [MPR] ⩾ 80%), especially celecoxib, showed a negative association with CV events as well as more baseline comorbidities compared with not frequent user or non-user in AS individuals.42–44 Non-exposure to NSAIDs was associated with over four-fold higher all-cause mortality (odds ratio [OR] = 4.35, 95% CI [1.75, 10.77]) in an AS cohort in which circulatory disease contributed to 40% of mortality. 2 Therefore, effect of NSAIDs use on CV event in patients with axSpA remained controversial. In the current study, we did not find any association between time-varying NASIDs or CoX-2 inhibitor and CVE, further prospective study is required to address this question.
Similarly, data on the use of biologic DMARDs and CVE in axSpA is scarce. A meta-analysis observed no association between TNFi drug and MI. 45 In contrast, a recent retrospective cohort study found that baseline use of anti-TNF in SpA patients is associated with lower risk for major adverse cardiovascular event (MACE). 46 Nonetheless, this study included all types of SpA patients and details including time-varying anti-TNF use and disease activity over time were omitted. After analyzing the detailed use of biologics as well as inflammatory markers and over time, we did not observe any effect of drug use and CV events, suggesting that effective suppression of inflammation is the key in preventing progression of atherosclerosis.
There are some strengths in our study. First, this is the first longitudinal cohort to investigate the effect of time-varying inflammatory burden and treatments as predictors for future CVE in patients with axSpA. Second, we included nr-axSpA population and found no differences in CV outcomes compared with r-axSpA subgroup. Third, this is the first long-term study with a large sample size to confirm that the traditional/modified CV risk scores underestimated CVE. Nonetheless, our study has some limitations. First, age restrictions in some CV risk score restricted the use of these risk scores in the younger axSpA patients. Second, whether time-varying ASDAS may be a better predictor of CV events in these patients deserves further study. Third, carotid ultrasound was not performed in this cohort. Whether the presence of carotid plaques can predict cardiovascular event in axSpA would be worth addressing in future studies.
Summary
Increased inflammatory burden as reflected by elevated ESR levels (ESR ⩾ 20) over time was associated with increased risk of CVE, while the use of NSAIDs and DMARDs were not. These results highlight that tight control of inflammation may be an important target to reduce CV risk in axSpA patients.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221122401 – Supplemental material for High inflammatory burden predicts cardiovascular events in patients with axial spondyloarthritis: a long-term follow-up study
Supplemental material, sj-docx-1-tab-10.1177_1759720X221122401 for High inflammatory burden predicts cardiovascular events in patients with axial spondyloarthritis: a long-term follow-up study by Lin-Hong Shi, Steven H. Lam, Ho So, Edmund K. Li, Tena K. Li, Cheuk-Chun Szeto and Lai-Shan Tam in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X221122401 – Supplemental material for High inflammatory burden predicts cardiovascular events in patients with axial spondyloarthritis: a long-term follow-up study
Supplemental material, sj-docx-2-tab-10.1177_1759720X221122401 for High inflammatory burden predicts cardiovascular events in patients with axial spondyloarthritis: a long-term follow-up study by Lin-Hong Shi, Steven H. Lam, Ho So, Edmund K. Li, Tena K. Li, Cheuk-Chun Szeto and Lai-Shan Tam in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
