Abstract
With varying prevalence and manifestations depending on the underlying disease, thoracic involvement is one of the major factors determining morbidity and mortality in patients with rheumatic diseases (RDs). The most frequent pulmonary complication is interstitial lung disease (ILD), but other thoracic manifestations can also be present. Often, the only way to depict these extra-articular manifestations of disease is through imaging. Making an early diagnosis and thus aiding in the appropriate management requires radiologists to possess a comprehensive understanding of the typical imaging findings and the accompanying clinical context. In this review, the common features of thoracic involvement in RD are systematically described, with illustrations and explanatory drawings. ILD patterns, rheumatoid nodules, bronchial involvement, esophageal involvement, and pleural and pericardial disease are among the features discussed.
Background
Rheumatic diseases (RDs) are a broad and diverse category of systemic illnesses that are characterized by immune system dysregulation and inflammation, which can cause tissue damage and fibrosis. RDs often show extra-articular manifestations (EAMs), which may even occur before articular disease. Cardiovascular and pulmonary EAMs play a significant and potentially fatal role in patients with RDs. The most affected anatomical region is the thorax, and a significant percentage of individuals with RDs have lung involvement. While there is overlap, distinct patterns of pulmonary involvement are linked to each type of RD (Table 1), although they are not very specific. The most prevalent and severe type of lung involvement in RDs is interstitial lung disease (ILD). In individuals with RDs, ILD is a major cause of death. Imaging is often the only option for depicting these extra-articular features of the disease. In addition, imaging is utilized to evaluate treatment response (cure, partial response, and no response), which is crucial in determining the optimal course of action for the patient. 1
Distinct patterns of pulmonary involvement in RDs.

DAD, diffuse alveolar damage; LIP, lymphocytic interstitial pneumonia; NSIP, non-specific interstitial pneumonia; OB, obliterative bronchiolitis; OP, organizing pneumonia; RA, rheumatoid arthritis; RDs, rheumatic diseases; SLE, systemic lupus erythematosus; SSc, systemic sclerosis; UIP, usual interstitial pneumonia.
Findings and procedure details
Rheumatoid arthritis
While genetic and nongenetic variables account for roughly two-thirds and one-third of the risk, respectively, the precise pathophysiology of rheumatoid arthritis (RA) is still poorly understood. 2 Up to half of all RA patients develop EAMs, which are linked to higher rates of morbidity and death as well as significantly lower quality of life. 3 Compared to seronegative patients, seropositive patients are more likely to show EAMs. It is interesting to note that although inflammation is the main pathologic condition in joints, pulmonary disease is characterized by fibrosis rather than a clear first phase of inflammation. 4 The risk factors for developing ILD in RA patients, in addition to genetic predisposition (risk alleles), include age of onset, seropositivity (Rheumatoid Factor, Cyclic Citrullinated Peptide), high disease activity, and smoking. 5
Unlike in other RDs, usual interstitial pneumonia (UIP) is the most common pattern of RA-ILD, followed by non-specific interstitial pneumonia (NSIP). 6 Along with peripheral traction bronchiectasis and/or bronchiolectasis, the primary computed tomography (CT) characteristics of UIP include basilar and peripheral predominant reticular opacities (Figure 1) and lung architectural distortion. Honeycombing (Figure 2) is not always present. Making the distinction between idiopathic pulmonary fibrosis (IPF) and RA-associated interstitial pneumonia can be difficult because of their similar imaging presentations. Exuberant honeycombing, anterior upper lobe involvement (Figure 3), and the straight-edge sign are three characteristics that prefer RDs with typical interstitial pneumonia over IPF. 7 The straight-edge sign occurs when the fibrosis is delineated by a straight interface with the normal orthogonal lung, and when there is no significant superior extension along the lateral chest wall. 8 The histological findings of RA-UIP are similar to those observed in IPF, although prominent lymphocyte aggregation may sometimes be seen in RA-UIP. 5

Axial CT image of UIP in RA. Two key features of UIP are visible in this image, namely peripheral basal reticulations (red) and honeycombing (black). Reticulations are a pattern of linear opacification in the lung, and in UIP they are typically located in the periphery and the basal regions of the lungs. Honeycombing refers to clustered cystic air spaces with irregularly thickened walls, often with a subpleural, peripheral, and basal distribution. 9

Honeycombing (black) is a descriptive term on CT for fibrotic walled cysts. In severe cases, honeycombing can also be visible on a chest radiograph, as in the upper image. The lower axial CT image also shows ground-glass opacities (brown). A ground-glass opacification is a region of increased attenuation in the lung with maintained bronchial and vascular markings. Importantly, ground-glass opacity is a non-specific sign and has a wide etiology. 9

Axial CT image of UIP in a man in his mid-50s with RA. Apart from the traction bronchiectasis, peripheral reticulations, and ground-glass opacities, there is honeycombing present in both upper lobes. Note the anterior upper lobe sign (arrows), the fibrosis is mainly concentrated along the anterior aspect of the upper lobes which makes RDs with typical interstitial pneumonia a more likely diagnosis than idiopathic pulmonary fibrosis.
Around 20% of individuals develop rheumatoid nodules, which are highly specific for RA and more common in men and cigarette smokers. 10 In 80% of instances, lung nodules are related to subcutaneous nodules. Rheumatoid lung nodules on CT scans are lobulated or round, ranging in size from a few millimeters to a few centimeters, and are frequently found in a peripheral distribution, with a predominance in the mid-upper lung (Figure 4). They can appear as a single nodule, although they are usually numerous, and can include calcifications. Nodules might become larger and more numerous, but they can also stay the same for years, or even disappear on their own. 11 A smooth border, calcification, peripheral location, pleural contact, satellite nodules, and cavitation are CT features that support a diagnosis of rheumatoid nodules over malignancy. 12
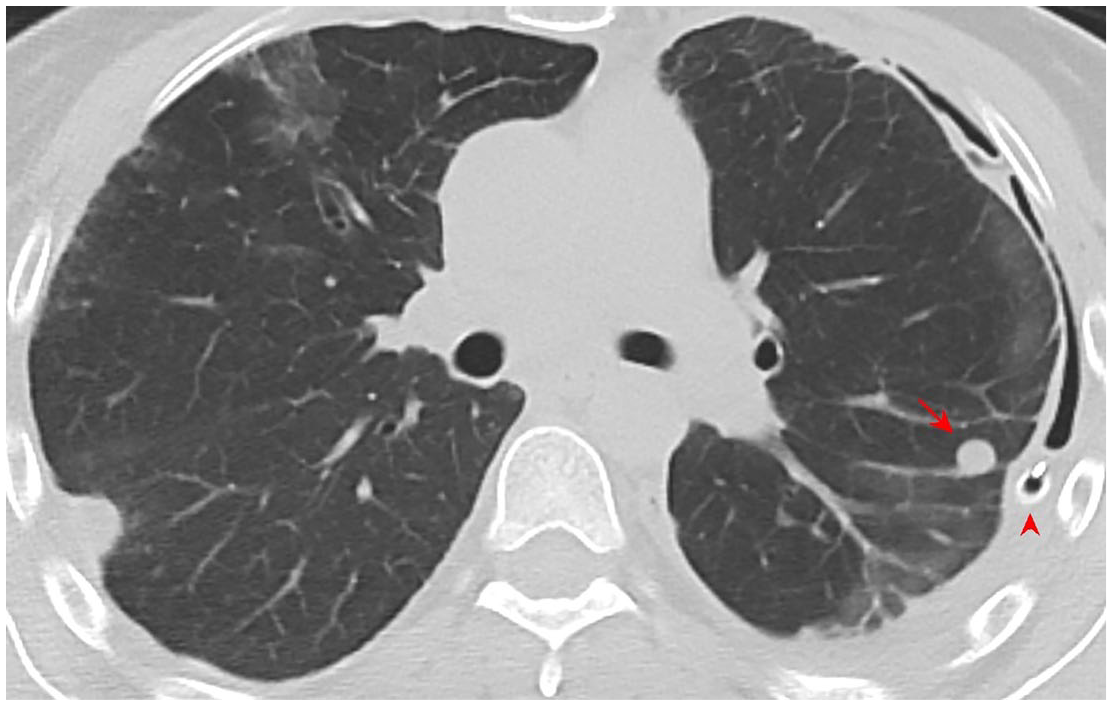
Axial CT image of a rheumatoid nodule in a woman in her late 30s with RA. The nodule (arrow) is round, a few millimeters in diameter, and follows a peripheral distribution, which is typical. This patient also has the complication of recurring pneumothorax, for which an intercostal catheter has been placed on the left (arrowhead).
In 39%–60% of RA patients, there is evidence of airway involvement. The most prevalent type of upper airway involvement is cricoarytenoid joint arthritis, which is typically clinically silent. 13 Lower airway diseases may include bronchial hyperresponsiveness, bronchiectasis, or bronchiolitis (Figure 5). 14 The incidence rises with disease duration. 15 Obliterative bronchiolitis, a type of bronchiolitis, can be a pulmonary manifestation of RA and is a small airway disease characterized by the destruction of bronchiolar epithelium and airflow obstruction.

Sagittal CT image in a man in his mid-40s with RA. There is a zone of bronchiolitis in the right upper lobe. Two main features of bronchiolitis are present: centrilobular small nodules in a tree-in-bud pattern and mosaic attenuation. Centrilobular lung nodules are small lung nodules that are located centrally within the secondary pulmonary lobules, whereas a tree-in-bud pattern describes multiple areas of these centrilobular lung nodules with a linear branching pattern, resembling a budding tree. Mosaic attenuation is a term used for a patchwork of regions of differing lung attenuation. Irregular pleural thickening can also be seen (blue). 9
Though typically asymptomatic, pleural illness is frequent in RA patients. The pleural effusion exhibits the distinctive clinical features of an exudate. Low glucose levels (<40 mg/dL), pleural fluid acidosis (pH < 7.20), high lactate dehydrogenase levels (>700 IU/L), high cholesterol levels (>65 mg/dL), and negative bacterial smears or cultures are typical findings of rheumatoid sterile empyematous pleural effusion. 16 Effusions in the acute context are usually unilateral, tiny, and accompanied by pleural enhancement. Prolonged inflammation of the pleura causes thickening (Figure 5) and loculation of the fluid within the parietal and visceral pleura, resembling empyema. 17 Severe pleuritis can cause fibrothorax with restricted pulmonary physiology if left untreated or if it recurs. 18
In RA, pulmonary arterial hypertension (PAH) is rarely an isolated finding, which is then typically observed in elderly patients with chronic illness. 19 Much more frequently, PAH secondary to ILD in RA might result in right heart failure and chronic respiratory failure. 20 Dilated main pulmonary artery diameter greater than 29 mm, main pulmonary artery diameter to ascending aorta diameter ratio greater than 1, right heart chamber enlargement, and right ventricular hypertrophy are CT findings suggestive of PAH. The lungs may show mosaic attenuation, implying variations in pulmonary perfusion across different pulmonary regions. 21
Systemic sclerosis
Systemic sclerosis (SSc) is a multisystemic chronic disorder in which the overproduction of collagen causes structural abnormalities in the visceral organs and thickening of the skin. The incidence peaks in the third to fifth decade of life, with a predisposition toward females. 22 Early-stage lung fibrosis in SSc-ILD is caused by vascular injury, inflammation, and immune system activation, which are the initial changes in the pathophysiology of the disease. 23 According to recent studies, lung ultrasound (LUS) could be a viable option in the future in the screening process for ILD-SSc in patients who are asymptomatic or exhibit nonspecific symptoms. By evaluating B-lines, the sonographic sign of lung interstitial involvement, LUS can assess ILD. 24
SSc patients also have a higher tendency to develop lung cancer, usually adenocarcinoma, with a relative risk of malignancy ranging from 1.8 to 6.5. It most commonly affects patients with lung fibrosis. 25
NSIP (Figures 6 and 7) is the most common SSc-ILD pattern, followed by UIP, which may be found more commonly in individuals with an SSc and RA overlap syndrome. 26 CT in NSIP shows traction bronchiectasis, basilar and peripheral predominant ground-glass opacity (GGO), and fine reticulations (Figure 7) with or without immediate subpleural sparing, although subpleural sparing is more typical. 1 Traction bronchiectasis is frequently more central, while fibrosis is usually uniform and symmetric. When compared to IPF, SSc-ILD often exhibits a larger degree of ground-glass attenuation and reticular abnormalities are typically less coarse (Leslie). Treatment seldom reverses the ground glass opacities typical of NSIP, indicating that fine fibrosis rather than alveolitis may be their cause. 27

Axial CT image of NSIP in a woman in her mid-50s with SSc, illustrating the key feature namely peripheral reticulations (red). 9
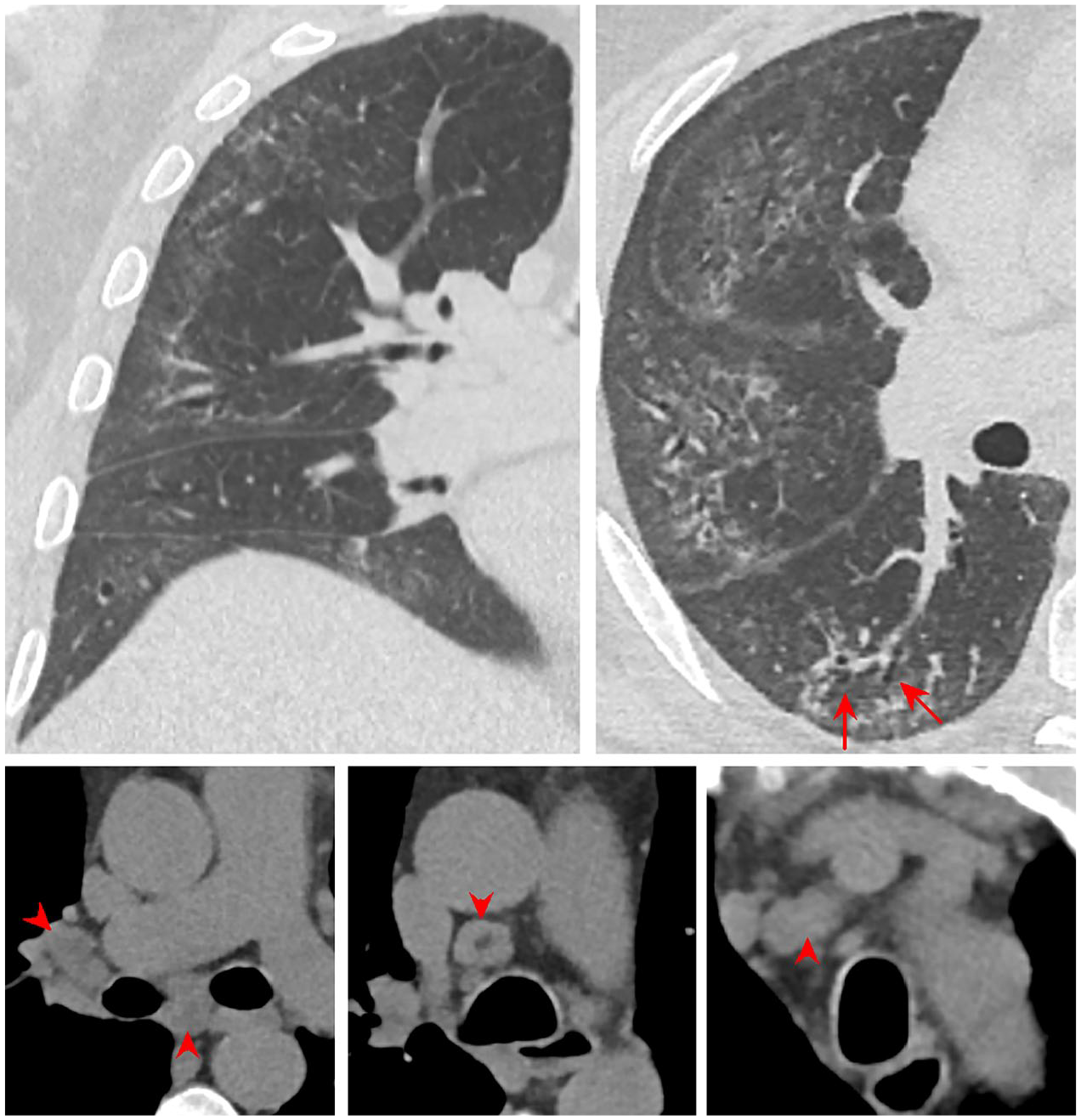
Coronal and axial CT images of NSIP in a man in his late 40s with SSc. Multiple key features of NSIP are visible: traction bronchiectasis (arrows), peripheral ground-glass opacities, and reticulations with sparing of the immediate subpleural space. There is also right paratracheal lymphadenopathy and right hilar lymphadenopathy (arrowheads).
Compared to other RDs, airway involvement in SSc is uncommon. 28 Likewise, pleural involvement in SSc affects only about 5% of patients, most often those with a diffuse cutaneous form, and is less common than pericardial disease. 29
PAH (Figure 8) is the predominant manifestation of pulmonary vascular involvement in RDs, and it primarily affects SSc patients, being detectable in 8%–12% of cases. In contrast to ILD, PAH rarely is the presenting manifestation of the disease; instead, it frequently happens 10–15 years after SSc is diagnosed. 27
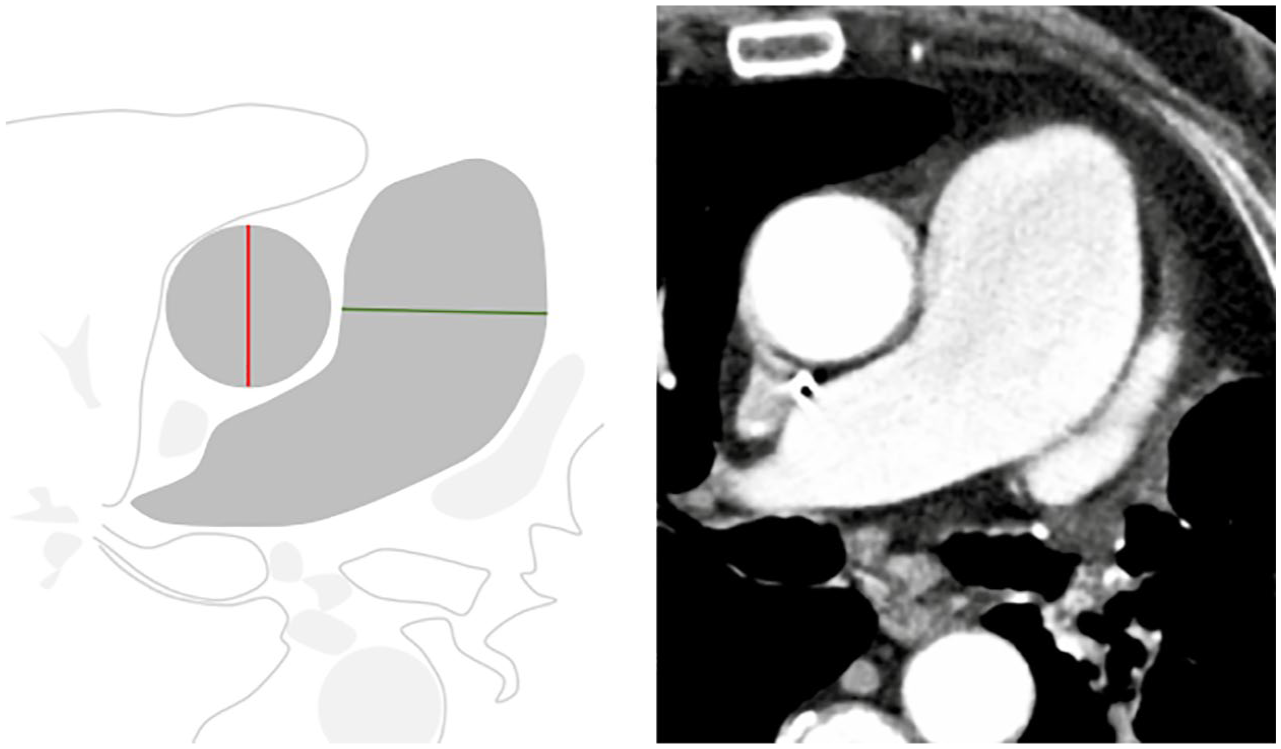
PAH in a man in his mid-60s with SSc. On the axial CT slice, the diameter of the main pulmonary artery (green line) is much larger compared to the diameter of the aorta (red line), indicating chronic PAH. 9
Esophageal involvement (Figure 9) is frequent, with up to 75%–90% of patients noting abnormalities on esophageal mobility testing. 30 This causes a dilated esophagus and a wide gastro-esophageal junction, which can lead to reflux and the potential complication of aspiration pneumonia. 31

Esophageal involvement in SSc. Barium swallow shows the absence of peristaltic waves in the lower two-thirds of the esophagus. On coronal CT, this esophageal dilation (red) is clearly visible. 9
Furthermore, more than 50% of individuals have mediastinal lymphadenopathy (Figure 5), which may be a response to interstitial abnormalities. 32
Idiopathic inflammatory myopathies
Because the spectrum of idiopathic inflammatory myopathies (IIMs) is very heterogeneous, different conditions within this spectrum are linked with different frequencies of ILD. Three main subgroups of IIMs have been identified based on clinical, histopathologic, and immunologic features: polymyositis (PM), adult and juvenile dermatomyositis (DM), and inclusion body myositis. 33 It is unclear exactly how lung involvement in IIMs develops. However, some data indicate that the lung may be the location of illness initiation due to frequent exposure to environmental immunogenic stimuli, at least in the subgroups with a high prevalence of lung involvement. 34
Autoantibodies play a significant role in the diagnosis of myositis. Myositis-specific antibodies (MSAs) are predictive of ILD and prognosis of the patients. The antisynthetase antibodies (ARS-Abs) and the MSAs directed against the RNA helicase encoded by melanoma differentiation-associated gene 5 (anti-MDA-5) are the MSAs with the most significant impact on the lungs. Antisynthetase syndrome is a phenomenon characterized predominantly by some combination of ILD, myositis, arthritis, Raynaud’s phenomenon, fevers, and mechanic’s hands in the presence of ARS-Abs. It is associated with chronic ILD. 35
Reticulation and/or ground-glass opacities, with or without consolidation and without honeycombing, are the most prevalent high-resolution CT (HRCT) patterns in PM or DM. These patterns correlate best with NSIP. 36 Organizing pneumonia (OP) (Figure 10), whether or not in combination with NSIP, is also possible. OP is a typical manifestation of anti-MDA-5 and is associated with a high risk of rapidly progressive ILD with a diffuse alveolar damage pattern. 35 Among the frequent CT findings of OP are ground-glass opacities, nodules, peripheral and peribronchovascular consolidation, and a reverse halo sign. 37 In addition, OP may show signs of overlap with NSIP and IPF. In this instance, the lung is mostly affected by reticular opacities with architectural distortion, while ground-glass opacities and consolidations are detected relatively less frequently. 38 Furthermore, as a result of the rupture of alveoli or bullae, pneumothorax or pneumomediastinum can occur early in myositis. 39 Malignancy, weak respiratory muscles, pulmonary hypertension, and aspiration pneumonia are other possible pulmonary problems. 33
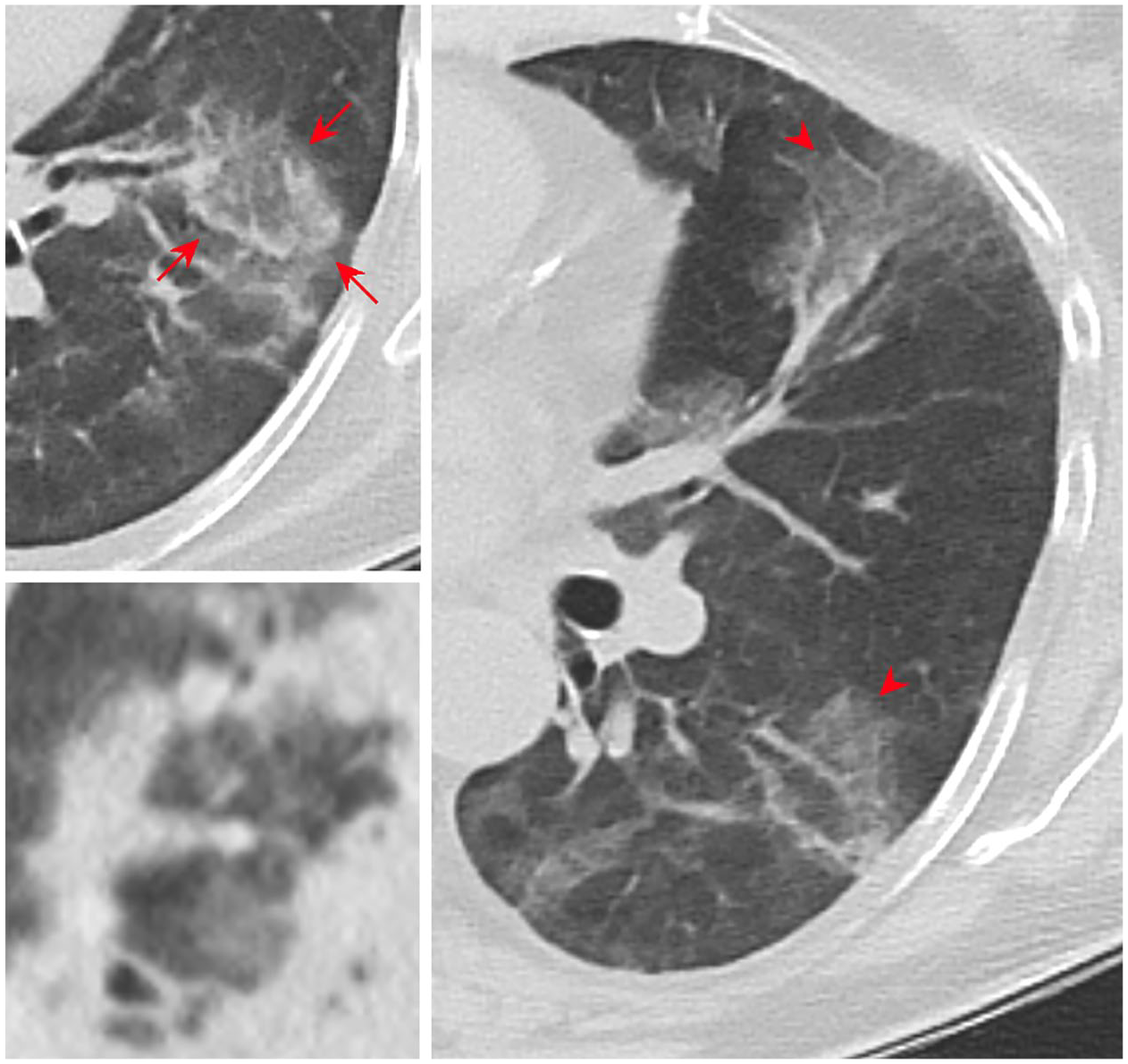
A man in his early 40s with OP. Axial CT demonstrates ground-glass opacities (arrowheads) with a peripheral and peribronchovascular distribution and the reversed halo sign in the upper and lower left image (arrows). The reversed halo sign or atoll sign shows ground-glass opacity with a peripheral ring of consolidation around it, which can be complete or incomplete.
Sjögren’s syndrome
Sjögren’s syndrome (SS) is a chronic autoimmune disease marked by inflammatory alterations in the exocrine glands, which clinically manifests itself as dry mouth and eyes. Around 30% of patients have systemic symptoms. Patients with systemic lupus erythematosus (SLE), SSc, RA, and other autoimmune illnesses can all have SS as a secondary condition, but SS can also occur as a primary condition on its own. 40
ILD primarily arises after SS onset, and as the illness progresses, the prevalence of ILD also increases. 41 Historically, lymphocytic interstitial pneumonia (LIP) (Figure 11) has been associated with SS, followed by NSIP, UIP, and OP. In LIP thin-walled cysts with a predominance in the lower lobe that are close to vessels (perivascular distribution) can be found with or without ground-glass opacities or septal thickening. 37 Bronchovascular thickening and centrilobular nodules are less frequent. Mixed alveolar and interstitial infiltrates are typical; these infiltrates frequently have a nodular or finely reticular pattern. 42 New studies, however, suggest that the predominant pattern in SS may in fact be NSIP, and not LIP as traditionally thought. These different findings reflect the heterogeneity of SS itself. 43

LIP in a woman in her early 50s with SS. The axial CT shows thin-walled cysts (arrows) with a lower lobe predominance and a perivascular distribution. These are caused by multifocal lung destruction, typical of SS. Furthermore, there are some ground-glass opacities and interstitial thickening along lymph channels (arrowheads).
The hallmark of respiratory involvement in SS is airway disease. 44 Chronic active inflammation is accompanied by lymphoplasmacytic infiltration and fibrotic alterations of central and peripheral bronchi and bronchioles. It manifests as bronchiectasis or bronchiolectasis, bronchial wall thickening, and areas of centrilobular opacities. 45 On expiratory CT scans, mosaic attenuation—a sign of obstructive bronchiolitis—can occasionally be seen. 46 Nodular centrilobular or ground-glass attenuation, modest thickening of interlobular septa, and bronchovascular bundles, and, rarely, air cysts are among the CT findings associated with follicular bronchiolitis, occurring in patients with SS. 47
Involvement of the pleura is uncommon. 48 In addition, a higher prevalence of lymphoma is observed with voluminous mediastinal adenopathy. 1 The most frequent neoplasm in individuals with primary SS is mucosa-associated lymphoid tissue (MALT) lymphoma. 49 MALT lymphoma in the lung appears on CT as solitary or multiple nodules or masses, sometimes with areas of ground-glass attenuation or airspace consolidation. 50 MALT lymphomas typically appear along a bronchovascular bundle, and traction bronchiectasis may be present alongside them. Furthermore, there may be interlobular septal thickening within or around the lesion. 51
Systemic lupus erythematosus
SLE is an autoimmune disease defined by immune complex deposition against nuclear antigens and tissue-binding autoantibodies. There is a significant predisposition for African Americans and women, with a peak frequency between the ages of 20 and 50. Between 50% and 60% of patients have pleural and pulmonary involvement. 52 The vast majority of the pulmonary involvement is due to serositis. SLE rarely has parenchymal involvement, impacting only about 10% of patients. The most prevalent histopathologic pattern is NSIP, although LIP, OP, and UIP are also possible. 53
Very rarely, patients may experience acute respiratory failure due to diffuse alveolar hemorrhage (DAH), a condition that has a 50% fatality rate. 54 In terms of risk factors for the development of DAH, clinically and serologically active disease is the most significant. 41 Another rare complication of SLE with a potentially devastating outcome is lupus pneumonitis, which is reported in 1%–4% of patients. Typical clinical features are fever, cough, dyspnea, hemoptysis, and hypoxemia. This can even be the first manifestation of previously unrecognized SLE. The radiological findings resemble acute interstitial pneumonia, showing bilateral GGO or patchy consolidation predominantly in the lower lobes, with or without pleural effusion. 55
Airway disease is an unusual SLE manifestation. Bronchial wall thickening and bronchiectasis are found on chest HRCT in roughly 20% of SLE patients, but they are often clinically silent. 56 Likewise, the prevalence of SLE-PAH is uncommon, occurring in only about 5% of patients. 27
The most frequent pulmonary symptom of SLE is pleuritis (Figure 12). 57 Up to 50% of SLE patients experience a clinically significant pleural effusion at some point over the course of their disease, and these effusions can either be uni- or bilateral. 41 Pleural effusion by itself is a nonspecific finding on imaging. Diagnostic thoracentesis guided by CT or US can help distinguish it from other causes of pleural effusion. 58 High titers (>1:160) of antinuclear antibodies in the pleural fluid support the diagnosis of lupus pleuritis in patients without a previous diagnosis of SLE. 59

An axial CT image of pericardial (arrows) and pleural (arrowheads) effusion in a woman in her early 40s with SLE. The pleural effusion is unilateral.
Patients with SLE frequently develop cardiac disease, in which the coronary arteries, myocardium, valves, conduction system, and pericardium may be affected. The most frequent symptom (30%) is pericarditis, which is typically latent. 60 Furthermore, SLE significantly increases the risk of pulmonary embolism, with a relative risk of 4.9 compared to individuals from the general population. 61
Mixed connective tissue disease
Mixed connective tissue disease (MCTD) is a distinct clinical entity with an overlap of SLE, SSC, and inflammatory myositis. The most relevant immunolaboratory marker of MCTD is the anti-ribonucleoprotein antibody, which is specific for this condition. 62 Raynaud’s phenomenon, polyarthritis, myositis, sclerodactyly, and esophageal dysmotility are common clinical characteristics of MCTD; renal disease is less common than in other systemic connective tissue diseases. 63 An increasing amount of data suggests that ILD and PAH are very common in patients with MCTD. ILD and PAH are major complications linked to increased mortality. 64 The most common ILD pattern is NSIP, while UIP, OP, and traction bronchiectasis are less common. 63
Extra-pulmonary findings
Routine imaging of the chest visualizes anatomical regions adjacent to the lungs. Therefore, chest imaging may demonstrate findings of RDs in these areas, which might have clinical significance and need to be reported. An example of this is upper abdomen small bowel involvement in SSc. A common sign related to small bowel involvement is the hide-bound sign, which describes the narrow spacing between the valvulae conniventes in a dilated small bowel (Figure 13).

Small bowel involvement in a woman in her mid-50s with SSc. This axial CT is a great example of the hide-bound sign: luminal dilation (diameter >3 cm) and crowding of the folds of the mucosal valves (red). Asymmetric smooth muscle atrophy of the inner circular muscularis layer in relation to the outer longitudinal layer is the cause of this sign. The bowel becomes foreshortened as a result of the longitudinal layer contracting, and the valvulae conniventes are closely packed. 9
Furthermore, it stands to reason that the ribs, spine, sternomanubrial joint, shoulder (Figure 14), and chest wall may all be affected by RA or other RDs. Subchondral cysts, sclerosis, ankylosis, and erosions are all visible on radiographs and CT scans.

Shoulder lesions in a man in his late 60s with RA. There are multiple erosions (red), for example, on the greater tuberosity and anatomic neck of the humerus. Furthermore, numerous subchondral cysts (dark blue) are visible and there is osteolysis of the outer 1/3 of the clavicle (purple, top image). Subsequently, the glenohumeral joint narrows (light blue, top image), there is superior migration of the humeral head (red arrows, lower image), and the subacromial space is reduced. 9
Conclusion
Thoracic manifestations of RDs are common, potentially fatal, and often precede articular disease. Radiologists must have a thorough awareness of the typical imaging findings along with the supporting clinical context to make an early diagnosis and to help guide appropriate management.
