Abstract
Background:
The incidence of various types of vasculitis conditions over time, specifically during coronavirus disease 2019 (COVID-19), is unknown.
Objectives:
We aimed to assess recent trends in vasculitides and the effect of the COVID-19 pandemic on these trends.
Design:
We conducted a retrospective analysis of Israel’s largest Health Maintenance Organization, which covers over 4.7 million patients and represents 55% of the country.
Methods:
We calculated the age- and sex-adjusted incidence of giant cell arteritis (GCA), Takayasu, ANCA-associated vasculitis (AAV), IgA vasculitis, cryoglobulinemia, and Behcet’s disease (BD) during 2007–2021. We analyzed associations of COVID-19 with the incidence of each of the examined conditions.
Results:
During 2007–2021, the adjusted annual incidence decreased from 7.9 (95% confidence interval (CI) 3.5–17.9) to 1.5 (95% CI 0.7–3.6) per 100,000 for GCA, from 5.2 (95% CI 2.7–11.1) to 1.5 (95% CI 0.7–3.3) per million for IgA vasculitis, and from 6.3 (95% CI 3.0–13.5) to 1.0 (0.5–2.5) per 100,000 for BD. The relative risks for these conditions decreased: 0.92 (95% CI 0.91–0.93), 0.93 (95% CI 0.89–0.98), and 0.90 (95% CI 0.85–0.94), respectively. The incidences of Takayasu, AAV, and cryoglobulinemia remained unchanged. The COVID-19 pandemic was not associated with changes in the incidence of any examined vasculitides.
Conclusion:
The incidences of GCA, IgA vasculitis, and BD decreased substantially in Israel during 15 years and were unaffected by the COVID-19 pandemic. Future studies should focus on possible environmental contributions to these findings.
Background
The vasculitides are a group of conditions characterized by a systemic inflammatory process of blood vessel layers that may result from end-organ ischemia. The revised International Chapel Hill Consensus Conference (CHCC) from 2012 classifies vasculitides according to the predominant size of the vessels involved. 1 Among the vasculitides are large-vessel vasculitis (giant cell arteritis (GCA) and Takayasu), medium-vessel vasculitis (polyarteritis nodosa (PAN)), small-vessel vasculitis (e.g., granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA), eosinophilic granulomatosis with polyangiitis (EGPA), IgA vasculitis, and cryoglobulinemic vasculitis). In addition, Behcet’s disease (BD) can affect any size of artery or vein.
Incidences of vasculitides have been shown to vary with time and geography. For instance, the incidence of GCA declined from 1997 to 2019 in Sweden. 2 On the other hand, the annual incidence of ANCA-associated vasculitis (AAV) in Norway was reported to rise from 17.9 to 29.2 cases per million between 1999 and 2013. 3 Similar increases were observed in other areas of the world.4,5 The changing incidence of vasculitides is attributed to the introduction of the CHCC criteria, the availability of commercial antibody tests and imaging studies, and the overall rise in population age. 6
Following the coronavirus disease 2019 (COVID-19) pandemic, exposure to the SARS-CoV-2 virus and vaccine have been suggested to trigger vasculitides.7,8 IgA vasculitis, AAV, and cryoglobulinemic vasculitis were associated with new-onset diagnosis and relapse after acute infection of SARS-CoV-2 or receiving its vaccine.9–11 Most of these data are based on case series or small-scale studies focusing on a single vasculitis condition. In the current study, we aimed to evaluate the incidences of the major vasculitides over time in a large-scale cohort and to analyze the possible effects of the COVID-19 pandemic on the incidences of vasculitides.
Methods
Population
We extracted data from Clalit Health Services (CHS), Israel’s largest health maintenance organization. CHS insures more than 50% of the Israeli population (4.7 million patients). The electronic individual medical records are available and updated in real time at the patient level. The data included patient demographics, medical history, laboratory findings, hospitalizations, outpatient clinic visits, community and hospital diagnoses, and medication purchases. The CHS population can be considered a reasonable reflection of the overall Israeli population. 12 Data were extracted using the Clalit Research Data sharing platform powered by MDClone.
We calculated the crude and estimated incidences of the vasculitides in individuals insured by CHS. The data were extracted from the annual publications of the Israeli Central Bureau of Statistics. We used the same source to calculate the number of patients insured by CHS per year and by age group.
Case identification
We inspected the records of individuals aged >18 years during 2007–2018 diagnosed with vasculitis according to ICD-9 codes of GCA, Takayasu arteritis, AAV, IgA vasculitis, cryoglobulinemia, and BD (Supplemental Table 1). AAV included GPA, EGPA, MPA, and PAN. As the ICD-9 codes for MPA and PAN are identical, distinguishing between these conditions was not feasible, thus including PAN as well. The COVID-19 period was defined as the years 2020–2021. Diagnoses of vasculitis were only considered if a medication pertinent to vasculitis treatment was purchased in the year following the diagnosis. The drugs included glucocorticoids (of any type), disease-modifying antirheumatic drugs (including methotrexate 10–25 mg/week, azathioprine 50–150 mg/day, leflunomide 10–20 mg/day, colchicine 0.5–2 mg/day, apremilat 60 mg/day), rituximab, other biological therapies (e.g., TNF-alpha inhibitors and interleukin-6 inhibitors), and cyclophosphamide.
Statistical analysis
The data were described using percentages, means, and standard deviations (for normally distributed variables) and medians and interquartile ranges (for non-normally distributed variables). Based on the CHS and Israeli Central Bureau of Statistics databases, we calculated the insured population in CHS by age per year. Then, we applied generalized linear models using negative binomial regression to calculate the relative risks (RRs) of the estimated annual incidence of each vasculitis condition during the study period (the RR was calculated per trend of the entire study period). Each model was adjusted for sex and age, and the annual estimate of the population at risk (e.g., the general population older >50 years for GCA) was used as an offset. The results are presented as annual incidences and 95% confidence intervals (CIs). We restricted the analysis to age >50 years for GCA and <40 years for Takayasu. To avoid underestimating the incidence due to age restriction, we conducted a sensitivity analysis for the GCA and Takayasu, including all cases without age restriction. To analyze associations of the pandemic with incidences of the types of vasculitis, we incorporated into the regression an indicator of the COVID-19 period (2020–2021) and assessed each vasculitis diagnosis separately compared to the pre-COVID-19 period (2018–2019). For the COVID-19 period, we calculated the RR (and 95% CI) for being diagnosed with a new onset of each vasculitis condition compared to the non-COVID-19 period. The analysis was performed using SPSS version 28 (IBM Corp., Armonk, NY, USA) and Microsoft Excel. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 13
Results
Our data retrieval identified 5212 individuals with vasculitides: 2445 GCA, 128 Takayasu, 749 AAV, 547 IgA, 237 cryoglobulinemia, and 1106 BD (Table 1). The group with GCA was the oldest at presentation, and the group with BD was the youngest (63.1 ± 7.9 and 34.1 ± 12.4 years, respectively). The Takayasu group showed the highest proportions of females and smokers (71.9% and 50.8%, respectively). The IgA vasculitis group showed the highest rates of diabetes (37.5%), chronic kidney disease (29.8%), coronary artery disease (14.3%), and stroke (21.8%). Almost all the patients received glucocorticoid treatment, and the highest rates of 5-year mortality were for GCA and IgA vasculitis (21.6% and 29.4%, respectively).
General characteristics* of individuals in Israel diagnosed with vasculitides during 2007–2021.

Demographics were extracted from the year of diagnosis, and comorbidities were extracted from the last year of follow-up.
AAV, ANCA-associated vasculitis; DMARDs, disease-modifying antirheumatic drugs; GCA, giant cell arteritis; IQR, interquartile range; SD, standard deviation; SES, socioeconomic status.
Crude and estimated mean incidences, adjusted for age and sex, for each type of vasculitis examined are shown in Table 2 and Figure 1, respectively. For the years 2007 and 2021, the respective adjusted incidences per 100,000 were 7.9 (95% CI 3.5–17.9) and 1.5 (95% CI 0.7–3.6) for GCA, 3.5 (95% CI 1.2–10.0) and 2.7 (95% CI 1.0–7.4) for AAV, and 6.3 (95% CI 3.0–13.5) and 1.0 (95% CI 0.5–2.25) for BD. For the years 2007 and 2021, the respective adjusted incidences per 1 million were 1.5 (95% CI 0.7–3.8) and 2.9 (95% CI 1.5–5.3) for Takayasu, 5.2 (95% CI 2.7–11.1) and 1.5 (95% CI 0.7–3.3) for IgA vasculitis, and 1.5 (95% CI 0.7–3.7) and 1.2 (0.6–3.0) for cryoglobulinemia. The incidences of GCA (RR 0.92, 95% CI 0.91–0.93), IgA vasculitis (RR 0.93 95% CI 0.89–0.98), and BD (RR 0.90 95% CI 0.85–0.94) decreased during 2007–2021. The incidences of the remaining conditions remained unchanged over the study period. Our sensitivity analysis of GCA and Takayasu without age restriction is shown in Supplemental Figure 1A and B. This analysis showed that the incidence and trend of GCA decreased over the study period (p = 0.04), while the incidence of trend of Takayasu remained the same (p = 0.5).
Crude incidences in CHS of types of vasculitis by the year of diagnosis for the years 2007–2021.
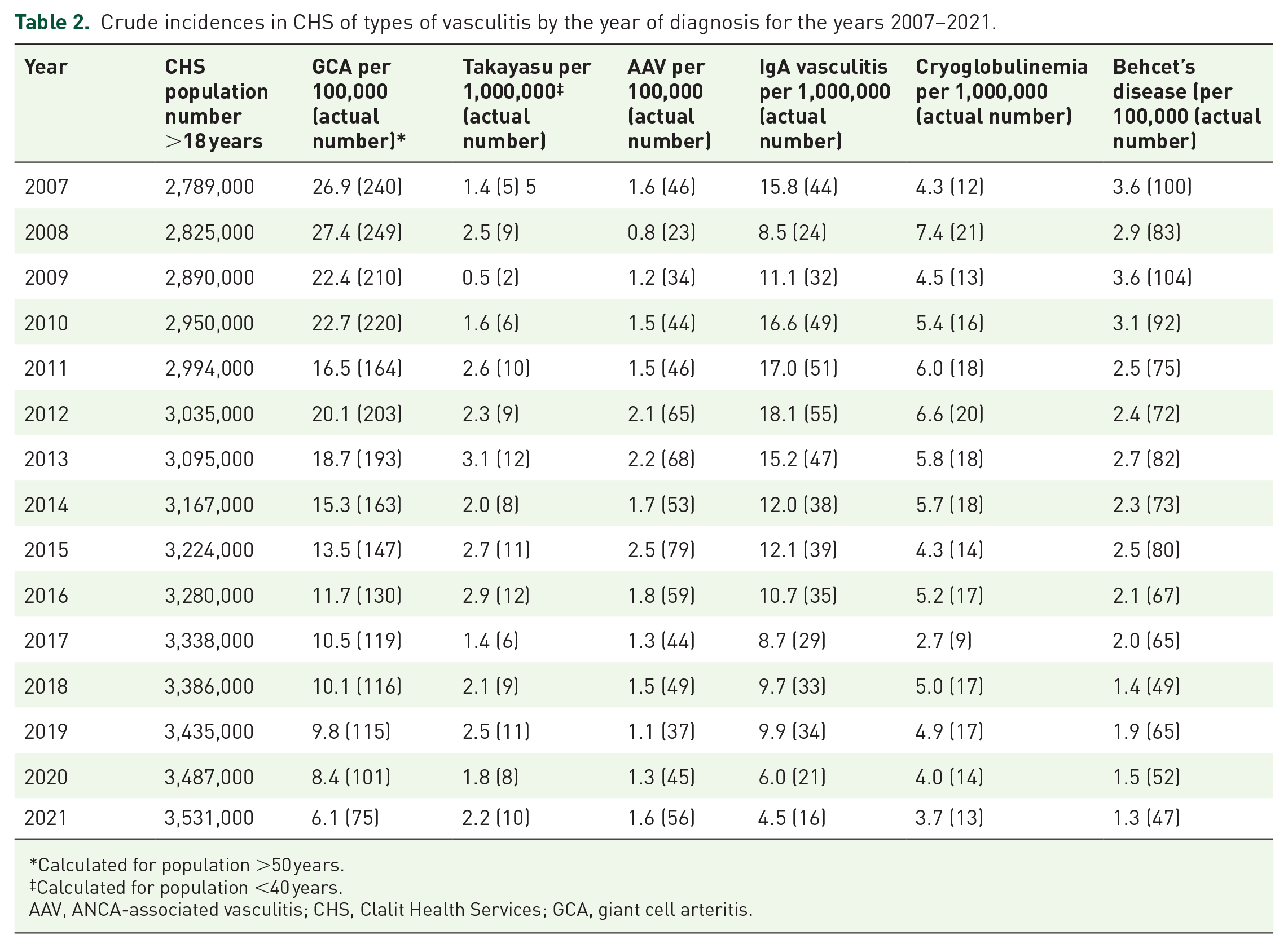
Calculated for population >50 years.
Calculated for population <40 years.
AAV, ANCA-associated vasculitis; CHS, Clalit Health Services; GCA, giant cell arteritis.

The incidences of vasculitis conditions during 2007–2021, per vasculitis type. The dotted line represents the mean trend; closed bars represent the 95% CI. (a) Giant cell arteritis (RR 0.92, 95% CI 0.91–0.93). (b) Takayasu (RR 1.03, 95% CI 0.96–1.13). (c) ANCA-associated vasculitis (RR 0.97, 95% CI 0.90–1.04). (d) IgA vasculitis (RR 0.93 95% CI 0.89–0.98). (e) Cryoglobulinemia (RR 0.97, 95% CI 0.92–1.02). (f) Behcet’s disease (RR 0.90 95% CI 0.85–0.94).
Associations of the COVID-19 period with the new onset of the vasculitides examined are shown in Table 3. After applying multivariable-adjusted analysis, the COVID-19 period was not associated with changes in the incidence of any of the vasculitides.
Associations of the COVID-19 period (2020–2021 vs 2018–2019) with the new onset of vasculitis by type*.

The models were adjusted for sex, age, year, and population size.
CI, confidence interval; COVID-19, coronavirus disease 2019.
Discussion
Our principal finding is that the incidences of GCA, IgA vasculitis, and BD disease decreased in Israel over a recent 15-year period. Our results are based on a large-scale national cohort that remained consistent after controlling for age, sex, and time. Our analysis found no association between the COVID-19 period and the incidence of any of the vasculitides examined.
This study has several strengths. Our cohort is based on large CHS real-life data that were previously validated for other rheumatologic conditions. 14 To increase the validity of our cohort, we included only individuals for whom a vasculitis ICD-9 code was recorded and for whom a relevant treatment was prescribed during the year of diagnosis. Our final data represent a national cohort, in contrast to the single-center study and regional studies that were presented in previous publications. The comprehensive CHS database enabled a relatively long follow-up, to capture comorbidities and medical treatments. Epidemiological studies of vasculitides have been based on various methodologies: cohorts of diagnosed patients in specific centers or regions, and registry-based databases.
Moreover, the entry criteria of those studies showed wide variation, including their case definitions by ICD-9 diagnoses, classification criteria, the receipt of relevant treatments, and positive biopsy or imaging results. Importantly, most of the studies were conducted in Europe and North America, thus underrepresenting other regions in the world. 6 Our findings corroborate the results of a study conducted in Jerusalem in 2016. 15 Nesher et al. reported similar incidences of AAV (11.2 per million) and Takayasu (2.1 per million), but a lower incidence of GCA (8.1 per 100,000). The latter may be attributed to the inclusion of only hospitalized patients.
The decrease in the incidence of GCA observed in our cohort contrasts with the increase in GCA diagnoses reported in the United Kingdom during 2011–2020, regardless of the classification criteria (ACR 1990 vs ACR/EULAR 2022). 16 The authors suggested that the increment could be due to the introduction of the GCA-fast track in their institute and the mounting use of ultrasound. On the other hand, other studies from Scandinavian countries and Canada found that the incidence of GCA had leveled out or even decreased during the period examined, especially when the diagnosis was based on temporal artery biopsy.2,17,18 The decreasing incidence was attributed to the lowering trend of biopsy-based diagnosis (parallel to the mounting use of magnetic resonance imaging and positron emission tomography-computed tomography). As diagnoses in our cohort were based on GCA code and treatment, the rate of temporal artery biopsies was irrelevant to this finding. An enormous ethnic change occurred in Israel during the 1990s, as more than 1 million former residents of the Soviet Union, with a relatively high elderly population, immigrated to Israel. 19 This population’s aging (and consequently dying) may explain our cohort’s decrease in GCA incidence. In addition, the expanding usage of ultrasound, magnetic resonance imaging, and positron emission tomography-computed tomography may increase an imaging-dependent diagnosis of GCA instead of relying only on clinical signs, which may lower the incidence of this condition.
Moreover, the age- and sex-adjusted incidence in our cohort during the last 5 years of our study period was similar to the incidence reported from the Mediterranean region, further supporting this speculation. 20 The claim of a possible relationship between COVID-19 infection or vaccination and GCA is based mostly on anecdotal reports. 21 Only one study evaluated the impact of COVID-19 on GCA nationwide, namely in Spain. Although the authors reported a slight increase in the incidence of GCA during the COVID-19 period, their analysis was based only on GCA-related hospitalizations and did not capture ambulatory cases. 22
IgA vasculitis is less common in adults than in children. The few studies that evaluated the incidence of this condition among persons aged >15 years reported a wide range of annual incidences, from 1.5 per million (Spain) to 50 per 100,000 (Slovenia).23,24 No study evaluated time trends in IgA vasculitis incidence over time. On the other hand, one observational study compared incidences before and during COVID-19 in a national cohort of children and adults with IgA vasculitis from France. The authors reported a decrease in the incidence of IgA vasculitis in children and adults during the pandemic. They speculated that this may have been due to the use of precautionary measures, which reduced the trigger of viral infections during the pandemic. 25 Global pharmacovigilance data from 24 countries showed no increased risk of developing IgA vasculitis after receiving the COVID-19 vaccine. 26
The incidence of BD is highly variable across countries; the highest rate was reported along the ancient Silk Road (e.g., Turkey, Iran, and Jordan). 27 However, the majority of the research reported prevalence rather than incidences. In South Korea, the annual incidence was 8.15 per 100,000 in 2004, which decreased to 1.51 per 100,000 in 2017. 28 The authors suggested that this change may be attributed to environmental factors such as reducing the burden of infectious diseases and improving oral hygiene. As expected, the incidence of BD in European countries was lower. The annual incidence in Sweden was 0.2 per 100,000, which was higher among non-Swedish ancestry and even lower in Poland.29,30 From this perspective, our findings of the annual incidence of 5 to 1 per 100,000, with a gradual decline over 15 years, align with the location of Israel in the Silk Road region and the decreased incidence reported from South Korea. A COVID-19 infection was associated with an increased risk for new-onset BD and a flare, as was reported in four patients following the COVID-19 vaccine.31,32
We acknowledge several limitations in this study. Our analysis is based on data from Israel and may not apply to other regions worldwide. In addition, our data were assembled using ICD-9 codes extracted from an electronic health database. Hence, due to the nature of this study, we could not validate our registry. However, to strengthen the validity of our cohort, we included only patients who had purchased relevant treatment for vasculitis during the first year of diagnosis. For instance, most of our patients received treatment with glucocorticoids in addition to disease-modifying antirheumatic drugs. Thus, we assume that the vast majority of the patients who were included had vasculitis. We acknowledge that relying on medication purchases does not guarantee concomitant use of these therapies. Yet, with relatively high accuracy, multiple large-scale real-world registries have been assembled based on the combination of vasculitis codes and relevant drug utilization.33–35 Lastly, we did not capture environmental covariates (e.g., viral infections and air pollution) that may have confounded the temporal trend of our analysis. We also did not evaluate differences in the incidence of vasculitis conditions in specific ethnic groups (as has been done previously), due to the nature of our database.36,37
Conclusion
In conclusion, our results show that incidences of GCA, IgA vasculitis, and BD in Israel decreased between 2007 and 2021. By contrast, Takayasu, AAV, and cryoglobulinemia incidence remained unchanged throughout this period. Although we did not find associations between COVID-19 infection and the incidence of any types of vasculitides examined, future studies should focus on the effects of other environmental factors on the emergence of vasculitides.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X241274032 – Supplemental material for The incidence of vasculitides in Israel from 2007 to 2021 and during the COVID-19 pandemic
Supplemental material, sj-docx-1-tab-10.1177_1759720X241274032 for The incidence of vasculitides in Israel from 2007 to 2021 and during the COVID-19 pandemic by Lior Zeller, Ran Ben David, Lena Novack, Ran Abuhasira, Mahmoud Abu-Shakra, Yuval Miskin and Iftach Sagy in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X241274032 – Supplemental material for The incidence of vasculitides in Israel from 2007 to 2021 and during the COVID-19 pandemic
Supplemental material, sj-docx-2-tab-10.1177_1759720X241274032 for The incidence of vasculitides in Israel from 2007 to 2021 and during the COVID-19 pandemic by Lior Zeller, Ran Ben David, Lena Novack, Ran Abuhasira, Mahmoud Abu-Shakra, Yuval Miskin and Iftach Sagy in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
