Abstract
In the past decade, unprecedented progress has been made in understanding the pathogenesis, diagnosis, assessment, and treatment of anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitides (AAVs). International collaborations and input from several fields (e.g. immunology, rheumatology, and nephrology) have been critical for analyzing demographics, disease manifestations, and outcomes in clinical research studies. Such efforts opened new avenues for generating novel questions and rationale to design better clinical trials. In addition, clinical research has been a source of several biological discoveries and the starting point for knowledge seeking on the pathophysiology of AAV. Interestingly, the blending of clinical and basic research provides a platform for personalized medicine. Despite recent revisions on AAV classification, the incorporation of new findings on disease genetics and immunologic responses may soon result in changes in clinical practice. These advances will enhance the selection of more specific and targeted therapies. However, current unmet needs in the management of AAV are still sizable and heavily impact long-term survival. Especially, frequent relapses, damage accrual, and high morbidity contribute to poor outcomes. Finally, the lack of defined biomarkers for disease activity and the prognosis is a permanent challenge in AAV research. Our work provides an overview of the current state of the art in AAV literature and suggests bridges for the remaining knowledge gaps. It offers potential future directions for the clinical assessment, management, and research in the field toward a more personalized medicine approach.
Introduction
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitides (AAVs) are a group of small vessel vasculitis disorders characterized by neutrophil-driven inflammation of blood vessels leading to endothelial injury and tissue damage. 1 Microscopic polyangiitis (MPA), granulomatosis with polyangiitis (GPA), and eosinophilic granulomatosis with polyangiitis (EGPA) are the three types of AAV with different definitions according to the Chapel Hill nomenclature (Table 1).2–4 In MPA, kidney involvement is predominant. In contrast, GPA and EGPA are characterized by extravascular granulomatous inflammation, predominantly involving the respiratory tract.1,2 Severe disease occurs with capillaritis, manifesting more commonly as glomerulonephritis (GN) or alveolar hemorrhage. 5 The loss of tolerance to neutrophilic proteins, namely proteinase 3 (PR3) and myeloperoxidase (MPO), plays a central role in AAV pathogenesis.1,2 Immunofluorescence shows minimal deposition of immunoglobulins and complement, hence the so-called ‘pauci-immune’ vasculitis.1,2 AAV are rare diseases with an estimated prevalence of 200–400 cases per million people, and there was an apparent increase in incidence over time due to higher awareness for the diagnosis and higher yield in ANCA testing methodologies.6–9
Definitions, clinical features, and classification criteria of ANCA-associated vasculitis (AAV).
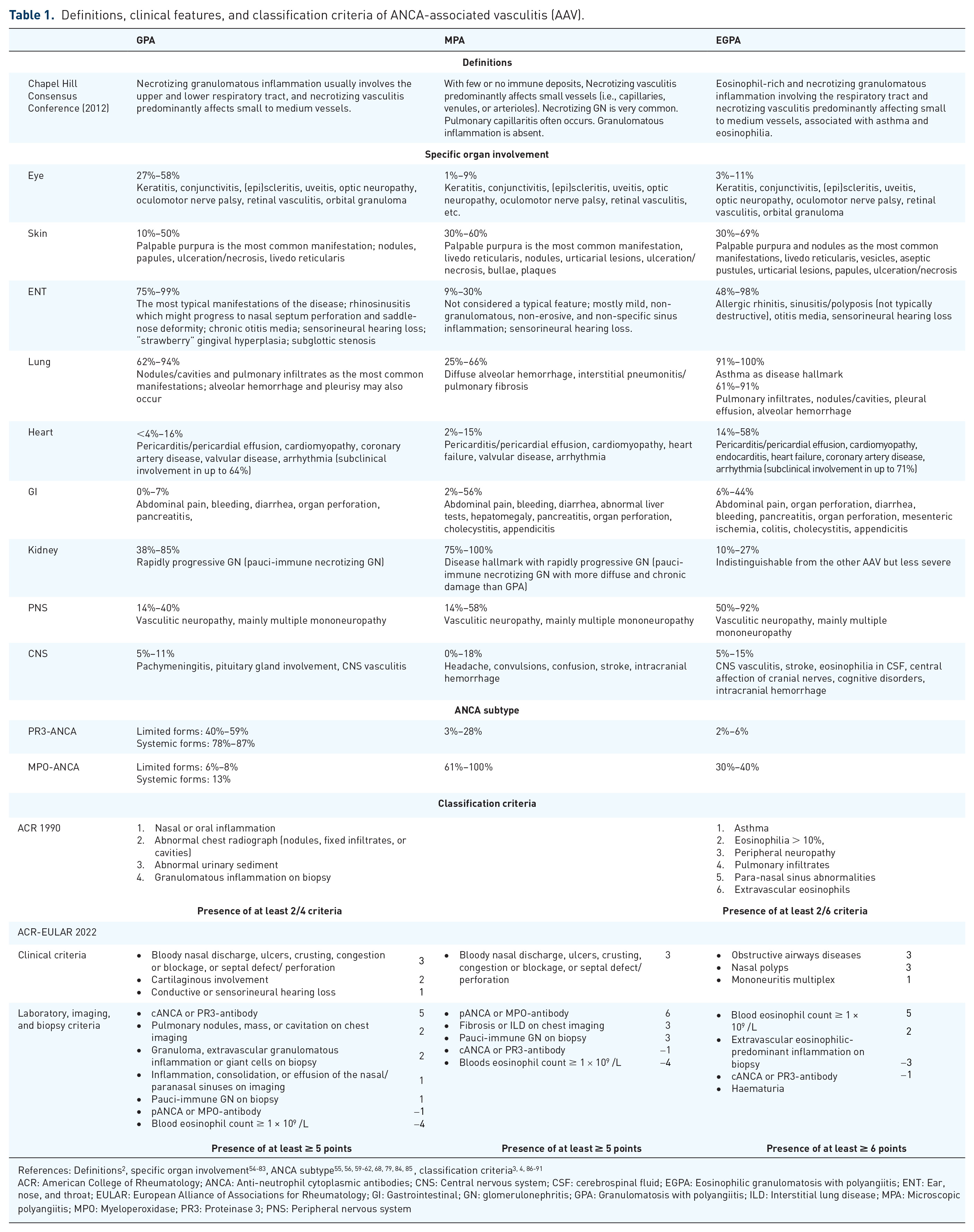
References: Definitions 2 , specific organ involvement54-83, ANCA subtype55, 56, 59-62, 68, 79, 84, 85, classification criteria3, 4, 86-91
ACR: American College of Rheumatology; ANCA: Anti-neutrophil cytoplasmic antibodies; CNS: Central nervous system; CSF: cerebrospinal fluid; EGPA: Eosinophilic granulomatosis with polyangiitis; ENT: Ear, nose, and throat; EULAR: European Alliance of Associations for Rheumatology; GI: Gastrointestinal; GN: glomerulonephritis; GPA: Granulomatosis with polyangiitis; ILD: Interstitial lung disease; MPA: Microscopic polyangiitis; MPO: Myeloperoxidase; PR3: Proteinase 3; PNS: Peripheral nervous system
In the last decade, several research goals in AAV have been attained. AAV genetic background is now better characterized.10–12 Due to efficient immunosuppressive agents, the prognosis for patients with AAV has evolved from a fatal outcome to a potentially treatable disease. 13 After the PEXIVAS and ADVOCATE trials, it became apparent that reducing exposure to glucocorticoids (GCs) is possible and safe.14,15 The benefits of using plasma exchange (PLEX) are currently being questioned. 16 Moreover, remission-maintenance treatment with rituximab (RTX) is now recommended as the primary strategy for relapse prevention.17,18 However, several critical needs remain unmet in the management of AAV. Many patients still progress to kidney failure (KF), associated with increased morbidity and mortality.19–22 The risk of relapse is high, contributing to damage accrual and decreased survival. 23 In addition, especially during remission-induction treatment, the incidence of infectious complications related to immunosuppressants is high. 1 Finally, remission-maintenance strategies are not protocolized or guided by specific biomarkers, which does not help to subside the exposure to unnecessary treatments. 16
Our work shows how pathogenesis, disease classification, and assessment can be combined for a unified approach. Consequently, we expect improvements in phenotypic and biologic disease characterization and in selecting specific treatments to potentially step towards personalized medicine and mitigate some of the knowledge gaps that remain in AAV (Figure 1).
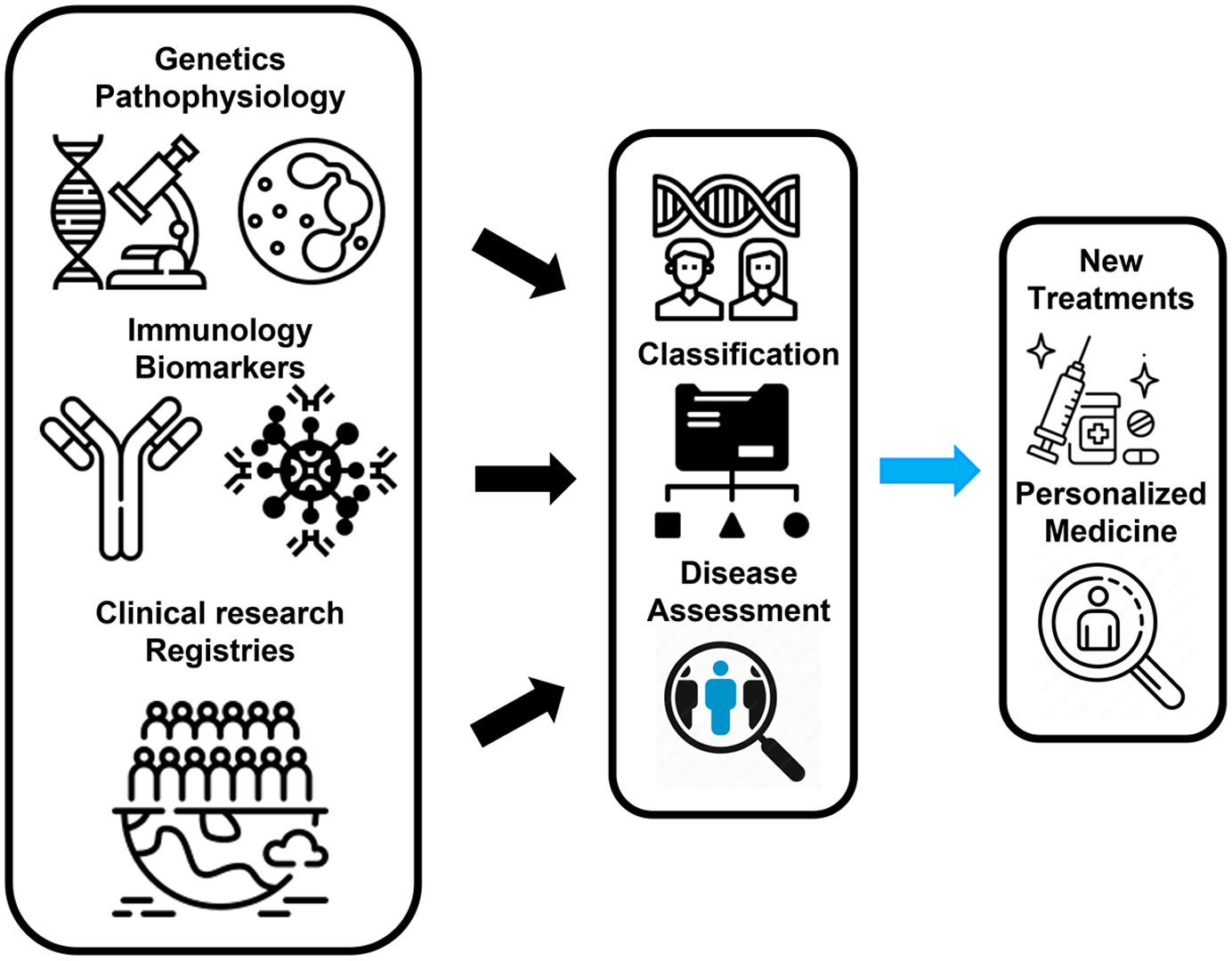
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis framework. Integrating genetic and immunologic background on the phenotypical characterization of patients with ANCA-associated vasculitides (AAVs) will potentially add precision to the selection and development of new treatments. In addition, new classification criteria, adjusted to current practice, will aid correct recruitment of patients into clinical trials and research studies, ultimately improving patients’ management. Finally, the continuous development of AAV registries will allow for more meaningful research based on a multidisciplinary approach and using real-world data. These will potentially converge to personalized medicine in AAV.
Pathogenesis
The pathogenic hallmark of AAV is the loss of immunologic T- and B-cell tolerance to neutrophilic proteins, namely PR3 or MPO. 1 The loss of tolerance is multifactorial and occurs in the presence of risk factors, such as genetic background and age, in combination with environmental factors and, more commonly, in the context of inflammation or infection. 1 MPO and PR3 are released from neutrophils and presented for T-cell recognition resulting in proinflammatory cytokine production and recruitment of effector leukocytes. 1 After the loss of tolerance, the generated ANCA activate the neutrophils in the endothelium microvasculature, leading to local injury and inflammation. 1 Endothelial and tissue injury ensues, facilitating extravascular inflammation, fibrosis, and progressive loss of function. 1
Genetics
Genome-wide association studies (GWASs) have shown that GPA and MPA are genetically distinct. In contrast, EGPA encompasses two genetically different subtypes (MPO-ANCA-positive EGPA and ANCA-negative EGPA). Shared genetic risk factors for vasculitis might underlie the susceptibility to the disease.10–12 Major histocompatibility complex (MHC) genes and non-MHC genes lead to an increased susceptibility to AAV. In PR3-AAV, the presentation of PR3 antigen to the immune system is ruled by the HLA-DPB1*04:01 allele, and increased plasma levels of PR3 result from the balance between α1-antitrypsin (SERPINA1) and PR3 (PRTN3) gene expression, contributing the immune system awareness and reactivity to PR3. 10 MPO-AAV and MPA have been mainly associated with HLA-DQ. 10 In EGPA, the GWAS studies allowed the differentiation of EGPA into two distinct subtypes. The authors identified 11 loci associated with the disease. 11 Furthermore, the general risk for vasculitis seems to be conveyed by polymorphisms in the PTPN22 gene and further modified by epigenetics.12,24
Infection
The loss of tolerance is promoted by infection through the priming of neutrophils for ANCA-induced activation, molecular mimicry, and autoantigen exposure included in neutrophil extracellular traps (NETs).1,25,26 The presence of infectious triggers in AAV pathogenesis has been suggested in observational studies and in particular the contribution of Staphylococcus aureus as a trigger for relapse in GPA or as a source of molecular mimicry in MPO-AAV.27,28
Loss of tolerance
In combination with the appropriate genetic background and as a response to an adequate trigger, the loss of immunologic T- and B-cell tolerance to PR3 or MPO is necessary for the pathogenesis of AAV. 1 This occurs before the onset of AAV symptoms. 1 Loss of T-cell tolerance results in the differentiation of autoantigen-specific T-cells into a T-helper phenotype, including T follicular helper (Tfh), type 1 T helper (Th1), and interleukin 17A (IL-17A)-producing T helper (Th17) cells.1,29–31 Furthermore, CD4 + promotes ANCA production, generates effector memory T-ells, and is present in local organ lesions (with CD8 +). The immune response will be perpetuated, particularly when there is reduced expression of T-cell exhaustion markers, associated with an increased risk of relapse.1,32,33
The emergence of autoreactive B-cells and plasma cells occurs after the loss of B-cell tolerance, and this has been identified in AAV patients. Autoreactive PR3 + and MPO + B-cells represent the source of high-affinity pathogenic autoantibodies in AAV.34–36 The maturation of B-cells occurs in germinal centers of secondary lymphoid tissues or tertiary lymphoid organ structures of inflamed target tissues, which promotes the interaction of the recruited autoreactive B-cells with the Tfh. The Tfh are a specialized subset of CD4 + T-cells with a critical role in the germinal center formation, contributing to high-affinity maturation, clonal selection, and class switch of B-cells leading to expansion, differentiation, and positive selection of the memory subsets.37–39 Among B-cells, a small fraction is defined as autoreactive B-cells (i.e. cells bearing a B-cell receptor that recognizes autoantigens). 34 In addition, detection of in situ PR3+ B-cells in inflamed tissues has been pursued based on the hypothesis that granuloma formation in AAV might represent ectopic lymphoid structures, potentially leading to autoantibody production.40,41 Clinical studies have been highlighting the role of CD19+ B-cells as an immunologic biomarker for disease monitoring since B-cell reappearance after immunosuppressive treatment precludes the occurrence of relapses in most patients with AAV.42,43
ANCA
The clinically relevant neutrophil antigens that ANCA targets are PR3 and MPO, and only rarely different targets such as bactericidal permeability–increasing protein (BPI), elastase, lysosome-associated membrane protein-2 (LAMP-2), cathepsin G, lysozyme, and lactoferrin are documented.44,45 Clinical in vitro and in vivo studies have supported the hypothesis of the pathogenic role for ANCA in the development of AAV. 46 This evidence is more robust for MPO-ANCA than for PR3-ANCA, which has shown the same proinflammatory effects in in vitro experimentation. 47
Neutrophil priming, activation by ANCA, and promotion of antigen recognition
The so-called effector phase in AAV is best documented by the presence of capillaritis (such as GN). The effector phase happens in two steps. 47 First, neutrophils are primed by low-level exposure to proinflammatory cytokines, such as tumor necrosis factor (TNF) and IL-1, by pathogen-associated molecular pattern (PAMP) and damage-associated molecular pattern (DAMP) engagement with toll-like receptors, (TLRs) and by the binding of C5a to the C5a receptor on neutrophils. 1 Priming of neutrophils results in surface expression of MPO, PR3, and other substances of neutrophil granules (e.g. lactoferrin, gelatinase, and elastase) and promotes neutrophil adhesion to the endothelial surface of blood vessels. 1 In the second step, neutrophils are activated by interactions with ANCA by binding to neutrophils Fc receptors and/or to antibody substrate, leading to the rapid release of reactive oxygen species (respiratory burst), preformed proteases, mediators of inflammation, and chemotactic factors for neutrophils and other cells of the immune system, ultimately leading to inflammation, endothelial injury, and tissue damage.47,48 ANCA-stimulated neutrophils can also release chromatin fiber leading to NETs formation, in which autoantigens MPO and PR3 can be stored, further representing a persistent source of autoantigens contributing to inflammation maintenance. 49
Complement
ANCA has been shown to have an in vitro cytolytic effect on endothelial cells through the activation of neutrophils and the interaction with the complement system’s alternative pathway (especially with C5a and the C5a receptor). 50 The importance of complement was shown in mouse models and renal histologic samples.51,52 C5-deficient mice failed to develop GN and vasculitis. Similarly, C5-deficient and factor B-deficient mice were completely protected from the disease. In contrast, the wild-type and C4-deficient mice were not protected from developing disease, reinforcing the role of complement activation via the alternative pathway in the pathogenesis of AAV. Furthermore, mice lacking the C5a receptor (C5aR, also known as CD88) were protected from AAV-GN development. The description of immunologic phenotypes and genetic background, might help to a more precise classification of patients, better correlated with outcomes, contributing to a more accurate prognostic assessment. This will potentially be helpful for the selection of therapeutic targets in AAV.
Diagnosis and classification
There are no AAV diagnostic criteria published so far. Hence, current diagnosis is mostly based on clinical features and supported, whenever possible, by the presence of ANCA and typical histological findings. Patients may present with various symptoms, from constitutional features (e.g. malaise, fatigue, weight loss, fever, arthralgia, and myalgia) to specific organ-related manifestations. 53 Although there are many overlapping features between the three subtypes of AAV, differences in organ manifestations and ANCA specificity may offer excellent clues to help in their distinction (Table 1).
Diagnosis and clinicopathologic entities
GPA is more often associated with upper and lower respiratory tract involvement, particularly with ear, nose, and throat (ENT) manifestations such as nasal crusting, epistaxis, sinusitis, otitis, and hearing loss. 69 Continued nasal inflammation may lead to severe damage with perforation of the nasal septum and saddle nose deformity, a very specific feature of GPA. 92 Moreover, eye involvement may commonly occur, in some cases with the presence of orbital pseudotumor, another clinical feature extremely suggestive of GPA. 54 While some patients may only present with localized forms of the disease (i.e., mainly limited to the upper respiratory tract with no life-threatening manifestations), others may progress to a more systemic clinical picture, usually involving the lungs and/or kidneys.55,84 Serologic testing showing positive ANCA with a cytoplasmic pattern (C-ANCA) directed against PR3 is strongly associated with GPA.
In MPA, renal involvement is a significant feature of the disease. 56 Some patients may also present with lung involvement, mainly diffuse alveolar hemorrhage (DAH), and pulmonary fibrosis has been increasingly recognized in MPA, sometimes as the initial manifestation of the disease.93,94 ANCA positivity with a perinuclear pattern (P-ANCA) directed against MPO is typically seen in patients with MPA, although PR3-ANCA may also be present. Of note, it is essential to highlight that either MPA or GPA may present with positive PR3-ANCA or positive MPO-ANCA, making the differential diagnosis between both diseases challenging and, in some cases, impossible to achieve. By contrast, EGPA is classically characterized by late-onset asthma, nasal polyposis, and eosinophilia of the peripheral blood and/or tissue. 60 Cardiac involvement is a recognized hallmark of EGPA, with endomyocarditis representing its most severe form of manifestation. 95 Renal disease is rarely seen, and ANCA is only detected in up to 40% of patients, usually MPO-ANCA.62,96,97
In some patients, the combination of highly suggestive clinical features and the detection of ANCA may be sufficient to make the diagnosis of AAV and begin treatment. Nevertheless, histologic confirmation of AAV should be sought whenever possible. Necrotizing vasculitis, with no or few immune deposits, predominantly affecting small vessels, is the most typical finding in AAV. 2 However, the diagnostic sensitivity of biopsy may be very low and highly dependent on the location it is performed. It is estimated that nasal and sinus biopsies may contribute to the diagnosis of vasculitis in only 28% and 37% of cases, respectively. 98 Moreover, the diagnostic yield of transbronchial biopsies in GPA is below 50%. 99 The combination of superficial peroneal nerve and peroneus brevis muscle biopsies has shown an estimated sensitivity of around 60–75% for vasculitis neuropathy. However, it can lead to a definitive sensory deficit at the procedure site.100,101 Skin biopsies are easy to perform but often show non-specific findings such as leukocytoclastic vasculitis. When renal involvement is suspected, kidney biopsy is advisable to confirm pauci-immune GN and exclude other causes of renal disease (e.g. drug toxicity) and assess prognosis in terms of renal recovery. 102 In 2010, a histologic classification for renal AAV was established by a group of pathologists and nephrologists from the European Vasculitis Society (EUVAS). 103 Patient biopsies were divided into four classes (1) focal (⩾50% normal glomeruli), (2) crescentic (⩾50% glomeruli with cellular crescents), (3) sclerotic (⩾50% sclerotic glomeruli), and (4) mixed (<50% normal, <50% crescentic, and <50% sclerotic glomeruli), with a subsequent validation study confirming the prognostic value of this histopathologic classification for 1- and 5-year renal outcomes. 103 Since then, several other validation studies have been published worldwide.102,104–112 In addition, the renal risk score for ANCA-associated GN was developed based in clinical and histologic characteristics and highlights the contribution of unaffected glomeruli to renal recovery. 113 More recently, a kidney biopsy chronicity grading score – the Mayo Clinic Chronicity Score – has also been developed and validated, additionally showing an impact on prognostic prediction. 114
Classification criteria
Classification criteria define a homogeneous group of patients with a specific disease for correct recruitment into clinical research studies. 115 Classification criteria for GPA and EGPA were established by the American College of Rheumatology (ACR) in 1990 (Table 1) to differentiate these cases from other forms of vasculitides but not from non-vasculitic diseases.3,4 Therefore, these criteria should not be used for diagnostic purposes and have been proven to perform poorly when used in this manner. 116 In addition, the 1990 ACR criteria were established before the routine testing of ANCA and the widespread use of advanced imaging modalities. More importantly, the criteria did not include separate classification criteria for MPA. Between 2011 and 2017, a multinational, observational study was conducted to develop diagnostic criteria and update classification criteria for systemic vasculitis: the Diagnostic and Classification Criteria for Vasculitis (DCVAS) study. 117 It recruited 6991 patients from 136 different sites and 32 countries. The final ACR-EULAR endorsed classification criteria for AAV have now been published (Table 1).86–91
There is an ongoing debate on classifying patients with AAV based on their clinical phenotype versus ANCA specificity (PR3 vs MPO). 118 Patients with positive PR3-ANCA have been shown to have a different response to treatment, relapse rate, and long-term survival when compared to patients with positive MPO-ANCA.13,19,23,119–123 Furthermore, data from GWAS have found genetic distinctions to be more closely aligned with ANCA status than clinicopathologic manifestations of the disease in patients with MPA and GPA.10–12 However, it is still unclear which fraction of patients with EGPA or negative ANCA would fit in this ANCA-based classification. 124 In addition, despite the controversy, no clinical trials to date have used this different classification system as the sole inclusion criteria to recruit patients with AAV. ANCA serology is usually just included as one of the eligibility items. 125
It is possible that in the future, diagnosis and classification of patients with AAV may include genetic findings paired with ANCA specificity. However, at least for the next few years, the new classification criteria are expected to optimize the way we include patients with AAV in clinical trials and research studies, hence improving their management and outcomes.
Disease assessment
AAV is a chronic and relapsing inflammatory disease requiring careful assessment of its activity, damage, and prognosis to ensure the appropriate use of potentially toxic therapies and accurate monitoring of disease progression. Moreover, patients with AAV perceive the burden of their illness differently from clinicians, which should be considered when evaluating these patients.53,126 The regular use of assessment tools allows for a structured approach in clinical practice and is helpful to ensure homogeneous definitions of response to treatment and outcomes in clinical trials.69,127,128
Activity
After establishing the diagnosis of AAV, defining organ involvement is critical to evaluate response to treatment and outcomes (i.e. remission and relapse). The Birmingham Vasculitis Activity Score (BVAS) is endorsed by the Outcome Measures in Rheumatology Clinical Trials (OMERACT) group to assess the level of disease activity, allowing the comparison between different disease states for individual patients or across groups of patients.129–132 The most recent version of this tool (BVAS v3) consists of a list of items that typically occur in patients with active systemic vasculitis and provides an overall measure of disease activity using a score from 0 to 63. 131 Each item is only recorded as present if the clinician judges it to be due to active vasculitis.
Damage
After controlling disease activity, persistent organ lesions might ensue because of disease extension, cumulative relapses, treatment adverse effects, and comorbidities. 127 The resultant damage can be quantified using the Vasculitis Damage Index (VDI), assessed on a scale of 0 to 64 items, which can only be applied 3 months after the diagnosis of vasculitis has been established.127,133 When monitoring patients with AAV, it is essential to distinguish clinical manifestations resulting from damage or activity to avoid unnecessary immunosuppressive treatment. 127 Moreover, estimating damage helps to record the presence of comorbidities. 127
Function, quality of life, and patient-reported outcomes
Independent of disease activity and damage, the performance of daily life activities can be impaired in AAV. The assessment of function encompasses the overall impact of the disease on physical, social, and psychologic functions, including quality of life and employment. 127 Patients with AAV show a reduction in quality of life like that found in other chronic diseases. Therefore, these patients are suitable to be evaluated by generic instruments like Short Form 36 (SF-36). 134 Recently, a disease-specific measurement tool for patients with AAV became available to fully capture patient-related outcomes (AAV-PRO).135,136 However, it still requires further validation using real world cohorts, which is expected to occur in the next few years.
Prognosis
A cumulative VDI score ⩾5 has been associated with increased mortality risk after 2 years. Higher levels of BVAS at disease presentation have been linked with worst prognostic outcomes.13,133,137 Nevertheless, in patients with AAV, the prognosis is most frequently assessed using the 5-Factor Score (FFS).138,139 This prognostic tool was first validated in 1996 for patients with EGPA, polyarteritis nodosa (PAN), and MPA, and revised in 2009 to also include patients with GPA.138,139 The original FFS encompasses five positive baseline items: creatinine >1.58 mg/dL, proteinuria > 1 g/24 h, and central nervous system, gastrointestinal, and cardiac involvement. 138 The revised FFS includes one negative item (ear, nose, and throat involvement) in addition to the following four positive items: age > 65 years, creatinine ⩾150 µmol/L, and central nervous system and cardiac involvement. 139 Mortality rate at 5 years was estimated at 12%, 26%, or 46% for the original FFS, and 9%, 21%, and 40% for the revised FFS if the patient punctuates 0, 1, or 2, respectively.138,139
Laboratory testing and imaging techniques
Conventional inflammatory markers – C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) – are of limited value for assessing disease activity in AAV due to the lack of specificity. It has been challenging to determine the utility of ANCA serial testing for relapse prediction. In the RAVE trial, patients with PR3-ANCA, relapsing disease, or who had the diagnosis of GPA were at a higher risk of relapse when compared to patients with MPO-ANCA, new disease onset, or who were diagnosed with MPA.42,123,140 The most recent guidelines on AAV do not recommend monitoring ANCA titres. 16 This was mainly based on the conclusions from a meta-analysis that included studies in which the relapse risk was assessed in cohorts that combined patients with MPO- and PR3-AAV.16,141 Previous studies have shown that rise in ANCA titres correlated better with relapses in patients with AAV-GN than in patients without renal disease.142,143 Therefore, the potential use of ANCA as a biomarker for guiding treatment strategies of remission-maintenance and relapse monitoring may depend on organ involvement and ANCA specificity. Finally, routine use of imaging techniques has shown to be of small value for monitoring patients with AAV. 128
Composite measures
The development of composite assessment tools has been desirable to evaluate response to treatment in AAV, particularly in the setting of clinical trials.144,145 The evaluation of patient-related outcomes (PROs) and damage measures in AAV is lacking in many clinical trials. 144 Therefore, the development of a composite assessment tool for vasculitis has recently become a focus of the OMERACT Vasculitis Working Group. 145 The objective is to generate an instrument capable of capturing the entire burden of the disease across multiple domains, paired with the ability to detect response to treatment during different disease states. 145 A systematic review, a Delphi exercise, and a planned methodology were performed and discussed, and domains and high-quality instruments were defined. 145 The next step is to determine a scoring system that allows outcome measurement. 145 It is possible that in future clinical trials, patients’ perspectives will be better captured on the measures of efficacy used.
Treatment
Following diagnosis or reasonable suspicion of AAV, treatment should be initiated as soon as possible. 146 In order to tailor therapy for each patient, a careful evaluation should be carried out to assess the individual risk of infection (including concurrent viral and bacterial infections), immunodeficiency, and comorbidities associated with stratification of the impact of GC treatment (e.g. diabetes, osteopenia/osteoporosis, psychiatric diseases). The treatment of AAV consists of two phases: remission-induction and remission-maintenance. Inducing long-term remission is one of the main goals of immunosuppressive therapy, which might be disrupted by the clinical reappearance of disease activity (relapse). 144 For this reason, after controlling disease manifestations, remission-maintenance treatment is instituted. The recommendations for each phase of treatment are currently stratified according to the severity of the disease and the AAV subtype (GPA/MPA vs EGPA). Severe disease can be defined as vasculitis with life- or organ-threatening manifestations and non-severe disease as vasculitis without life- or organ-threatening manifestations.16,140 In addition, assessment of disease severity may also be aided by the BVAS and FFS (e.g. FFS ⩾ 1 and FFS = 0 for severe and non-severe disease, respectively).129,130,147
Remission-induction treatment
The main goal of the remission-induction treatment is to suppress and control inflammation as fast and steadily as possible to minimize organ damage. During the last two decades, research in AAV management has taken significant steps toward reducing immunosuppression toxicity, with the decreasing of cyclophosphamide (CYC) cumulative doses (CYCLOPS, CORTAGE), progressive reduction of GCs (PEXIVAS, LoVAS) and introduction of new immunosuppressants like RTX (RAVE, RITUXVAS).14,140,148–152
GPA and MPA
In severe AAV, current standard therapy includes a combination of GCs and RTX or CYC, as proposed by the most recent guidelines (Figure 2).16,146,153 Following the RAVE and RITUXVAS trials, the use of RTX for remission-induction treatment has been favored over CYC, particularly in cases of relapsing or PR3-ANCA positive disease, to preserve fertility, in frail older adults, in children and adolescents, or when GC-sparing is strongly needed.16,140,146,152,153 When CYC is the treatment of choice, intravenous (IV) pulses are favored to reduce cumulative doses. 16 In patients with markedly reduced or rapidly declining estimated glomerular filtration rate (eGFR) (serum creatinine [SCr] > 4 mg/dL [354 µmol/L]) and/or KF (eGFR < 15.0 mL/min per 1.73 m) 2 , some debate remains regarding the choice of RTX versus CYC for remission-induction.16,146,153 The efficacy of RTX in patients with KF has not been directly tested in clinical trials. However, some studies have reported that both RTX and CYC therapies seem to be equivalent in severe kidney disease.154,155 A post hoc analysis of the RAVE trial showed that patients enrolled with an eGFR < 30.0 mL/min per 1.73 m2 at baseline responded similarly to RTX (18 patients) and CYC (14 patients). 156 There was no statistically significant difference in the mean eGFR increase over the 18 months of follow-up. 156 In addition, in a cohort of 251 patients with an eGFR < 30.0 mL/min per 1.73 m2, a propensity score matching analysis that adjusted for the severity of kidney disease (eGFR < 15.0 mL/min per 1.73 m) 2 showed no differences between the frequency of remission and renal events between patients who received RTX versus CYC for remission-induction (64 vs 161 patients, respectively). 154 In another study that included 37 patients with eGFR < 20.0 mL/min per 1.73 m2, there were no differences in remission, renal recovery from KF, or death when patients were treated with RTX and GC, with or without CYC (25 vs 12 patients, respectively). 155 Similarly, it is unclear whether the combination of RTX with CYC would benefit the treatment of these patients.
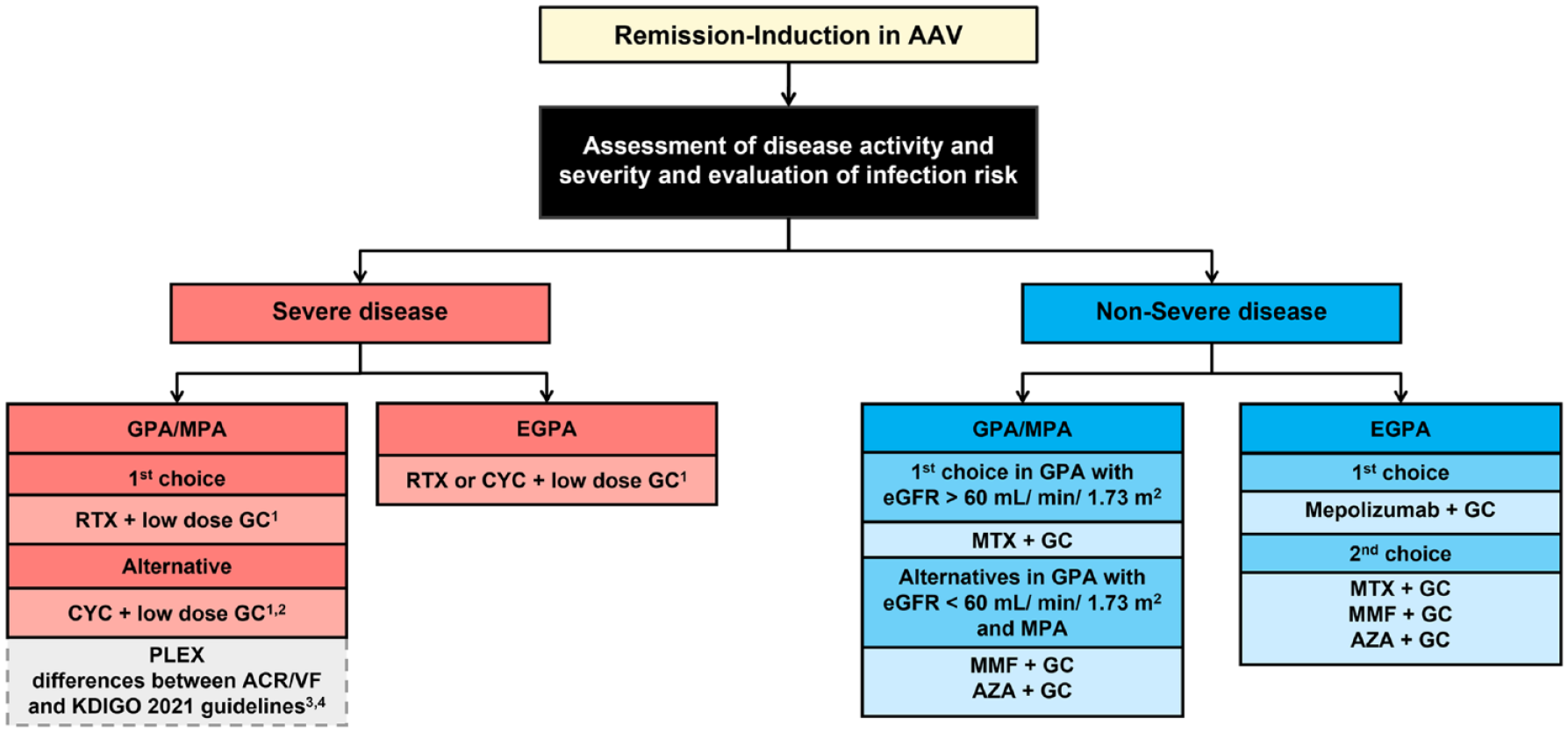
Algorithm for remission-induction treatment in ANCA-associated vasculitis (AAV).
In the last decade, several combined schemes of CYC and RTX have emerged to reduce both CYC and GC cumulative doses.152,157–160 Kavita Gulati et al. 158 recently reported a reduction in KF at 36 months from 67% to 52% with a low-dose CYC and GC regimen with two doses of RTX and PLEX. In addition infection rates were similar. 158 The SMARTVAS (Rituximab/Cyclophosphamide and Minimal Dose Glucocorticoid in AAV) study, in which the induction regimen consisted of two doses of RTX, 3 months of low-dose CYC, and a short course of GCs, provided an important breakthrough in achieving a substantial reduction of the CYC and GC doses (median of 3–3.2 g and median of 1–1.2 g after 1–2 weeks, respectively). It reported similar outcomes to other EUVAS trials, although with fewer GC-related adverse events, namely severe infections, and diabetes. 159 Previously, similar results were found in the CYCLowVasc study that included patients with more severe kidney disease. 157 Results from randomized clinical trials to further support these combined options are awaited. 161 Consequently, reflecting the controversy regarding the treatment of patients with severe kidney disease, the main guidelines have different positions on this subject: the Kidney Disease Improving Global Outcomes (KDIGO) 2021 guidelines for the management of glomerular diseases favor the use of CYC alone or combined with RTX, whereas in the 2021 ACR/Vasculitis Foundation (VF) guidelines for the management of AAV, the use of CYC as the preferred induction treatment for these patients is considered controversial, and the combination of both induction agents is not endorsed due to the currently limited data.16,153 New EULAR recommendations, which are expected to be published by the end of 2022, will incorporate other studies and perhaps expand on current guidelines.
GCs have an essential role in the AAV remission-induction therapy. Methylprednisolone IV pulses of 500–1000 mg per day or high doses of oral prednisone (1 mg/kg/day) are used as an adjuvant to rapidly reduce inflammation until the biologic effect of the immunosuppressive agent occurs.16,146,153 The need for methylprednisolone IV pulses to control disease activity, instead of only high doses of oral prednisone, has been recently questioned following a retrospective study which reported that its avoidance significantly reduced diabetes and severe infections without compromising efficacy. 162 The PEXIVAS trial provided strong evidence for a new reduced prednisone tapering regimen which proved to be efficient and equivalent in the achievement of remission and is currently recommended for remission-induction. 14
Advances in AAV pathogenesis research have shed light on the crucial role of the alternative complement pathway and allowed the development of new drugs that can potentially replace GCs. 50 In the ADVOCATE trial, the anti-C5a receptor (avacopan, 30 mg twice daily) proved to be noninferior at 26 weeks and superior at 52 weeks compared to GCs, with improved kidney outcomes. 15 This was reflected on the increase of eGFR and decrease in albuminuria, consistent with what has been previously reported in phase 2 trials.163–165 In addition, there was a decrease in relapses within the first 52 weeks from 21% to 10% when compared with a prednisone tapering regimen, and quality of life patient-reported outcomes significantly improved. 15 However, there are still uncertainties about the best duration of treatment and its efficacy in patients under kidney replacement therapy and/or eGFR below 15 mL/min. 15 Two recent case series using avacopan have confirmed its steroid-sparing effect and safety profile, including in one patient with an eGFR of 11 mL/min who was able to recover stable kidney function (eGFR 23 mL/min) at 12 months.166,167 Nevertheless, further studies documenting the results of avacopan in patients with a long-term follow-up in real-life settings are still needed.
The use of PLEX in DAH or rapidly progressive crescentic GN decreased considerably following the PEXIVAS study, which did not show a benefit in mortality or the incidence of KF compared to standard remission-induction treatment. 14 Strong evidence favoring PLEX in DAH is lacking, with different studies showing no difference in mortality, even in patients with hypoxemia. 168 A cohort study used propensity score matching analysis that adjusted for the severity of kidney disease (eGFR < 15.0 mL/min per 1.73 m2) and showed no differences between the frequency and risk for renal events between patients who received PLEX vs those who did not (51 vs 200 patients, respectively). 154 However, in patients with severe kidney disease (SCr > 5.7 mg/dL) studies have reported conflicting results regarding the role of PLEX and international community is divided.14,169–172 These controversies are reflected in the most recent guidelines, with KDIGO considering PLEX in patients with SCr > 5.7 mg/dL, rapidly increasing SCr or who require dialysis, and in patients with DAH who have hypoxemia, while ACR/VF recommend against its routine use, but may consider PLEX in critically ill patients at high risk for progression to KF.16,153
IV immunoglobulin may also be a therapeutic option as adjunctive therapy for patients with refractory or relapsing AAV. It may induce rapid improvement in disease activity and related biomarkers.16,173,174
In non-severe disease, methotrexate (MTX) and mycophenolate mofetil (MMF) are considered the alternative options to CYC, based on the NORAM and MYCYC trials, respectively (Figure 2).146,175–177 Azathioprine (AZA) may present as an alternative for remission-induction in cases of pregnancy or intolerance to MTX or MMF. 17
EGPA
Patients with EGPA were not included in the main AAV clinical trials since the disease pathophysiology is substantially different from GPA and MPA, with higher rates of ANCA negativity and eosinophilic-driven inflammation. Hence, recommendations for severe EGPA disease are like those employed in GPA/MPA (Figure 2). 16 For non-severe EGPA, mepolizumab is recommended as the first choice of treatment for remission-induction in new-onset, relapsing and refractory disease. Mepolizumab improved remission rates and reduced the risk of relapse.16,178
Ongoing clinical trials of remission-induction in AAV
Table 2 summarizes the most relevant clinical trials, already terminated or currently ongoing, for remission-induction in AAV.
Clinical trials of remission-induction treatment in ANCA-associated vasculitis (AAV).
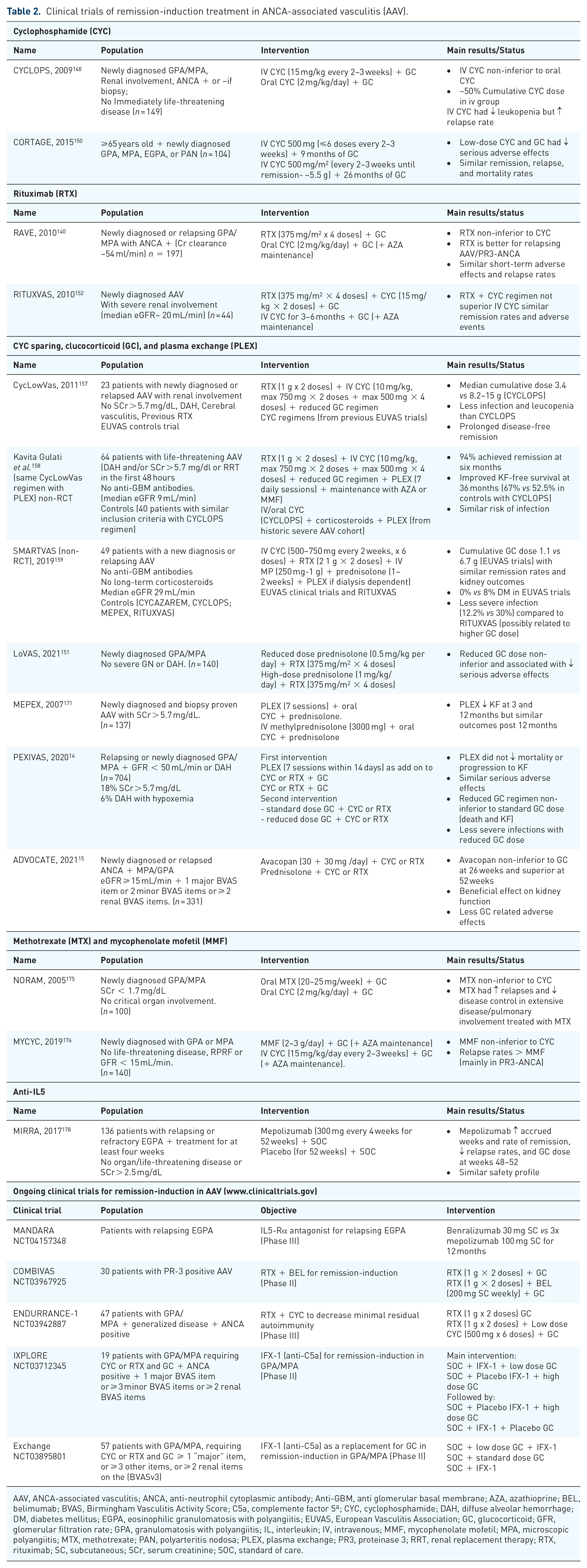
AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; Anti-GBM, anti glomerular basal membrane; AZA, azathioprine; BEL, belimumab; BVAS, Birmingham Vasculitis Activity Score; C5a, complemente factor 5ª; CYC, cyclophosphamide; DAH, diffuse alveolar hemorrhage; DM, diabetes mellitus; EGPA, eosinophilic granulomatosis with polyangiitis; EUVAS, European Vasculitis Association; GC, glucocorticoid; GFR, glomerular filtration rate; GPA, granulomatosis with polyangiitis; IL, interleukin; IV, intravenous; MMF, mycophenolate mofetil; MPA, microscopic polyangiitis; MTX, methotrexate; PAN, polyarteritis nodosa; PLEX, plasma exchange; PR3, proteinase 3; RRT, renal replacement therapy; RTX, rituximab; SC, subcutaneous; SCr, serum creatinine; SOC, standard of care.
In patients with positive PR3-ANCA (GPA or MPA), treatment with belimumab plus RTX (COMBIVAS) is being compared with RTX alone with regard to improvement in biologic endpoints, functional outcomes, and clinical status (ClinicalTrials.gov: NCT03967925). The combination of RTX with CYC is also being compared to RTX alone in the ENDURRANCE-1 study to achieve a favorable immunologic state of minimal residual autoimmunity and reduce the need for retreatment (ClinicalTrials.gov Identifier: NCT03942887). In addition, the IXPLORE (ClinicalTrials.gov Identifier: NCT03712345) and the Ixchange phase II trials (ClinicalTrials.gov Identifier: NCT03895801) focus on GC replacement by a monoclonal antibody specifically binding to C5a (IFX-1-vilobelimab) in patients with GPA and MPA. Moreover, there is an ongoing pilot study assessing the efficacy and safety of tofacitinib in active AAV (ClinicalTrials.gov: NCT04973033). In patients with EGPA, benralizumab, an anti-IL-5 monoclonal antibody, is currently being compared to mepolizumab in a phase III trial in efficacy and safety (ClinicalTrials.gov: NCT04157348).
Remission-maintenance treatment
The choice of remission-maintenance treatment is challenging and attempts to define strict criteria or biomarkers for its guidance have been insufficient. The main objective of the remission-induction phase is to prevent relapse while avoiding long-term drug toxicities and resultant comorbidities. 1 Several factors should be considered while choosing the best regimen: the remission-induction regimen used, disease severity, patient comorbidities, drug contraindications, and potential toxicities.16,146,153
GPA and MPA
The most recent guidelines on the treatment of AAV recommend remission-maintenance regimens according to disease severity (Figure 3).

Algorithm for remission-maintenance treatment in ANCA-associated vasculitis (AAV).AAV, ANCA-associated vasculitis; ANCA, anti-neutrophil cytoplasmic antibody; AZA, azathioprine; CYC, cyclophosphamide; eGFR, estimated glomerular filtration rate; EGPA, eosinophilic granulomatosis with polyangiitis; GC, glucocorticoid; GPA, granulomatosis with polyangiitis; LFN, leflunomide; MMF, mycophenolate mofetil; MPA, microscopic polyangiitis; MTX, methotrexate; RTX, rituximab.1The GC tapering is the standard of care at this point since avacopan was only recently approved as GC sparing strategy.
In patients with severe GPA/MPA, RTX has the best evidence for the maintenance of remission when compared with AZA.17,18 On the MAINRITSAN trial, the efficacy of RTX (500 mg on days 0 and 14, and months 6, 12, and 18) for remission-maintenance was compared to AZA (tapering regimen of 2 mg/kg/d for 12 months; 1.5 mg/kg for 6 months; 1 mg/kg for 4 months) in patients who previously received CYC for remission-induction. 17 The authors showed lower relapse rates at 28-months in the RTX arm. 17 Subsequently, the RITAZAREM trial showed that RTX was efficient in reinducing and maintaining remission in patients with relapsing GPA/MPA. 18 RTX for remission-maintenance has been used either in fixed intervals or guided by B-cell reconstitution (CD19 + monitoring) and/or ANCA reappearance. The MAINRITSAN2 trial showed no differences between the two strategies. 179 However, the most recent ACR/VF guidelines endorsed the fixed regimen. 17 The possible maintenance doses for RTX reported include: (1) IV 500 mg every 6 months (FDA-approved), (2) IV 1000 mg every 4 months, and (3) IV 1000 mg every 6 months.17,18 Comparative studies are still warranted. 17 MTX or AZA are viable alternatives for cases in which RTX cannot be used (e.g. due to limited access, intolerance), with both drugs exhibiting a similar efficacy and safety profile. 180 Other immunosuppressants like MMF and leflunomide may also be considered.16,146,181,182 In patients that remain dialysis-dependent following remission-induction, there is still some debate regarding the institution of remission-maintenance therapy. Recent KDIGO guidelines recommend avoiding further immunosuppression, especially in patients with MPO-ANCA vasculitis. The relapse rate is low and risk of infection related to therapy is high. The ongoing Maintaining or Stopping Immunosuppressive Therapy in Patients With ANCA Vasculitis and End-stage Renal Disease (MASTER-ANCA) will provide future guidance. In non-severe GPA/MPA, it is recommended to continue the same treatment used for remission-induction (Figure 3).16,146
EGPA
Evidence regarding remission-maintenance therapy for EGPA is not as strong as for GPA/MPA.16,146 In patients with severe disease manifestations who have attained remission after induction with CYC, maintenance treatment with MTX, AZA, or MMF is preferred over RTX or mepolizumab (Figure 2). 16 By contrast, the use of mepolizumab over MTX, AZA, and MMF is indicated for remission-maintenance in cases of non-severe disease (Figure 3). 16 In the last couple of years, RTX has frequently been used in EGPA as a GC-sparing agent. It is currently the treatment of choice for remission-induction and maintenance in cases of relapsing disease with severe manifestations.16,146,183,184 In patients who present with non-severe relapses while receiving MTX, AZA, or MMF, adding mepolizumab should be considered. 16
Duration of remission-maintenance treatment
The optimal time of the remission-maintenance treatment is still a matter of debate. The general recommendations are for at least 18–24 months of treatment.16,146 Results of the MAINRITSAN 3 study showed that in patients who underwent RTX maintenance treatment and were in sustained remission for 2 years, two additional years of treatment with RTX decreased the relapse rate. 185
Ongoing clinical trials of remission-maintenance in AAV
Table 3 summarizes the most relevant clinical trials on remission-maintenance for AAV.
Clinical trials of remission-maintenance treatment in ANCA-associated vasculitis.

AAV, ANCA-associated vasculitis; ABA, abatacept; ANCA, anti-neutrophil cytoplasmic antibody; AZA, azathioprine; BEL, belimumab; BVAS, Birmingham Vasculitis Activity Score; CYC, cyclophosphamide; EGPA, eosinophilic granulomatosis with polyangiitis; GC, glucocorticoid; GPA, granulomatosis with polyangiitis; HCQ, hydroxychloroquine; IV, intravenous; KF, kidney failure; LEF, leflunomide; LFN, leflunomide; MMF, mycophenolate mofetil; MPA, microscopic polyangiitis; MTX, methotrexate; RTX, rituximab; SC, subcutaneous; SCr, serum creatinine; SOC, standard of care; TOF, tofacitinib; UIP, usual interstitial pneumonia.
The TAPIR (ClinicalTrials.gov: NCT01933724) and the MAINEPSAN trials (ClinicalTrials.gov: NCT03290456) focus on assessing the best duration of GC treatment to maintain remission in patients with GPA and with GPA or MPA, respectively. The HAVEN trial (ClinicalTrials.gov: NCT04316494) will provide information regarding the potential role of hydroxychloroquine in reducing disease activity in AAV. The STATVAS study (ClinicalTrials.gov Identifier: NCT02117453) will assess whether rosuvastatin can reduce AAV subclinical atherosclerosis. The ABROGATE trial is currently recruiting patients with relapsing, non-severe GPA (ClinicalTrials.gov: NCT02108860) to evaluate the efficacy of abatacept (CTL4-Ig) in achieving GC-free remission. The role of tofacitinib (ClinicalTrials.gov: NCT04944524) is also being assessed in patients with non-severe GPA (new-onset or relapsing patients) in comparison to MTX as a remission-maintenance treatment. In addition, treatment of specific organ involvement is now being studied in clinical trials due to the impact on morbidity and survival in AAV. Pirfenidone, an oral antifibrotic agent, used to reduce the progression of idiopathic pulmonary fibrosis,191–193 has been evaluated to treat pulmonary fibrosis in a pilot study including patients with MPO-ANCA, with or without AAV (ClinicalTrials.gov Identifier: NCT03385668); results are still pending. For patients with newly diagnosed or relapsing EGPA, RTX is under further study as a maintenance agent compared to AZA in the MAINRITSEG trial (ClinicalTrials.gov Identifier: NCT03164473). Other treatment approaches focusing on tailoring treatment to avoid relapse are also being explored. For instance, there is a clinical trial that focuses on determining CD5 + regulatory B cells as patients with low levels have a higher need to keep remission-maintenance treatment to avoid relapse. (ClinicalTrials.gov Identifier: NCT03906227).
Potential new management strategies
The ability to target immunologic pathways will become increasingly possible. Hence the rationale for investigation is expected to be driven by pathophysiology. The analysis of specific populations might help with personalized care. Decreasing immunosuppressant drug toxicity, damage accrual, and morbidity are at the center of drug development.
The treatment to target has been explored using different strategies. The complement inhibition has now been studied as an adjuvant of remission-induction. It has been shown that avacopan has the potential for GC sparing. 15 New drugs targeting the complement system, such as IFX-1, a monoclonal antibody specifically binding to C5a, are under development. Inhibition of MPO has been recently proposed as a valuable target, as shown in a preclinical crescentic GN study where it suppressed kidney damage without augmenting adaptive immune responses. 194 Emerging cell therapies with tolerogenic dendritic cells, regulatory T cells (CAR-T cells), and stem cells (namely human amniotic epithelial cells) will probably emerge due to their selective immunosuppressive capacity. 195 In addition, the use of cytokines for the treatment of autoimmune rheumatic diseases has been studied. 196 It was shown that administration of low dose IL-2 allows expansion of T reg repertoire without effector T cell activation. 196 This could be a potential approach to maintaining immune tolerance, particularly in antibody-mediated diseases. The loss of tolerance to particular antigens is generally the starting point of antibody generation.196,197 When and whether these newer strategies will be translated into improved management of patients with AAV in clinical practice is uncertain. In EGPA, depemokimab, an IL-5 receptor antagonistic monoclonal antibody, is currently being investigated to treat patients with severe eosinophilic asthma with an eosinophilic phenotype (ClinicalTrials. gov: NCT04718103 and NCT04718389).
Although most efforts in AAV research have focused on immunomodulating agents, therapies that address cellular and molecular mechanisms of tissue repair and regeneration, reducing evolution to tissue fibrosis, are also unmet needs in the AAV treatment approach. Blockade of the renin-angiotensin-aldosterone system (RAAS) by angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) has been central in retarding progression of chronic kidney disease (CKD). However, patients still progress to KF, and research studies targeting the critical components of the fibrogenic pathway are currently ongoing, with a particular interest in TGF-β1 BMP-7, CTGF, CC motif chemokines, PDEs, Nox1/4, ET-1 and TNF-α.198,199 The role of antifibrotic drugs in different kidney diseases is being evaluated in several clinical trials. However, their effect on AAV is still unknown.198,199 More recently, sodium-glucose co-transporter 2 inhibitors (SGLT2i), which are reno-protective in diabetic and nondiabetic kidney disease, could be a potential adjunctive therapy in AAV. 200 However, to date, patients with AAV have been excluded from these clinical trials.
Registries
Collaborative networks have been synergic in the study of rare diseases. The clinical heterogeneity of AAV is particularly prone to benefit from registries and data accrued from different real-world practices. Furthermore, incidence, prevalence, and geographic differences regarding risk factors are better characterized using this overview type. Therefore, registries are a vital source of clinical data and can be particularly useful in facilitating research in disease biomarkers, optimizing recruitment for clinical trials, and deepening our understanding of the natural course of AAV. 201 There are currently various vasculitis registries available or in development in different European and North American countries, for example: France [the French Vasculitis Study Group (FVSG)]; UK and Ireland [the UK and Ireland Vasculitis Rare Disease Group (UKIVAS)]; Spain [Registro Español de Vasculitis Sistémicas (REVAS)]; Poland (the Polish Vasculitis [POLVAS] registry); Czech Republic (Czech Registry of AAV); Norway [Norwegian Vasculitis Register & Biobank (NorVas)]; Portugal [Portuguese vasculitis registry [Reuma.(Reuma. ]);)]; Germany, Austria, and Switzerland [Joint Vasculitis Registry in German-speaking countries (GeVas)]; Greece (AAV Patient Registry); USA and Canada [Vasculitis Patient-Powered Research Network (VPPRN)].121,202–215 However, these existing registries exhibit significant differences in terms of content, data collection (retrospective vs prospective), stages of development, and aims, primarily influenced by the medical specialties involved (e.g. nephrology, rheumatology, internal medicine, immunology) and local resources. 201
Ideally, AAV databases should have a common language and terminology, and patients should be recruited by all health care providers involved in management of AAV. Harmonizing data collection is crucial for attaining homogeneous communication and the correct merging of information between registries. Two initiatives are already underway to align vasculitis registries across Europe: 1) ‘Model registry’ initiative to define a long list of items using a REDCap (Research Electronic Data Capture) platform, led by the EUVAS working group for registries; 2) FAIRVASC, a collaboration between the EUVAS and the European Reference Network for Rare Primary Immunodeficiency, Autoinflammatory and Autoimmune diseases (ERN – RITA) which aims to use semantic-web technologies to link vasculitis registries, establishing a common platform and reaching agreement on data governance.201,216
Improvements in registries interoperability and compatible joint exports of data, will facilitate research in AAV with an accrual of a sizable number of patients.
Conclusion
AAV is a group of challenging and complex conditions with overlapping clinical and laboratory manifestation patterns. Our increasing understanding of pathogenesis and early diagnosis has helped us choose a more tailored treatment and reshape the use of potential biomarkers for monitoring these patients. Mortality has significantly improved over the years, and survival rates are now close to 80% after remission-induction. Nevertheless, patients with AAV still have a relapsing course that impacts long-term survival and accrual of morbidity. Integrating genetic and immunologic backgrounds in the phenotypical characterization of patients with AAV will potentially add precision to the selection and development of new treatments. In addition, new classification criteria, adjusted to current practice, will aid correct recruitment of patients into clinical trials and research studies, ultimately improving patients’ management. Finally, the continuous development of AAV registries will allow for more meaningful research based on a multidisciplinary approach and using real-world data. Although 10 years from now, the future of AAV is still uncertain, knowledge in this field is rapidly evolving. Thus, patients are expected to achieve higher rates of sustained remission with more individualized therapies while being exposed to less treatment toxicity.
