Abstract
IgA vasculitis (IgAV), formerly known as Henoch-Schönlein purpura, is the most common cause of systemic vasculitis in childhood. Given its potential life-threatening systemic complications, early and accurate diagnosis as well as management of IgAV represent a major challenge for health care professionals. This study was carried out to attain an evidence-based expert consensus on a treat-to-target management approach for IgAV using Delphi technique. The preliminary scientific committee identified a total of 16 key clinical questions according to the patient, intervention, comparison, and outcomes (PICO) approach. An evidence-based, systematic, literature review was conducted to compile evidence for the IgAV management. The core leadership team identified researchers and clinicians with expertise in IgAV management in Egypt upon which experts were gathered from different governorates and health centers across Egypt. Delphi process was implemented (two rounds) to reach a consensus. An online questionnaire was sent to expert panel (n = 26) who participated in the two rounds. After completing round 2, a total of 20 recommendation items, categorized into two sections were obtained. Agreement with the recommendations (rank 7–9) ranged from 91.7–100%. Consensus was reached (i.e. ⩾75% of respondents strongly agreed or agreed) on the wording of all the 20 clinical standards identified by the scientific committee. Algorithms for the diagnosis and management have been suggested. This was an expert, consensus recommendations for the diagnosis and treatment of IgAV and IgA vasculitic nephritis, based on best available evidence and expert opinion. The guideline presented a strategy of care with a pathway to achieve a state of remission as early as possible.
Plain Language Summary
Given its potential life-threatening systemic complications, early and accurate diagnosis of immunoglobulin A vasculitis represents a major challenge for health care professionals. This work provided cornerstone principles for the management of the condition. Adopting PICO approach and implementing Delphi process a consensus was reached on evidence-based treat-to-target treatment recommendations. This will endorse enhancement and consistency of care of this cohort of patients in standard practice.
Keywords
Introduction
IgA vasculitis (IgAV), formerly known as Henoch-Schönlein purpura (HSP), is the commonest cause of vasculitis in childhood, 1 with an incidence of 3–26.7 cases per 100 000 population. 2 Pathologically, it is an acute immunoglobulin A (IgA) immune-mediated disorder characterized by small vessel vasculitis of the skin, joints, kidney, gastrointestinal tract, and, rarely, the lungs and the central nervous system. IgAV is a relatively self-limiting disorder in children and generally resolves without permanent complications. 3 Renal and gastrointestinal complications may occur. Persistent renal affection and the presence of crescentic glomerulonephritis on renal biopsy may occur in a small minority of patients who may progress to end-stage renal disease later in life. Renal damage is considered the most common cause of morbidity and mortality in IgAV.4,5
Given its potential life-threatening systemic complications, early and accurate diagnosis represents a major challenge for health care professionals. It is also vital to initiate the appropriate management and to closely monitor the IgAV disease activity, aiming at persistent disease remission status. 6 Locally in Egypt and in concordance with the international experience, IgAV represents a similar challenge. In a multi-center study in Egypt, Immunoglobulin A vasculitis was the third commonest form of vasculitis reported among Egyptian patients diagnosed with vasculitis. 7 However, there is paucity of international guidelines for management of IgAV, and up till now, there are no Egyptian agreed, evidence-based, treat-to-target (T2T) recommendations regarding the appropriate diagnosis and management strategy of IgAV in children. Given the heterogeneity of the ethnic constitution of Egyptian population, IgAV remains major contest, particularly in the context of discrepancies in the therapeutic strategies encountered. This was the driving force behind the development of guidelines specific for Egyptian children with IgAV.
The development of a comprehensive process for yielding consensus among experts in vasculitis has been agreed to be the best approach for establishing a T2T for IgAV and for assessing the possibility of its implementation in standard day-to-day clinical practice. Therefore, Delphi technique was applied to analyze the level of consensus on T2 T strategy in IgAV among specialists practicing in Egypt. This project was an initiative led by the Egyptian college of pediatric rheumatology.
Methods
Design
A qualitative synthesis of scientific evidence and consensus based on clinical experience and existing scientific evidence was used to formulate the study design. This was a multistep process which followed the ‘Clinical, Evidence-based, and Guidelines’ (CEG) initiative protocol aiming at setting up an actionable clinical gold standard for T2T management of IgAV in children. The project is an initiative led by the Egyptian College of Pediatric rheumatology to set up a gold standard for IgAV management in Egypt.
Development stages
Core team
It is formed of four experts with recognized experience in pediatric rheumatology, vasculitis as well as nephrology. The core team supervised and coordinated the teamwork, assisted with developing the scope of the project and initial patient/ population, intervention, comparison, and outcomes (PICO) clinical questions, reached a consensus on the key questions to include in the guidelines, nominated the expert panel and drafting the manuscript.
Literature review team
Led by an experienced literature review consultant, the literature review was conducted with the assistance of an expert in methodology. The team completed the literature search, data abstraction as well as the quality of evidence rating. 8 Following the revision, each of the experts responsible for the literature review provided recommendations regarding each section based on evidence, when that was available, or on their own experience. The level of evidence was determined for each section using the Oxford Center for Evidence-based Medicine (CEBM) system. 9
Inclusion criteria
Articles included were systematic reviews, randomized controlled trials (RCTs), uncontrolled trials, observational studies including cohort, case–control, and cross-sectional studies.
Exclusion criteria
Editorials, commentaries, conference abstracts, and non-evidence-based narrative/personal reviews were excluded.
Expert panel
The core leadership team nominated 26 participants. The criteria for their selection included practice in the Egyptian Health System have professional knowledge and experience (at least 8 years of experience) in the field of pediatric rheumatology, management of vasculitis, and renal disorders as well as active participation in scientific research on pediatric rheumatic diseases. The expert panel assisted with developing the scope of the project, refining the PICO questions and voted on the recommendations.
Key questions used to develop the guideline
This guideline was centered on a string of structured key questions that define the target population, classification criteria, the diagnostic test, intervention, or exposure under investigation, the comparison(s) used, and the outcomes used to assess efficacy, effectiveness, or risk. The evidence to answer the clinical questions was gathered according to the following steps: structuring of clinical questions, formulation of questions, find the evidence, critical analysis/evaluation as well as selection of evidence, presentation of outcomes, and recommendations. These questions, shown in Table 1 form the principles of the systematic literature review and subsequently the clinical care standards.
Key questions used to develop the guideline.

Developing the clinical care standards framework
According to the answers to the formulated key clinical questions and the review of literature, a structured model was developed to enable standardized categorization of guideline components. For each recommendation’s component, the format in which the information/recommendations will be presented and obtained has been identified.
Delphi process
The Delphi method is a structured approach broadly used to collect valuable information on a specific issue. It relies on the key hypothesis that, in general, predictions from a group are more accurate than those gathered from individuals. Therefore, the Delphi method aims at constructing consensus forecasts from a consortium of specialists in a structured iterative style. Its technique is based on a sequence of cycles or ‘rounds’ addressed to specialists. The Delphi process generally entails the following phases: (1) a panel of specialists is gathered. (2) Predicting tasks/challenges are set and shared with the experts. (3) Experts give back their preliminary scores and justifications. These are recorded and recapitulated to deliver feedback. (4) Feedback is presented to the experts, who reviewed their forecasts considering the feedback. This step may be iterated until a satisfactory level of consensus is reached. (5) Final forecasts are constructed by aggregating the experts’ forecasts. The key features of this method are the anonymity of participants and the controlled feedback.10–12
Consensus process
Two Delphi rounds were carried out to establish consensus regarding the T2T (treat-to-target) strategy in IgAV. Once the main aspects of this strategy were identified, a discussion group has defined the aspects to be included in the questionnaire with the scientific committee. The structured Delphi approach ensures that the opinions of participants are equally considered, and it is particularly useful for geographically diverse centers as in Egypt. The Delphi process was conducted through online questionnaires. The first round of the electronic questionnaire included 20 items involved in the T2T strategy of IgAV.
Voting process
Live online-delivered voting was carried out in two rounds that were strictly time limited. All members of the task force were invited to participate and were pre-informed of the time of opening and closure of each round of votes. Unique access links were sent out, and anonymous votes were gathered and processed. Comments on re-phrasing, potential ambiguity, unidentified overlaps were gathered regarding each statement at the same time in the voting process. Only the members of the task force had the right to vote on the statements.
Rating
Each statement was rated between 1 and 9 with 1 being ‘complete disagreement’ and 9 being ‘complete agreement’. Generally, 1–3, 4–6, and 7–9 represent disagreement, uncertainty, and agreement, respectively. There is no requirement to vote on all statements, and the members are encouraged to abstain if they feel that a statement falls outside their area of expertise. Therefore, an ‘uncertainty’ vote represents ‘inconvenience about the accuracy of the recommendation’. All statements are permitted for the entry of remarks which were evaluated by the scientific committee after each round of voting. In the second voting round, the members were further encouraged to leave notes wherever they vote ‘disagree’. This will allow the panel to recognize an example of misinterpretation of statement and overturn the vote on that statement.
Definition of consensus
Description of consensus was determined before data analyses. It was agreed that consensus would be reached if at least 75% of contestants achieved agreement (score 7–9) or disagreement (score 1–3).11–14 A statement was retired if it had a ‘low’ level of agreement or a mean vote below 3. Statements whose score came in the uncertainty rate, (4–6), were amended in view of the comments. The agreement levels on each recommendation statement were identified as ‘high’ if after the second round of votes, all votes on a statement fell into the agreement bracket (7–9).–14
Chronogram of Delphi rounds
The first round took place between 14–18 April 2021 (5 days). The aspects about which the contestants did not achieve consensus in round ‘1’ were revised in view of the experts’ notes and contained in round ‘2’. The second round occurred (1 week after round ‘1’) and lasted for 3 days, between 20 and 22 April 2021.
Ethical aspects
This study was carried out in accordance with the Helsinki Declaration. The ‘Clinical, Evidence-based, Guidelines’ (CEG) initiative protocol has been ethically approved (ethical approval code: 34842/8/21). As per the Egyptian national Ethical Committee regulations, verbal informed consent was gathered from all the contestants sharing in this work. All the contributors were kept anonymous, in accordance with the data protection scheme.
Results
Literature research and evidence selection
In the study selection process, we found 6122 potentially relevant studies by search strategy. About 5998 were excluded by screening of title and abstracts (studies did not examine population or intervention of interest, did not match study design of interest, or did not report outcome measures of interest). Therefore, relevant 124 studies were included for full article review. Around 101 studies were excluded as citations did not provide evidence matching a PICO. Therefore, we included 23 studies in this work.
Expert panel characteristics
The Delphi form was sent to expert panel (n = 26), who shared in the two rounds. Experts were drawn from different health centers and counties across Egypt: Cairo University (30.4%), Ain Shams university (15.2%), Tanta university (7.6%), Benha university (3.8%), Alexandria university (3.8%), Suez Canal University (7.6%), Zagazig university (7.6%), Minia university (3.8%), Mansoura university (3.8%), Fayoum university (3.8%), Assiut university (3.8%), Menofeya university (3.8%), and Sohag university (3.8%).
Delphi round 1
Round ‘1’ response rate was 100% (26/26). Consensus was achieved on the inclusion of clinical standards on 85% of the items (i.e. ⩾75% of respondents strongly agreed or agreed). There were notes made concerning the wording of some of the statements. Comments (excluding minor editing suggestions) were more frequent for key points, investigations, abdominal pain, cerebral vasculitis, and pulmonary hemorrhage. Diversity of opinion was greatest for the item ‘dose of pulsed steroid therapy’. One statement was retired for similarities to other statements. The number of statements which were added after round 1 were 1, 2, 1, 1, 1, 2, 1, 1, 2, and 3 statements in key points, investigations, imaging, general principals, abdominal pain, skin affection, cerebral vasculitis, pulmonary hemorrhage, renal affection, and recurrence sections, respectively. Several statements were revised after round 1; most edited statements were in treatment of nephritis section (five statements), two statements were edited in investigations and general principles sections, and one statement was edited in joint affection, skin affection, cerebral vasculitis, and pulmonary hemorrhage.
Delphi round 2
Round ‘2’ response rate was 100% (26/26). Frequency of high rank recommendation (rank 7–9) ranged from 91.7–100 %. One statement was retired for similarity with another statement. Consensus was achieved (i.e. ⩾75% of respondents strongly agreed or agreed) on all the clinical standards. Table 2 shows all the statements with their assigned level of evidence, in accordance with the Oxford Center for Evidence-Based Medicine (CEBM) criteria as well as mean ± standard deviation and level of agreement. 9 There was unanimous agreement (>80% agreement) for the statements’ wording.
Levels of evidence.

Recommendations for management of children with IgAV
At the end of round 2, a total of 20 recommendation items, categorized into two sections (eight recommendations in diagnosis section and 12 in management), were obtained. Diagnostic recommendations used in this study are summarized in Table 3. A breakdown is presented in Table 4.
Diagnostic recommendations.

Breakdown of statements of recommendations, its individual rank by experts opinion and level of agreement.

ARBS, angiotensin II receptor blockers; ACE-I, angiotensin-converting enzyme inhibitors; ANA, antinuclear antibodies; ASOT, Anti Streptolysin O Titre; B.P, blood pressure; cANCA, cytoplasmic antineutrophil cytoplasmic antibodies; CBC, complete blood count; COVID, coronavirus disease; CRP, C-reactive protein; dsDNA, double stranded deoxyribonucleic acid; FMF, familial mediterranean fever; eGFR, estimated glomerular filtration rate; G6PD, glucose-6-phosphate dehydrogenase; Gl, glucose, H, high level of agreement; HPF, high power field; ICU, intensive care unit; IgAV, IgA vasculitis; ISKDC, International Study of Kidney Disease in Children; IV, intravenous, LE, Level of evidence according to the Oxford Center for Evidence-Based Medicine (CEBM) criteria; MEFV, Mediterranean fever gene; NSAIDS, non-steroidal anti-inflammatory drugs; pANCA, Perinuclear antineutrophil cytoplasmic antibodies; SD, standard deviation; UP: UC, urinary protein-to-urinary creatinine ratio; US, Ultrasound.
Application of the primary recommendations to clinical practice guidelines
Flow chart for the study selection process is shown in Figure 1, whereas Figures 2 and 3 show the monitoring and management approach.

Flow chart for the study selection process.

Monitoring of immunoglobulin A vasculitis.
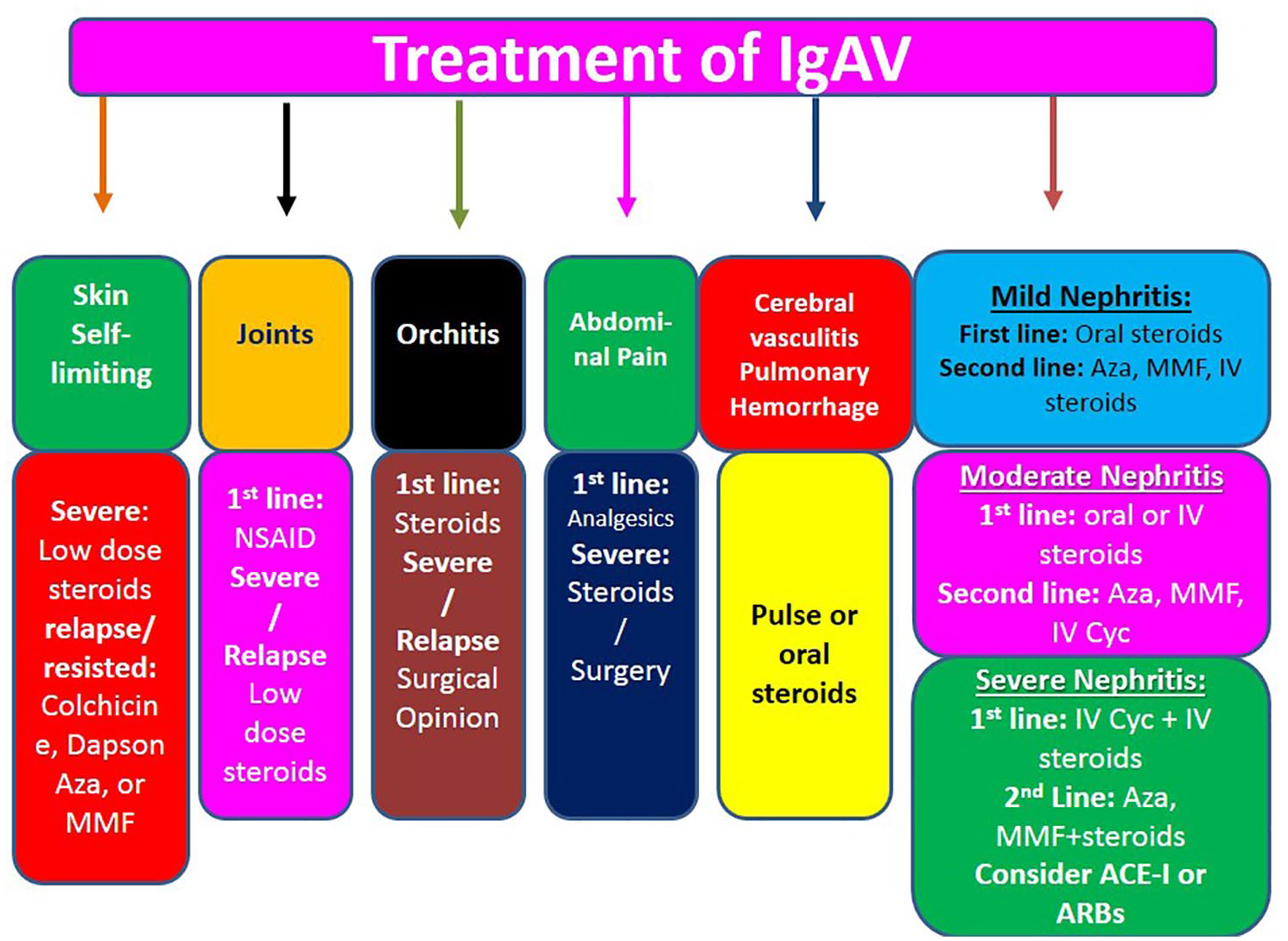
Systemic treatment plan for immunoglobulin A vasculitis.
Discussion
IgA is one of the most common forms of systemic vasculitis in children. In a population-based study carried out in the United Kingdom, the approximate annual incidence was 20 per 100,000 in children <17 years of age, with a peak incidence of 70 per 100,000 in children between the ages of 4 and 6 years. 15 In a multi-center study carried out in Egypt 7 which included 630 patients, vasculitis linked to HCV infection and Behçet’s disease were the most commonly types of vasculitis diagnosed. This accounted for 24% and 23.5% of the studied cases, respectively, followed by IgAV at 16%. As far as the vasculitis cases geographic distribution, the frequency of IgAV was higher among patients from Lower Egypt. 7 Bearing in mind the early finding that 90% of IgAV cases occur in the pediatric age group, 2 this reflects the importance of having guidelines for IgAV in Egypt. The 2020 Egypt census revealed that number of population is 104,124,440, of whom 33.62% are children in the age range of 0–14 and 18.01% in the range of 15–24. 16 This work was an initiative by the Egyptian College of Pediatric rheumatology aiming at providing an expert, consensus recommendations for the diagnosis and treatment of IgAV and IgAV nephritis.
The role of genes in the disease pathogenesis and the possibility of genetic predisposition in individuals with IgAV has been reported in earlier studies.17,18 This was attributed to the association with the HLA class II region that in people of European background is mainly related to HLA-DRB1*01 allele. 17 Other studies19,20 revealed that the cytokines signaling pathway genes are crucial factors of the genetic network underlying the pathogenesis of the disease. However, while, the influence of the interleukin (IL)33-IL1 receptor-like (IL1RL)1 signaling pathway on the increased risk of several immune-mediated diseases has been described; 19 B cell-activating factor (BAFF, also known as B-lymphocyte stimulator or BlyS) and a proliferation-inducing ligand (APRIL), which are cytokines expressed by antigen-presenting cells that play a crucial role in the development of B-lymphocytes, were reported not contributing to the genetic network underlying IgAV. 20
The ACR criteria published in 1990 21 for HSP mandates the presence of at least two of the following parameters: (I). Age at disease onset lower than or equal to 20 years; (II). Palpable purpura; (III). Acute abdominal pain; (IV). Hemorrhagic diarrhea with bowel ischemia; (V). Exclusion of other hematological disorders. In contrast, the new classification criteria for Henoch-Schonlein purpura (HSP) by the EULAR/PRINTO/Pediatric Rheumatology European Society-endorsed Ankara 2008 criteria (SHARE) 6 removed the age at onset, included predominant IgA deposition, and added arthritis and renal affection to the classification criteria. This guideline defined patient populations based on EULAR/SHARE classification criteria. The decision was made as SHARE recommendations were developed based on a large international patients’ registry and were specifically validated for childhood-onset disease. However, as there is a variety of gastrointestinal manifestations in IgAV, which not only include diffuse abdominal pain but also gastrointestinal bleeding (such as hematemesis, melena, hematochezia, or silent gastrointestinal bleeding) and vomiting, the Egyptian guidelines recommended diagnostic work-up as well as standard investigations to cover this variety of gastrointestinal affection.
In general, the prognosis of IgAV is good, with the exception of those with significant renal involvement. Therefore, timely and effective treatment aiming at disease control should be considered as a priority to avoid the end-stage renal progression. This guideline has taken the extra step, defined treatment outcomes as stable or partial remission and provided a T2T algorithm for both renal as well as extra-renal affection. Thus, endorsed goal-directed therapy strategy to tailor the treatment according to the patient’s disease activity status and, in the meantime, assist the clinicians in selecting the most appropriate initial therapy as well as second and third lines of management. Though there has not been published T2T guidelines for IgAV in children, this approach has been highly recommended in different inflammatory arthritic conditions. 22
In contrast to the EULAR/SHARE recommendations 6 which were mainly therapy based, the Egyptian guidelines adopted an organ-specific management strategy. The guidelines presented a comprehensive approach showing how to treat individual patients with mild-severe forms of systemic organ affection including renal disorders. The Egyptian guidelines regarding the management of renal disorders are also in agreement with the Kidney Disease: Improving Global Outcomes (KDIGO) 23 which is mainly based on expert opinion. The KDIGO group endorsed angiotensin-converting enzyme inhibitor for persistent proteinuria for its valuable impact on proteinuria and the mesangial cell. However, while KDIGO does not suggest the use of immunosuppression, except for the use of cyclophsphamide for >50% crescents, the Egyptian guidelines advised the use of effective immunosuppression to prevent/minimize the risk of occurrence of chronic kidney disease or renal failure, particularly in the acute vasculitic stages. This agrees with earlier published studies which emphasized the need for such lines of therapy.24–26
Considering the lack of high-level evidence for management of IgAV, expert consensus recommendations for the diagnosis and treatment of IgAV and IgAV nephritis is expected to play an important role. The Delphi method has proven to be a reliable tool in attaining such consensus and setting the path for future-orientated research. 27 Delphi methodology pursues the opinion of a cohort of experts to measure the level of agreement and to solve disagreement on a disputed matter. 28 When the experts were questioned about the likelihood of employing a well-defined target in IgAV, there was a wide consensus. In Delphi technique, consensus is usually achieved when agreement or disagreement ranges from 50% to 80%.28,29 In this work, a total of eight recommendations for diagnosis and 12 for treatment, the agreement ranged between 91.7% and 100%, reflecting a strong tendency among the Egyptian health care professionals to have a T2T approach for IgAV management. These findings agree with the outcomes of the EULAR/Share consensus-based recommendations for the diagnosis and management of IgAV. 6
Although relapses are common in IgAV, specifically focused information on relapses is scarce and results are often discordant. In this regard, earlier studies reported the frequency of relapses in the range of 2.7–51.7%.30,31 However, in view of the discrepancy in the frequency and clinical spectrum of relapses in IgAV, there have not been much data regarding the management of these cases, and treatment protocols for such cases are not yet established. As the Egyptian guidelines addressed the specific organ affection in IgAV, it gave a plan regarding the appropriate management in cases of relapse; particularly the treatment of joint, gastrointestinal manifestations and the history of previous infection at the time of the diagnosis of IgAV which have been identified as independent predictive factors for IgAV relapses. 32 The guidelines endorsed the use of musculoskeletal ultrasound as a valuable tool for the detection of early musculoskeletal findings in patients with joint manifestations. Also, criteria for admission and early referral to pediatric nephrologist were identified. Furthermore, the Egyptian guidelines advised the use of plasmapheresis and IVIG in resisted cases. Also, rituximab was suggested as an alternative therapy in refractory or relapsing IgAV. In concordance, the recommendations optimized the use of steroids, colchicine, dapson, and mycophenolate mofetil in relapsed skin cases.
The main strengths of the study are related to the diversity as well as the expertise of the participants, the high levels of consensus achieved, and the agreement with the most recently published recommendations. Also, the adoption of the PICO methodology approach as well as the T2T outcome as the main pillars of this work. Limitations of the guideline: the guideline reflects the best data available at the time the report was prepared. Also, the guideline refers specifically to IgAV in children and cannot be extrapolated to adults living with the disease. Furthermore, in view of the absence of head-to-head comparative studies identified in the literature review, indirect comparisons among trials/therapies were used for the purpose of this work. Although framed for Egyptian children with MAS, we hope that these guidelines will be valuable for pediatric rheumatologists across the globe. Caution should be exercised in interpreting the data; the results of future studies may require alteration of the conclusions or recommendations stated in this report. It may be necessary or even desirable to depart from the guidelines in the interests of specific patients and special circumstances. Just as adherence to guidelines may not constitute defence against a claim of negligence, so deviation from them should not necessarily be deemed negligent.
Conclusion
This was an expert, consensus recommendations for the diagnosis and treatment of IgAV and IgAV nephritis, based on best available evidence and expert opinion. The guidelines fill a gap in the literature as it presents a T2T approach for IgAV and a guide to treat-relapsed cases. These recommendations should facilitate the best methods of diagnosis and management of this condition, also this will help to increase consistency in practice and promote the highest standards of care.
Footnotes
Author contributions
All authors contributed in the study methodology, analysis, and interpretation of the data and outcomes as well as the manuscript writing, reading, and approval of the final version.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
