Abstract
Background:
We analysed clinical and biochemical parameters in predicting severe gastrointestinal (GI) manifestations in childhood IgA vasculitis (IgAV) and the risk of developing renal complications.
Methods:
A national multicentric retrospective study included children with IgAV reviewed in five Croatian University Centres for paediatric rheumatology in the period 2009–2019.
Results:
Out of 611 children, 281 (45.99%) had at least one GI manifestation, while 42 of 281 (14.95%) had the most severe GI manifestations. Using logistic regression several clinical risk factors for the severe GI manifestations were identified: generalized rash [odds ratio (OR) 2.09 (95% confidence interval (CI) 1.09–4.01)], rash extended on upper extremities (OR 2.77 (95% CI 1.43–5.34)] or face [OR 3.69 (95% CI 1.42–9.43)] and nephritis (IgAVN) [OR 4.35 (95% CI 2.23–8.50)], as well as lower values of prothrombin time (OR 0.05 (95% CI 0.01–0.62)], fibrinogen [OR 0.45 (95% CI 0.29–0.70)] and IgM [OR 0.10 (95% I 0.03–0.35)]] among the laboratory parameters. Patients with severe GI involvement more frequently had relapse of the disease [OR 2.14 (CI 1.04–4.39)] and recurrent rash [OR 2.61 (CI 1.27–5.38)]. Multivariate logistic regression found that the combination of age, GI symptoms at the beginning of IgAV and severity of GI symptoms were statistically significant predictors of IgAVN. Patients in whom IgAV has started with GI symptoms [OR 6.60 (95% CI 1.67–26.06)], older children [OR 1.22 (95% CI 1.02–1.46)] with severe GI form of IgAV (OR 5.90 (95% CI 1.12–31.15)] were particularly high-risk for developing IgAVN.
Conclusion:
We detected a group of older children with the onset of GI symptoms before other IgAV symptoms and severe GI form of the IgAV, with significantly higher risk for acute and chronic complications of IgAV.
Introduction
IgA vasculitis (IgAV), or Henoch–Schönlein’s purpura, is the most frequent systemic vasculitis in childhood with an incidence fluctuating from three to 27 cases per 100,000 children.1,2 Although the disease is most often self-limiting, various acute and chronic complications are possible.3,4 Gastrointestinal (GI) involvement is present in more than 50% children with IgAV and in about 10–20% of patients with GI manifestations serious complications such as intussusception, bowel perforation and massive bleeding occur.5,6 While the most common acute complications of the disease are related to the GI system, nephritis (IgAVN) is the most important chronic complication of IgAV and thus the main prognostic factor.7–10
Therefore, we conducted a multicentre retrospective study to analyse clinical and biochemical parameters in patients with IgAV in order to better describe the distinctive nature of patients with GI manifestations, to try to identify risk factors for severe GI manifestations in IgAV and to determine risk factors for IgAVN in a group of patients with GI manifestations.
Methods
Patient selection and study design
This multicentre retrospective study included patients under the age of 18 years with IgAV reviewed in five Croatian University Centres for paediatric rheumatology in the period between 2009 and 2019. Diagnosis of IgAV was based on the criteria defined by the European League Against Rheumatism, the Paediatric Rheumatology International Trials Organization and the Paediatric Rheumatology European Society. 11 The study was approved by the Ethics Committee of the University of Zagreb School of Medicine. Patient(s) or their guardian(s)/legally authorized representative(s) provided written informed consent for the publication of patient information in the present study. The STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) checklist for cohort, case–control and cross-sectional studies is presented as a Supplemental file online.
Data collection
Clinical data and laboratory parameters were collected from medical records. Clinical data included sex; age at disease onset; time of follow-up; prodromal infections before the appearance of purpura; isolated pathogen in patients with prodromal infections; number of relapses; joint, renal, scrotum and central nervous system involvement; distribution and severity of skin changes: rash localized on lower extremities, rash extended to the trunk, upper extremities or face, generalized rash, recurrent rash; and type of GI manifestation. A generalized rash was defined as a diffuse eruption of purpura or petechiae over the trunk and extremities, involving the neck and face. In patients with severe skin lesions and atypically distributed rash from the onset of the disease a skin biopsy was performed to confirm the diagnosis of IgAV and to exclude other vasculitides.
All GI manifestations were divided into four subgroups: subgroup one included abdominal pain with or without vomiting and diarrhoea; subgroup two included positive faecal occult blood test without other GI symptoms; subgroup three included abdominal pain with or without vomiting and diarrhoea and with positive faecal occult blood test; subgroup four included intussusception, intestinal perforation, haematochezia and/or massive GI bleeding. Subgroups one and two were considered mild GI manifestations, subgroup three moderate and subgroup four severe GI manifestations.
Relapse was defined as a new flare of clinical signs attributable to IgAV in a patient previously diagnosed with IgAV after an asymptomatic period of at least 1 month.
Patients with IgAVN were divided into two groups: the first group consisted of patients who had IgAVN at the time of diagnosis of IgAV or developed it within 1 month of the first symptoms of IgAV, and the second group consisted of patients who developed IgAVN more than a month after the onset of the first symptoms of IgAV. In patients with severe proteinuria (>2.5 g/day of proteins in 24 h urine collection), persistent isolated proteinuria (1–2.5 g/day of proteins in 24 h urine collection for more than 3 months) as well as in those with nephritic or nephrotic syndrome at any time point or acute progressive glomerular nephritis, renal biopsy was performed.
Laboratory findings included: erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), haemoglobin, haematocrit, leukocytes, platelets, serum urea, serum creatinine, estimated glomerular filtration rate (eGFR), urinalysis and urine albumin:urine creatinine ratio in a spot urine sample, fibrinogen, D-dimer, prothrombin time (PT), activated partial thromboplastin time (APTT), total serum protein, serum albumin, immunoglobulins (IgA, IgG, IgM and IgE), serum complement C3, complement C4, antistreptolysin O titre, and faecal occult blood test. In the case of pathological urine findings quantification of proteinuria in 24 h urine collection was performed. Samples of venous blood, urine and stool were taken from all patients at the disease diagnosis.
Statistical analysis
Statistical analysis was performed using the R software. 12 Numerical data are descriptively presented with medians and interquartile ranges, while categorical data are presented as absolute and relative frequencies. The normality of the distribution was evaluated using the Kolmogorov–Smirnov test. Differences between categorical variables between the group of patients with IgAV and GI manifestations and the group of patients with IgAV without GI manifestations were examined using the χ2, binomial and Fisher’s exact test, and among the numerical ones using the Mann–Whitney U test. Spearman correlation matrices were utilized to interpret co-variations between the parameters studied.
Logistic regression analysis was used to find predictors for the development of the most severe forms of GI manifestations. Patients who had no GI symptoms at all were compared with those who developed severe forms of GI manifestations. As well, the association of GI symptoms with renal disease was also examined by univariate followed by a multivariate logistic regression analysis based on highly significant features (p < 0.01) obtained from the univariate model.
The level of statistical significance was determined with p < 0.05.
Results
Patients
Out of 611 children with IgAV, 320 were males, 291 were females. The median (range) age at diagnosis was 6.33 (4.50–8.92) years, the median (range) follow-up time was 49 (7–120) months. Out of 611 children, 281 (45.99%) had at least one GI manifestation, 475 (77.74%) had joint involvement, 130 (21.28%) had IgAVN, 29 (9.06% of male patients) had orchitis and three (0.49%) had central nervous system involvement. In 20 patients (3.27%) a skin biopsy was performed to confirm the diagnosis of IgAV and renal biopsy was done in 40 patients (30.77% of patients with IgAVN).
Among patients with IgAVN, 109 (17.84%) had nephritis at the time of diagnosis of IgAV or within 1 month of the first symptoms of IgAV, and in 21 patients (3.44%) IgAVN developed more than a month after the onset of the first symptoms of IgAV. The median (range) time for IgAVN development was 4 (0–390) days.
A statistically significant difference was observed between gender proportions (p = 0.003). Among patients with GI symptoms there were 1.44 times more males (n = 166) than females (n = 115).
Clinical features at diagnosis
Patients with GI symptoms had fewer infections that preceded the appearance of purpura compared with patients without GI symptoms (59.80% versus 70.90%, p = 0.005). Observing by type of infection, statistical significance was found in the difference of respiratory infections: patients with GI symptoms had fewer respiratory infections (35.60%) compared with those without (45.20%, p = 0.02), while regarding intestinal infections there was no difference, as well as in the success of isolation of the pathogens.
Patients suffering from IgAV with GI symptoms were found to be significantly more likely to have rash extended on the trunk (61.90% versus 48.50%, p = 0.001) and upper extremities (35.20% versus 24.70%, p = 0.006), as well as generalized rash (38.80% versus 28.30%, p = 0.008). No difference in the frequency of recurrent rash was observed.
When the involvement of organic systems other than GI was compared between IgAV patients with GI symptoms and without GI symptoms, a statistically significant difference was found in the frequency of renal involvement (p = 0.012), while there was no difference in involvement of the joints, scrotum and central nervous system.
No difference was found in the number of relapses of IgAV depending on the presence of GI symptoms.
In patients with GI symptoms, the most common first symptom of IgAV was a rash (66.50%), followed by articular manifestations (22.80%), and in 10.70% of patients the disease began with GI symptoms. About 20% of patients in whom the first symptom of the disease was a GI manifestation or joint manifestation developed the most severe GI complications, which is statistically significantly more than in patients in whom the disease began with a rash (p < 0.001).
In our cohort 143 (50.89%) patients had mild, 96 (34.16%) had moderate, and 42 out of 281 children (14.95%) had severe GI manifestations.
In patients with more severe GI symptoms the frequency of glucocorticoid administration was significantly higher (Table 1). However, univariate logistic regression showed that glucocorticoid therapy did not change the risk of developing IgAVN more than a month after the onset of the first symptoms of IgAV [odds ratio (OR) 2.10, confidence interval (CI) 0.61–7.19, p = 0.24] (Table 2).
Application of glucocorticoid therapy according to the severity of GI disease.

Patients with abdominal pain with or without vomiting and diarrhoea.
Patients with positive faecal occult blood test without other GI symptoms.
Patients with abdominal pain with or without vomiting and diarrhoea and with positive faecal occult blood test.
Patients with intussusception, intestinal perforation, haematochezia and/or massive GI bleeding.
GI, gastrointestinal.
Results of univariate logistic regression analysis of the association of clinical features, symptoms, signs and laboratory findings with the development of IgAVN in patients with IgAV and GI symptoms.
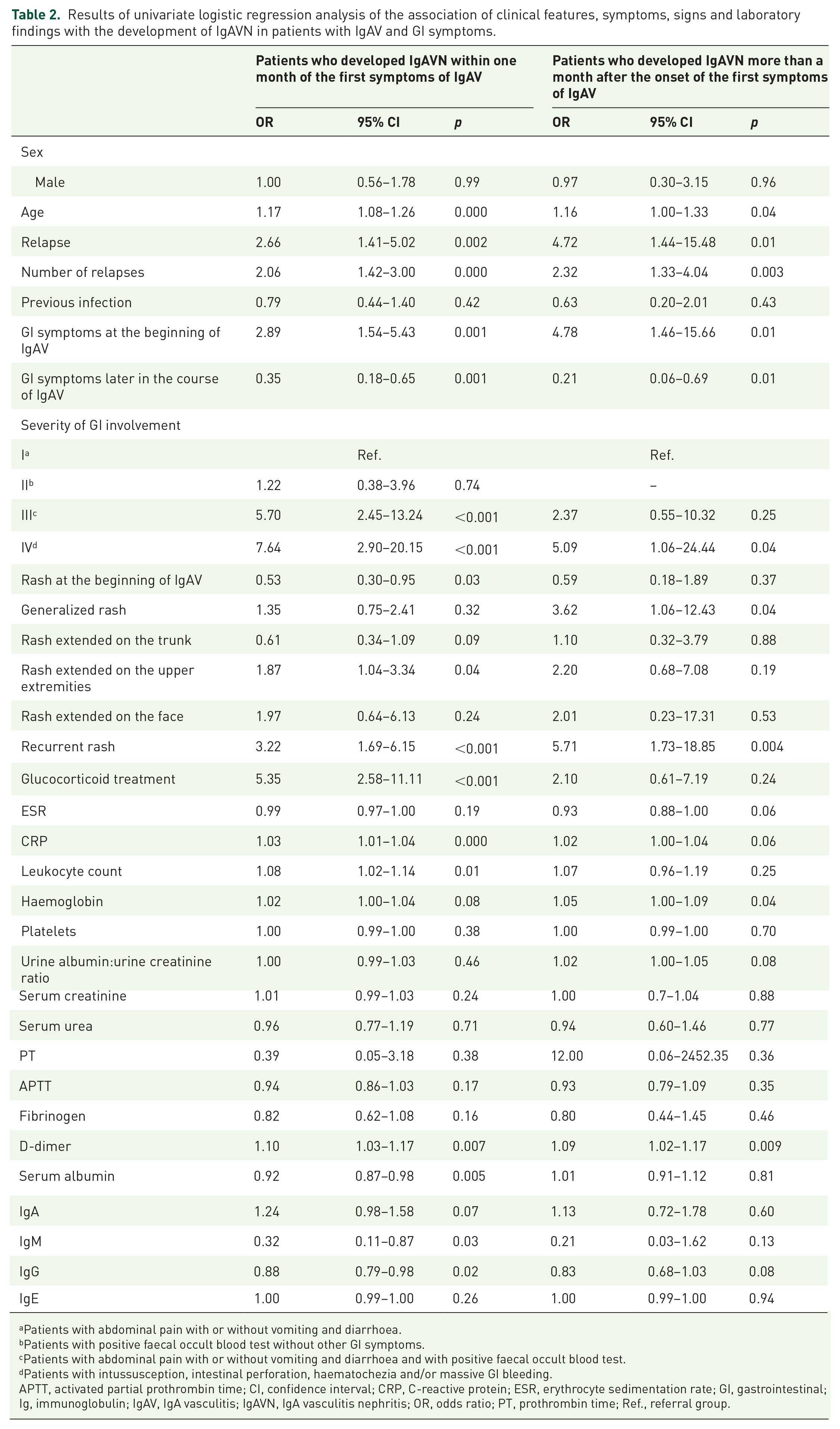
Patients with abdominal pain with or without vomiting and diarrhoea.
Patients with positive faecal occult blood test without other GI symptoms.
Patients with abdominal pain with or without vomiting and diarrhoea and with positive faecal occult blood test.
Patients with intussusception, intestinal perforation, haematochezia and/or massive GI bleeding.
APTT, activated partial prothrombin time; CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; GI, gastrointestinal; Ig, immunoglobulin; IgAV, IgA vasculitis; IgAVN, IgA vasculitis nephritis; OR, odds ratio; PT, prothrombin time; Ref., referral group.
In the univariate model, several parameters were associated with the development of severe GI manifestations: generalized rash (OR 2.09, CI 1.09–4.01, p = 0.03), rash extended on upper extremities (OR 2.77, CI 1.43–5.34, p = 0.002), rash extended to the face (OR 3.69, CI 1.42–9.43, p = 0.007) and IgAVN (OR 4.35, CI 2.23–8.50, p < 0.001). Patients with severe GI involvement more frequently had relapse of the disease (OR 2.14, CI 1.04–4.39, p = 0.04) and recurrent rash (OR 2.61, CI 1.27–5.38, p = 0.009).
Results of univariate logistic regression showed that the presence of GI symptoms increases the chance of developing IgAVN (OR 1.68, CI 1.14–2.48, p = 0.009). Within the group of patients with GI symptoms, the following patients had the highest risk for developing IgAVN: patients with severe and moderate GI manifestations; patients with recurrent rash; patients in whom the first symptom of IgAV was a GI symptom; patients with GI symptoms and one or more relapses of IgAV; older children (Table 2).
It has been shown that patients with generalized rash (extended to the face) were more likely to develop IgAVN more than a month after the onset of the first symptoms of IgAV (Table 2).
Multivariate analysis in patients with IgAV and GI symptoms found that the combination of age, GI symptoms at the beginning of IgAV and severity of GI symptoms were statistically significant predictors of IgAVN, both in patients who developed IgAVN within one month of the first symptoms of IgAV, and in patients who developed IgAVN later (Table 3).
Results of multivariate logistic regression of IgAVN development in patients with IgAV and GI symptoms who developed IgAVN within one month of the first symptoms of IgAV (a) and more than a month after the onset of the first symptoms of IgAV (b).

Patients with positive faecal occult blood test without other GI symptoms.
Patients with abdominal pain with or without vomiting and diarrhoea and with positive faecal occult blood test.
Patients with intussusception, intestinal perforation, haematochezia and/or massive GI bleeding.
CI, confidence interval; CRP, C-reactive protein; GI, gastrointestinal; Ig, immunoglobulin; IgAV, IgA vasculitis; IgAVN, IgA vasculitis nephritis; OR, odds ratio.
If the first symptom of IgAV was a GI symptom, the patient had 2.45 times (CI 1.05–5.68, p = 0.04) higher chance of developing IgAVN within the first month and even 6.60 times (CI 1.67–26.06, p = 0.007) higher chance of developing IgAVN later in the course of IgAV compared with patients in whom the IgAV did not begin with GI symptoms.
This risk for early-onset IgAVN was 4.66 times (CI 1.64–13.24, p = 0.004) higher in patients who had moderate and severe GI manifestations. The risk for late-onset IgAVN was 5.90 times (CI 1.12–31.15, p = 0.04) higher in patients with moderate and severe GI manifestations.
With each year of life, the risk of developing IgAVN in our cohort of children with IgAV increased by 1.22 times (CI 1.02–1.46, p = 0.03).
Patients with early-onset IgAVN 1.70 times (CI 1.08–2.67, p = 0.02) more frequently had relapse of the disease. If patients received glucocorticoids because of GI manifestations, the chance for IgAVN development within one month increased 2.88 times (CI 1.12–7.44, p = 0.03) but the risk for late-onset IgAVN did not change.
Laboratory findings
Patients with IgAV and GI symptoms had statistically significant higher values of CRP, leukocyte count, erythrocytes, platelets, haemoglobin, haematocrit and D-dimer concentrations and lower levels of IgG and IgM (Table 4). Faecal occult blood test was positive in 66.50% of patients with GI symptoms.
Comparison of laboratory parameters according to GI system involvement.

Mann–Whitney U test.
CRP, C-reactive protein; GI, gastrointestinal; Ig, immunoglobulin.
When a statistical analysis of laboratory parameters in dependence on the intensity of GI symptoms was performed, it was shown that patients with the most severe GI manifestations had statistically significantly higher values of 24-h urine protein levels and D-dimer concentrations, while the values of fibrinogen, total serum protein, albumin, IgG, IgM and C3 levels were significantly lower in comparison with children whose GI manifestations were less severe (Table 5).
Comparison of laboratory parameters according to the severity of gastrointestinal involvement.

Kruskal–Wallis test.
Patients with abdominal pain with or without vomiting and diarrhoea.
Patients with positive faecal occult blood test without other GI symptoms.
Patients with abdominal pain with or without vomiting and diarrhoea and with positive faecal occult blood test.
Patients with intussusception, intestinal perforation, haematochezia and/or massive GI bleeding.
Ig, immunoglobulin.
After Spearman correlation analysis, positive strong correlations of ESR with CRP and fibrinogen were observed, as well as age with haemoglobin levels and creatinine. Also, there was positive strong correlation between IgG and IgA levels (Supplemental material Table 1 online).
Univariate logistic regression analysis showed that patients with lower values of PT (i.e. tendency toward prolonged PT; normal values ⩾70%; OR 0.05, CI 0.01–0.62, p = 0.02), fibrinogen (OR 0.45, CI 0.29–0.70, p < 0.001) and IgM (OR 0.10, CI 0.03–0.35, p < 0.001) had a higher chance of developing a severe GI form of the disease. Although other laboratory parameters showed statistical significance, their change had a negligible impact on increasing the risk of severe GI symptoms.
Multivariate analysis, however, did not confirm the results of univariate logistic regression.
In the univariate model, several laboratory parameters in patients with GI symptoms of IgAV were associated with the development of IgAVN: higher values of CRP, leukocytes, haemoglobin, D-dimer and lower values of serum albumin, IgG and IgM levels (Table 2).
After a multivariate analysis, only CRP and IgG were shown to be predictors of IgAVN development in patients with IgAV and GI symptoms (Table 3). CRP increase by one unit increased the odds of IgAVN 1.02 times (CI 1.01–1.04, p = 0.006), and the reduction in IgG for one unit increased the risk 1.14 times (OR 0.88, CI 0.76–1.01, p = 0.07).
Discussion
The most important observation in our study is that certain patients with IgAV and GI symptoms have an increased risk of developing IgAVN: if GI symptoms occurred before other symptoms of IgAV, those patients were 2.45 times more likely to develop IgAVN within the first month and even 6.60 times more likely of developing IgAVN later in the course of IgAV compared with patients in whom the IgAV began with skin or joint symptoms. Moreover, the more severe the GI manifestation, the greater is the likelihood of renal involvement.
Previous studies and one meta-analysis have shown that boys in the age group older than 4–8 years who suffered from IgAV were at higher risk of renal involvement.13–18 Our research has shown that older children are at additional risk for developing IgAVN if they had moderate to severe GI symptoms, and the risk increased further with the number of relapses of IgAV.
Considering the association of GI symptoms in IgAV and IgAVN, there are conflicting results from the literature. A meta-analysis from 2016, which included 13 studies and 2398 children with IgAV, of whom 974 had renal involvement, showed that GI symptoms were strongly related to renal involvement. 12 Several other studies have shown similar results.15,19,20 Nonetheless, the results of other research have not confirmed this association.14,16,21
Although, therefore, the possible relationship between GI symptoms and renal disease in IgAV has been described in the literature, the potential pathogenic mechanisms of this association are generally not commented on. It can be hypothesized that patients with more pronounced GI symptoms have a more pronounced inflammatory response resulting in infiltration of leukocytes, which in turn secrete inflammatory agents that lead to tissue damage. This would be supported by the results of several studies according to which leukocytosis and neutrophilia are risk factors for severe renal impairment in IgAV, but are also related to severe GI disease.13,22,23
Furthermore, our observation that patients suffering from IgAV with GI symptoms were found to be significantly more likely to have rash extended on the trunk and upper extremities, as well as generalized rash, can also attempt to explain that patients with GI symptoms have a more pronounced inflammatory response leading to more extensive involvement of blood vessels in the skin by the inflammatory process.
However, other possible pathogenic mechanisms, besides enhanced inflammation, could underlie the association between the GI system and the kidney in IgAV. For example, various GI diseases such as inflammatory bowel disease or coeliac disease, as well as GI manifestations in autoimmune diseases, such as type 1 diabetes, are known to be associated with an increased risk of renal disease. 24 The possible pathogenic mechanisms of this association have not been elucidated, but the following mechanisms are considered: disruption of the GI wall, microbial dysbiosis and translocation of bacterial endotoxins that have proinflammatory and nephrotoxic features. 24 It is possible that some of these mechanisms may also be found in IgAV with GI symptoms.
This is consistent with our findings that patients with IgAV and GI symptoms had higher values of inflammatory parameters compared with patients without GI symptoms, as also shown by Hong et al., 23 but although these differences were statistically significant, the values of inflammatory parameters alone did not show clinical significance between these two groups of patients in our cohort.
Regarding other laboratory parameters, we found that patients suffering from IgAV with GI symptoms had statistically but not clinically significant higher values of erythrocytes, haemoglobin and haematocrit in comparison with patients without GI symptoms. Whilst similar studies did not show such association, 25 we believe that these findings may be the result of haemoconcentration due to dehydration in patients with GI symptoms. Namely, most patients (85.05%) had mild and moderate GI involvement, predominantly accompanied with abdominal pain, nausea and vomiting, causing them to refuse to take food and fluids, resulting in dehydration. However, the differences in erythrocytes, haemoglobin and haematocrit values were not clinically relevant, that is, in practical terms they were 1 g/l in haemoglobin value or 0.1% in haematocrit value.
There are conflicting results in the literature regarding serum IgA, IgG and IgM levels in patients with IgAV.26–30 We noticed that the patients who had the most severe GI manifestations had the lowest serum levels of IgG and IgM compared with patients whose GI manifestations were less severe and with patients without GI symptoms. Moreover, univariate logistic regression analysis showed that a decrease in serum IgM levels is a risk factor for the development of severe forms of GI symptoms in patients with IgAV. Based on the above, patients with IgAV could have immune disorders outside the IgA compartment, and there could be interactions between IgA and other immunoglobulins, which could be associated with a higher likelihood of complications. An alternative hypothesis is that due to GI inflammation there may be stool loss of IgM via mesenteric vasculitis, accounting for the lower IgM levels, but these observations need to be confirmed in further studies.
Similar to Nagamori et al., 22 we observed that patients with GI symptoms and IgAV in our cohort had lower values of albumin and total serum proteins compared with patients without GI symptoms. The lower the values of these parameters, the higher the intensity of GI symptoms. These patients also had statistically significant higher values of 24-h urine protein levels in comparison with patients whose GI manifestations were less severe. Interestingly, a similar trend was observed in patients with inflammatory bowel disease: inflammatory bowel disease patients with active disease have demonstrated higher urinary albumin levels compared with those in the remission phase.29,30
Previous studies have shown activation of coagulation including hyperfibrinolysis secondary to the endothelial damage in patients with IgAV.31,32 Yilmaz et al. 32 have even demonstrated that fibrinogen levels were inversely correlated with GI system score. In our cohort, patients with the most severe GI symptoms had significantly lower fibrinogen values compared with patients without GI symptoms. So far, two possible mechanisms of hypofibrinogenaemia in patients with IgAV have been described in the literature. 31 Even though none of the patients in our study had pathological PT values, a tendency toward lower values (prolonged PT) was observed in patients with more severe GI symptoms. This also suggests that in patients, especially with more severe forms of IgAV, coagulation disorder may be present. 31
The complement system has an important role in the pathogenesis of IgAV.33,34 Notwithstanding we did not observe a statistically significant difference in C3 and C4 complement concentrations in patients with and without GI symptoms in IgAV, patients with the most severe GI manifestations had statistically significantly lower C3 concentrations compared with patients with mild GI symptoms and those without GI symptoms. Low serum C3 and C4 have been reported in the acute stage of IgAV, as a result of complement activation, but did not correlate with the severity of IgAV or renal involvement.35,36 Further research is needed on the possible association of individual components of complement with GI complications.
Another interesting observation from our study is that patients with severe GI involvement as well as those with early-onset IgAVN more frequently had relapse of the disease. Relapses are a well-known complication of IgAV. 37 In one epidemiological study that analysed the frequency, clinical features and predictors of relapses, it was found that the best predictive factors for relapse were joint and gastrointestinal manifestations at IgAV diagnosis, while a history of previous infection was a protective factor for relapses. 37 The exact mechanism of relapse is unknown, but an association with more severe gastrointestinal manifestations and IgAVN may be consistent with the finding that in patients with favourable IgAV outcome the disease is self-limiting, while in patients with continuous inflammation or several inflammatory bursts the disease progresses.
While some authors pointed out that an upper respiratory tract infection, especially those caused with group A beta-haemolytic streptococcus as a trigger in children with IgAV, may lead to a higher incidence of IgAVN, 38 we did not find that prodromal infections before the onset of IgAV were predictors of IgAVN development. Also, our patients with GI symptoms had fewer respiratory infections than patients without GI manifestations. Previous infections were a kind of protective factor for GI involvement. This is in concordance with observations from Calvo-Río et al. 37 about infections as protective factor against relapses of IgAV because of their transient nature, which may lead to a more limited form of the disease.
As shown in our and other research, in most patients with IgA, GI involvement is mild and short-lived, thus treatment is usually supportive.39,40 However, data about optimal treatment of patients with severe GI manifestations are scarce. 40 Published results have not unequivocally shown the benefits of glucocorticoid treatment in patients with severe GI involvement. Nevertheless, in these patients treatment with glucocorticoids could be considered. 40 In our cohort of patients, the frequency of glucocorticoid administration was significantly higher in a group of patients with the most severe GI manifestations: almost all of our patients with such manifestations (95.24%) received systemic glucocorticoids and they were administered as intravenous pulses for three consecutive days, which is also according to the recently published recommendations for diagnosis and treatment of IgAV.40 Furthermore, three children (1.07% of IgAV patients with GI manifestations) required surgical intervention because of ileo-ileal or ileo-colic intussusception and in four patients (1.42%) blood transfusions were necessary because of severe anaemia as a consequence of massive GI bleeding. Second line treatments, such as the use of mycophenolate mofetil, intravenous cyclophosphamide or intravenous immunoglobulin, as well as endoscopic haemostasis, were not used since all patients with GI bleeding favourably responded to therapy with glucocorticoids, similar to many other studies. 41 Although glucocorticoids have been shown to be very successful in treating our patients with severe gastrointestinal manifestations, they have not influenced the risk of developing late-onset IgAVN, and similar results have been shown in controlled studies, therefore prophylactic use of glucocorticoids in IgAVN prevention is not indicated. 40 In patients who were treated with glucocorticoids because of GI manifestations we have observed higher chance for early-onset IgAVN, probably due to the previously explained association between GI symptoms and renal disease, in that more pronounced inflammatory response results in more severe GI manifestations and, consequently, by still insufficiently elucidated mechanisms, it also leads to increased local inflammation in the kidneys.
A potential limitation of our study is its retrospective nature, so it is possible that some information has been omitted, along with the presence of mis-classification or information bias, and conclusion on causal relationships is difficult to assess.
We also did not take into account different genetic factors that also have significance in the aetiopathogenesis of IgAV, while some of them could be associated with different disease phenotypes. For example, it was found that interleukin 1 receptor antagonist gene polymorphism was related to severe renal involvement in patients with IgAV, 42 interleukin 8 gene polymorphism was associated with increased risk of IgAN, 43 and vascular endothelial growth factor gene functional variants with susceptibility to nephritis in IgAV patients. 44 On the other hand, intercellular adhesion molecule-1 gene polymorphisms decreased risk of developing severe GI complications in patients with IgAV. 45 Future studies of various genetic factors, combined with insights from proteomics, will open a new and exciting approach to diagnostic and therapeutic possibilities in IgAV. 46
Conclusions
We detected a group of patients with IgAV and GI symptoms that differed in their clinical and biochemical characteristics from patients without GI symptoms. The most important features of these group of patients were older children, the onset of GI symptoms before other IgAV symptoms and severe GI form of the IgAV. These patients were found to be significantly more likely to develop renal disease and thus cumulatively have a higher risk of acute and chronic complications of IgAV. Clinicians should pay special attention to this group of patients with IgAV, follow them up closely and regularly investigate their renal status with blood pressure measurement, urinalysis and eGFR.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X211024828 – Supplemental material for Gastrointestinal involvement and its association with the risk for nephritis in IgA vasculitis
Supplemental material, sj-docx-1-tab-10.1177_1759720X211024828 for Gastrointestinal involvement and its association with the risk for nephritis in IgA vasculitis by Mario Sestan, Nastasia Kifer, Marijan Frkovic, Matej Sapina, Sasa Srsen, Mateja Batnozic Varga, Aleksandar Ovuka, Martina Held, Ana Gudelj Gracanin, Ana Kozmar, Stela Bulimbasic, Marijana Coric, Gordana Laskarin, Alenka Gagro and Marija Jelusic in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
Some results of this research were previously presented at the 26th Paediatric Rheumatology European Society (PReS) e-Congress in congress abstract publication: Sestan M, Kifer N, Srsen S, et al. Gastrointestinal manifestations and their association with the risk for renal disease in patients with Henoch–Schönlein’s purpura. Pediatr Rheumatol 2020; 18: P292. ![]()
Author contributions
MSe and NK drafted the initial manuscript, collected data, reviewed the literature, examined the patients, performed rheumatologic assessments and created a database of patients. MF critically reviewed the manuscript for important intellectual content and contributed to the acquisition and interpretation of data for the study. MSa designed the data collection instruments, analysed the data, interpreted the results and revised the draft. SS, MBV, AO, MH and AGG contributed to the acquisition and interpretation of clinical data for the study,; they examined the patients and performed rheumatologic assessments. AK, SB and MC contributed to the acquisition and interpretation of laboratory data for the study, reviewed and revised the manuscript. GL and AG critically reviewed the manuscript for important intellectual content and contributed to the acquisition and interpretation of data for the study. MJ conceptualized and designed the study, coordinated and supervised data collection, and critically reviewed the manuscript for important intellectual content. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been fully supported by Croatian Science Foundation under the project IP-2019-04-8822.
Ethics statement
The study was approved by the Ethics Committee of the University of Zagreb School of Medicine, Zagreb, Croatia (date: 18 September 2019; Protocol Number – Class: 641-01/19-02/01).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
