Abstract
Background:
Patients with active axial spondyloarthritis (axSpA) exhibit more absences and lower levels of productivity in the workplace and household than the general population, which can improve upon treatment.
Objectives:
The objective of this study is to determine the long-term impact of achieving different levels of clinical response or disease activity on workplace and household productivity in patients with axSpA.
Design:
RAPID-axSpA (NCT01087762) was a 204-week phase III trial evaluating the safety and efficacy of certolizumab pegol (CZP) in adult patients with active axSpA.
Methods:
The impact of axSpA on workplace and household productivity was evaluated using the validated arthritis-specific Work Productivity Survey. Outcomes included the percentage of patients achieving Assessment of SpondyloArthritis International Society (ASAS) response and Ankylosing Spondylitis Disease Activity Score (ASDAS) thresholds. This post hoc study used a generalised estimating equations model to determine the association between the threshold of clinical response achieved and patient productivity.
Results:
Of 218 CZP-randomised patients, 65.1% completed week 204. At baseline, 72.0% were employed outside the home. Of the patients who were unemployed, 42.6% were unable to work due to arthritis. Achievement of higher treatment response thresholds, such as clinical remission, was associated with fewer days affected by workplace absenteeism (ASAS-partial remission: 4.0 days, ASAS40: 8.6 days, ASAS20 but not reaching ASAS40 response: 29.4 days, ASAS20 non-response: 69.2 days; ASDAS-inactive disease: 5.0 days, ASDAS-low disease activity: 15.6 days, ASDAS-high disease activity: 32.7 days, ASDAS-very high disease activity: 93.4 days). Similar associations were found for workplace presenteeism, and household absenteeism and presenteeism.
Conclusions:
Over 4 years, achievement of higher clinical response thresholds and lower levels of disease activity was associated with fewer cumulative days affected by absenteeism or presenteeism, with clinical remission associated with the greatest improvements in productivity. This highlights the importance of targeting these thresholds to limit the burden of axSpA on society and on patients’ daily lives.
Introduction
Axial spondyloarthritis (axSpA) is a chronic, immune-mediated, inflammatory disease characterised primarily by axial inflammation, in particular in the sacroiliac joints and in the spine. 1 Chronic back pain, stiffness and fatigue are key patient-reported symptoms; patients also often present with peripheral and extra-musculoskeletal manifestations.2–5
There are two subtypes of axSpA: radiographic axSpA [r-axSpA, also known as ankylosing spondylitis (AS)] and non-radiographic axSpA (nr-axSpA). 3 Structural damage in patients with r-axSpA usually develops first in the sacroiliac joints and often progresses to the spine. The development of structural damage together with active inflammation in the hips and spine is relevant for long-term disease outcomes.6,7 Patients with nr-axSpA have no or limited evidence (not fulfilling the requirement of classification as r-axSpA) of radiographic damage, but they commonly have axial inflammation, elevated C-reactive protein (CRP) levels and/or erythrocyte sedimentation rates.3,8,9 Patients with either form of axSpA experience pain, stiffness, fatigue, limitations in mobility and physical function and subsequently impaired quality of life (QoL).7,10–16
Symptoms, such as chronic back pain, stiffness, fatigue, poor sleep, limited spinal mobility and impaired physical function, experienced by patients with axSpA contribute to increased absences from work 17 ; 21% of patients with axSpA reported leaving the labour force within 10 years of diagnosis, and withdrawal from work was shown to be three times higher than in the general population. 18 As well as full days of work missed (absenteeism), axSpA has also been associated with productivity losses at work. Presenteeism, defined as a ⩾ 50% reduction in productivity, is often seen at higher rates than absenteeism in patients with axSpA.19,20 It is therefore important to consider both absenteeism and presenteeism when quantifying the impact of axSpA on productivity or the impact of treatment on improving productivity.
While the impact of axSpA on work productivity is well understood, few studies have assessed how the disease affects additional aspects of daily living, such as productivity in household activities.21–23 Impairments in productivity due to axSpA represent a considerable burden on patients, families and caregivers and, as evidenced by modelling studies, also translate to an economic burden.19,20
Data from the phase III RAPID-axSpA trial previously reported that treatment with certolizumab pegol (CZP), an Fc-free, PEGylated tumour necrosis factor-α inhibitor (TNFi), resulted in improvements in household and work productivity in patients with axSpA, as well as increased patient participation in social and leisure activities.20,24,25 Improvements were seen as early as week 4 and were maintained throughout the placebo-controlled period and through to week 96. 20 However, the association between the achievement of higher clinical response thresholds or lower levels of disease activity and improvements in work and household productivity over long-term biologic treatment has not been previously evaluated.
Here, we present the results of a post hoc study examining the association between the achievement of higher thresholds of clinical response and lower levels of disease activity with improvements in workplace and household productivity in patients with axSpA over 4 years of CZP treatment in the phase III RAPID-axSpA trial. 26
Methods
Study design
RAPID-axSpA (NCT01087762) was a 204-week randomised, multi-centre phase III trial, double-blind and placebo-controlled to week 24, dose-blind to week 48 and open-label to week 204, which evaluated the efficacy and safety of CZP in adult patients with active axSpA. Patients had a clinical diagnosis of axSpA, either nr-axSpA or r-axSpA, and had inadequate response or intolerance to at least one non-steroidal anti-inflammatory drug (NSAID). Detailed inclusion and exclusion criteria were reported previously. 26
At baseline, patients were randomised 1:1:1 to placebo, subcutaneous CZP 200 mg every 2 weeks (Q2W) or subcutaneous CZP 400 mg every 4 weeks (Q4W) until week 24 (following subcutaneous CZP 400 mg at weeks 0, 2 and 4 as a loading dose). This post hoc study considers only those patients randomised to CZP at baseline, as they continued with their assigned dose throughout the trial, including the open-label period from weeks 48 to 204.
Outcomes
Trial outcomes reported through week 204 included the percentage of patients achieving Assessment of SpondyloArthritis International Society (ASAS) 20, ASAS40 and ASAS-partial remission (PR), as well as Ankylosing Spondylitis Disease Activity Score (ASDAS) disease activity states and ASDAS improvement thresholds.2,9 The ASAS20 response is defined as a relative improvement of at least 20% and absolute improvement of at least one unit in at least three of four domains. The ASAS40 response is defined as a relative improvement of at least 40% and an absolute improvement of at least two units in at least three of the four domains and no worsening at all in the remaining domain. ASAS-PR is a measure of clinical remission defined as a score of ⩽2 units in all four domains. 27
The ASDAS is a composite measure of disease activity in axSpA, comprised of objective and subjective elements, including total back pain, peripheral pain or swelling, duration of morning stiffness, the Patient’s Global Assessment of Disease Activity and CRP levels.28–31 ASDAS improvement level categories are ASDAS-Clinically Important Improvement (ASDAS-CII) and ASDAS-Major Improvement (MI). ASDAS-CII is defined by an ASDAS reduction of ⩾1.1 relative to baseline, and ASDAS-MI is an ASDAS reduction of ⩾2.0 relative to baseline. ASDAS-Inactive Disease (ID) is a measurement of clinical remission, and is defined as ASDAS < 1.3. 29 ASDAS-Low Disease Activity (LDA) is an acceptable goal of treatment in axSpA and is defined as ASDAS ⩾ 1.3 to <2.1. ASDAS-High Disease Activity (HDA) and ASDAS-Very High Disease Activity (VHDA) are defined as ASDAS ⩾ 2.1 to ⩽3.5 and ASDAS > 3.5, respectively.29,31
ASDAS and ASAS responses were calculated at 14 and 15 time points up to and including week 48, respectively, and every 12 weeks thereafter through study completion at week 204/early withdrawal. Trial outcomes were analysed as non-overlapping thresholds in the post hoc analysis; further details on these categories are detailed in section ‘Statistical analysis’.
The impact of axSpA on patients productivity was evaluated by assessing the cumulative number of days that arthritis affected workplace and household absenteeism and presenteeism, using the arthritis-specific Work Productivity Survey (WPS), which has been validated for use in an adult-onset axSpA population. 22 The WPS was self-reported but interviewer administered; questions from the WPS are detailed in Osterhaus et al. 32 The WPS was completed at the baseline visit and every subsequent 4 weeks until week 156, then every 12 weeks. The WPS considers the preceding 4 weeks before completion; workplace productivity questions were only applicable for those employed at the end of each 4-week period. All patients were eligible to answer questions about household productivity. As the WPS was administered every 12 weeks after week 156, the subsequent WPS assessments were given a weight of 3 to provide a balanced estimate across the cumulative period.
Statistical analysis
The clinical response of patients was assessed using ASAS and ASDAS criteria. In this post hoc analysis, patients were stratified into non-overlapping groups by achievement of different thresholds of clinical response at each time point: ASAS20 non-response, ASAS20–<40 (patients achieving ASAS20 response but not reaching ASAS40 response), and ASAS40; ASDAS non-response (<ASDAS-CII), ASDAS-CII–<ASDAS-MI (patients achieving ASDAS-CII response but not reaching ASDAS-MI response) and ASDAS-MI. Measures of clinical remission, ASAS-PR and ASDAS-ID, were also separately assessed, compared with ASAS-PR non-response and ASDAS-LDA, ASDAS-HDA and ASDAS-VHDA, respectively. For example, the ASAS20 non-response group includes only those who did not meet the ASAS20 response criteria, and those in the ASAS20–<40 group met the ASAS20 response criteria but did not meet the ASAS40 response criteria.
Data from the WPS responses were used to estimate the mean cumulative number of days affected by arthritis from baseline through to each time point using a weighted generalised estimating equations (GEE) model. The mean cumulative number of days affected by workplace absenteeism, workplace presenteeism, household absenteeism and household presenteeism was estimated using data from WPS questions 2, 3, 5 and 6, respectively.
The GEE model used a time point × (clinical threshold) outcome interaction to consider the association between the threshold of clinical response achieved (e.g. ASAS40) and productivity in patients with axSpA. The model included all observations for each patient at different time points, using a linear link function and an independent working correlation matrix. The model estimated the absenteeism or presenteeism for each clinical response or disease activity group separately; model results should be interpreted as the mean cumulative number of days of absenteeism or presenteeism for a theoretical patient population which had a constant threshold of clinical response across the 4-year study. The model did not adjust for confounding factors, such as gender, geographical region or type of work performed (e.g. manual versus non-manual). As the model was based on observed cases, the inverse probability of study continuation before week 204 was used as the weighting to adjust for patient dropout (missing data). Those patients with a low probability of remaining in the study at one visit were therefore more heavily weighted during the next visit, to account for any dropouts that did occur. Using a stabilised inverse probability weighting (IPW) ensured that the theoretical patient population considered in the model maintained the same size throughout the study. Due to the post hoc nature of this analysis, no p values were calculated.
The probability of each patient remaining in the study was calculated at each time point based on the time since the study started, geographic region, age at baseline, sex, prior TNFi use, employment status at last visit and mean ASDAS at the last visit (Supplemental Table 1).
Results
Patient disposition and baseline characteristics
At baseline, 218 patients with axSpA were randomised to receive CZP either 200 mg Q2W or 400 mg Q4W after loading. 26 Overall baseline demographics are shown in Table 1; patient disposition and discontinuation are shown in Supplemental Figure 1. Of the 218 patients who received CZP treatment from week 0, 203 (93.1%) patients completed the double-blind period to week 24, 191 (87.6%) patients completed the dose-blind period to week 48 and 142 (65.1%) completed the open-label period to week 204 (Supplemental Figure 1). Approximately 99% of patients remaining in the study completed the WPS at each time point (Supplemental Table 2).
Baseline patient demographics and disease characteristics.
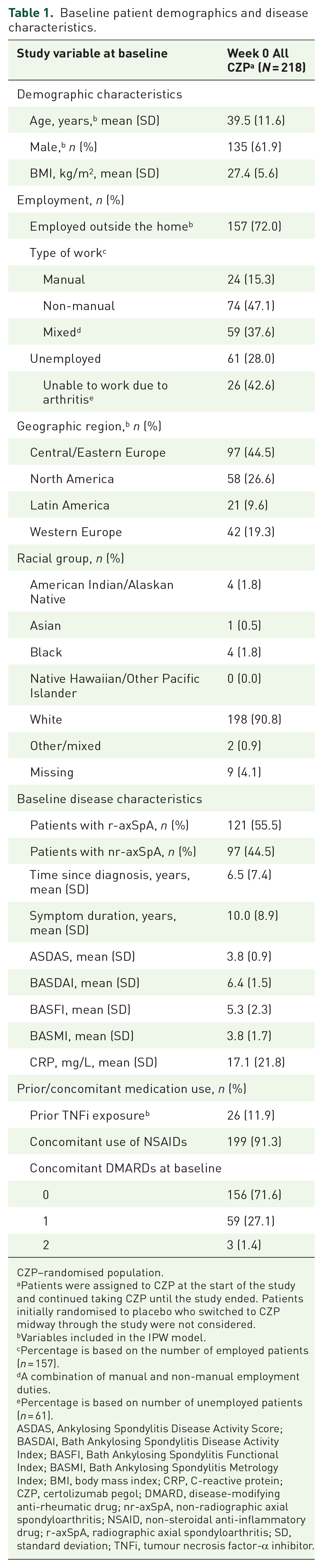
CZP–randomised population.
Patients were assigned to CZP at the start of the study and continued taking CZP until the study ended. Patients initially randomised to placebo who switched to CZP midway through the study were not considered.
Variables included in the IPW model.
Percentage is based on the number of employed patients (n = 157).
A combination of manual and non-manual employment duties.
Percentage is based on number of unemployed patients (n = 61).
ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; BMI, body mass index; CRP, C-reactive protein; CZP, certolizumab pegol; DMARD, disease-modifying anti-rheumatic drug; nr-axSpA, non-radiographic axial spondyloarthritis; NSAID, non-steroidal anti-inflammatory drug; r-axSpA, radiographic axial spondyloarthritis; SD, standard deviation; TNFi, tumour necrosis factor-α inhibitor.
Thresholds of clinical response and remission
The proportion of patients who achieved higher thresholds of clinical response increased over time during the study (ASAS40 week 24: 54.4%; week 204: 67.8%; ASDAS-MI week 24: 44.3%; week 204: 49.0%) and the percentage of non-responders decreased from week 24 through week 204 (Figure 1). The same trend was observed for achievement of ASAS-PR, while the proportions of patients achieving ASDAS-ID and ASDAS-LDA were sustained (Supplemental Figure 2). Disease activity states over time are depicted in Supplemental Figure 3.

ASAS and ASDAS outcome measures through week 204 (OC).
Workplace productivity
Patient’s workplace productivity appeared to improve over the course of the 4-year study (Supplemental Table 3). Achievement of higher thresholds of clinical response or of lower states of disease activity was associated with improved productivity, with fewer days of workplace absenteeism and presenteeism reported.
Achievement of higher thresholds of ASAS response was associated with numerically fewer cumulative absent workplace days as early as week 4 and were sustained through week 204 [ASAS40: 8.6 days (95% CI: 3.7, 13.5), ASAS20–<40: 29.4 days (95% CI: 8.3, 50.5); Figure 2(a)]. Fewer workplace days were affected by presenteeism in patients achieving higher clinical thresholds at week 4 and through week 204 [ASAS40: 26.3 days (95% CI: 15.7, 36.9), ASAS20–<40: 79.4 days (95% CI: 48.4, 110.4); Figure 2(b)]. Achievement of ASAS-PR was associated with fewer absent workplace days compared with non-achievement of ASAS-PR [4.0 days (95% CI: 0.2, 7.9) versus 32.0 days (95% CI: 18.8, 45.3), respectively] at week 204 (Supplemental Figure 4A).
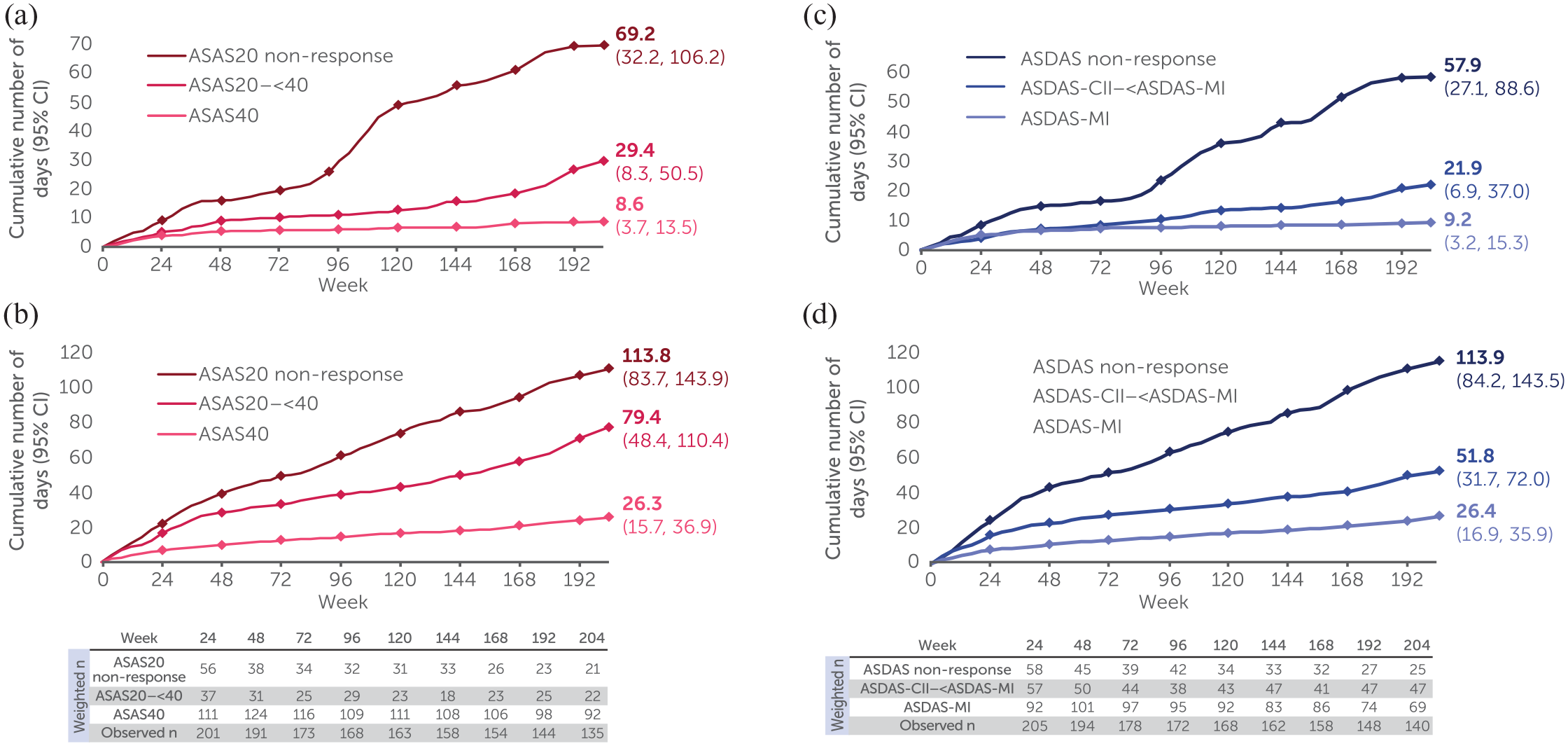
Workplace absenteeism and presenteeism by ASAS and ASDAS thresholds (OC).
Findings were consistent when using non-overlapping ASDAS improvement scores as measures of clinical response, where achievement of higher thresholds was associated with a numerically similar number of workplace days affected by absenteeism at week 4 and fewer days affected at week 204 [ASDAS-MI: 9.2 days (95% CI: 3.2, 15.3); ASDAS-CII–<ASDAS-MI: 21.9 days (95% CI: 6.9, 37.0); Figure 2(c)]. Fewer workplace days were affected by presenteeism in patients achieving ASDAS-MI versus ASDAS-CII–<ASDAS-MI at week 4 and were sustained through week 204 [ASDAS-MI: 26.4 days (95% CI: 16.9, 35.9); ASDAS-CII–<ASDAS-MI: 51.8 days (95% CI: 31.7, 72.0); Figure 2(d)].
Achievement of ASDAS-ID and ASDAS-LDA was associated with fewer numbers of absent workplace days compared with ASDAS-HDA and ASDAS-VHDA [ASDAS-ID: 5.0 days (95% CI: 0.1, 10.0), ASDAS-LDA: 15.6 days (95% CI: 3.7, 27.5); ASDAS-HDA: 32.7 days (95% CI: 18.6, 46.8); ASDAS-VHDA: 93.4 days (95% CI: 49.6, 137.2); Supplemental Figure 4C].
Household productivity
Patient’s household work productivity improved over the course of the 4-year study (Supplemental Table 3). Achievement of higher thresholds of clinical response using both ASAS and ASDAS was also associated with decreased impairment of productivity in household work, with fewer days affected by household absenteeism and presenteeism.
Achievement of ASAS40 response was associated with fewer days affected by household absenteeism and presenteeism from week 4 through week 204 [absenteeism: ASAS40: 21.7 days (95% CI: 14.4, 29.0), ASAS20–<40: 143.4 days (95% CI: 86.2, 200.5); presenteeism: ASAS40: 44.8 days (95% CI: 28.6, 61.1), ASAS20–<40: 179.4 days (95% CI: 115.8, 242.9); Figure 3(a) and (b)]. On the other hand, patients who achieved ASAS-PR had fewer absent household workplace days compared with those who did not achieve ASAS-PR [4.3 days (95% CI: 2.2, 6.5) versus 111.1 days (95% CI: 75.9, 146.4), respectively] at week 204 (Supplemental Figure 5A).

Household work absenteeism and presenteeism by ASAS and ASDAS thresholds (OC).
Findings were consistent when using ASDAS improvement scores; achievement of ASDAS-MI was associated with fewer household work days affected by absenteeism and presenteeism at week 4 and through week 204 [absenteeism: ASDAS-MI: 17.9 days (95% CI: 11.4, 24.3), ASDAS-CII–<ASDAS-MI: 69.5 days (95% CI: 44.2, 94.8); presenteeism: ASDAS-MI: 47.3 days (95% CI: 26.5, 68.1), ASDAS-CII–<ASDAS-MI: 92.2 days (95% CI: 63.1, 121.4); Figure 3(c) and (d)].
Achievement of ASDAS-ID through week 204 was associated with fewer absent household workplace days compared with ASDAS-LDA [ASDAS-ID: 8.2 days (95% CI: 4.1, 12.3), ASDAS-LDA: 30.7 days (95% CI: 18.0, 43.5)]. At week 204, patients with ASDAS-HDA and ASDAS-VHDA missed 121.1 cumulative workplace days (95% CI: 85.9, 156.4) and 321.2 cumulative workplace days (95% CI: 154.2, 488.1), respectively (Supplemental Figure 5C).
Discussion
This study demonstrates the association between the achievement of higher thresholds of treatment response and therefore lower levels of disease activity in axSpA with improvements in patient workplace and household productivity. Patients who reached the most stringent thresholds of clinical outcomes such as clinical remission, evaluated by ASAS-PR and ASDAS-ID, had the fewest cumulative number of days affected by absenteeism or presenteeism through week 204. Achievement of ASAS-PR was associated with the fewest days of affected productivity compared with all measures used in this study, and therefore could be a relevant goal in clinical practice. Furthermore, there were clear differences, as demonstrated by non-overlapping CI values, in the reduction of impairment in productivity achieved between the treatment-responsive patients who reached varying thresholds of clinical response (ASAS40 versus ASAS20–<40, ASDAS-MI versus ASDAS-CII–<ASDAS-MI). Although the difference in missed workplace days between patients achieving varying thresholds of clinical response is small at week 4 (ASAS40: 0.4 day versus ASAS20–<40: 0.9 day), this translates into a substantial difference in the long term (week 204; ASAS40: 8.6 days versus ASAS20–<40: 29.4 days). As achievement of ASDAS-ID may be a difficult treatment target to reach, ASDAS-LDA is considered as an alternative acceptable target for patients with axSpA. 33 This post hoc study highlights the benefits of aiming for the most stringent thresholds of clinical response (remission or LDA) in patients with axSpA, as these are associated with long-term patient productivity at work and at home.
To our knowledge, these are the first published data on the cumulative days of work and household productivity gained over 4 years of axSpA treatment with a biologic therapy. Although previous studies in axSpA have assessed the impact of the disease on patient productivity,34–36 this study quantified the long-term benefits in productivity associated with treatment. For example, the mean age of patients in this study was 39.5 (SD: 11.6) years. Assuming a retirement age of 65, these patients would typically have around 25 years of productive work left. Assessing work productivity in this young, working-age population demonstrated the benefits of reaching these stringent thresholds of clinical remission to help guide future treatment strategies.
Although the impact of existing structural damage in the sacroiliac joints on productivity was not evaluated in this study, this has previously been reported to have little impact on QoL and productivity.6,37 Of the patient population in the RAPID-axSpA study, 55.5% had r-axSpA, indicating a balanced patient population between r-axSpA and nr-axSpA. On average, patients with r-axSpA experience greater spinal inflammation, structural damage and functional disability than those with nr-axSpA. 3 However, the burden of disease on QoL and productivity is similar between r-axSpA and nr-axSpA patients, with both patient populations experiencing similar rates of absenteeism, presenteeism and work productivity.37,38 Moreover, the clinical response rates upon CZP treatment in the RAPID-axSpA study were similar between r-axSpA and nr-axSpA patients. Therefore, it is expected that radiographic damage in sacroiliac joints would have little effect on productivity. The results presented here consider a balanced population and can therefore be considered in treatment strategies for patients with r-axSpA and nr-axSpA.
The association between the achievement of treatment targets and improved workplace and household productivity was assessed using a validated, arthritis-specific WPS and examined using a GEE model. Using a stabilised IPW ensured that both the theoretical patient population maintained the same size throughout the study, and that patients who discontinued the study (who were less likely to achieve thresholds of clinical responses and more likely to have reduced productivity) were still considered within the subsequent weeks of the study to prevent skewing of the data towards patients who remained in the study.
One limitation of the statistical analysis used in this study was the assumption that a patient experienced a constant disease state in the RAPID-axSpA trial through week 204. In reality, the treatment response of a patient can fluctuate while on treatment so the number of days of improvement seen between thresholds may not be as high. Supplemental Figure 3 shows the variety of disease states a patient experienced over the course of treatment. Understanding that individual patients’ responses to treatment can vary supports the use of a treat-to-target approach to aim for achievement of the highest thresholds of response (clinical remission or LDA), to further improve patient participation and productivity in workplace and household activities.39,40
It is important to note that not all the questions in the WPS were used in the statistical analysis; for example, patient employment status was not assessed. The work productivity analysis focused on patients who declare that they are working; future studies could evaluate the impact of disease activity on the ability to be employed, as well as compare those in manual job with those in non-manual jobs. Furthermore, future studies could record additional aspects of patient lifestyle not captured by the WPS, such as improvements in working conditions, to further assess the relationship between achievement of thresholds of clinical response and improvements in productivity.
This study improves the understanding of how treatment strategies can improve productivity; the clinical benefit has been demonstrated, and future work should seek to understand the economic impact. Economic modelling of axSpA has thus far focused on the indirect costs of reduced patient productivity in the workplace.19,41,42 A study conducted across three European countries estimated the mean annual societal direct costs for each patient were EUR 2640, with worse physical function and higher disease activity as important determinants of cost. 43 More specifically, a study carried out in the United Kingdom estimated the annual cost of workplace absenteeism and presenteeism per patient with axSpA was GBP 411 and GBP 3425, respectively. 44 This study has demonstrated that achievement of clinical thresholds of response and LDA using pharmacological therapy for axSpA is associated with long-term improvements in workplace and household productivity. Future modelling should aim to develop an understanding of the indirect economic gain of improved productivity in this patient population, due to the achievement and maintenance of high thresholds of clinical response with long-term therapy.
Conclusion
Over 4 years of CZP treatment in patients with axSpA, achievement of higher thresholds of clinical response, such as remission and LDA, was associated with a reduced burden on workplace and household productivity. The relationship between lower levels of disease activity and higher thresholds of clinical response and a reduction in impairment in productivity indicates the importance of targeting these thresholds to limit the burden of axSpA on patients’ daily lives.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X231189079 – Supplemental material for Achievement of higher thresholds of clinical responses and lower levels of disease activity is associated with improvements in workplace and household productivity in patients with axial spondyloarthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X231189079 for Achievement of higher thresholds of clinical responses and lower levels of disease activity is associated with improvements in workplace and household productivity in patients with axial spondyloarthritis by Martin Rudwaleit, Pedro M. Machado, Vanessa Taieb, Natasha de Peyrecave, Bengt Hoepken and Lianne S. Gensler in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors thank the patients, the investi-gators and their teams who took part in this study. The authors also acknowledge Simone E. Auteri, MSc EMS, PhD, UCB Pharma, Italy, for publication coordination and editorial assistance and Laura Mawdsley, MSc, Sona Popat, BA, and Ben McNally, PhD, from Costello Medical, UK, for medical writing and editorial assistance based on the authors’ input and direction. The authors also thank Tommi Nurminen for contributing to the development of the analyses, and David Deerling, Domenico Magazzu and Otis Rimmer for contributing to the programming of these analyses.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
