Abstract
Hidradenitis suppurativa is a chronic inflammatory disease of the skin with a suppurative-cicatricial outcome affecting the infundibular component of the pilo-sebaceous unit. The lesions are typically localized in the intertriginous and apocrine gland-rich areas. Hidradenitis suppurativa mainly affects patients at a young age and is very often refractory to conventional medical treatment. During pregnancy, the management of hidradenitis suppurativa becomes demanding due to the need of safety therapies. Certolizumab pegol is a pegylated monoclonal TNF-α inhibitor that lacks the fragment crystallizable region preventing active placental transfer and could be used in pregnancy and lactation if clinically needed and is label for chronic plaque psoriasis, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease. Herein, we describe a case of a 32-year-old pregnant woman with Hurley stage III hidradenitis suppurativa successfully treated with certolizumab pegol.
Introduction
Hidradenitis suppurativa (HS) is a chronic inflammatory disorder that is clinically characterized by deep-seated nodules, abscesses, fistulae, sinus tracts and scars in the intertriginous and apocrine gland-rich areas including axillary, inguinal and submammary folds. 1 A recent meta-analysis estimated an overall prevalence of HS 0.4% in the general population. The highest incidence is between 18 and 40 years, with a prevalence in women two times higher than in men.2,3
The clinical course of HS during pregnancy is unpredictable, with the majority of patients experiencing a disease worsening and consequently a major impact on quality of life. 4
The pharmacological treatment of HS during pregnancy could be challenging because some treatments are contraindicated such as retinoids because of well-known teratogenicity, others are better avoided including tetracycline and rifampicin that are classified by Food and Drug Administration as categories D and C, respectively, whereas efficacy and safety data in pregnancy are lacking for other therapies such as adalimumab. 5 In addition, the current European Guidelines does not provide recommendations for the treatment of HS in pregnancy; hence, there is a high need of data for HS treatments in pregnancy. 6
Certolizumab pegol is a pegylated monoclonal TNF-α inhibitor that lacks the fragment crystallizable region preventing active placental transfer and could be used in pregnancy and lactation if clinically needed and is label for chronic plaque psoriasis, rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease. 7
Here, we report a 32-year-old woman with severe worsening of HS during pregnancy successfully treated with certolizumab pegol.
Case report
A 32-year-old woman was referred to the outpatient clinic of the University Hospital of Verona because of severe worsening of HS occurring during the 20-week of pregnancy. Physical examination revealed multiple nodules and painful abscesses draining purulent material affecting the axillae, submammary, inguinal, and perianal area (Figure 1(a) and (b)) corresponding to Hurley stage III and physician global assessment of 4 out of 5. In addition, the numerical rating scale for pain (pain NRS) was 9 out of 10 and the Dermatology Life and Quality Index (DLQI) was 28 out of 30. After consultation with the gynecologist, the patient was first treated with oral clindamycin 300 mg twice a day for 4 weeks without achieving any clinically relevant benefit. Consequently, treatment with certolizumab pegol was initiated with the dose approved for chronic plaque psoriasis, that is, 400 mg subcutaneous at 0, 2 e 4 weeks and 200 mg every 2 weeks thereafter. At week 8, a meaningful improvement of axillary and inguinal lesions was appreciated (physician global assessment = 3) as well as a reduction in pain (NRS = 5 out of 10) and improvement in quality of life (DLQI = 8 out of 30) (Figure 2(a) and (b)). Certolizumab was administered during all the pregnancy and continued during lactation and thereafter with a follow-up of 2 years. During the last weeks of the pregnancy, the patient developed gestational diabetes and was treated with insulin. At 37 weeks of gestation, after premature rupture of the membranes, she gave birth without any complications. No episodes of infections were reported during pregnancy and lactation. The patient is a nonsmoker, has a body mass index of 26 kg/m2, and has no comorbidities, including polycystic ovary syndrome, associated with HS. Before becoming pregnant, the patient was treated with adalimumab 40 mg per week for approximately 2 years with partial clinical benefit. When the pregnancy became known, this treatment was suspended as a precaution. The off-label use of certolizumab pegol for HS was approved by the Ethical Committee of Verona.
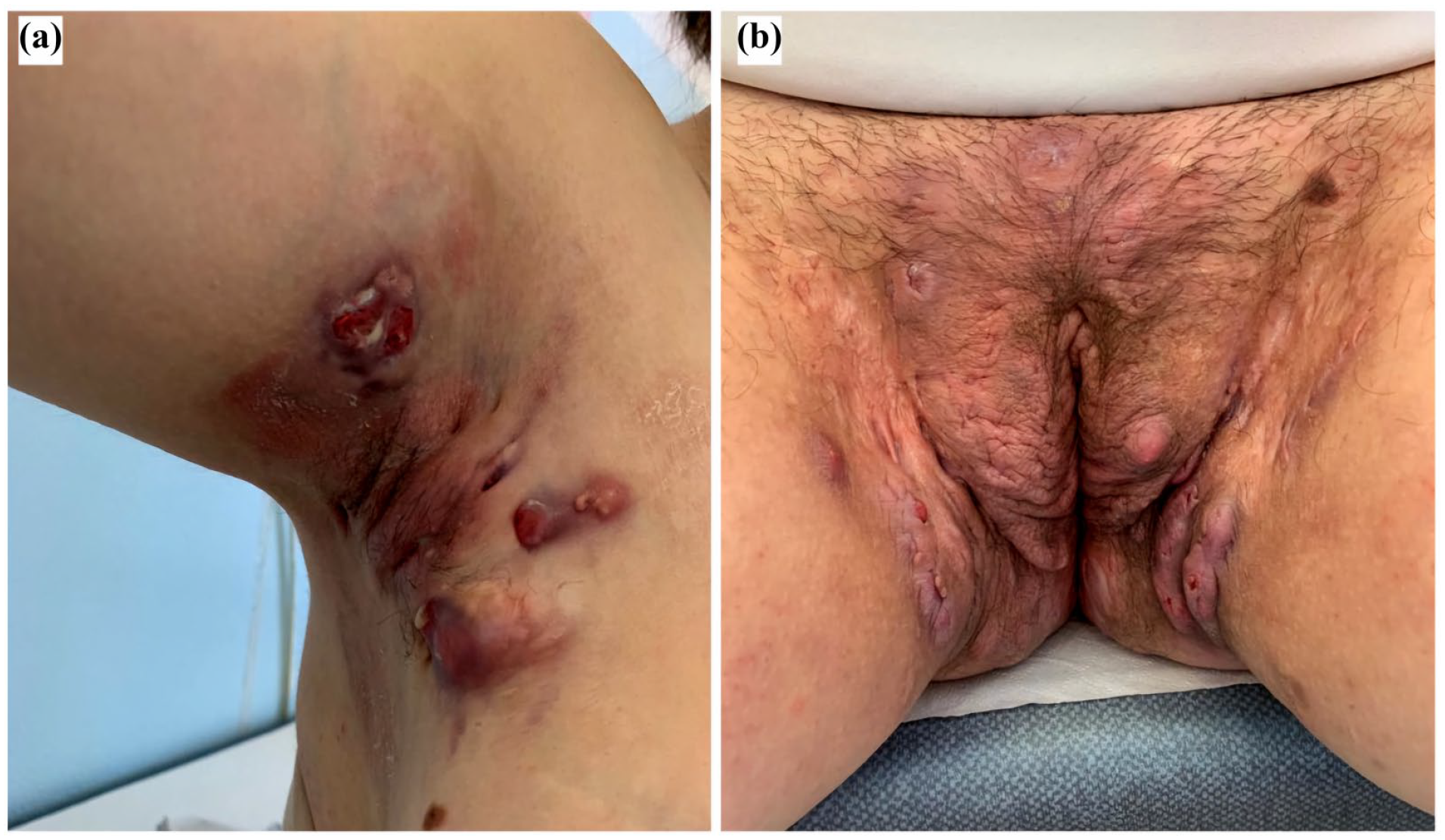
(a, b) At baseline, multiple exudating nodules, abscesses, and retracting scars in the right axilla and groin.

(a, b) After 6 weeks of therapy with certolizumab pegol, significant improvement of lesions: absence of nodules, and persistence of residual sinus tracts and retracting scars.
Discussion
The main finding of this case is that certolizumab pegol was effective and safe in a young patient with severe worsening of HS during pregnancy, after failure of oral antibiotic therapy with clindamycin. Since the therapy with certolizumab was effective during pregnancy and breastfeeding, it was continued during the subsequent period with a 2-year follow-up.
Certolizumab pegol is a pegylated monoclonal TNF-α inhibitor that lacks the fragment crystallizable region preventing active placental transfer and could be used in pregnancy and lactation if clinically needed.7,8 Our finding is consistent with other reports in the literature.9,10
Repetto et al. 9 described two cases in which certolizumab 200 mg was administered every 2 weeks with clinically significant improvement after 3 months of treatment. Wohlmuth-Wieser et al. 10 reported that certolizumab was administered at an initial dose of 400 mg every 2 weeks, but since the disease control was insufficient, the dose was therefore increased to 400 mg subcutaneous weekly at 32 weeks of gestation successfully. After delivery, they opted to switch her back to adalimumab.
The course of HS in pregnancy is unpredictable, with exacerbations in 60%–80% of cases according to literature data.11,12 Increased estrogen, insulin resistance, and free testosterone levels may induce a pro-inflammatory effect. In addition, pregnancy weight gain increases skin maceration at intertriginous sites and promotes TNF alpha secretion, leading to increased inflammation and exacerbation of HS. 12 Moreover, breastfeeding may be impaired in case of HS affecting the apocrine glands of the mammary.4,5,12 Certolizumab is a safe drug for the fetus during pregnancy because it does not cross the placenta, but it may be associated with an increased risk of infections in the mother. 13 Furthermore, therapy with TNF inhibitors, including certolizumab, can cause weight gain. 14
Specific guidelines/recommendations for the treatment of HS in pregnancy are needed. Further studies could confirm the efficacy and safety of certolizumab in HS during pregnancy.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paolo Gisondi has been consultant in advisory board and/or served as a speaker for AbbVie, Biogen, Almirall, Eli Lilly, Janssen, LEO Pharma, Novartis, Pfizer, Pierre Fabre, Sanofi, and UCB Pharma; Giampiero Girolomoni has received personal fees from AbbVie, Almirall, Amgen, Boehringer-Ingelheim, Bristol-Myers Squibb, Eli-Lilly, Leo Pharma, Merck Serono, Novartis, Pfizer, Pierre Fabre, Samsung bioepis and Sanofi; Francesco Bellinato, Elena Montalto, Tea Curic, Elisabetta Danese none declared.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the European Union—Next Generation EU—NRRP M6C2—Investment 2.1 Enhancement and strengthening of biomedical research in the NHS.
Patient consent
The patient gave permission for the clinical case to be written up and for the photographs to be published. Data are available upon request to corresponding author.
