Abstract
Background:
Axial spondyloarthritis (axSpA) is a chronic inflammatory disease which primarily affects the axial skeleton resulting in chronic back pain and stiffness. According to the guideline, the first-line treatment includes non-steroidal anti-inflammatory drugs (NSAIDs), conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) and non-pharmacological treatment. Second line treatment involves biological disease-modifying antirheumatic drugs (bDMARDs) such as tumour necrosis factor and interleukin-17 inhibitors.
Objectives:
The aim of this social media listening research project was to analyse switches of medication and the reasons thereof to gain valuable insights into real-life journeys of patients suffering from axSpA.
Methods:
Publicly available posts in German-speaking disease-specific forums were scanned for disease-specific keywords and commonly used drugs by axSpA patients on the Permea platform. Posts containing at least two key words were selected and switches between medications were manually labelled. A total of 287 scraped posts between 01 July 2010 and 04 Feb 2022 were analysed.
Results:
The largest group of described medication switches was initially using bDMARDs. Switches to a different bDMARD, termination of medication and switches to glucocorticoids were most frequently named. Patients on NSAIDs switched to glucocorticoids, a different NSAID or bDMARD, whereas patients on csDMARDs most frequently changed to bDMARDs. In all medication groups the main reason for switching was insufficient efficacy and side effects. Additionally, for the medication groups bDMARDs, csDMARDs and corticosteroids, pregnancy and lactation were given as a reason for switching, whereas patients in the NSAID group never mentioned pregnancy and breastfeeding as a reason for switching treatment.
Conclusion:
Our analysis shows medication switches based on real-life patient experiences shared with peers in a social listening setting. We also show medication switches differing from advised guidelines. Gathering real-life insights into patients’ journey dealing with chronic diseases allows us to understand, and thereby improve patient care and treatment.
Introduction
Axial spondyloarthritis (axSpA) is an immune-mediated inflammatory disease primarily affecting axial skeleton – sacroiliac joints and spine. Depending on the absence or presence of structural damage visible on conventional radiography of sacroiliac joints (definite radiographic sacroiliitis), axSpA is subdivided into non-radiographic and radiographic axSpA, the latter also referred to as ankylosing spondyloarthritis (AS). AxSpA is often called also Morbus Bechterew in German-speaking regions. Chronic or recurrent inflammation in the spine can lead in severe cases to complete stiffening of the spine. Onset usually occurs around the age of 30, but can appear roughly 5 years earlier in HLA-B27-positive (Human Leukocyte Antigen-B27-positive) patients than in HLA-B27-negative patients.1,2 The ratio of males to females with AS is about 2–3:1, whereas in non-radiographic spondyloarthritis, the gender distribution seems to be more balanced. Additionally, further differences in terms of disease progression and manifestation have been observed between sexes. 3
Clinically, axSpA manifests as chronic back pain and stiffness found mainly in the lower back, although any part of the spine can be affected. Patients might also suffer from morning stiffness in the spine, which usually improves with exercise and sport but not with rest. 4 Peripheral manifestations such as arthritis, enthesitis, dactylitis occur in 30–50% of patients during the course of the disease. 4 Uveitis, inflammatory bowel disease and psoriasis represent the typical extra-musculoskeletal manifestations of axSpA. 5
The clinical management of axSpA is based on recommendations made by the Assessment of Spondyloarthritis international Society and European Alliance of Associations for Rheumatology. 6 Non-pharmacological treatments such as physiotherapy, exercise, rehabilitation, education and support groups play an important role and should consistently be integrated into a patient’s lifestyle. Independent of the predominant manifestation, axial or peripheral, the use of non-steroidal anti-inflammatory drugs (NSAIDs) as first-line therapy is recommended. In addition to this, local steroids and conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) such as sulfasalazine can be used for predominantly peripheral manifestations. In terms of second-line therapy, tumour necrosis factor (TNF) inhibitors and interleukin-17 inhibitors (IL-17 inhibitors) as well as recently approved Janus kinase inhibitors (JAK inhibitors) can be used. As additional therapy or in special clinical situations, the use of analgesics – and, in some cases, surgery – might also be considered.
Evidence suggests that lower levels of adherence to a medication regime is associated with substantial morbidity, mortality, and economic costs across a number of chronic and infectious diseases.7–10 The replacement of one medication for another can take place for a variety of different reasons, including but not limited to adverse side effects, lack or loss of efficacy, pregnancy and breastfeeding period.
Up to date, there is only limited information available regarding the ‘switch behavior’ of patients when it comes to the field of axSpA. Several publications describe the switches within a medication class itself (i.e. a different TNF inhibitors 11 ). However, there is currently a lack of information on switches between the medication groups and the reasons behind these decisions.
In the present work, we analyse experiences and voices of people dealing with axSpA posted on social media platforms where they seek advice and exchange information with peers regarding their diagnosis and treatment. We focused on German language only, as there is at the time of writing no data on axSpA-specific social listening analyses of a local non-English European market so far. Within the confines of a social listening setting, we anonymously evaluate the switches between medication classes and the reasons for these changes using our General Data protection Regulation (GDPR)-compliant data analytics platform Permea. This approach not only allows us to capture patient responses outside of the strict framework of clinical trials and medical reports, but – more importantly – provides insights into the real-life journeys and experiences of people affected by axSpA. Our findings will contribute to a broader understanding of how people cope with axSpA and help to improve future treatment.
Methods
Data source
The acquisition of public posts that are relevant for our analysis was performed in two steps. First, we ran a query with disease-specific keywords (Supplemental 1) on Permea platform, that continuously aggregates posts from 1304 sources like forums or twitter in a legally compliant way. The query also included all published posts of two disease-specific forums (Supplemental 1). To select relevant posts from the hereby scraped collection of 19,973 posts, a list of key words describing common drugs used by AS patients such as active ingredients, generic names, brand names and abbreviations was manually curated and compiled (Supplemental 2). Posts containing at least two keywords were selected. Subsequently, all posts were extracted whose length was within the 25th and 75th percentiles of the length of all posts. This resulted in a data set of 1030 posts for further analysis.
The scraped set of posts was manually evaluated by three human raters. Each rater independently curated, after a short introduction to the task, 342 to 346 posts. The web-based open-source tool Label Studio (version 1.0.1), normally used to label training data for machine learning, was set up with a predefined set of medication and possible reasons to ease the curation workflow. A rater could select the medication and reason(s) for switches based on user-defined information in each post. If a post did not contain information about medication switches, it was excluded from further analysis. If the reason for transition is unclear, then the ‘unknown’ category was selected. In case a post specified multiple switches and corresponding reasons, three most recent transitions were individually documented.
A total of 287 scraped posts between 01 July 2010 and 04 Feb 2022 were included in our analysis. A list of the forums where all posts included in the further analysis originate from can be found in Supplemental 3.
Processed data was used to calculate the percentage of posts with similar reason mentions. Similar active ingredient transitions were aggregated (Table 1), and the relative frequency of each reported problem or reason was calculated.
Medication groups based on active ingredients.

bDMARD, biological disease modifying antirheumatic drug; csDMARD, conventional synthetic disease modifying antirheumatic drug; NSAID, non-steroidal anti-inflammatory drug.
Software and tools
Post texts from publicly available forums were scraped using Brandwatch. The obtained data was then ingested into the Permea platform and processed in GDPR-compliant manner. Further processing of the dataset was performed using Python (version 3.9.6). The manually labelling of the posts was performed using the web-based open-source tool Label Studio (version 1.0.1). For further analysis of the data, we used Python (version 3.9.6). Statistical analysis was performed using Prism 9 (version 9.4.0). Figures were designed using R Studio (version 2022.02.3), the online open-source tool SankeyMATIC and Prism 9 (version 9.4.0).
Statistical analysis
A Fisher’s exact test was performed to detect significant differences in the relative frequencies of medication switches and in reasons for medication switches. A p-value less than 0.05 was considered to be significant.
Results
Our analysis was divided into two parts. First, we examined which medication class changes the patients described in their posts. Here we examined the individual changes as well as the different steps in medication changes. In the second part, we examined the reasons given for the changes.
Medication flow in patients
First, we analysed the total of 347 medication switches changes described in the posts. We found that the largest group of drugs from which the switching originated were biological disease modifying antirheumatic drugs (bDMARDs), with a total of 116 (33.43%) switches described. The largest proportion of changes described here occurred within the same group with 76 (65.52%) changes to another bDMARD, followed by 15 (12.93%) descriptions of a discontinuation of any medication, 10 (8.62%) to glucocorticoids and 7 (6.03%) switches to csDMARDs (Figure 1).

Shows the reported switches between different medication groups and switches from or to unknown medication and no medication. The arrows show the direction of the medication switches.
The second largest initial medication group was formed by NSAIDs (n = 98, 28.24%). The most common switch described by patients was to glucocorticoids (n = 31, 31.63%), followed by a change within the NSAID group (n = 26, 26.53%) and bDMARDs (n = 21, 21.43%).
In the csDMARD group (54 of 347 changes, 15.56%), the most frequent change was to the bDMARDs group (n = 36, 66. 67%). In addition, we also looked at switching between different groups of bDMARDs (data not shown). We could distinguish between IL-17 and TNF inhibitors. All described therapy changes to an IL-17 inhibitor (n = 11) originated from a TNF inhibitor. A switch from an IL-17 inhibitor to a TNF inhibitor was described in only two cases. Of all switches within the class of biologics (n = 181), 22% switched to another TNF inhibitor. Details about switches between different groups can be found in Supplemental 4. Details about switching between and from biologics can be found in Supplemental 6.
In the following, we focused our analysis on different steps of medication switches. Here we analysed in which order the patients described several switches and focused the analysis on the first and second switch of medication. We therefore analysed a total of 294 medication changes that were classified as the first or second switch of the patient. Figure 2 shows that 213 (72.45%) of the 294 changes were assigned to a first switch and 81 (27.55%) of the changes were attributed to a second switch. In the bDMARDs group, both the first and second reported switches were found to occur more often within the same medication class than to another group (e.g. bDMARDs to bDMARDs versus bDMARDs to no medication: 36 (60.00%) versus 10 (16.67%) of 60 switches at the first switch). No second switches were described within the analgesics, NSAID and csDMARD group. Details about all switches can be found in Supplemental 5.
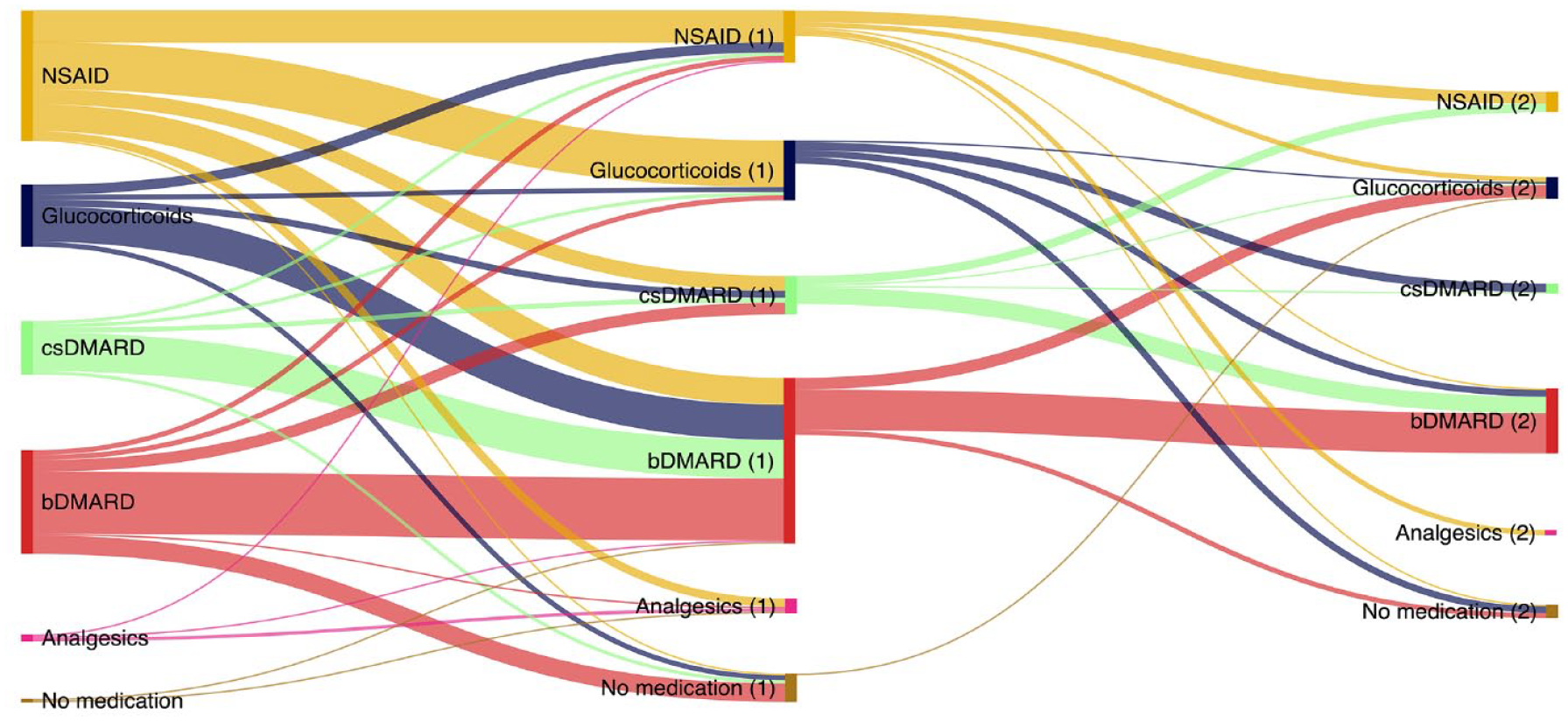
Shows the described medication switches divided in ‘First switches’ and ‘Second switches’. (1) Indicates a target of a First switch and a source of a Second switch and (2) indicates a target of a Second switch.
Accordingly, patients who started the first switch with a corticoid mostly switched to a bDMARD (e.g. glucocorticoids to bDMARDs (1) versus to NSAID (1): 21 versus 6 of 37). In the csDMARD group, this was also observed for both the first switch and the second switch (e.g. csDMARD to bDMARDs (1) versus to csDMARD (1): 23 versus 3 of 32). In the second step of switching csDMARDs, only a small difference was seen between switching to a bDMARD or a NSAID (csDMARD to bDMARD versus to NSAID, 10 versus 5 of 17).
In the initial NSAID group, the largest proportion of patients switched to the corticoid drug group in the first switch (n = 28, 35.90%). This switch was more frequent than to bDMARDs, csDMARDs and analgesics (e.g. from NSAID to bDMARDs versus to corticoid: 16 versus 28 of 78 switches). In the second switch, switching within NSAIDs was more frequent than switching to a bDMARD (7 versus 1 of 15 switches). No switch to a csDMARD was described.
Reasons for switches in medication
In the second part of our analysis, we examined the mentioned reasons for a switching from one to another medication group. Figure 3 shows the relative frequency of the reasons mentioned for switching in relation to the medication groups.

Shows the relative frequency of reasons for changes in medication based on the absolute number of mentioned reasons in one medication group. The medication groups show the medication that was switched away from.
The most frequently mentioned reason for a change of medication in the analgesics group was insufficient efficacy (55.6%, n = 5 out of 9), followed by side effects (33.3%, n = 3). For bDMARDs, insufficient efficacy was also the most frequently cited reason with 31.6% (n = 60 out of 190). The second most common reason for switching was side effects with 51 out of 190 mentions (26.8%).
In the csDMARD group, a total of 83 reasons for drug changes were identified in the posts. Here, insufficient efficacy (n = 33, 39.8%) and side effects (n = 19, 22.9%) were the most frequent reasons.
In the case of NSAIDs (non-steroidal anti-inflammatory drugs), insufficient efficacy (n = 66 of 147, 44.9%) and the side effects associated with taking them (n = 37, 25.2%) were also the most important reasons for a change in medication, in line with all the groups mentioned.
We also looked more closely at the subgroups bDMARDs and csDMARDs (see Figure 4(a) or (b)). We found that in case of bDMARDs (Figure 4(a)), side effects and lack of efficacy not only had the highest number of mentions in absolute terms but were also mentioned more often than all other reasons (e.g. side effects versus other disease: 51 versus 19 mentions). In addition, another disease (n = 19 of 190) was more often the reason for a change than a worsening of the underlying disease (n = 7), pregnancy or breastfeeding (n = 6), an intolerance (n = 6) or the way of drug application (n = 5). In 21 cases (18.42%), no reason was given for a switch.
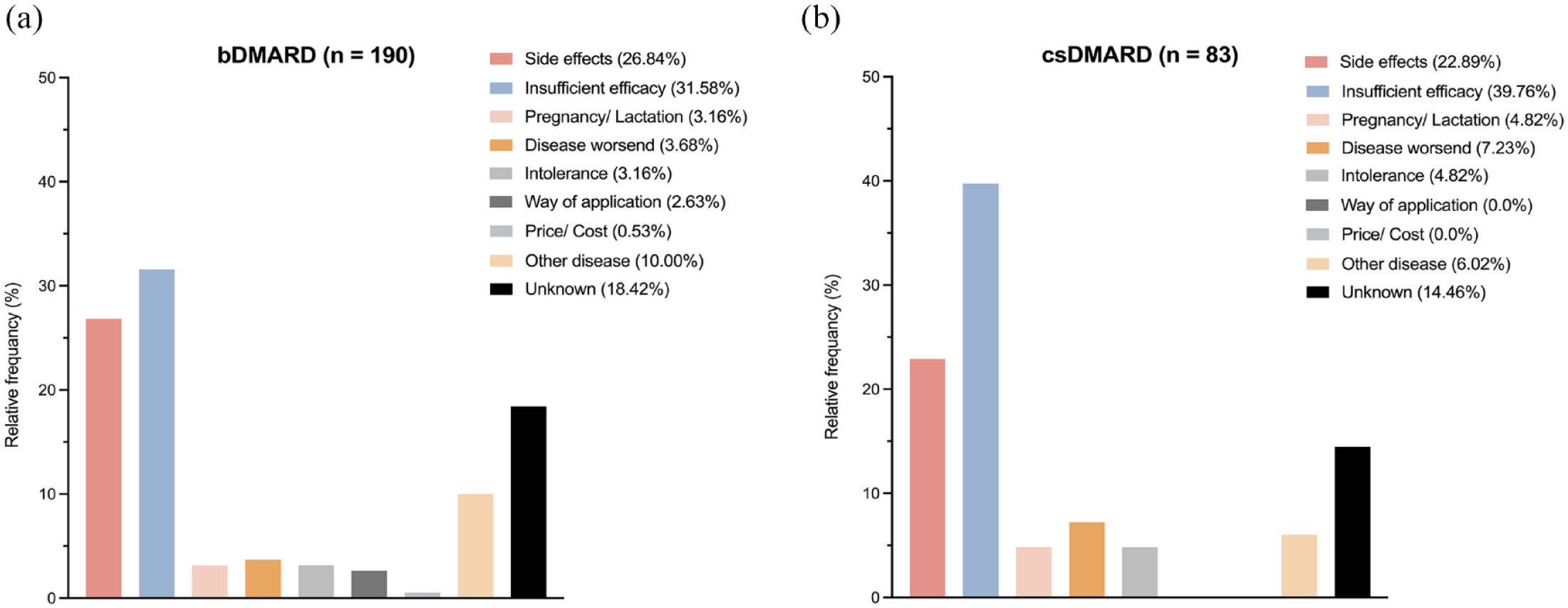
Shows the relative frequencies of mentioned reasons for changes in medication for the medication groups (a) bDMARDs and (b) csDMARDs. The relative frequency is based on the absolute number of mentioned reasons in the specific medication group.
In the group of csDMARDs (see Figure 4(b)), insufficient efficacy was also mentioned significantly more often as a reason for switching than all other reasons, and in particular also significantly more often than side effects (33 mentions versus 19 mentions). However, side effects (n = 19) were mentioned more often as a reason than other reasons, such as disease exacerbation (n = 6), pregnancy/breastfeeding (n = 4), intolerance (n = 4) or other diseases (n = 5). In the csDMARD group, however, no reason for switching was given in 11 cases (14.46%).
Discussion
The aim of this work was to examine medication changes of people suffering from axSpA by performing a social listening analysis. By taking a closer look at the real-life reasons for these changes we gain valuable insights into therapy regimen according to the patients themselves outside of the strict framework of clinical trials.
First, we looked at the medication transitions. The largest group of the described changes started with the medication group bDMARDs (116 of 347, 33.34%). Although most of the changes occurred within this group (76, 65.52%), that is, from one dDMARD to another dDMARD. Interestingly, the termination of any medication was the second largest part (n = 15, 12.93%), although this chronic disease requires lifelong therapy. Only as the third most frequent change target were the glucocorticoids described.
The group of patients using bDMARDs represents the largest proportion of described switches in our analysis which may be related to the severity of disease that is causal for treatment with this medication group. According to the treatment guidelines, bDMARDs are recommended as a second line treatment after NSAIDs failure. If there is an insufficient response or side effects after a minimum therapy period of 12 weeks, a change within the group of bDMARDs is recommended. 6 A higher severity of illness could be associated with a greater need for emotional support, which could be gained by sharing one’s own medical history and experiences with medication and the resulting reaction or support from others affected. For this reason, this group of patients could be particularly active in forums, which could explain why they represent the largest share in our analysis.
Switch between bDMARDs is common in axSpA. 11 The fact that most of the described switching takes place within the bDMARD group seems to be explained by the treatment recommendations and is also confirmed by our analysis, in which a large proportion of bDMARD users switched once or even twice to another bDMARD (data not shown). Until recently, the only biological agent to treat axSpA were TNF-inhibitors. 12 The recent introduction of the first IL-17 inhibitors have extended treatment options for patients who did not respond to TNF inhibition or experienced secondary failure. 13 The availability of treatment options may contribute to the preference of switching. Our analysis also showed that around 22% switched between TNF inhibitors, but only few switched from TNF to IL-17 inhibitors. This is likely to change with the increasing treatment options in the future. The posts analysed did not contain any reports on the use of targeted synthetic disease modifying antirheumatic drugs, such as JAK inhibitors, which also belong to a separate class in the treatment of axSpA. However, the proportion of patients in the bDMARDs group who describe a discontinuation of medication is particularly noteworthy and somewhat concerning. For the population switching to no medication after one or two different bDMARDs this could be due to a certain resignation or the lack of treatment options, if all recommended treatment options had already been tried, or the side effects became unbearable.
In the second largest group NSAID, the second largest proportion of switches occurred to corticosteroids. Here it does not appear to make sense that a complete change to an oral corticosteroid is involved; it is much more likely that this is an extension of the therapy regimen to include local injections with corticosteroids, which, however, may have been described inaccurately by the patients. It may also be possible that the patients stopped taking NSAIDs because they either used them only as an on-demand medication and no longer suffered from relevant symptoms or developed side effects by taking NSAID over a longer period of time. A complete change of therapy to an oral corticosteroid would not comply with the current therapy guidelines.
This part of our analysis already shows how important it is to understand the individual reasons for switching medication, especially the reasons why patients chose to discontinue treatment or why treatment guidelines recommendations were deviated from when treatment escalation was necessary.
To address this question, we analysed the main reasons patients reported for their medication switches.
In all medication groups, the main reasons for switching were insufficient efficacy and side effects. In the subgroups bDMARDs and csDMARDs were given significantly more often than any other reason (see Figure 4(a) or (b)).
Interestingly, only in the medication groups bDMARDs, csDMARDs and corticosteroids pregnancy and lactation were given as a reason for switching. In the NSAID group, pregnancy and breastfeeding were not once given as a reason for switching medicines. However, it is widely known that NSAIDs should especially be avoided taking in the third trimester of pregnancy to avoid negative effects for the unborn, such as premature obstruction of the foetal ductus arteriosus, oligohydramnios and renal injury. 14 In addition, studies have shown that the disease flares up in 25–80% of cases during pregnancy and in 30–100% of cases in the postpartum period. 15 This raises the question whether the patient refrained from taking medication during pregnancy and simply did not report this in the forums, or whether the medication was changed due to a worsening of the disease or a relapse and this is considered to be insufficient efficacy of the NSAIDs.
In the csDMARD group, pregnancy and lactation were cited as the reasons for a change in medication in 4.8% (n = 4) of cases. In the therapeutic guidelines, sulfasalazine in particular is recommended as the drug of choice when using csDMARDs. 6 Sulfasalazine is considered safe to use during pregnancy and breastfeeding, so that a change would not be necessary if patients were treated in accordance with the guidelines. 16 However, the number of mentions could be due to the use of methotrexate as a csDMARD, which is contraindicated during pregnancy and breastfeeding. 16 In addition, the use of methotrexate should be fundamentally questioned here, as the evidence on the benefit of methotrexate in the treatment of AS is questionable. 17 Nevertheless, the fears of pregnant women rather than evidence-based medical reasons could be the reason for a change of medication.
In the group of bDMARDs, pregnancy and breastfeeding is only mentioned as a reason for a change of medication in approximately 3.2% (n = 6) of cases. In particular, TNF-alpha inhibitors are considered safe for use during pregnancy and breastfeeding. Studies have shown that the outcome of pregnancy with TNF-alpha inhibitors does not differ from a pregnancy with no TNF-alpha inhibitors.18–21 However, studies show that infliximab and adalimumab in particular enter the foetal system at around 16 weeks of pregnancy and are detectable several weeks after delivery. 22 Although there is no evidence for a negative impact of these TNF-alpha inhibitors on pregnancy or foetus, due to safety reasons there might have been a wish for a change in medication.
Our analysis shows that by observing real-world patient experiences in relation to information that is partially hidden or not fully presented in a rigorous clinical trial setting, we can gain valuable insights into the reasons for switching medications or discontinuing treatment. Patients seek advice and support from their peers and exchange information which otherwise may be lost. Understanding the individual patient journey and the challenges it brings will help improve treatment regimens and in turn patients’ health.
Limitations
Our analysis had some limitations. Our analysis is based on postings on the internet by unknown individuals. There is no way to determine if these people are diagnosed with axSpA, but since the information is taken from axSpA-specific forums and preselected to the relevance based on specific keywords, it is fair to assume that the people posting their voices and experiences are dealing with the disease. Further, there is a possibility that there is a reporting bias on the internet. People that are impacted greater may report more than the individuals not suffering from side-effects or have a milder disease. Additionally, not all switches were labelled with the medication names which leads to ‘unknown’ switches. Also, no information on tsDMARDs (targeted-synthetic disease modifying antirheumatic drugs) switches could be generated what might be related to the very recent approval of this drug class for the indication axSpA.
Conclusion and outlook
Overall, our analysis demonstrates how important it is to understand the individual journey of patients dealing with chronic diseases. On the one hand we were able to show the medication switches based on real-life user experiences, but more importantly also showcase medication switches differing from advised guidelines. Understanding the reasons behind these switches can help improve patient care.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X231187189 – Supplemental material for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis
Supplemental material, sj-docx-1-tab-10.1177_1759720X231187189 for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis by Maria Kahn, Steffeni Papukchieva, Axel Fehr, Markus Eberl, Berenice Rösler, Justyna Veit, Benjamin Friedrich and Denis Poddubnyy in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X231187189 – Supplemental material for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis
Supplemental material, sj-docx-2-tab-10.1177_1759720X231187189 for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis by Maria Kahn, Steffeni Papukchieva, Axel Fehr, Markus Eberl, Berenice Rösler, Justyna Veit, Benjamin Friedrich and Denis Poddubnyy in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-3-tab-10.1177_1759720X231187189 – Supplemental material for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis
Supplemental material, sj-docx-3-tab-10.1177_1759720X231187189 for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis by Maria Kahn, Steffeni Papukchieva, Axel Fehr, Markus Eberl, Berenice Rösler, Justyna Veit, Benjamin Friedrich and Denis Poddubnyy in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-4-tab-10.1177_1759720X231187189 – Supplemental material for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis
Supplemental material, sj-docx-4-tab-10.1177_1759720X231187189 for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis by Maria Kahn, Steffeni Papukchieva, Axel Fehr, Markus Eberl, Berenice Rösler, Justyna Veit, Benjamin Friedrich and Denis Poddubnyy in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-5-tab-10.1177_1759720X231187189 – Supplemental material for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis
Supplemental material, sj-docx-5-tab-10.1177_1759720X231187189 for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis by Maria Kahn, Steffeni Papukchieva, Axel Fehr, Markus Eberl, Berenice Rösler, Justyna Veit, Benjamin Friedrich and Denis Poddubnyy in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-6-tab-10.1177_1759720X231187189 – Supplemental material for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis
Supplemental material, sj-docx-6-tab-10.1177_1759720X231187189 for Drug switching in axial spondyloarthritis patients in Germany – a social listening analysis by Maria Kahn, Steffeni Papukchieva, Axel Fehr, Markus Eberl, Berenice Rösler, Justyna Veit, Benjamin Friedrich and Denis Poddubnyy in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
