Abstract
Objectives:
To investigate whether concomitant autoimmune inflammatory rheumatic diseases (AIIRDs) represent a risk factor for denosumab discontinuation and to explore other possible predictors.
Design:
This is a real-life retrospective study conducted at our centre on consecutive patients who started treatment with denosumab from January 2014 to October 2021.
Methods:
Data on patients’ characteristics, denosumab prescriptions and reason for discontinuation were collected from their medical electronic records. A log-rank test was run to assess differences in the denosumab retention rate between the not AIIRD and AIIRD patients. A backward stepwise logistic regression was used to identify possible predictors of denosumab discontinuation. When available, BMD data of the lumbar spine and total hip were collected.
Results:
Three hundred and sixty-three patients were included (265 not AIIRD and 98 AIIRD; median follow-up, 44 months). Sixty-nine patients discontinued denosumab at any time point (4 due to patient’s decision, 3 due to medical decision, 62 were lost in follow-up). The log-rank test did not find a statistically significant difference for denosumab persistence between the two subgroups. In the binary logistic regression analysis, only older age at initiation and lower baseline serum 25-hydroxy vitamin D were confirmed as predictors for discontinuation. BMD significantly increased from baseline to the last prescription visit at both the lumbar spine and the total hip, without statistically significant differences in the not AIIRD and AIIRD patients.
Conclusion:
The present data seem to suggest that AIIRDs do not represent a risk factor for denosumab discontinuation. Furthermore, the presence of AIIRDs does not seem to impair its effectiveness in terms of BMD.
Introduction
Autoimmune inflammatory rheumatic diseases (AIIRDs) are one major determinant for systemic bone loss, and consequent increased risk of osteoporosis and fragility fracture. 1
Denosumab, a monoclonal antibody inhibiting the receptor activator of nuclear factor kappa-B ligand (RANKL), has proven to be an effective and safe osteo-active agent, and in subjects affected by post-menopausal osteoporosis, it is associated with a steady bone mineral density (BMD) increase over the years, without plateauing. 2 Furthermore, denosumab has been approved by the Food and Drug Administration and the European Medicines Agency for the treatment of glucocorticoid-induced osteoporosis. Unfortunately, its unsupervised discontinuation is followed by a quick rebound effect, characterized by most, if not all, of the treatment-related BMD gains and by a concerning increase in the risk of incident fragility fractures (especially multiple vertebral fractures).3,4 Previous studies reported denosumab short- to mid-term retention rates (12–36 months) from 91% to 82% in patients with primary osteoporosis;5,6 conversely, data on subjects with secondary osteoporosis are lacking (i.e. patients with AIIRDs). Given the concerns regarding the rebound phenomenon mentioned above, there is an increasing need to clarify the denosumab persistence in the different populations eligible for this treatment, to define subgroups at particular risk of unsupervised discontinuation. To investigate whether concomitant inflammatory rheumatic conditions represent a risk factor for this outcome, we conducted a real-life study on AIIRD patients and compared their denosumab retention rate with that of subjects affected by post-menopausal or senile osteoporosis and explored possible risk factors associated with denosumab discontinuation. In addition, we also investigated the BMD gains in the overall sample and in patients with or without AIIRDs.
Materials and methods
This is a retrospective study of prospectively collected data conducted at the Rheumatology Unit, University of Verona (Verona, Italy) on consecutive patients who started treatment with denosumab from January 2014 to October 2021. The patients’ names were extracted from our centre prescribers’ list retrieved on the Agenzia Italiana del Farmaco (AIFA) treatment plan registry. All the patients included in the database who signed informed consent were considered eligible for the study.
The inclusion criteria matched those stated by ‘Nota AIFA 79’, the regulatory tool that norms the national healthcare system coverage for the pharmacological treatment of osteoporosis in Italy. 7 The main criteria of Nota 79 include, in the setting of primary prevention, post-menopausal women or men older than 50 years receiving (or scheduled to receive) treatment with glucocorticoids (daily prednisone equivalent ⩾5 mg for at least 3 months), or with a T-score (lumbar or femoral) ⩽–3 with at least one risk factor for osteoporosis, or, finally, with a T-score (lumbar or femoral) ⩽4 for whom treatment with bisphosphonate is not suitable (due to contraindications or previous adverse events). In secondary prevention, it includes patients with at least one non-vertebral fracture and T-score (lumbar or femoral) ⩽–3, and patients with previous vertebral or femoral fractures.
For the enrolment in the present study, we also established the following exclusion criteria: patients with moderate or severe chronic kidney disease (CKD) [defined as estimated glomerular filtration rate (eGFR) <45 ml/min as measured by the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation], patients receiving renal replacement therapy, kidney transplant receivers, patients with active cancer, patients receiving anti-hormonal treatment for breast or prostate cancer, and patients affected by endocrinological conditions known to affect the bone.
All patients gave consent to treatment.
Data on patients’ characteristics, fracture history (vertebral, hip, humerus, wrist and pelvis), diagnosis of AIIRDs, previous treatment with bisphosphonates or teriparatide, concurrent treatment with corticosteroid, 25-hydroxyvitamin D (25(OH)D) serum concentrations at baseline, and denosumab first and last prescription date were collected from their medical electronic records (the reported follow-up duration matches the treatment duration). The medical records of the eligible patients were then searched to retrieve the most recent follow-up data on the occurrence and date of incident fragility fractures after denosumab initiation, as well as date and reason for its discontinuation.
The reasons for discontinuation were classified as discontinued due to medical decision, due to patients’ decision or lost in follow-up. Since for receiving the denosumab doses in Italy it is necessary for each patient to obtain a yearly renewal of the treatment plan from the prescriber, the definition of lost in follow-up was made upon the finding of a timeframe longer than 12 months from the last renewal to the search date. During each follow-up visit, the treatment adherence before the prescription renewal is routinely ascertained by the clinician. An attempt via phone call was made to contact the subjects classified as lost in follow-up. When possible, the reason for the failed referral was retrieved. Those who forgot the appointed were rescheduled, while those who were physically unable to reach the centre were offered the possibility of a virtual consult.
When available, BMD data expressed as T-scores of the lumbar spine and total hip were collected.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement (the checklist is available as Supplementary Material).
Statistical analysis
Normality for all variables was tested by Shapiro–Wilk’s test. Normally distributed variables are reported as mean ± standard deviation (SD) and non-normally distributed variables as median [interquartile range (IQR)].
Two-sided Student’s t test for independent samples test was used to estimate the absolute differences between subgroups for normally distributed variables and Mann–Whitney’s U test for non-normally distributed variables.
To describe denosumab persistency for the overall cohort, a Kaplan–Meier survival curve was generated. A log-rank test was run to assess differences in the denosumab retention rate between the not AIIRD and AIIRD patients.
A backward stepwise logistic regression was used to identify possible predictors of denosumab discontinuation out of the following covariates: age at initiation, gender, body mass index (BMI), baseline 25(OH)D serum concentrations, ongoing corticosteroid treatment, history of fragility fractures, history of previous treatment with bisphosphonates, baseline lumbar spine T-score and baseline total hip T-score.
The increase in BMD, expressed as T-score, from baseline of the overall cohort and its subgroups was tested through two-sided Student’s t test for paired samples. Chi-square was used to test for differences in proportions.
Two-sided p values of 0.05 or less were considered statistically significant. Data were analysed using SPSS software, version 22 (SPSS, Inc., Chicago, IL, USA).
Results
Three hundred and sixty-three patients were included, with a median follow-up time of 44 (24–59) months (min–max: 6–133). The characteristics of the sample are reported in Table 1. Two hundred and sixty-five subjects were classified as not AIIRD, while 98 had a diagnosis of AIIRD. The diagnosed AIIRDs were distributed as follows: 50 rheumatoid arthritis, 5 spondyloarthropathies, 16 connective tissue diseases, 16 polymyalgia rheumatica and 12 systemic vasculitides. There was a statistically significant difference in the two subgroups in the proportion of patients receiving glucocorticoids, prevalence of history of fragility fractures, previous treatment with bisphosphonates and months of follow-up.
Characteristics of the overall sample and divided according to the presence or absence of AIIRD.
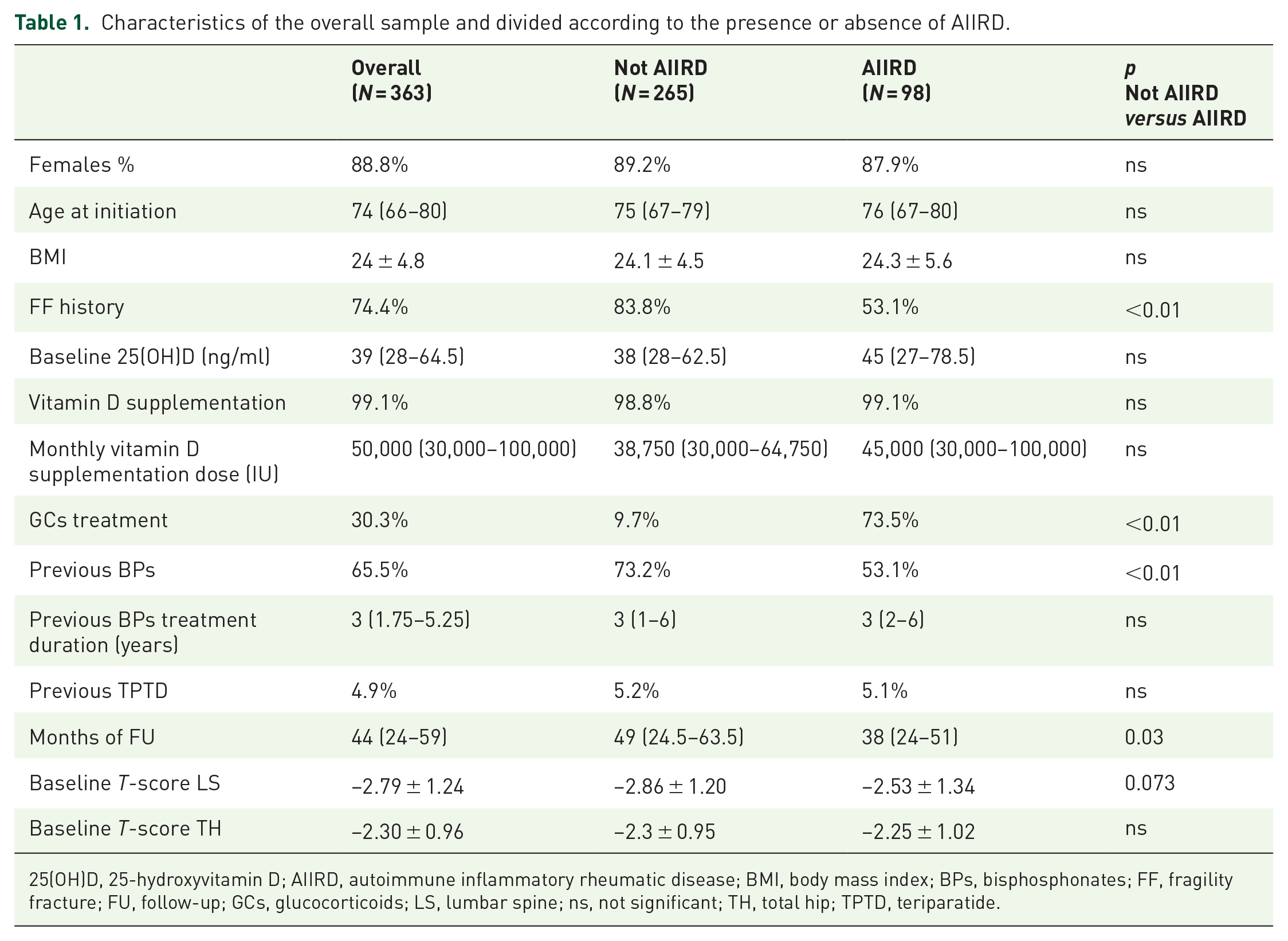
25(OH)D, 25-hydroxyvitamin D; AIIRD, autoimmune inflammatory rheumatic disease; BMI, body mass index; BPs, bisphosphonates; FF, fragility fracture; FU, follow-up; GCs, glucocorticoids; LS, lumbar spine; ns, not significant; TH, total hip; TPTD, teriparatide.
Sixty-nine patients eventually discontinued denosumab (4 due to patient’s decision, 3 due to medical decision, 62 were lost in follow-up). Of the 62 lost in follow-up, 58 answered our follow-up phone call (in person or their relatives): 32 patients were deceased, 10 were still on denosumab but switched to a different healthcare facility, 9 failed to refer due to the worsening of comorbid conditions and consequent logistic complications and 7 forgot to schedule the appointment (Supplementary Figure 1). None of the patients who discontinued denosumab but maintained follow-up reported clinical fractures (data from their electronic records), and, similarly, none of the patients contacted via phone call who discontinued denosumab reported clinical fractures.
The Kaplan–Meier curves for denosumab persistence (calculated from baseline to the last denosumab prescription) of the overall sample and for the not AIIRD and AIIRD subgroups are plotted in Figure 1. The log-rank test did not find a statistically significant difference for denosumab persistence between the not AIIRD and AIIRD subgroups (p = 0.975). Table 2 reports the univariate analysis for denosumab discontinuation as well as the binary logistic regression analysis. Starting with nine covariates that might theoretically be predictors of denosumab discontinuation, the stepwise logistic regression model reduced them to two. Increasing age was associated with an increased likelihood denosumab discontinuation, while increasing 25(OH)D baseline serum concentration was associated with its reduction.

Kaplan–Meier survival curves of denosumab persistence and 95% CI in the (a) overall cohort and divided according to the (b) not AIIRD or AIIRD subgroups.
Binary logistic regression analysis for factors associated with increased risk of denosumab discontinuation.
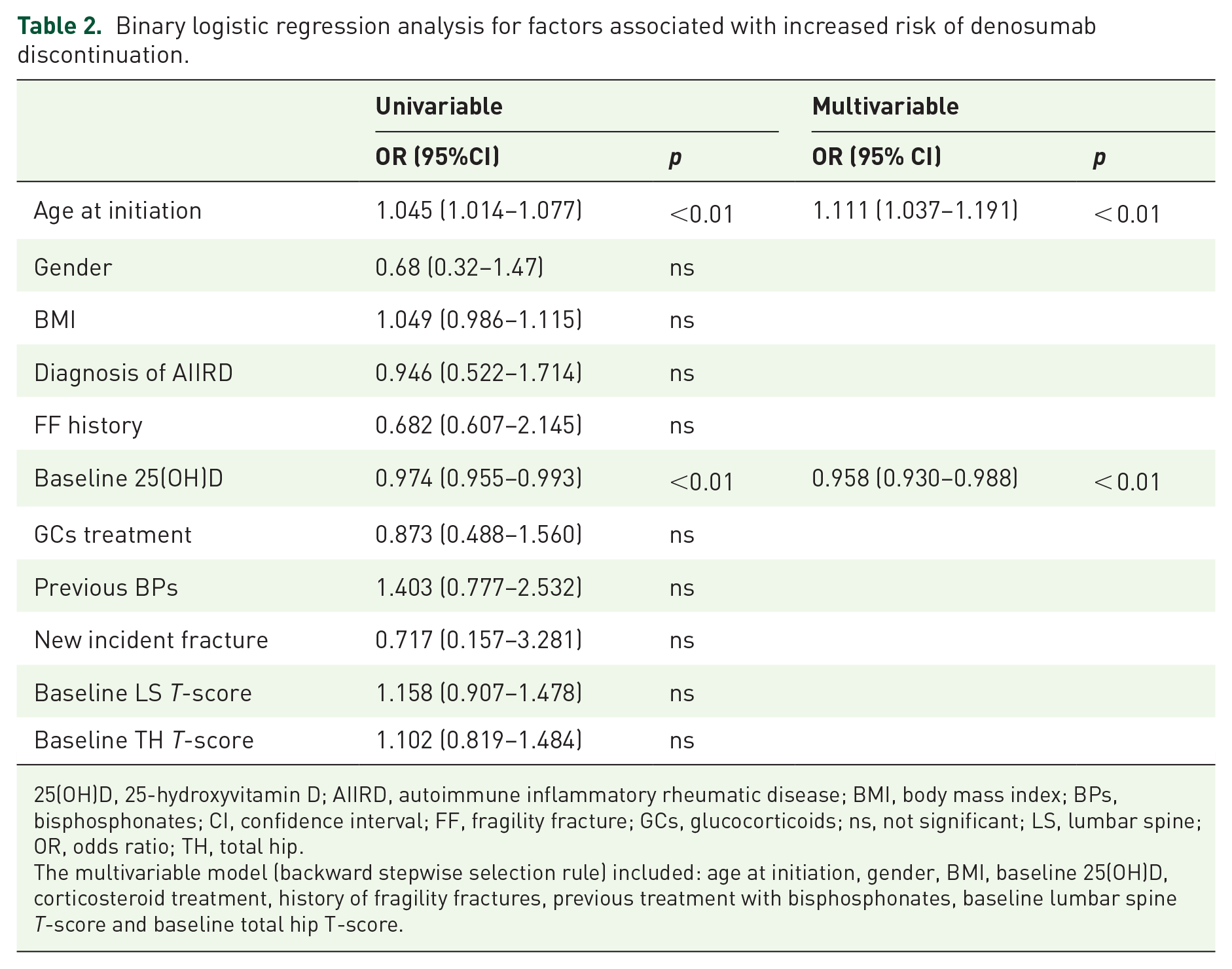
25(OH)D, 25-hydroxyvitamin D; AIIRD, autoimmune inflammatory rheumatic disease; BMI, body mass index; BPs, bisphosphonates; CI, confidence interval; FF, fragility fracture; GCs, glucocorticoids; ns, not significant; LS, lumbar spine; OR, odds ratio; TH, total hip.
The multivariable model (backward stepwise selection rule) included: age at initiation, gender, BMI, baseline 25(OH)D, corticosteroid treatment, history of fragility fractures, previous treatment with bisphosphonates, baseline lumbar spine T-score and baseline total hip T-score.
When the same analysis was run in the AIIRD subgroup, similar results were yielded (data not shown); in the multivariate model, only age remained a significant predictor for discontinuation: adjusted odds ratio (aOR) = 1.49, 95% confidence interval (CI): (1.06–2.10), p = 0.02.
BMD data at baseline and follow-up were available for 291 and 298 patients for the lumbar spine and total hip (225 and 66 in the not AIIRD and AIIRD groups at the lumbar spine, and 227 and 71 in the not AIIRD and AIIRD groups at the total hip, respectively). BMD significantly increased from baseline to the last available follow-up visit (and therefore last prescription) both at the lumbar spine and the total hip, in the overall cohort and in the two subgroups, without statistically significant differences in the not AIIRD and AIIRD patients. The absolute T-score variations are plotted in Figure 2. In terms of T-score percentage changes, we observed at the lumbar spine a mean increase of 21.9% (95% CI: 12.2–24.6) (p < 0.01 versus baseline) and at the total hip a mean increase of 9.4% (95% CI: 3.6–15.3) (p < 0.01 versus baseline), without significant differences in the two subgroups (data not shown). In addition, when we calculated the annualized BMD gains, no significant differences were found between the two subgroups (data not shown).
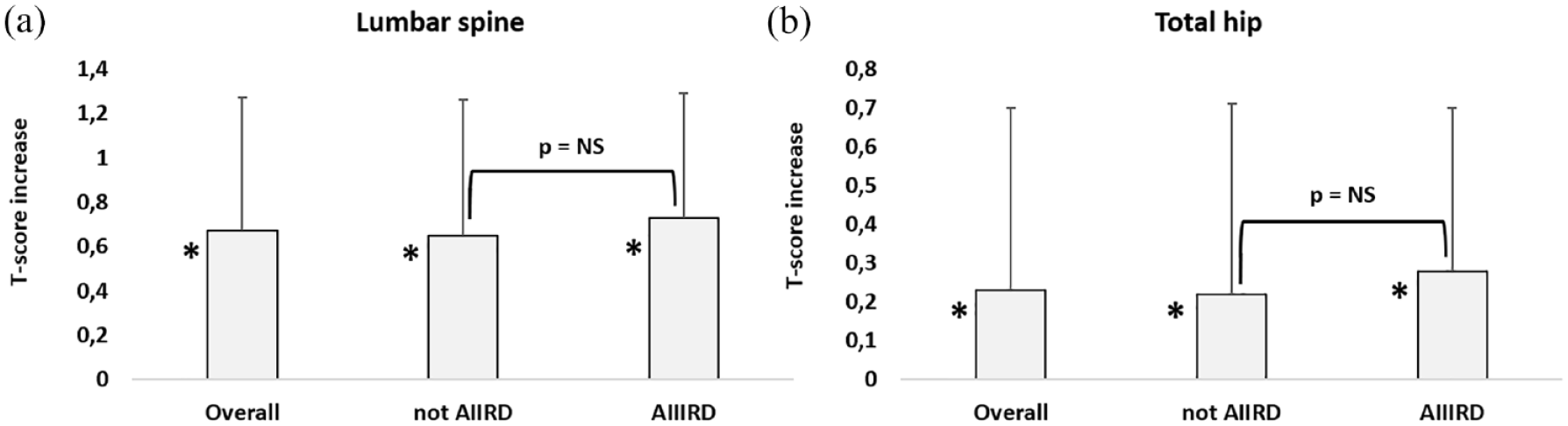
Mean BMD increases (expressed as T-scores) of the overall sample from baseline to the last prescription and the not AIIRD or AIIRD subgroup at (a) lumbar spine and (b) total hip. Error bars represent 95% CI.
Finally, 14 patients (3.8%) reported a new fracture during the observation period while on denosumab treatment (9 vertebral, 1 hip, 3 humerus and 1 wrist fractures: 11 in the not AIIRD and 3 in the AIIRD groups, p = ns).
Discussion
Our study investigated for the first time, in a real-life scenario, the denosumab retention rate in patients affected by AIIRD referring to a tertiary care rheumatology clinic. Our data suggest that a concurrent diagnosis of AIIRD does not represent a significant risk factor for denosumab discontinuation and therefore supports the viability of the drug also in this special population. Furthermore, our data also showed similar increases in the T-scores in the not AIIRD and AIIRD subgroups. This is a comforting remark since it suggests that concomitant inflammatory rheumatic disease may not impair the efficacy of the drug in terms of BMD gains.
In the last years, the interest on the treatment with denosumab in patients with AIIRDs has grown. Recent data from randomized controlled trails (RCTs) showed its effectiveness in inhibiting radiographic progression in patients with rheumatoid arthritis. 8 Moreover, it also demonstrated greater BMD gains when compared with alendronate and risedronate in the treatment of glucocorticoid-induced osteoporosis,9,10 a common treatment-induced complication occurring in many rheumatic conditions. A further advantage of denosumab over bisphosphonates is represented by its comforting safety data in subjects with CKD, 11 another significant comorbid condition in patients affected by several rheumatic diseases [i.e. systemic lupus erythematosus, scleroderma, antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, etc.]. However, in the last years, concerns have been raised on the risk of a rebound effect after its discontinuation, with the complete loss of the previously gained BMD and increased fracture risk, especially for longer treatment periods.3,12 The exact mechanism underlying of this phenomenon is still unclear, but the accumulation of immature osteoclast precursors during the treatment course 5 and the overshoot of RANKL after its discontinuation 12 are suspected to play a role.
Indeed, differently from bisphosphonates, unsupervised denosumab discontinuation and poor adherence do not result simply in a decreased effectiveness but may also represent a health concern, also in patients with AIIRD taking glucocorticoids. 13 Currently, there is insufficient evidence to strongly recommend one specific strategy to prevent this undesired outcome, although treatment with powerful bisphosphonates has been advocated (particularly zoledronic acid).3,14 Therefore, the investigation of the variables affecting the denosumab retention rates is of key importance to help clinicians in selecting the right candidates for this drug, and, on the contrary, to profile those who are at higher risk of unsupervised discontinuation. AIIRDs are complex conditions, often associated with burdensome comorbidities and polypharmacotherapy, features often associated with poor treatment compliance and adherence. 15 However, in our cohort, the presence of a rheumatic diagnosis did not prove to be a significant risk factor for decreased persistence when compared to a sample of subjects largely affected by primary (post-menopausal or senile) osteoporosis.
The mid-term (36 months) persistence of our overall cohort was roughly 85%, in line with previous data,5,16,17 while the long-term persistence seemed to stabilize around 75%, and it was similar in the AIIRD subgroup. Of all the analysed variables, the only ones associated with an increased risk for discontinuation were older age at initiation and lower baseline 25(OH)D serum concentrations. While the first relationship has already been documented,16,18 the second one is, to the best of our knowledge, an original finding. However, it is unlikely that the vitamin D status could have directly influenced the treatment adherence, and, in our opinion, higher 25(OH)D levels might simply be a marker of good adherence to the vitamin D supplementation that almost all our patients were already receiving, and therefore, a proxy for more compliant and thorough individuals.
In our study, the not AIIRD and AIIRD subgroups were comparable in terms of age, M:F ratio, BMI and BMD, but differed in the proportion of patients with fragility fracture history, glucocorticoid treatment and previous treatment with bisphosphonates. In our opinion, these differences are expected and reflect the inherent features of the AIIRD subgroup (i.e. glucocorticoid prevalence) and the different criteria of Nota 79 through which the two subgroups, on average, access the reimbursed treatment with denosumab. For instance, in the AIIRD subgroup, the initiation of the treatment might be more skewed towards the prevention of glucocorticoid-induced osteoporosis in primary prevention, while for the not AIIRD subgroup, the secondary prevention of new incident fragility fractures might have played a bigger role.
Our study has limitations. The first one is its retrospective design that exposes the data to several potential unaccounted confounders. Second is the limited sample size, especially of the AIIRD subgroup; given the heterogeneity of rheumatic conditions, our findings may not be generalizable to different cohorts, as the natural history of the different diseases can follow very different courses (i.e. rheumatoid arthritis versus ANCA-associated vasculitis). Third, the visiting schedule of subjects with primary osteoporosis is inevitably quite different from that of rheumatic patients, who often require a closer monitoring. For this reason, we cannot exclude that the observed adherence for the AIIRD subgroup was indeed reinforced by their more frequent consultations with the rheumatologist. However, these data were collected in a real-life scenario and without any interference on the usual scheduling. Finally, these data have been collected in a tertiary care rheumatology clinic and may be not immediately transposable to other care setting.
In conclusion, our real-life data may suggest that the presence of AIIRDs is not a risk factor for a worse denosumab persistence when compared with a population affected by primary osteoporosis. Moreover, similar BMD increases in the not AIIRD and AIIRD subgroups were observed. Currently, older age at initiation, also in rheumatic patients, seems to be the most significant risk factor for discontinuation, albeit the significance and effect sizes of other variables associated with age are still poorly investigated. Future research should focus on obtaining more granular data on the different AIIRD subtypes, also factoring in their most common complications (i.e. CKD, renal replacement therapy, institutionalization, severe recurrent infections, etc.).
Supplemental Material
sj-tif-1-tab-10.1177_1759720X221124543 – Supplemental material for Comparable long-term retention rates and effects on bone mineral density of denosumab treatment in patients with osteoporosis with or without autoimmune inflammatory rheumatic diseases: real-life data
Supplemental material, sj-tif-1-tab-10.1177_1759720X221124543 for Comparable long-term retention rates and effects on bone mineral density of denosumab treatment in patients with osteoporosis with or without autoimmune inflammatory rheumatic diseases: real-life data by Angelo Fassio, Davide Gatti, Davide Bertelle, Elena Fracassi, Giulia Zanetti, Ombretta Viapiana, Maurizio Rossini and Giovanni Adami in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
