Abstract
Background:
Rheumatoid arthritis (RA) and prolonged high-dose glucocorticoid (GC) treatment are established risk factors for osteoporosis.
Objectives:
In this study, we aimed to evaluate the therapeutic efficacy of denosumab according to the GC dose considered to increase the risk of glucocorticoid-induced osteoporosis (GIOP) in patients with RA.
Design:
A retrospective analysis of collected data on RA patients with osteoporosis starting denosumab.
Methods:
We included 418 patients with RA who were started on denosumab therapy and categorized them into those with and without GC intake ⩾2.5 mg/day for >3 months. The T-score and areal bone mineral density (aBMD) at the lumbar spine, total hip, and femur neck, as well as serum bone turnover markers, were measured at baseline and 12 months. We performed between-group and within-group comparisons of the BMD values at baseline and at 12 months.
Results:
Denosumab significantly increased the T-scores and aBMD at the lumbar spine, total hip, and femur neck after 12 months, regardless of GC intake. However, apart from the T-score at the lumbar spine, the other parameters did not show significant between-group differences. Similarly, in patients with anti-cyclic citrullinated peptide (CCP) antibody positivity or those treated with biologic or targeted synthetic disease-modifying antirheumatic drugs (DMARDs), although there were significant increases in the T-score and areal BMD at all sites in both groups, there were no significant between-group differences.
Conclusion:
Our findings suggest that the GC dose considered to increase the risk of GIOP did not significantly attenuate the therapeutic efficacy of denosumab in RA patients, including those positive for anti-CCP antibodies and users of biologic or targeted synthetic DMARDs.
Introduction
Inflammation in patients with rheumatoid arthritis (RA) disrupts the balance between bone formation and resorption, leading to osteoporosis. 1 RA is an established risk factor for osteoporotic fractures and has been used as an input variable in the fracture risk assessment tool. 2 Factors that contribute to bone loss in RA include disease activity, extended disease duration, erosive disease, positivity for autoantibodies, and joint damage resulting in reduced physical activity. 1 Additionally, glucocorticoids (GCs), which are commonly used to treat autoimmune diseases including RA, can contribute to osteoporosis (glucocorticoid-induced osteoporosis (GIOP)). 3 GCs initially enhance osteoclast function, followed by a decrease in bone formation due to apoptosis of osteoblasts and osteocytes. 3 Van Staa et al. 4 reported that GC doses ⩾2.5 mg/day increased the risk of fractures at both the spine and hip in patients with systemic GC treatment. Accordingly, the 2022 American College of Rheumatology (ACR) guidelines for the prevention and treatment of GIOP 5 recommend early screening for the risk of fractures following initiation of ⩾2.5 mg/day GC treatment for >3 months.
Denosumab is a fully human monoclonal antibody against the receptor activator of NFκB ligand (RANKL). 6 Denosumab prevents RANKL from activating RANK on osteoclasts, and reduced RANK–RANKL binding inhibits osteoclast function and survival. 6 Through this antiresorptive mechanism, denosumab increases bone mass and has demonstrated therapeutic efficacy against postmenopausal osteoporosis7–9 and GIOP.10,11 In addition, denosumab has been shown to increase bone mineral density (BMD) and inhibit joint erosion in patients with RA.12,13 According to these previous studies,12,13 the use of GC in patients with RA did not influence the effect of denosumab on the change rate in the BMD of the lumbar spine.
However, whether GC intake ⩾2.5 mg/day for >3 months impacts the therapeutic efficacy of denosumab remains unclear. Dore et al. 14 reported that denosumab increased the mean BMD of the lumbar spine and total hip as well as reduced bone turnover markers over 12 months in patients with RA, regardless of concomitant GC intake ⩾2.5 mg/day for ⩾3 months. However, the study of Dore et al. did not exclusively target patients with RA diagnosed with osteoporosis, and the mean baseline T-scores of concomitant GC users and nonusers were within the normal range. To our knowledge, no study has compared the therapeutic efficacy of denosumab based on GC intake ⩾2.5 mg/day for ⩾3 months in RA patients with osteoporosis.
The present study aimed to evaluate the impact of the GC dose considered to increase the risk of GIOP on the therapeutic efficacy of denosumab therapy against osteoporosis in patients with RA. Furthermore, we performed subgroup analysis in patients with anti-cyclic citrullinated peptide (CCP) antibody positivity and among users of biologic or targeted synthetic disease-modifying antirheumatic drugs (DMARDs).
Methods
Study population and design
We retrospectively obtained medical records data of patients with RA who visited Seoul St. Mary’s Hospital, a tertiary care university-affiliated hospital and referral center in Korea. We used the 2010 ACR/European League Against Rheumatism (EULAR) classification criteria 15 for RA diagnosis. The inclusion criteria comprised patients with RA who initiated denosumab treatment for osteoporosis. In contrast, the exclusion criteria included individuals with a history of fragility fractures, a denosumab dosing interval exceeding 7 months, and those who did not undergo BMD measurements. Data of 547 patients with RA who started on denosumab (60 mg through subcutaneous injection) between January 2016 and December 2023 due to osteoporosis, and without a history of fragility fractures, were initially screened. Osteoporosis diagnosis was defined based on the World Health Organization criteria. 16 We excluded 84 patients with >7-month intervals (considering pharmacokinetics of denosumab 17 ) between denosumab administrations and 45 who did not undergo BMD measurement. Finally, 418 patients with RA who received 2 or more doses of denosumab at 5- to 7-month intervals and underwent BMD measurement at the denosumab initiation and after 12 months were included. All patients underwent 25-hydroxyvitamin D and calcium level measurements every 6 months, with supplementation provided if the levels were <30 ng/mL for 25-hydroxyvitamin D or <8.6 mg/dL for calcium. The functional status of the included patients was class 1 or 2.
Patients were classified into those with and without a GC dosage ⩾2.5 mg/day prednisolone (or equivalent) continuously for >3 months between denosumab initiation (baseline) and 12 post-initiation months (increased GIOP risk and non-increased GIOP risk groups, respectively). Patients with GC intake <2.5 mg/day for >3 months, for ⩽3 months regardless of daily dosage, or without prednisolone were all included in the non-increased GIOP risk group. BMD values and levels of serum bone turnover markers at baseline and 12 months were retrieved from medical records. In addition, we collected data regarding age, sex, body mass index (BMI), disease duration of RA, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), prior GC dose, concurrent DMARD usage, 28-joint count disease activity scores (DAS28), 18 prior osteoporosis medication, rheumatoid factor (RF) positivity, and anti-CCP antibody positivity at baseline.
BMD and bone turnover marker measurements
The T-scores and areal BMD (aBMD) of the lumbar spine, total hip, and femur neck were assessed using dual-energy x-ray absorptiometry (DEXA) devices (Horizon W; Hologic, Marlborough, MA, USA) at baseline and 12 months. DEXA of the lumbar vertebra considered the lumbar 1–4 levels, with the exclusion of any vertebral bodies with compression fractures. For patients with avascular bone necrosis of the hip or joint replacement, the BMD of the other hip was used.
Serum bone turnover markers, including c-telopeptide of collagen type 1 (CTx) for bone resorption and total procollagen 1 N-terminal propeptide (P1NP) for bone formation, were measured through electrochemiluminescence immunoassay at baseline and 12 months.
Statistical analysis
The normality of the distribution of continuous variables was assessed using the Kolmogorov–Smirnov test. Continuous variables are expressed as mean and standard deviation or 95% confidence interval (CI), whereas categorical variables are expressed as frequency and percentage. Student’s t-test or Mann–Whitney U-test was used for between-group comparisons of continuous variables. Chi-squared test or Fisher’s exact test was used for between-group comparisons of categorical variables. A paired t-test or Wilcoxon signed-rank test was used to assess the significance of changes from baseline to 12 months within each group. To account for between-group comparisons of the T-score differences and aBMD changes from baseline to 12 months, analysis of covariance (ANCOVA) was performed with patient age, CRP, and baseline T-score and aBMD of total hip and femur neck (baseline variables significantly differ between the two groups) as covariates. As anti-CCP antibody positivity is an established risk factor for osteoporosis in patients with RA,19,20 we conducted a subgroup analysis of patients with anti-CCP antibody positivity. In addition, since biologic or targeted synthetic DMARDs have been suggested to improve bone metabolism and bone turnover in patients with RA,1,21 we performed subgroup analysis of such patients as well. All statistical analyses were performed using SPSS software version 24.0 (IBM Corporation, Chicago, IL, USA). Statistical significance was set at a p-value <0.05. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 22
Results
Characteristics of the study population
Table 1 presents the baseline characteristics of the 418 patients with RA. Among them, 282 and 136 patients were in the increased and non-increased GIOP risk groups, respectively. The non-increased GIOP risk group consisted of 23 patients with GC intake of <2.5 mg/day for >3 months or for ⩽3 months regardless of daily dosage, and 113 patients without GC. The concurrent GC dose and duration over 12 months were 1529.7 ± 699.3 mg and 11.5 ± 1.5 months for the increased GIOP risk group and 42.0 ± 112.8 mg and 0.7 ± 2.0 months for the non-increased GIOP risk group. Considering that the highest rate of bone loss occurs within the first 3–6 months of GC treatment, 5 there was no significant difference between the two groups in the proportion of GC users who started GC treatment within 6 months of denosumab initiation (11.0% vs 8.7%; p = 1.000). There were no significant between-group differences in terms of sex, BMI, disease duration of RA, ESR, percentage of DMARD use excluding methotrexate, DAS28 of biologic or targeted synthetic DMARD users, T-score and aBMD of the lumbar spine, prior use of osteoporosis medication, RF positivity, anti-CCP antibody positivity, and serum bone turnover markers. Age (64.7 ± 9.7 vs 62.7 ± 9.2 years; p = 0.04), CRP levels (0.428 ± 0.844 vs 0.228 ± 0.419 mg/dL; p = 0.001), prior 6 months GC dose (669.7 ± 379.5 vs 52.6 ± 116.5 mg; p < 0.001), and percentage of methotrexate use (75.9% vs 65.4%; p = 0.025) were higher in the increased GIOP risk group than in the non-increased GIOP risk group. Furthermore, the baseline T-scores and aBMD of the total hip (T-score, −2.11 ± 0.64 vs −1.93 ± 0.64; p = 0.01, aBMD, 0.6968 ± 0.0788 vs 0.7226 ± 0.0746 g/cm2; p = 0.002) and femur neck (T-score, −2.61 ± 0.55 vs −2.45 ± 0.61; p = 0.039, aBMD, 0.5825 ± 0.0800 vs 0.6111 ± 0.0798 g/cm2; p = 0.007) were lower in the increased GIOP risk group than in the non-increased GIOP risk group.
Baseline clinical and laboratory variables of included RA patients (n = 418).
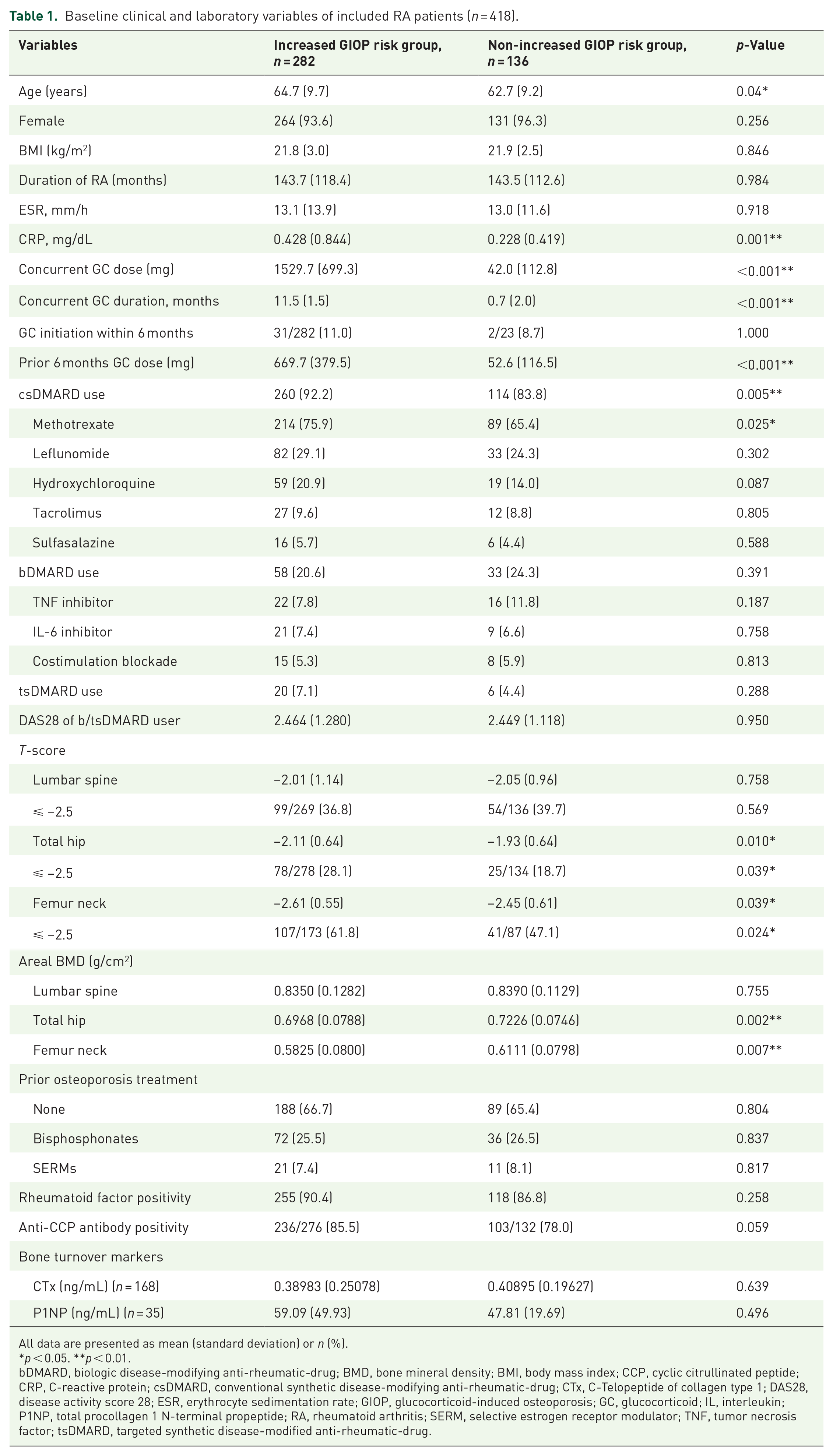
All data are presented as mean (standard deviation) or n (%).
p < 0.05. **p < 0.01.
bDMARD, biologic disease-modifying anti-rheumatic-drug; BMD, bone mineral density; BMI, body mass index; CCP, cyclic citrullinated peptide; CRP, C-reactive protein; csDMARD, conventional synthetic disease-modifying anti-rheumatic-drug; CTx, C-Telopeptide of collagen type 1; DAS28, disease activity score 28; ESR, erythrocyte sedimentation rate; GIOP, glucocorticoid-induced osteoporosis; GC, glucocorticoid; IL, interleukin; P1NP, total procollagen 1 N-terminal propeptide; RA, rheumatoid arthritis; SERM, selective estrogen receptor modulator; TNF, tumor necrosis factor; tsDMARD, targeted synthetic disease-modified anti-rheumatic-drug.
We conducted a subgroup analysis of 339 patients with anti-CCP antibody positivity (Supplemental Table 1). Among them, 236 and 103 patients were classified into the increased and non-increased GIOP risk groups, respectively. Except for age (64.9 ± 9.7 vs 62.6 ± 9.2 years; p = 0.042), CRP (0.437 ± 0.886 vs 0.254 ± 0.455 mg/dL; p = 0.013), and baseline T-score (−2.10 ± 0.65 vs −1.93 ± 0.62; p = 0.022) and aBMD (0.6973 ± 0.0794 vs 0.7201 ± 0.0737 g/cm2; p = 0.014) of the total hip, there were no significant between-group differences in the other parameters.
Subgroup analysis results of 117 patients treated with biologic or targeted synthetic DMARDs are presented in Supplemental Table 2. Among them, 78 and 39 patients were classified into the increased and non-increased GIOP risk groups, respectively. There was no significant between-group difference in DAS28 (Table 1). Excluding the baseline T-score (−2.18 ± 0.66 vs −1.89 ± 0.75; p = 0.037) and aBMD (0.6877 ± 0.0771 vs 0.7234 ± 0.0809 g/cm2; p = 0.025) of the total hip, there were no significant between-group differences in the other variables.
Comparison of therapeutic efficacy of denosumab according to GC dose considered to increase the risk of GIOP
T-score differences from baseline to 12 months of the lumbar spine were 0.14 (95% CI: 0.10–0.19) in the increased GIOP risk group and 0.22 (95% CI: 0.16–0.27) in the non-increased GIOP risk group. ANCOVA-adjusted values were 0.14 (95% CI: 0.10–0.18) and 0.22 (95% CI: 0.16–0.28), respectively (Figure 1(a)). aBMD changes from baseline to 12 months at the lumbar spine were 2.046% (95% CI: 1.463–2.629) in the increased GIOP risk group and 2.813% (95% CI: 2.080–3.546) in the non-increased GIOP risk group. ANCOVA-adjusted values were 2.032% (95% CI: 1.456–2.608) and 2.872% (95% CI: 2.059–3.685), respectively (Figure 1(b)). T-score differences from baseline to 12 months at the total hip were 0.10 (95% CI: 0.07–0.13) in the increased GIOP risk group and 0.14 (95% CI: 0.10–0.17) in the non-increased GIOP risk group. ANCOVA-adjusted values were 0.10 (95% CI: 0.07–0.12) and 0.14 (95% CI: 0.09–0.18), respectively (Figure 1(c)). aBMD changes from baseline to 12 months of the total hip were 1.731% (95% CI: 1.215–2.247) in the increased GIOP risk group and 2.141% (95% CI: 1.516–2.767) in the non-increased GIOP risk group. ANCOVA-adjusted values were 1.710% (95% CI: 1.219–2.202) and 2.192% (95% CI: 1.478–2.906), respectively (Figure 1(d)). T-score differences from baseline to 12 months at the femur neck were 0.12 (95% CI: 0.10–0.15) in the increased GIOP risk group and 0.12 (95% CI: 0.09–0.15) in the non-increased GIOP risk group. ANCOVA-adjusted values were 0.12 (95% CI: 0.09–0.15) and 0.11 (95% CI: 0.07–0.16), respectively (Figure 1(e)). aBMD changes from baseline to 12 months at the femur neck were 2.098% (95% CI: 1.608–2.588) in the increased GIOP risk group and 1.914% (95% CI: 1.117–2.710) in the non-increased GIOP risk group. ANCOVA-adjusted values were 2.154% (95% CI: 1.502–2.806) and 1.833% (95% CI: 0.906–2.760), respectively (Figure 1(f)). In both groups, there were significant increases in all parameters compared with the baseline values (all p < 0.01). The mean increase in the T-score and aBMD at the lumbar spine and total hip were greater in the non-increased GIOP risk group than in the increased GIOP risk group. However, apart from the T-score difference at the lumbar spine (p = 0.037), there were no significant between-group differences.
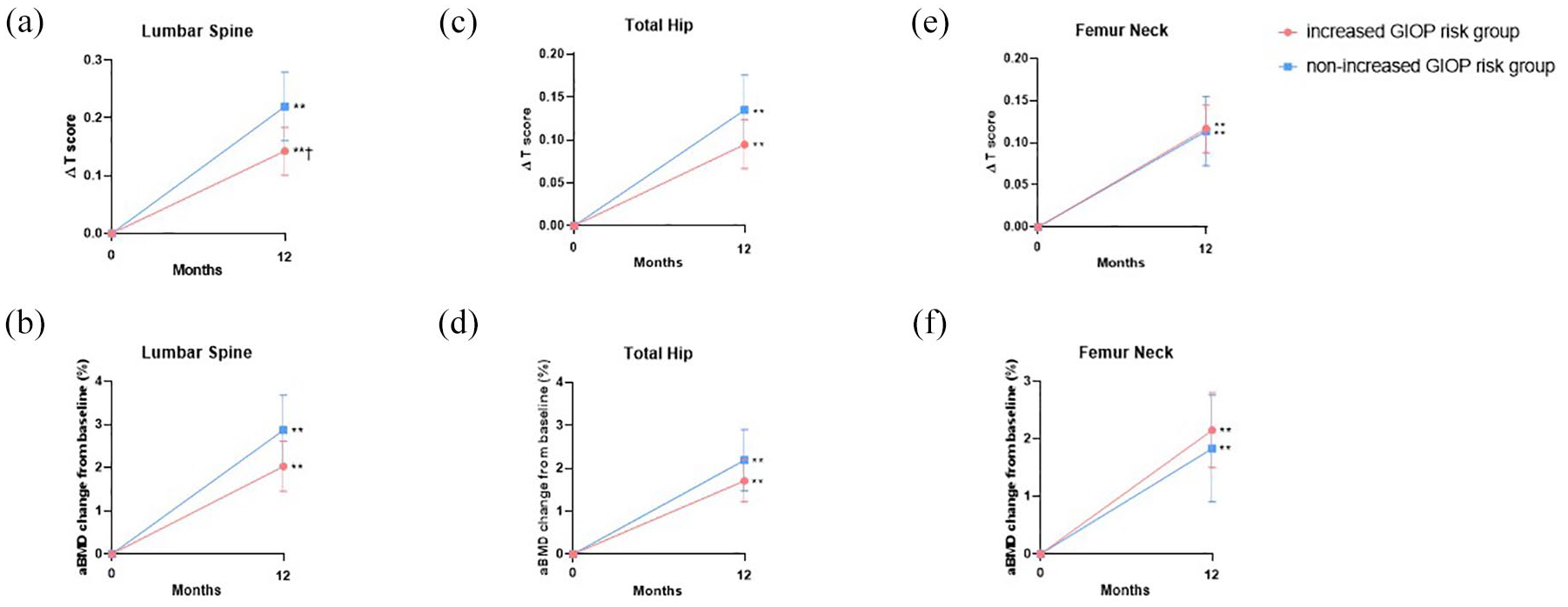
ANCOVA-adjusted mean T-score differences (∆T score) and aBMD changes from baseline to 12 months at the (a, b) lumbar spine, (c, d) total hip, and (e, f) femur neck in the included RA patients.
Comparison of therapeutic efficacy of denosumab according to GC dose considered to increase the risk of GIOP in patients with RA positive for anti-CCP antibodies
ANCOVA-adjusted T-score differences and aBMD changes in anti-CCP antibody-positive patients with RA are presented in Figure 2. T-score differences from baseline to 12 months measured by DEXA at the lumbar spine were 0.14 (95% CI: 0.10–0.19) and 0.22 (95% CI: 0.16–0.28) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 0.14 (95% CI: 0.10–0.19) and 0.22 (95% CI: 0.16–0.29; Figure 2(a)). aBMD changes from baseline to 12 months measured by DEXA at the lumbar spine were 2.156% (95% CI: 1.536–2.775) and 2.869% (95% CI: 2.008–3.730) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 2.140% (95% CI: 1.521–2.760) and 2.946% (95% CI: 2.026–3.866; Figure 2(b)). T-score differences from baseline to 12 months at the total hip were 0.10 (95% CI: 0.07–0.14) and 0.13 (95% CI: 0.09–0.17) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 0.10 (95% CI: 0.07–0.13) and 0.13 (95% CI: 0.09–0.18; Figure 2(c)). aBMD changes from baseline to 12 months at the total hip were 1.821% (95% CI: 1.263–2.379) and 2.070% (95% CI: 1.423–2.718) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 1.802% (95% CI: 1.287–2.318) and 2.124% (95% CI: 1.337–2.911; Figure 2(d)). T-score differences from baseline to 12 months at the femur neck were 0.12 (95% CI: 0.09–0.15) and 0.11 (95% CI: 0.08–0.13) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 0.11 (95% CI: 0.08–0.14) and 0.10 (95% CI: 0.06–0.14; Figure 2(e)). aBMD changes from baseline to 12 months at the femur neck were 2.114% (95% CI: 1.619–2.610) and 1.613% (95% CI: 0.882–2.343) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 2.212% (95% CI: 1.590–2.834) and 1.435% (95% CI: 0.495–2.375; Figure 2(f)). There were significant increases in the T-score and aBMD at the lumbar spine, total hip, and femur neck (all p < 0.01) in both groups. Similar to the total cohort, the mean increase in the T-score and aBMD at the lumbar spine and total hip were greater in the non-increased GIOP risk group than in the increased GIOP risk group. However, there were no significant between-group differences in the parameters.

ANCOVA-adjusted mean T-score differences (∆T score) and aBMD changes from baseline to 12 months at the (a, b) lumbar spine, (c, d) total hip, and (e, f) femur neck of RA patients positive for anti-CCP antibodies.
Comparison of therapeutic efficacy of denosumab according to GC dose considered to increase the risk of GIOP among biologic or targeted synthetic DMARD users
Figure 3 shows ANCOVA-adjusted T-score differences and aBMD changes in patients with RA who used biologic or targeted synthetic DMARDs. For the lumbar spine and femur neck, there were no covariates requiring adjustment. T-score differences from baseline to 12 months measured by DEXA at the lumbar spine were 0.16 (95% CI: 0.08–0.24) and 0.28 (95% CI: 0.18–0.37) in the increased and non-increased GIOP risk groups, respectively (Figure 3(a)). aBMD changes from baseline to 12 months measured by DEXA at the lumbar spine were 2.427% (95% CI: 1.294–3.559) and 3.375% (95% CI: 1.867–4.883) in the increased and non-increased GIOP risk groups, respectively (Figure 3(b)). T-score differences from baseline to 12 months at the total hip were 0.16 (95% CI: 0.10–0.22) and 0.15 (95% CI: 0.08–0.23) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 0.16 (95% CI: 0.10–0.22) and 0.16 (95% CI: 0.08–0.25; Figure 3(c)). aBMD changes from baseline to 12 months at the total hip were 2.820% (95% CI: 1.821–3.819) and 1.937% (95% CI: 1.032–2.842) in the increased and non-increased GIOP risk groups, respectively. ANCOVA-adjusted values were 2.795% (95% CI: 1.792–3.797) and 2.353% (95% CI: 0.915–3.791; Figure 3(d)). T-score differences from baseline to 12 months at the femur neck were 0.17 (95% CI: 0.12–0.22) and 0.13 (95% CI: 0.08–0.19) in the increased and non-increased GIOP risk groups, respectively (Figure 3(e)). aBMD changes from baseline to 12 months at the femur neck were 2.957% (95% CI: 2.021–3.894) and 2.084% (95% CI: 1.211–2.957) in the increased and non-increased GIOP risk groups, respectively (Figure 3(f)). Although there were significant increases in the T-score and aBMD at the lumbar spine, total hip, and femur neck (all p < 0.01) in both groups, there were no significant between-group differences.

ANCOVA-adjusted mean T-score differences (∆T score) and aBMD changes from baseline to 12 months at the (a, b) lumbar spine, (c, d) total hip, and (e, f) femur neck in RA patients treated with biologic or targeted synthetic DMARDs.
Changes in bone turnover markers
The levels of major serum bone turnover markers, CTx and P1NP, at baseline and 12 months were measured (Figure 4). As described in Table 1, there were no significant between-group differences in bone turnover markers at baseline. Serum CTx levels at 12 months were 0.192 ng/mL (95% CI: 0.157–0.227) and 0.229 ng/mL (95% CI: 0.173–0.285) in the increased and non-increased GIOP risk groups, respectively (Figure 4(a)). Serum P1NP levels at 12 months were 24.51 ng/mL (95% CI: 20.56–28.45) and 21.73 ng/mL (95% CI: 19.06–24.40) in the increased and non-increased GIOP risk groups, respectively (Figure 4(b)). Serum levels of CTx and P1NP were both significantly decreased at 12 months compared with the baseline values (all p < 0.01), regardless of the risk of GIOP. However, there were no significant ANCOVA-adjusted between-group differences.

ANCOVA-adjusted mean changes in bone turnover markers (a) CTx and (b) total P1NP from baseline to 12 months of included RA patients.
Discussion
Our findings demonstrated that denosumab significantly improved the T-score and aBMD at 12 months in patients with RA who had osteoporosis. However, there were no significant differences in the therapeutic efficacy of denosumab according to the GC dose considered to increase GIOP risk (Figure 1). These findings were consistent even in the subgroup analyses according to anti-CCP antibody positivity (Figure 2) or use of biologic or targeted synthetic DMARDs (Figure 3). Additionally, denosumab significantly reduced serum bone turnover markers at 12 months; however, there were no significant between-group differences in the degree of reduction (Figure 4).
Despite the older mean age and higher mean CRP levels in the increased GIOP risk group, GC did not significantly attenuate the therapeutic efficacy of denosumab. The only conventional synthetic DMARD that showed a significant between-group difference in the proportion of users was methotrexate, which has previously demonstrated a limited effect on bone metabolism in patients with RA.1,23,24 Therefore, the confounding effect of methotrexate was presumed to be minimal. There were no significant between-group differences in the ratios of users of biologic and targeted synthetic DMARDs, which are known to improve bone metabolism in patients with RA.1,21 Regarding the total hip and femur neck, the increased GIOP risk group had a lower baseline T-score and aBMD. However, there was no significant between-group difference in the therapeutic efficacy of denosumab at the total hip and femur neck. The only significant between-group difference was observed in the mean T-score difference of the lumbar spine. Previous systematic review studies on the therapeutic efficacy of denosumab in patients with RA12,13 have reported no significant difference in lumbar spine BMD improvements between patients treated with and without GC. This suggests that our findings regarding the lumbar spine T-score may have been influenced by the confounding factors of older age and higher CRP levels related to disease activity.
Anti-CCP antibodies are among the RA-specific risk factors for osteoporosis since they bind to and stimulate osteoclasts. 20 Anti-CCP antibodies have been associated with both local joint erosions and systemic bone loss in both early- and late-stage RA.19,25–30 Denosumab reduces osteoclast function and survival, and therefore, counteracts the mechanisms stimulated by anti-CCP antibodies. Denosumab may facilitate the prevention of fragility fractures and structural damage in patients with RA with positive anti-CCP antibodies.31,32 However, the impact of GC on the therapeutic efficacy of denosumab in this subgroup remains unclear. Our findings indicated that denosumab significantly increased the T-score and aBMD at the lumbar spine, total hip, and femur neck in patients with RA who were positive for anti-CCP antibodies. Furthermore, although the increased GIOP risk group had older age, higher CRP levels, and lower baseline T-score and aBMD values of the total hip, the therapeutic efficacy of denosumab showed no significant between-group difference.
Biologic DMARDs targeting certain cytokines and their signaling pathways might offer a protective effect against bone loss in patients with RA.1,21,33–38 Targeted synthetic DMARDs, including Janus kinase inhibitors, have been shown to reduce osteoclastogenesis by decreasing RANKL expression by osteoblasts.1,39–41 In our study, denosumab significantly improved the T-score and aBMD in patients with RA with osteoporosis who received biologic or targeted synthetic DMARDs. Although we cannot confirm whether this effect is attributable to DMARDs or denosumab, Hasegawa et al. 42 reported that concurrent use of denosumab and biologic DMARDs could effectively inhibit structural damage without increasing adverse events in patients with RA, compared with treatment by biologic DMARDs alone. Notably, we found that the GC dose considered to increase GIOP risk did not significantly influence the degree of improvement in T-score and aBMD in this subgroup. In the 2021 ACR guideline for the treatment of RA 43 and the 2022 update of EULAR recommendations for the management of RA with synthetic and biological DMARDs, 44 biologic or targeted synthetic DMARDs are recommended for patients with high or poorly controlled disease activity. In our study, there was no significant between-group difference in baseline DAS28 among patients who received biologic or targeted synthetic DMARDs; furthermore, GC did not significantly influence the therapeutic efficacy of denosumab.
Denosumab acts as an antiresorptive agent by decreasing bone resorption and/or inhibiting bone turnover. 6 It has been shown to significantly reduce serum levels of bone turnover markers in patients with RA or those receiving long-term GC treatment.10,13 To our knowledge, this is the first study to compare changes in bone turnover markers in patients with RA according to the intake of GC dose considered to increase GIOP risk. There was no significant between-difference in the denosumab-mediated reduction of CTx and P1NP, which ultimately failed to produce significant differences in the T-score and aBMD increase.
This study has some limitations. First, this was a single-center study. Therefore, potential selection bias could not be avoided. Second, although the increase in T-score and aBMD at the lumbar spine and total hip were numerically greater in the non-increased GIOP risk group, given the observational nature of the study, it might not have been properly powered, leading to no statistically significant difference. Finally, the analysis was limited to the period from the initial administration of denosumab to 12 post-administration months. To evaluate the long-term therapeutic efficacy of denosumab considering cumulative GC doses beyond 12 months, further longitudinal, prospective studies are needed.
Conclusion
In conclusion, our findings suggested that the GC dose considered to increase GIOP risk did not significantly attenuate the therapeutic efficacy of denosumab in increasing BMD or reducing bone turnover markers in patients with RA. These findings were consistent in the subgroup of patients with anti-CCP antibody positivity or users of biologic or targeted synthetic DMARDs. Denosumab appears to be an effective treatment option for osteoporosis in patients with RA, including those positive for anti-CCP antibodies and users of biologic or targeted synthetic DMARDs, regardless of the GC dosage.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251314712 – Supplemental material for Impact of glucocorticoids on the therapeutic efficacy of denosumab against osteoporosis in patients with rheumatoid arthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X251314712 for Impact of glucocorticoids on the therapeutic efficacy of denosumab against osteoporosis in patients with rheumatoid arthritis by Jiwon Yang, Youngjae Park, Jennifer Jooha Lee, Seung-Ki Kwok, Ji Hyeon Ju, Wan-Uk Kim and Sung-Hwan Park in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
