Abstract
Objectives:
The Simplified Disease Activity Index (SDAI) is a recommended composite score for assessing the remission status in patients with rheumatoid arthritis (RA). However, determination of C-reactive protein (CRP) levels takes several hours and sometimes days and limits the use of the SDAI in the clinical setting. The aim of this study was to validate the SDAI using a quick quantitative C-reactive protein (qCRP) assay (as SDAI-Q) in RA patients.
Design:
This is a multicenter, prospective, cross-sectional pilot study in RA patients.
Methods:
Adult patients (⩾18 years) with a clinical diagnosis of RA were recruited between January 2020 and September 2020 from five rheumatologic centers located in Berlin, Germany. SDAI, SDAI-Q, Clinical Disease Activity Index (CDAI), and DAS28 scores comprising CRP, qCRP, or erythrocyte sedimentation rate (ESR) were calculated. The agreement of disease activity categories was analyzed using cross tabulations and weighted Cohen’s kappa. The agreement of numerical values was analyzed with Bland–Altman plots and intraclass correlation coefficients (ICCs).
Results:
Overall, 100 RA patients were included in the statistical analysis. The mean value of qCRP (7.89 ± 16.98 mg/l) was slightly higher than that of routine laboratory CRP (6.97 ± 15.02 mg/l). Comparing SDAI and SDAI-Q, all patients were assigned to identical disease activity categories. Agreement of disease activity categories by CDAI and SDAI/SDAI-Q was observed in 93% with a weighted Cohen’s kappa of 0.929 (95% confidence interval (CI) = 0.878; 0.981).
Conclusion:
The SDAI-Q showed an absolute agreement regarding the assignment of disease activity categories in comparison with the conventional SDAI. Therefore, the SDAI-Q may facilitate the application of a treat-to-target concept in clinical trials and clinical routine as a quickly available disease activity score incorporating CRP as an objective parameter.
Introduction
Rheumatoid arthritis (RA) is a chronic, autoimmune joint disease with a prevalence of 0.5–1.0% in North America and Europe.1,2 Typically, RA patients suffer from polyarthritis and morning stiffness particularly affecting the small joints of hands and feet with a symmetric distribution. 3 Moreover, also extra-articular manifestations like pulmonary involvement, vasculitis, or rheumatoid nodules are possible. 1 Insufficient control of the inflammatory process leads to subsequent structural joint damage that causes irreversible impairment of physical function.4–8
The treat-to-target (T2T) concept in rheumatology comprises regular monitoring of disease activity using composite scores as well as therapeutic modifications based on the current disease activity level to achieve the targeted disease activity level. 9 Over the past years, significant evidence favoring the application of a T2T concept in RA patients has been accumulated.9,10 Implementation of a T2T strategy showed improved physical function,11–13 enhanced health-related quality of life, 11 and cost-effectiveness11,14 compared with routine care. Thus, current treatment and management guidelines have given a clear recommendation to apply the T2T strategy in RA patients, aiming at clinical remission or –whether remission cannot be achieved – low disease activity (LDA) as an alternative target.15,16 Furthermore, it has been recommended to use validated composite scores for regular disease activity assessments. 15
According to American College of Rheumatology/European Alliance of Associations for Rheumatology (ACR/EULAR) remission criteria, the Simplified Disease Activity Index (SDAI) represents the most suitable composite score to define and assess the state of remission in clinical trials. 17 SDAI is calculated by summation of patient and physician global disease activity assessment on a visual analogue scale from 0 to 10 cm, tender and swollen joint count (out of 28 joints), and the C-reactive protein (CRP) value in mg/dl. 18 The Clinical Disease Activity Index (CDAI) was introduced as an alternative disease activity score for clinical routine, constituting of the same variables as SDAI but omitting the CRP value. 19 As current, routine laboratory CRP values are mostly not immediately available in the outpatient rheumatological setting, the ACR/EULAR committee suggested CDAI as an alternative composite index for the remission definition in clinical practice. 17
The CDAI showed a very good agreement with SDAI regarding disease activity categories 20 and CRP contributed on average only 5% to SDAI values 19 in the CDAI validation studies. A composite score involving CRP as an objective inflammatory parameter will better reflect the real disease activity than a purely clinical score in certain situations, however. This might be especially the case for patients with pain hypersensitivity (fibromyalgia) or indolent RA patients with a few subjective symptoms in which an objective inflammatory parameter may help to recognize and distinguish genuinely and apparently increased disease activity. Therefore, CDAI may lead to an over- or under-estimation of disease activity in aforementioned patient groups. Furthermore, a large register study found discrepant disease activity categories by SDAI and CDAI between 12.8% and 20.4%, and the authors concluded that acute-phase reactants may contribute more strongly to composite scores than originally assumed. 21
Validated, quick quantitative C-reactive protein (qCRP) assays can provide a current CRP value within a few minutes after blood collection. In the past, we have already validated the Ankylosing Spondylitis Disease Activity Score (ASDAS) using a qCRP assay (called ASDAS-Q) in a single-center pilot study 22 and in a multicenter study with a higher sample size. 23 This qCRP assay can be also used for SDAI calculation (as SDAI-Q). As a timely available composite index containing an acute-phase reactant, the SDAI-Q may combine the advantages of the conventional SDAI as well as the advantages of the CDAI, but has not been validated yet.
The Disease Activity Score 28 (DAS28) is no more recommended for the definition of remission, 17 among others, as the DAS28 remission state was shown to allow significant residual inflammation and performed worse in predicting good radiographic outcomes in comparison with other remission definitions.17,24 DAS28 is still frequently used in clinical practice, however. Thus, a qCRP-based DAS28 (DAS28-qCRP) might be helpful for some rheumatologists.
This study aimed to validate the SDAI-Q in a prospective, multicenter, cross-sectional pilot study in early and established RA patients receiving different medical treatments. Owing to its wide distribution in clinical practice, the performance of DAS28-qCRP in comparison with DAS28-CRP was also investigated.
Patients and methods
Patients and assessments
The current research project was conducted as part of a joint, higher level, multicenter research project to validate different disease activity scores using a qCRP assay, including the validation of the ASDAS based on a qCRP assay. 23
This cross-sectional, multicenter pilot study was performed in Berlin, Germany. Five rheumatologic centers participated in this project, including two specialized academic university hospitals and three rheumatologic outpatient practices. The study recruitment took place between January 2020 and September 2020. All adult (⩾18 years) patients with a clinical diagnosis of RA, who had given written informed consent, were considered eligible for study participation. Rheumatologists were encouraged to include consecutive patients. Exclusion criteria for study participation were a previously known clinically significant anemia, meaning a hemoglobin-concentration <10 g/dl (<6.2 mmol/l) or signs of an acute infection. 23 A structured rheumatologic assessment was performed in all study patients, including joint counts, patients’ global assessment of disease activity, evaluator’s global assessment of disease activity, and laboratory assessment. Patient’s and evaluator’s global assessments of disease activity were assessed on a visual analogue scale (VAS) from 0 cm = inactive to 10 cm = very active.
Measurement of routine laboratory CRP was performed in different certified laboratories by a conventional method (usually, a turbidimetric assay). Erythrocyte sedimentation rate (ESR) measurement was conducted locally. Measurement of qCRP values was performed with the ‘QuikRead go’ instrument (Aidian Oy, Espoo, Finland, software versions 7.5.1 and 7.6.2) and the reagent kits ‘QuikRead go wrCRP + Hb’ (Aidian Oy). In most of the patients, whole blood from the blood collection system could be used for the qCRP evaluation, which would have been disposed otherwise. Instead of whole blood from the collection system, the qCRP measurement could also be performed with ethylenediaminetetraacetic acid (EDTA) or heparin whole blood, when the collection system was no more available or blood in the collection system was already coagulated. 23
The qCRP measurement range was dependent on the hematocrit concentration and according to the manufacturer is 0.5–200 mg/l for a hematocrit concentration of 40–45%. 25 Measurement of qCRP was available for samples having a hematocrit concentration between 15% and 75%. 25 Studies validating the ‘QuikRead go’ products were performed according to the Clinical and Laboratory Standards Institute (CLSI) guidelines, EP5-A3.25,26 The qCRP measurement is based on an immunoturbidimetric method and was executed according to the assay instructions: 25 Ten microliters of whole blood were transferred into a glass capillary and from this capillary transferred into a special cuvette containing the reagent solution. The cuvette was closed with a reagent cap and then put into the ‘QuikRead go’ instrument 25 and the whole measurement can be performed in approximately 2 min. The precision of qCRP measurement was evaluated by the manufacturer in a multisite precision study. 27
The qCRP measurement device indicated all qCRP values with one decimal place. Quality controls were implemented once weekly using the ‘QuikRead go wrCRP Control’ (Aidian Oy) to verify correct measurement. 25
After all necessary parameters were available, disease activity scores (SDAI, 18 SDAI-Q, CDAI, 19 DAS28-CRP, 28 DAS28-qCRP, DAS28-ESR 29 ) were calculated using the routine laboratory CRP value, qCRP value, and ESR value. For CRP and qCRP values below the lowest detection limit, a value of 0.0 mg/l was used for further statistical analyses. SDAI and SDAI-Q were calculated according to the formula of Smolen et al., 18 CDAI according to the formula of Aletaha et al., 19 DAS28-ESR according to the formula of Prevoo et al., 29 and DAS28-CRP and DAS28-qCRP according to Fransen et al. 28 For the analysis of identical numerical values (see section ‘Outcomes’), all disease activity score values were rounded to one decimal place.
Using SDAI and SDAI-Q, patients were assigned to the following disease activity categories: remission = SDAI ⩽3.3; low disease activity (LDA) = SDAI >3.3 and ⩽11; moderate disease activity (MDA) = SDAI >11 and ⩽26; and high disease activity (HDA) = SDAI >26. 30 Disease activity classification for CDAI was as follows: remission = CDAI ⩽2.8; LDA = CDAI >2.8 and ⩽10; MDA = CDAI >10 and ⩽22; and HDA = CDAI >22. 20 For DAS28-CRP and DAS28-qCRP, the modified DAS28-CRP thresholds from Fleischmann et al.31,32 were used as follows: remission = DAS28-CRP <2.4; LDA = DAS28-CRP ⩾2.4 and ⩽2.9; MDA = DAS28-CRP >2.9 and ⩽4.6; and HDA = DAS28-CRP >4.6. For DAS28-ESR, the original published disease activity criteria were applied: remission = DAS28-ESR <2.6; LDA = DAS28-ESR⩾2.6 and ⩽3.2; MDA = DAS28-ESR >3.2 and ⩽ 5.1; and HDA = DAS28-ESR >5.1.33,34
Outcomes
The primary outcome of this study was the proportion of patients with identical disease activity category assignment by SDAI and SDAI-Q.
Secondary outcome measures were as follows:
the proportion of patients with identical disease activity categories by SDAI and CDAI and by SDAI-Q and CDAI;
the proportion of identical numerical values for SDAI-Q and SDAI, SDAI and CDAI as well as SDAI-Q and CDAI;
the proportion of patients with identical disease activity categories by DAS28-CRP and DAS28-qCRP and by DAS28-ESR and DAS28-CRP;
the proportion of identical numerical values for qCRP and routine laboratory CRP.
Identical numerical values of the different disease activity scores and CRP/qCRP were assumed if the numerical values were matching up to the first decimal place. To analyze the proportion of identical numerical values, values of SDAI, SDAI-Q, CDAI, and different DAS28 scores were rounded to one decimal place.
Sample size
Owing to the pilot project nature of this study, we aimed for a sample size of 100 RA patients to generate results that further projects could build on based on an adequate sample size calculation.
Statistical analysis
Demographic data, clinical and laboratory information as well as disease activity scores were analyzed using methods of descriptive statistics. The intake of non-steroidal anti-inflammatory drugs (NSAIDs) was quantified by calculating the NSAID equivalent score (modified formula according to Dougados et al. 35 ) 23
To analyze the agreement of different disease activity categories, cross tabulations and weighted Cohen’s kappa were calculated for SDAI and SDAI-Q, SDAI and CDAI, SDAI-Q and CDAI, DAS28-CRP and DAS28-qCRP as well as DAS28-CRP and DAS28-ESR.
Bland–Altman plots and intraclass correlation coefficient (ICC) were used to analyze the concordance regarding the numerical values of different disease activity scores and CRP versus qCRP values. For the calculation of ICC and their 95% confidence intervals (CIs), a mean-rating (k = 2), absolute agreement, two-way mixed-effects model was applied. The statistical analyses were implemented using SPSS Statistics (IBM, Armonk, New York, USA, version number 27) and Microsoft Excel (Microsoft Corporation, Redmond, Washington, USA, Office 2019). 23
Patients with missing routine laboratory CRP or qCRP values were not included in the statistical analysis. For patients with missing ESR (and thus missing DAS28-ESR) values, a general statistical analysis was performed, but outcomes including ESR or DAS28-ESR values were only analyzed for patients with available ESR and DAS28-ESR values.
The study was reported according to the STROBE statement. 36 This work was previously presented as a poster at the EULAR congress 2021, 37 at the ‘Deutscher Rheumatologiekongress’ 2021 38 and at the ACR congress 2021. 39
Results
Demographic and clinical characteristics
Altogether, 104 RA patients were eligible and have given written informed consent for study participation. Four of these patients could not be included in the statistical analysis because of missing qCRP values. Availability of both routine laboratory CRP and qCRP values was required to analyze the primary outcome measure. A total of 100 RA patients were included in the statistical analysis with all clinical data, routine laboratory CRP and qCRP values being available, while ESR measurement and DAS28-ESR were available for 99 of these patients. Patients with a previously known anemia were not considered for study participation, so no patient was excluded because of this reason.
Clinical, demographic, and treatment data are shown in Table 1 and Supplementary Table S1. Mean age of the included patients was 60.9 years; mean disease duration 11.4 years and 73 patients were female; 63 patients were rheumatoid factor (RF) positive, 57 patients were positive for anti-cyclic citrullinated peptide antibodies (ACPA) and 49 patients positive for both RF and ACPA. In this study, 40 patients received systemic glucocorticosteroids (GCs), 32 patients were treated with NSAIDs, 75 patients were treated with at least one conventional synthetic disease-modifying anti-rheumatic drug (csDMARD), 15 patients were treated with targeted synthetic disease-modifying anti-rheumatic drug (tsDMARDs) and 39 patients received biological disease-modifying anti-rheumatic drug (bDMARD) therapy.
Demographic, clinical, laboratory, and treatment characteristics.

ACPA, anti-cyclic citrullinated peptide antibodies; bDMARDs, biological disease-modifying anti-rheumatic drugs; CDAI, Clinical Disease Activity Index; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying anti-rheumatic drugs; DAS28, Disease Activity Score 28; DMARDs, disease-modifying anti-rheumatic drugs; ESR, erythrocyte sedimentation rate; GC, glucocorticosteroid; JAK-I, Janus kinase inhibitor; NSAIDs, non-steroidal anti-inflammatory drugs; qCRP, quick quantitative C-reactive protein; RF, rheumatoid factor; SD, standard deviation; SDAI, Simplified Disease Activity Index; SDAI-Q, SDAI calculated with a qCRP assay; tsDMARDs, targeted synthetic disease-modifying anti-rheumatic drugs.
n = 100; for ESR and DAS28-ESR, n = 99.
ACPA and RF status were quoted according to the local laboratory of each center.
Comparison of routine laboratory CRP and qCRP
The mean value of the routine laboratory CRP and qCRP was 7.0 ± 15.0 mg/l and 7.9 ± 17.0 mg/l, respectively (Table 1). Both measurement methods provided identical numerical CRP values in 12 of 100 patients. The mean difference of both CRP measurement methods was 0.9 ± 2.7 mg/l; observed differences between routine laboratory CRP and qCRP ranged from −4.3 to 17.2 mg/l. A Bland–Altman plot for the routine laboratory CRP and qCRP shows the agreement of numerical values by both measurement methods (Figure 1).
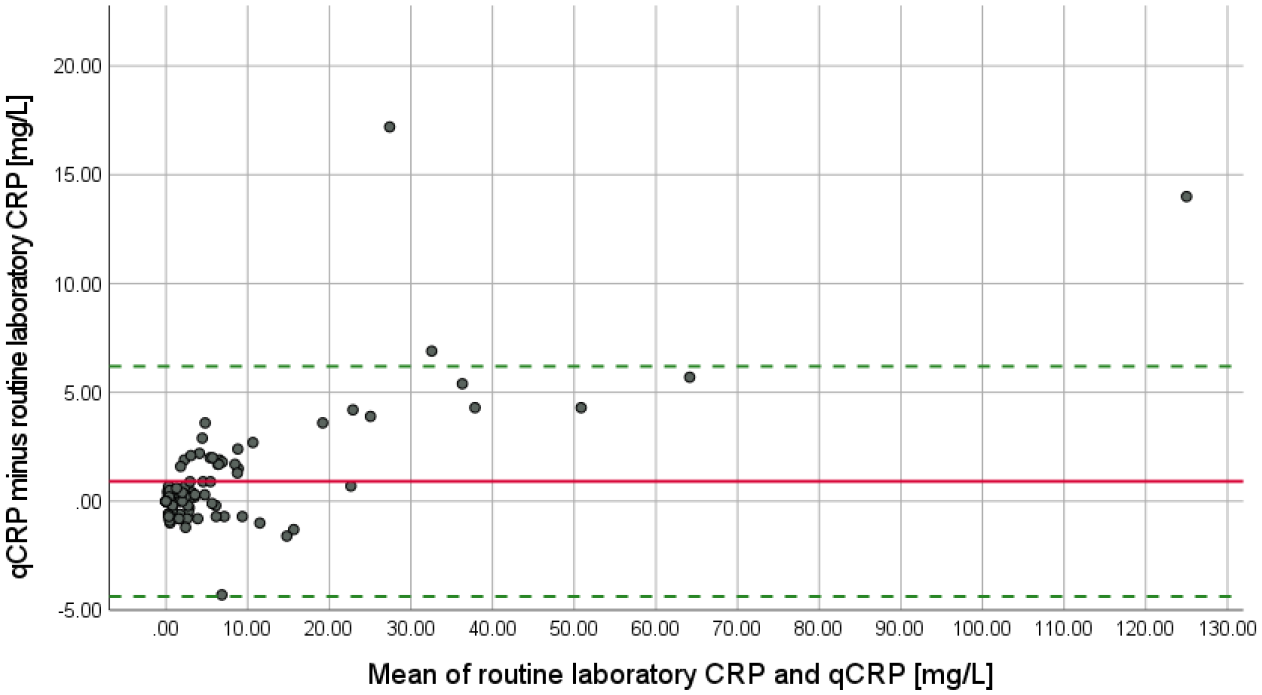
Bland–Altman plot for routine laboratory CRP and qCRP. The solid line marks the mean difference between routine laboratory CRP and qCRP (0.915 mg/l). The dotted lines indicate the 95% limits of agreement (−4.372; 6.202 mg/l).
The 95% limits of agreement were located at −4.4 and 6.2 mg/l. ICC for routine laboratory CRP and qCRP was 0.992 (95% CI = 0.987; 0.995) (Table 2).
ICC for different CRP measurement methods and disease activity scores.
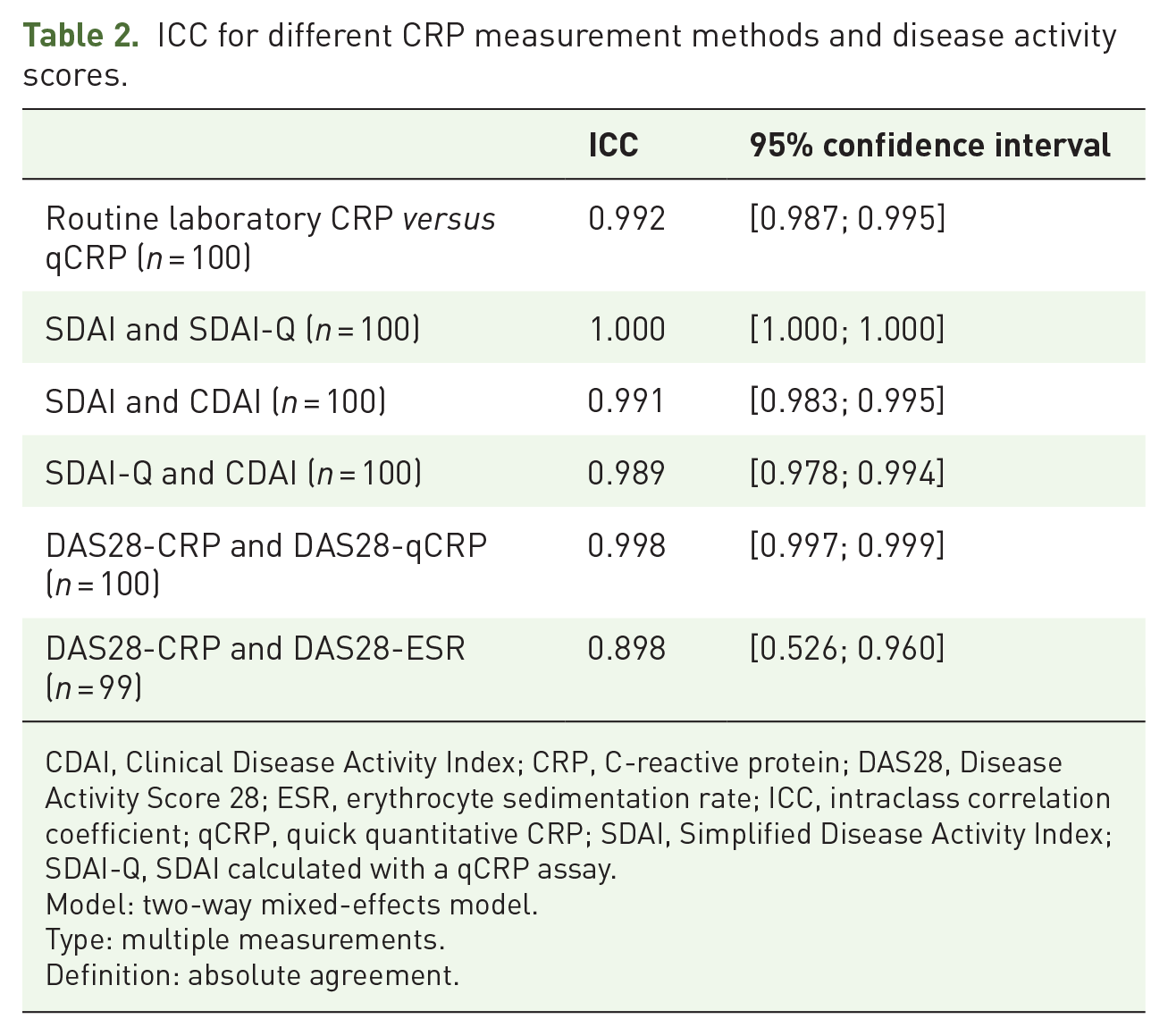
CDAI, Clinical Disease Activity Index; CRP, C-reactive protein; DAS28, Disease Activity Score 28; ESR, erythrocyte sedimentation rate; ICC, intraclass correlation coefficient; qCRP, quick quantitative CRP; SDAI, Simplified Disease Activity Index; SDAI-Q, SDAI calculated with a qCRP assay.
Model: two-way mixed-effects model.
Type: multiple measurements.
Definition: absolute agreement.
Comparison of SDAI, SDAI-Q, and CDAI (disease activity categories, numerical values)
Comparing SDAI and SDAI-Q, all patients were assigned to identical disease activity categories as shown in Table 3A. Weighted Cohen’s kappa was 1.000 (95% CI = 1.000; 1.000).
(A) Disease activity categories by SDAI-Q versus SDAI and (B) disease activity categories by SDAI-Q versus CDAI.

CDAI, Clinical Disease Activity Index; CI, confidence interval; SDAI, Simplified Disease Activity Index; SDAI-Q, SDAI calculated with a quick quantitative C-reactive protein assay.
Fields highlighted in green indicate matching disease activity categories according to both methods. Fields highlighted in red indicate that disease activity categories do not match. Percentage values refer to the entire study population included in statistical analysis.
The SDAI-Q provided identical values as the SDAI in 37 of 100 patients when values were rounded to one decimal. Mean of the differences between SDAI and SDAI-Q was 0.1 ± 0.3 with observed differences ranging from −0.4 and 1.7. ICC for SDAI and SDAI-Q was 1.000 (95% CI = 1.000; 1.000) (Table 2). The agreement of numerical values by SDAI and SDAI-Q is graphically illustrated in a Bland–Altman plot (Figure 2).

Bland–Altman plot for SDAI and SDAI-Q. The solid line marks the mean difference between SDAI and SDAI-Q (0.092). The dotted lines indicate the 95% limits of agreement (−0.437; 0.620). X- and Y-axis indicate the ‘mean of SDAI-Q and SDAI’ as well as ‘SDAI-Q minus SDAI’ in SDAI points.
Table 3B shows that 7 of 100 patients differed in their disease activity categories with CDAI compared with SDAI/SDAI-Q. In five of these seven cases, patients were assigned to a higher disease activity category when using the CDAI – all of these five patients had CRP and qCRP values of ⩽6.6 mg/l and CDAI values close to the cut-offs for remission/LDA (2.8) or LDA/MDA (10). The two patients with lower disease activity categories by CDAI compared with SDAI/SDAI-Q had high CRP and qCRP values (⩾29.1 mg/l) (Supplementary Table S2). Weighted Cohen’s kappa for disease activity categories by CDAI and SDAI/SDAI-Q was 0.929 (95% CI = 0.878; 0.981) (Table 3B).
Identical values by CDAI and SDAI were seen in 15 patients and by CDAI and SDAI-Q in 18 patients when values were rounded to one decimal. Mean of the differences between CDAI and SDAI was 0.7 ± 1.5 with differences ranging between 0.0 and 11.8; for CDAI and SDAI-Q, the mean of the differences was 0.8 ± 1.7 with differences ranging from 0.0 to 13.2. ICC for SDAI and CDAI was 0.991 (95% CI = 0.983; 0.995) and for SDAI-Q and CDAI 0.989 (95% CI = 0.978; 0.994), also shown in Table 2. A Bland–Altman plot illustrates the agreement of numerical values by SDAI-Q and CDAI (Figure 3).

Bland–Altman plot for SDAI-Q and CDAI.
Comparison of DAS28-CRP, DAS28-qCRP, and DAS28-ESR (disease activity categories, numerical values)
In 99 of all 100 patients, DAS28-qCRP provided identical disease activity categories in comparison with DAS28-CRP with a weighted Cohen’s kappa of 0.991 (95% CI = 0.973; 1.000) (Supplementary Table S3). The patient with different disease activity categories showed a lower disease activity category when using the DAS28-qCRP: LDA by DAS28-qCRP (2.74) versus MDA by DAS28-CRP (2.95); routine laboratory CRP was 0.8 mg/l and the qCRP value was below the lowest detection limit. Identical numerical values with DAS28-CRP and DAS28-qCRP occurred in 43 of 100 patients when values were rounded to one decimal. The mean difference of DAS28-CRP and DAS28-qCRP was 0.01 ± 0.10, with differences ranging from −0.25 to 0.23.
With DAS28-ESR, 29 of 99 patients (29.3%) showed a different disease activity category in comparison with DAS28-CRP with a weighted Cohen’s kappa of 0.707 (95% CI = 0.611; 0.802) (Supplementary Table S4). Within this group, 21 patients had a higher disease activity category with DAS28-ESR, one patient showed a discrepancy of two disease activity categories higher (MDA versus remission) with DAS28-ESR compared with DAS28-CRP. In eight patients, the DAS28-CRP (using the modified Fleischmann cut-off values) resulted in a higher disease activity category in comparison with DAS28-ESR and one patient was assigned to a disease activity category of two levels higher by DAS28-CRP. Identical numerical values of DAS28-ESR and DAS28-CRP were seen in 3 of 99 (3.0%) patients when values were rounded to one decimal. The mean difference of DAS28-CRP and DAS28-ESR was 0.50 ± 0.55, with the mean of DAS28-ESR being slightly higher and showing differences ranging from −1.71 to 1.49.
ICC values of different DAS28 scores are presented in Table 2. Bland–Altman plots illustrating the agreement of DAS28-CRP and DAS28-qCRP values as well as DAS28-CRP and DAS28-ESR values are shown in Supplementary Figures S1 and S2.
Discussion
This study investigated the performance of disease activity scores based on a validated qCRP measurement (SDAI-Q, DAS28-qCRP) compared with scores using routine laboratory CRP in a typical cohort of RA patients. Regarding the assignment to disease activity categories, the SDAI-Q showed an absolute agreement with the conventional SDAI. The DAS28-qCRP provided an almost perfect agreement of disease activity categories compared with the conventional DAS28-CRP.
According to this study, the qCRP measurement tended to yield slightly higher numerical values than routine laboratory CRP with a mean difference of 0.9 mg/l. This was especially the case for generally high CRP values (illustrated in the respective Bland–Altman plot, Figure 1): above the mean value of 20 mg/l for both CRP measurement methods, qCRP always resulted in higher numerical values than routine laboratory CRP. This observation had no effect on the disease activity assignment by SDAI-Q in comparison with SDAI, however, as all patients achieved the same disease activity category. In the only patient with different disease activity categories by DAS28-qCRP and DAS28-CRP (DAS28-qCRP: 2.74, LDA; DAS28-CRP: 2.95; MDA), routine laboratory CRP here showed a higher numerical value of 0.8 mg/l in comparison with the qCRP that was beneath the lowest detection limit. Remarkably, the DAS28-CRP value of this patient lies with 2.95 very close to the cut-off value between LDA and MDA of 2.90 according to Fleischmann et al. 32 Overall, the discrepancy between qCRP and routine laboratory CRP demonstrated no relevant clinical impact on the allocation of disease activity categories in our cohort of RA patients.
According to our data, the CDAI showed a very good agreement with SDAI/SDAI-Q regarding the assignment to identical disease activity categories (93 of 100 patients). In five patients, CDAI resulted in a one level higher disease activity category than SDAI/SDAI-Q. All of these patients had low CRP values and therefore similar SDAI, SDAI-Q, and CDAI values, all close to the cut-off values between remission versus LDA and LDA versus MDA (Supplementary Table S2). This different classification of these five patients could be well explained by the slightly lower cut-off values of CDAI in comparison with SDAI. Higher disease activity categories by SDAI/SDAI-Q compared with CDAI were seen in two patients with high CRP values. Consequently, a relevant numerical difference between SDAI and CDAI values occurred in these two patients. Summarizing, CDAI showed a very good agreement with SDAI in the majority of RA patients, but led to a different estimation of disease activity in 7.0% which was not the case for SDAI-Q.
A randomized controlled trial by Hodkinson et al. investigated the performance of a T2T strategy targeting LDA using SDAI or CDAI in a resource-constrained setting. The authors found no significant differences in achieving DAS28 remission and LDA at 12 months in both groups, but did not compare the agreement of disease activity categories by SDAI and CDAI in individual patients. 40 In the study establishing CDAI cut-off values, Aletaha and Smolen 20 showed identical disease activity categories by SDAI and CDAI in 90.4% of patients, a similar proportion compared with our results. Another study by Martins et al. investigated the concordance of disease activity categories between SDAI and CDAI in a large cohort of RA patients. The study showed a disagreement of SDAI and CDAI disease activity classification between 12.8% and 20.4% depending on the analyzed disease activity category, 21 notably higher than in this study.
The agreement of disease activity categories by DAS28-qCRP in comparison with DAS28-CRP was almost perfect (99%). In this study, we used modified DAS28-CRP cut-off values according to Fleischmann et al., which accounted for the systematically lower numerical values of DAS28-CRP compared with DAS28-ESR31,32 as observed in this study (Table 1) and several other studies.31,41,42 Nevertheless, a substantial proportion of patients (29%) was classified differently using DAS28-ESR compared with DAS28-CRP, and more patients were assigned to the ‘remission’ status using DAS28-CRP. Consequently, DAS28-CRP and DAS28-ESR should not be used interchangeably and the DAS28-qCRP may serve as a timely available alternative to DAS28-CRP. It is important to note that the DAS28 scores in general are no longer recommended for assessing remission in RA patients, however.17,32
The limitations of this study include that all study centers were located in Berlin, having 3.7 million inhabitants. 43 Patients were recruited from different private outpatient practices, and specialized academic centers and different local laboratories were involved, however. As a consequence, we would not assume different results with study centers located at different places in Germany. 23 Further notable limitations are the cross-sectional design of this study as well as the rather small sample size of 100 RA patients. Nevertheless, our recent work can be seen as a proof of concept, and the performance of the SDAI-Q should be further investigated in larger studies and also assessed for its sensitivity to change in longitudinal studies. Another limitation of the presented study is that patients with a pre-known clinically significant anemia (hemoglobin-concentration below 10 g/dl) before study enrollment could not be included in the study because of the exclusion criteria. The rationale for this exclusion criterion was to avoid additional blood sampling in patients with a previously known anemia, when whole blood from the collection system was no more available for the qCRP measurement. A further potential limitation of this study is that the qCRP assay used in this study gives accurate results only for hematocrit concentrations between 15% and 75% and that the qCRP measurement range depends on the hematocrit concentration, 25 whereas an exclusion because of a hematocrit concentration out of this range will only rarely occur with limited clinical relevance.
Conclusion
In conclusion, the SDAI-Q could be validated in this study, showing absolute agreement with SDAI regarding disease activity classification. Determination of qCRP only requires one drop of blood (e.g. from the finger berry before the physician’s appointment) and can provide current CRP values within a few minutes after sampling, making SDAI-Q a timely available, easy to perform disease activity score involving an acute-phase reactant. The SDAI-Q may therefore facilitate the implementation of a T2T approach in clinical trials and can be also used in clinical routine.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221114107 – Supplemental material for Validation of the Simplified Disease Activity Index (SDAI) with a quick quantitative C-reactive protein assay (SDAI-Q) in patients with rheumatoid arthritis: a prospective multicenter cross-sectional study
Supplemental material, sj-docx-1-tab-10.1177_1759720X221114107 for Validation of the Simplified Disease Activity Index (SDAI) with a quick quantitative C-reactive protein assay (SDAI-Q) in patients with rheumatoid arthritis: a prospective multicenter cross-sectional study by Julia Schally, Henning Christian Brandt, Jan Brandt-Jürgens, Gerd R. Burmester, Hildrun Haibel, Henriette Käding, Kirsten Karberg, Susanne Lüders, Burkhard Muche, Mikhail Protopopov, Valeria Rios Rodriguez, Murat Torgutalp, Maryna Verba, Silke Zinke, Denis Poddubnyy and Fabian Proft in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors deeply thank Braun T, Doerwald C, Deter N, Höppner C, Igel S, Lackinger J, Lorenz C, Lunkwitz K, Mandt B, and Zernicke J for their practical support and coordinating the study.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

