Abstract
Background:
Inflammatory processes play a central role in rheumatoid arthritis (RA), involving complex interactions among cytokines and blood cell components. Current clinical tools, including the Disease Activity Score (DAS28), and biomarkers such as erythrocyte sedimentation rate and C-reactive protein (CRP), have limitations in monitoring disease activity.
Objectives:
This study aimed to assess the utility of routine hematological parameters as potential biomarkers for diagnosing RA and evaluating disease activity in drug-naïve patients.
Methods:
In this single-center retrospective study, 486 RA patients and 301 age- and sex-matched healthy controls were analyzed. Drug-naïve RA patients (not treated with disease-modifying antirheumatic drugs or glucocorticoids) were selected according to the 2010 American College of Rheumatology/European League Against Rheumatism criteria. Hematological parameters (white blood cell count and differential, hemoglobin (HGB), platelet count (PLT), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), red cell distribution width (RDW), and mean platelet volume (MPV)) were obtained and calculated from complete blood counts. Disease activity was assessed using the 28-joint DAS28 with CRP. Diagnostic accuracy and association with disease activity were evaluated using receiver operating characteristic (ROC) analysis, correlation tests, and logistic regression.
Results:
Compared with controls, RA patients exhibited distinct hematological profiles, with significantly higher NLR, PLR, and RDW, and lower MPV (all p < 0.001). Multivariate logistic regression identified MPV (odds ratio (OR): 1.315, 95% confidence interval (CI): 1.085–1.594, p = 0.005) and age (OR: 0.984, 95% CI: 0.969–0.999, p = 0.038) as independent factors associated with moderate-to-high disease activity. MPV showed significant inverse correlations with DAS28-CRP (r = −0.098, p = 0.031) and CRP (r = −0.148, p = 0.001). ROC analysis demonstrated that NLR had a modest performance (area under the curve (AUC): 0.676) for distinguishing RA patients from controls, and its performance in differentiating disease activity levels was weak (AUC: 0.598).
Conclusion:
This study suggests that hematological indices, particularly NLR, PLR, and MPV, hold potential as supplementary biomarkers in RA, but not standalone diagnostic tools. MPV emerged as an independent marker associated with higher disease activity, yet its clinical significance requires further validation. These readily available parameters may be integrated as complementary components of a broader diagnostic and monitoring strategy.
Plain language summary
Rheumatoid arthritis (RA) is an inflammatory rheumatic disease. Diagnosing it and monitoring how active it is often relies on clinical exams and specialized blood tests, which can be costly or not readily available. Simple, routine blood cell counts are cheap and available everywhere, but their role in RA is not fully defined. This study investigated whether these simple blood test results could be used as biomarkers to help identify RA and determine how active the disease is, specifically in patients who have not yet started any treatment. We compared the blood test results of 486 treatment-naive RA patients with those of 301 healthy individuals. We looked at different types of white blood cells, platelets, and their ratios. We then analyzed how these results related to standard measures of RA disease activity. We found that RA patients have a distinct “inflammatory signature” in their blood tests. Specifically, ratios like the Neutrophil-to-Lymphocyte Ratio (NLR) and Platelet-to-Lymphocyte Ratio (PLR) were significantly higher in RA patients than in healthy people. This means these simple ratios could be useful as an initial, low-cost tool to support an RA diagnosis. A key finding was that Mean Platelet Volume (MPV) was independently linked to more active disease. This research shows that inexpensive and readily available blood tests, particularly the NLR, PLR, and MPV, provide valuable information about the inflammation in RA. They can serve as useful supporting tools for diagnosis. Using these existing parameters could lead to more efficient and cost-effective management of rheumatoid arthritis.
Introduction
Rheumatoid arthritis (RA) is the most prevalent chronic form of inflammatory arthritis, affecting approximately 1% of the global population. 1 Inflammation represents a pivotal component of RA pathogenesis, with inflammatory markers typically elevated and correlated with increased disease activity.2–4
Chronic inflammatory diseases are characterized by immune system dysregulation and persistent inflammation. Immune system components, including neutrophils (NEU), lymphocytes (LYM), monocytes (MONO), and platelets (PLT), play critical roles in modulating systemic inflammation and undergo alterations in both number and composition during inflammatory states. The interactions between cytokines and blood cells are fundamental to immune system regulation and coordination. Local and systemic inflammatory processes induce cellular activation, and the bidirectional communication between cytokines and blood cell counts amplifies this inflammatory response. Neutrophils and platelets contribute to the production of inflammatory cytokines, which, in turn, activate these cells. 5 The increased presence of neutrophils and platelets may be attributed to the inflammatory milieu in RA, characterized by elevated levels of inflammatory cytokines. 6 Changes in lymphocyte counts, particularly T cells, can influence the levels of regulatory cytokines 7 that maintain immune balance and prevent excessive inflammation. Monocytes, which differentiate into macrophages and dendritic cells, are associated with the production of cytokines such as interleukin-1β, interleukin-6 (IL-6), and tumor necrosis factor-alpha. 8 Platelets are rich in proinflammatory mediators and can release bioactive microparticles, with IL-6 playing a key role in this process. 9
Several clinical tools are currently used to monitor disease activity, including the Disease Activity Score, the 28-joint Disease Activity Score (DAS28), and the Simplified Disease Activity Index. 10 Despite the widespread use of erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) in evaluating disease activity and guiding RA treatment, these markers have notable limitations. 11 A previous study assessing the correlation between histological evidence and ESR and CRP reported that their sensitivities in detecting synovial inflammation were 71.3% and 64.1%, respectively. 12 The DAS28 has been widely applied in clinical trials to monitor disease activity in patients with RA. However, each parameter is influenced by inherent confounders, as the tender and swollen joint count (SJC) scores depend on individual pain tolerance and the examiner’s assessment skills. Moreover, the patient’s global health score represents a subjective measure. 12 Therefore, additional alternative approaches are needed to better reflect the degree of inflammation in patients with RA. Recognizing these limitations, ongoing research continues to seek novel biomarkers that can predict disease activity and progression.
Considering that a complete blood count (CBC) is a simple and inexpensive laboratory test, the use of CBC-derived parameters could be valuable in assessing disease activity in RA. Previous studies have reported lower hemoglobin (HGB) levels 13 and mean platelet volume (MPV),14,15 but higher neutrophil-to-lymphocyte ratio (NLR)15,16 and platelet counts 14 in patients with RA exhibiting high disease activity compared with those in disease remission. One study also identified an elevated NLR as a predictor of RA relapse. 16 Recently, hematological parameters such as the NLR, platelet-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio (LMR) have been increasingly used as markers of systemic inflammation. Several studies have demonstrated that NLR and PLR were significantly higher in patients with RA than in healthy controls.17,18 Moreover, NLR and PLR were positively correlated with disease activity and inflammatory parameters and were found to predict treatment responses in patients with RA. 19 An association has also been reported between LMR and disease activity in RA. 20 Furthermore, alterations in the volume and distribution of key blood cell types, including platelets and red blood cells, are known to reflect the presence of a proinflammatory state and oxidative stress.21–25 However, these studies were limited by small sample sizes and demonstrated only modest predictive performance.
Given these findings, it remains unclear whether the routinely performed CBC, a test ordered for nearly every patient at each clinical visit, can provide additional insight into the diagnosis and activity of RA.
Therefore, this study aimed to determine whether routine hematological markers and systemic inflammatory ratios (such as NLR and PLR) can serve as useful biomarkers for both diagnosing RA and assessing disease activity in drug-naïve patients.
Methods
Study design and participants
This single-center retrospective study included 486 patients diagnosed with RA between March 2024 and March 2025 at Balıkesir Ataturk City Hospital, a tertiary care center. A control group consisting of 301 individuals was also included.
All patients with RA were evaluated by an experienced rheumatologist and diagnosed according to the 2010 American College of Rheumatology/European League Against Rheumatism classification criteria. 26 The sample size was determined based on previous studies27–29 to ensure adequate statistical power for detecting significant associations between hematological markers and disease severity in patients with RA (with 90% power and alpha of 0.05, a total of 360 participants (180 per group) are required). All participants were newly diagnosed at our clinic. Patients who had not previously received disease-modifying antirheumatic drugs (DMARDs) or glucocorticoids were included in the study and were defined as drug-naïve. Prior use of nonsteroidal anti-inflammatory drugs was not considered an exclusion criterion.
The control group consisted of patients evaluated in the rheumatology clinic for degenerative musculoskeletal conditions without any inflammatory rheumatic disease. Age- and sex-matching were applied during selection. Regression analysis confirmed that the study groups were well-matched for age and sex, as there was no statistically significant multicollinearity between these variables (Tolerance: 1.0, Variance Inflation Factor (VIF): 1.0 for both), indicating their independence in the model.
Exclusion criteria included patients with other autoimmune diseases, such as systemic lupus erythematosus, vasculitis, scleroderma, or seronegative spondyloarthropathy. Patients with acute or chronic infections, hematological abnormalities, or chronic renal failure were also excluded, as these conditions could affect hematological indices.
All laboratory tests were performed in the hospital’s institutional laboratory.
This study adhered to the ethical principles outlined in the Declaration of Helsinki and was approved by the Human Research Ethics Committee of Balıkesir Atatürk City Hospital (approval no: 2025/07/73, date: July 24, 2025).
Data and sample collection
Demographic, clinical, and laboratory data were anonymously extracted from patient records. Data from consecutive patients newly diagnosed with RA at our center and not previously treated between March 1, 2024, and March 1, 2025, were analyzed.
A comprehensive physical examination was performed by an experienced rheumatologist, and parameters including the SJC, tender joint count (TJC), and patient global assessment using a visual analog scale (VAS) were recorded. The DAS28-CRP was calculated for each patient, incorporating TJC, SJC, CRP, and patient global health scores. 30 Disease activity was classified as moderate to high (DAS28 ⩾3.2) or low (DAS28 <3.2). Venous blood samples (8 mL, with and without anticoagulant) were collected from each patient for hematological parameter analysis and serological testing.
The primary outcomes included hematological parameters such as white blood cell (WBC) count and differential counts (neutrophil, lymphocyte, monocyte, HGB, and platelet counts); NLR; PLR; red cell distribution width (RDW); and MPV. Platelet histograms generated from the automated hematology analyzer were used to derive MPV as a marker of platelet activation. All CBC analyses were performed within 1 week before or after DAS28-CRP measurement using an automated hematology analyzer (Sysmex XN 1000, Kobe, Japan).
Rheumatoid factor (RF) levels were measured using nephelometric analysis (Siemens Dade Behring N2, Marbrug, Germany), with values above 15 IU/mL considered positive. Anti-cyclic citrullinated peptide (anti-CCP) antibodies were measured by enzyme-linked immunosorbent assay (Cobas e411, Roche Diagnostics, Mannheim, Germany), with levels above 17 IU/mL defined as positive. CRP was also measured by ELISA, and results were expressed in mg/dL.
Extra-articular manifestations included ocular involvement (episcleritis, scleritis, keratitis, uveitis, keratoconjunctivitis sicca), subcutaneous nodules, pleuritis, interstitial lung disease, pulmonary nodules, pericarditis, neurological involvement (entrapment neuropathy, peripheral neuropathy), and vasculitis.
Comorbidities evaluated in the study included hypertension, diabetes mellitus, asthma/chronic obstructive pulmonary disease, coronary artery disease, and malignancy.
Statistical analysis
Data were analyzed using IBM SPSS Statistics for Windows, version 20.0 (IBM Corp., Armonk, NY, USA). Continuous variables are presented as median, range (minimum–maximum), or as frequencies (number of cases) and percentages, as appropriate. The normality of data distribution was assessed using the Kolmogorov–Smirnov test. Comparisons between two subgroups were performed using Student’s t test or the nonparametric Mann–Whitney U test, as appropriate.
For comparisons among more than two groups, one-way analysis of variance was applied. Correlations between laboratory variables were evaluated using Spearman’s or Pearson’s correlation coefficients. Receiver operating characteristic (ROC) curves were constructed for significant hematological parameters, and the area under the curve (AUC) was calculated to determine the optimal cutoff value for each parameter. A p-value <0.05 was considered statistically significant.
The reporting of this study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 31 (Supplemental Material).
Results
The study included 486 patients with RA and 301 control subjects. The demographic and clinical characteristics of the participants are presented in Table 1.
Clinical characteristics and laboratory data of patients with RA and comparison with the control group.

CCP, cyclic citrullinated peptide; CRP, C-reactive protein; DAS28, Disease Activity Score 28; HGB, hemoglobin; LYM, lymphocyte; MONO, monocyte; MPV, mean platelet volume; NEU, neutrophil; NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; PLT, platelet; RA, rheumatoid arthritis; RDW, red cell distribution width; RF, rheumatoid factor; SJC, swollen joint count; TJC, tender joint count; VAS, visual analog scale; WBC, white blood cell count.
Demographics and clinical characteristics
The median age of patients with RA was 58 years (interquartile range (IQR): 50–67), while the control group had a median age of 58 years (IQR: 50–64). There was no statistically significant difference in age between the two groups (p = 0.175). Among the RA group, 365 patients (75%) were females and 121 (25%) were males. In the control group, 220 participants (73%) were females and 81 (27%) were males, with no significant difference in sex distribution between the groups (p = 0.304).
The median symptom duration among patients with RA was 1 year (IQR: 0.3–2). A total of 342 patients (70.4%) tested positive for RF and/or anti-CCP antibodies and were classified as having seropositive RA.
Extra-articular manifestations were observed in 61 patients (12.6%). Regarding disease activity, 359 patients (73.9%) were classified as having moderate-to-high disease activity based on DAS28-CRP scores.
Disease activity and laboratory findings
The median TJC was 4 (IQR: 3–6), and the median SJC was 4 (IQR: 2–5) among patients with RA. The median VAS score for pain was 6 (IQR: 6–7). The median DAS28-CRP was 3.61 (IQR: 3.23–4.12). Among patients with RA, the median CRP level was 1.1 mg/dL (IQR: 0.8–2.3).
Hematological parameters differed significantly between patients with RA and the control group. Patients with RA had significantly higher counts of WBC, NEU, MONO, and PLT, along with lower LYM counts and HGB levels compared with controls (all p < 0.001). In addition, the MPV was significantly lower, whereas the RDW was significantly higher in patients with RA.
Other inflammatory indices, including the NLR, were significantly higher in patients with RA (median 2.36 vs 1.0, p < 0.0001). The PLR was also markedly higher in patients with RA (median 138.29 vs 112.15, p < 0.0001).
Comparison of disease activity subgroups and controls
To explore the association between hematological parameters and disease activity, patients were stratified according to disease activity status. Post hoc Bonferroni analysis showed that several parameters—including NEU, MONO, PLT, and MPV—differed significantly between patients with moderate-to-high disease activity and the control group. Notably, MPV and NEU also differed significantly between patients with low and moderate-to-high disease activity (p = 0.014 and p = 0.015, respectively). NLR and PLR were not significant in distinguishing disease activity levels. Elevated monocyte and platelet counts differentiated patients with RA from controls, suggesting an ongoing inflammatory state (Table 2).
Post hoc Bonferroni test was performed to determine the differences between the disease activity levels of RA patients and the control group.
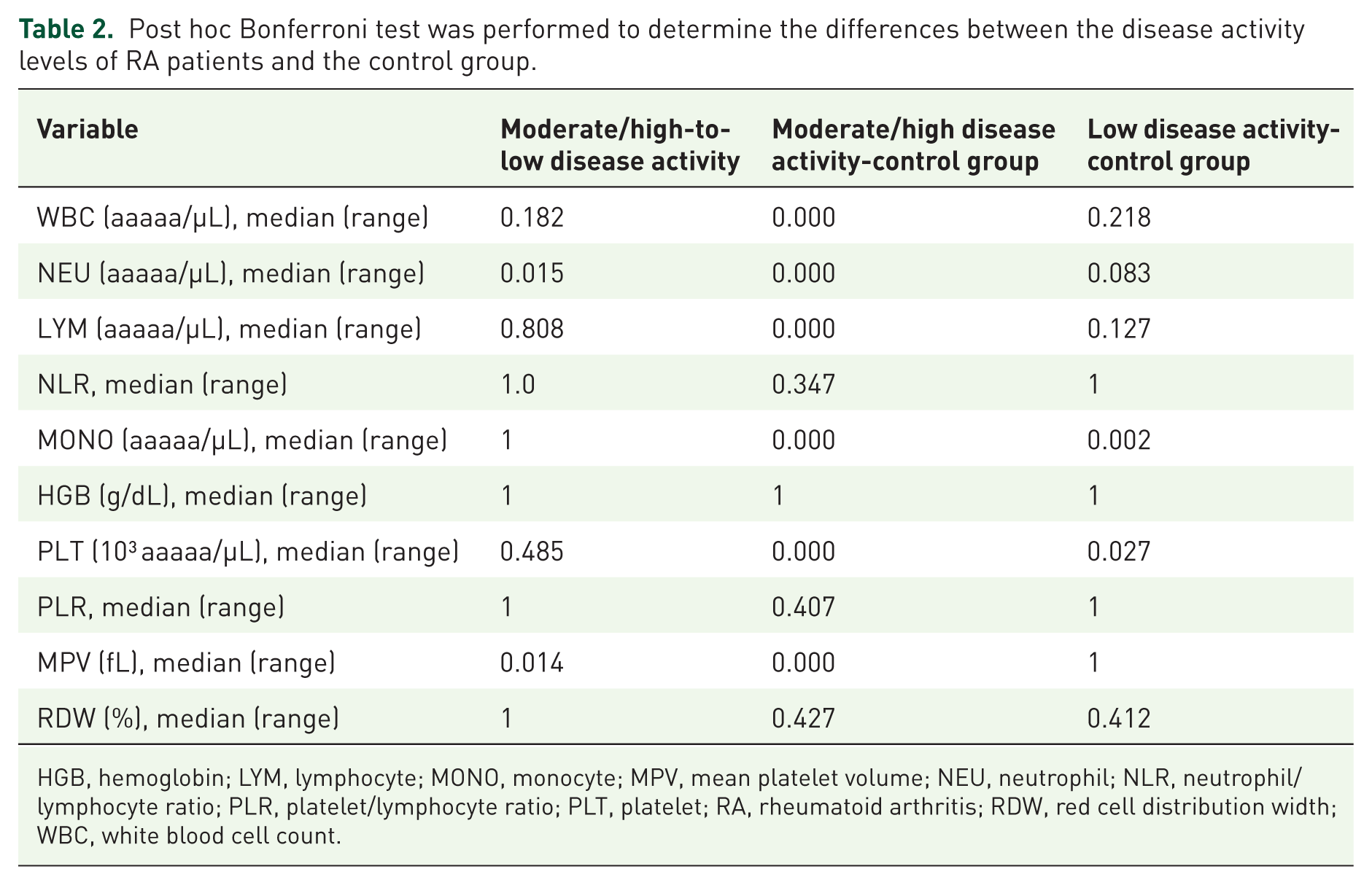
HGB, hemoglobin; LYM, lymphocyte; MONO, monocyte; MPV, mean platelet volume; NEU, neutrophil; NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; PLT, platelet; RA, rheumatoid arthritis; RDW, red cell distribution width; WBC, white blood cell count.
Univariate and multivariable logistic regression analyses of factors associated with moderate-to-high disease activity in RA patients
Univariate and multivariable logistic regression analyses were conducted to identify factors independently associated with moderate-to-high disease activity (Table 3). Age (odds ratio (OR) = 0.98; 95% confidence interval (CI): 0.97–1.00; p = 0.035) and MPV (OR = 1.29; 95% CI: 1.04–1.60; p = 0.020) were significantly associated with disease activity. After adjustment for confounding variables, MPV remained independently associated with moderate-to-high disease activity (adjusted OR = 1.32; 95% CI: 1.09–1.59; p = 0.005). Increasing age was independently associated with a lower likelihood of moderate-to-high disease activity (adjusted OR = 0.98; 95% CI: 0.97–1.00; p = 0.038).
Univariate and multivariable logistic regression analyses of factors associated with moderate-to-high disease activity in RA patients.

CI, confidence interval; HGB, hemoglobin; LYM, lymphocyte; MONO, monocyte; MPV, mean platelet volume; NEU, neutrophil; NLR, neutrophil/lymphocyte ratio; OR, odds ratio; PLR, platelet/lymphocyte ratio; PLT, platelet; RA, rheumatoid arthritis; RDW, red cell distribution width; WBC, white blood cell count. p values in bold are statistically significant.
Correlation of hematological indices with disease activity (DAS28-CRP) and CRP
The relationships between hematological parameters and disease activity, as measured by DAS28-CRP, were evaluated using Spearman’s correlation analysis. As summarized in Table 4, several parameters demonstrated weak correlations with disease activity. Lymphocyte count showed a weak negative correlation with DAS28-CRP (ρ = −0.130, p = 0.004), as did MPV (ρ = −0.098, p = 0.031). In correlation analyses with CRP levels, both neutrophil count (ρ = 0.322, p < 0.001) and NLR (ρ = 0.319, p < 0.001) showed moderate positive correlations with CRP, representing the strongest relationships observed in the study. Scatter plots of these associations are presented in Figures 1 and 2.
Correlation of hematological parameters with DAS28-CRP and CRP in RA patients.
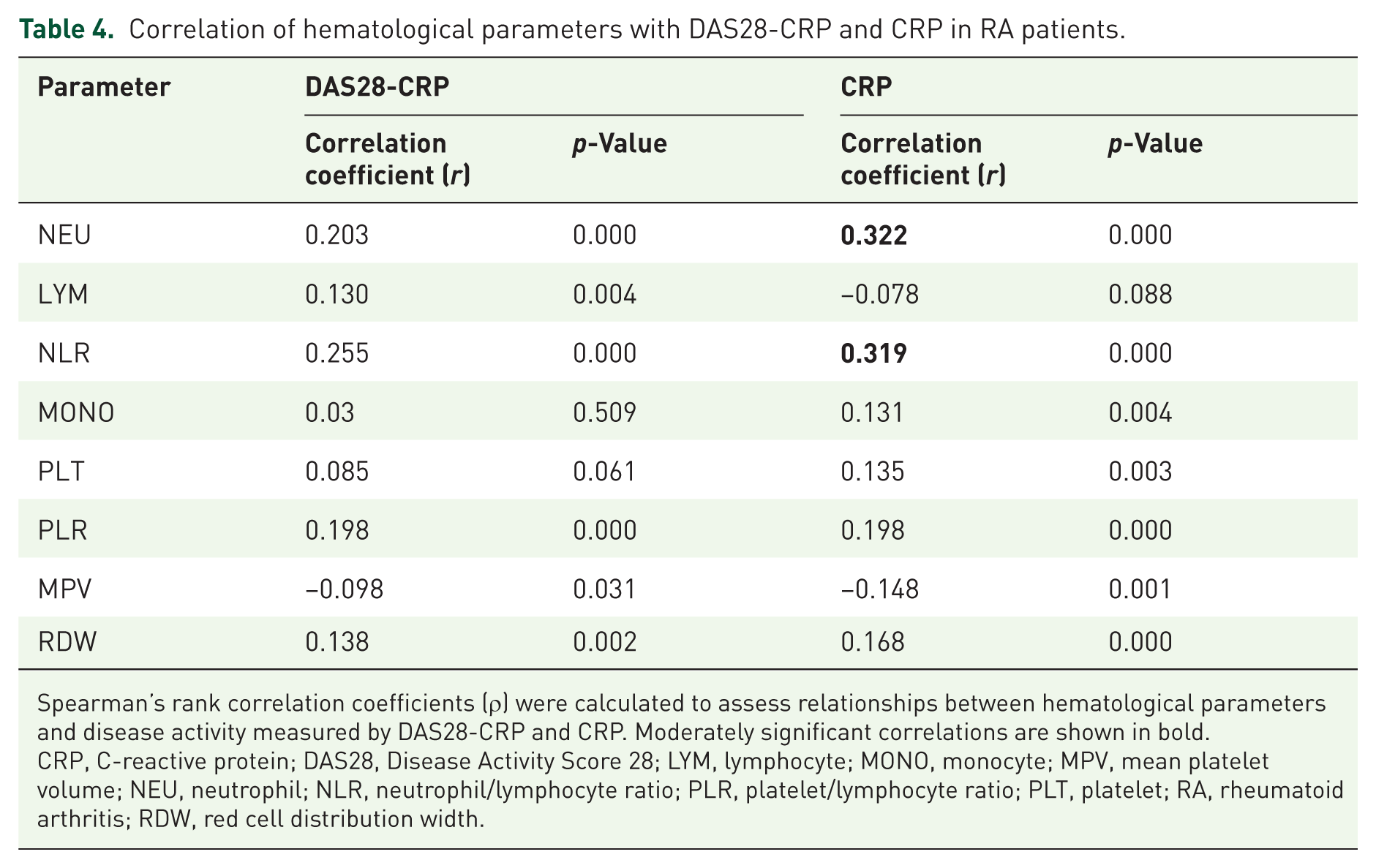
Spearman’s rank correlation coefficients (ρ) were calculated to assess relationships between hematological parameters and disease activity measured by DAS28-CRP and CRP. Moderately significant correlations are shown in bold.
CRP, C-reactive protein; DAS28, Disease Activity Score 28; LYM, lymphocyte; MONO, monocyte; MPV, mean platelet volume; NEU, neutrophil; NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; PLT, platelet; RA, rheumatoid arthritis; RDW, red cell distribution width.

Spearman correlation analysis was applied to estimate the associations of hematological parameters with DAS28-CRP in RA patients. Scatter plot correlations of DAS28-CRP with NLR (a), PLR (b), and MPV (c). Correlation coefficients and p-values are presented in every scatter plot.

Spearman’s correlation analysis was applied to estimate relationships in RA patients. Scatter plot correlations of CRP (mg/dL) with NLR (a), PLR (b), and MPV (c). Correlation coefficients and p-values are presented in every scatter plot.
Diagnostic and discriminative performance of hematological parameters
ROC curve analysis was performed to assess the diagnostic utility of various hematological parameters in RA (Figure 3). Among these parameters, the NLR demonstrated the highest discriminatory ability, with an AUC of 0.676 (95% CI: 0.639–0.714; cutoff = 2.33), followed by the PLR with an AUC of 0.662 (95% CI: 0.624–0.700; cutoff = 131.11). NEU, MONO, and RDW exhibited weak diagnostic performance, with AUC values ranging from 0.612 to 0.622. By contrast, LYM, HGB, and MPV were associated with AUC values below 0.5, indicating an inverse relationship with RA (Table 5).

ROC curves comparing the discriminatory ability of hematological indices to distinguish RA from healthy controls. Sensitivity and 1-specificity values are displayed.
ROC curve analysis of hematological indices as key factors in differentiating RA from healthy controls.

AUC, area under curve; CI, confidence interval; HGB, hemoglobin; LYM, lymphocyte; MONO, monocyte; MPV, mean platelet volume; NEU, neutrophil; NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; PLT, platelet; RA, rheumatoid arthritis; RDW, red cell distribution width; ROC, receiver operating characteristic.
For differentiating disease activity states within the RA cohort, the performance of all parameters was modest (Figure 4). The NLR again showed the highest, though only fair, discriminative ability (AUC = 0.598; 95% CI: 0.540–0.656; p = 0.001), followed by NEU (AUC = 0.576) and RDW (AUC = 0.572; Table 6). These results suggest limited discriminative power overall.

ROC curves comparing the discriminatory ability of hematological indices to distinguish moderate-to-high disease activity of RA patients. Sensitivity and 1-specificity values are displayed.
ROC curve analysis of hematological indices as key factors in differentiating moderate-to-high disease activity of RA patients.

AUC, area under curve; CI, confidence interval; HGB, hemoglobin; LYM, lymphocyte; MONO, monocyte; MPV, mean platelet volume; NEU, neutrophil; NLR, neutrophil/lymphocyte ratio; PLR, platelet/lymphocyte ratio; PLT, platelet; RA, rheumatoid arthritis; RDW, red cell distribution width; ROC, receiver operating characteristic.
Other parameters, including PLR, MPV, LYM, HGB, and MONO, showed AUC values below 0.5, indicating no significant association with disease activity.
Discussion
This study is the first to evaluate hematological parameters in drug-naïve patients with RA. All patients included in our study were newly diagnosed in our clinic and had not received DMARDs or glucocorticoids prior to enrollment. Consequently, drug-related alterations in hematological parameters were effectively excluded.
This retrospective study comprehensively assessed routine hematological indices as potential biomarkers in drug-naïve patients with RA. The findings revealed a distinct hematological profile characterized by elevated neutrophils, NLR, PLR, and RDW, along with decreased lymphocytes, HGB, and MPV compared with healthy controls. Notably, MPV emerged as an independent factor associated with moderate-to-high disease activity. These hematological alterations are consistent with the systemic inflammatory state characteristic of RA. Elevated neutrophil counts and NLR reflect activation of the innate immune response, while thrombocytosis and reduced MPV are recognized as acute-phase reactants.32–35
Systemic inflammatory ratios, particularly the NLR and PLR, demonstrated limited diagnostic potential for distinguishing patients with RA from healthy controls, with NLR showing the highest AUC value of 0.676 over other hematological parameters, with a modest discriminative performance. This finding aligns with previous literature that identifies these ratios as inexpensive and readily accessible indicators of systemic inflammation. A meta-analysis reported that both NLR and PLR were higher in patients with RA than in healthy controls. The standardized mean differences in NLR were greater in individuals with elevated ESR and CRP levels; however, these differences were not statistically significant in meta-regression analysis. Similar results were observed for PLR. Nevertheless, as noted in that study’s limitations, the influence of immunosuppressive therapy was not assessed, and the relationship of NLR and PLR with RA disease severity was not investigated. 18 In a multicenter study, NLR and PLR were significantly higher in patients with RA than in both other rheumatic diseases and control groups. 36 NLR demonstrated diagnostic value for RA with a cutoff value of 2.13. The authors reported that all participants had clinically active disease, but details regarding the medications used were not provided. In our study, the cutoff value for NLR was slightly higher than that reported in this multicenter analysis. However, the diagnostic utility of these ratios for differentiating disease activity levels within the RA group was limited, suggesting that they are effective in detecting the presence of RA but less sensitive to variations in disease activity. In a cross-sectional study with similar results, ESR and CRP were weakly and positively correlated with both NLR and PLR. However, the sensitivity and specificity of NLR and PLR were lower than those of ESR and CRP for distinguishing active disease. 37
One of the most important findings of our study is that MPV showed a significant inverse correlation with both CRP and DAS28-CRP. Our results are consistent with previous reports. Şahin et al. 38 demonstrated that MPV was inversely and significantly correlated with ESR and CRP, and negatively correlated with the DAS28 score in patients with RA. Kim and Kim 39 observed that MPV was lower in patients with active RA compared with those with osteoarthritis, and that MPV increased following treatment. Similarly, Gasparyan et al. 40 found that low MPV values were associated with disease exacerbation and that anti-tumor necrosis factor alpha therapy increased MPV. Under physiological conditions, an increase in platelet production is typically accompanied by a reduction in their mean volume. 41 However, during ongoing inflammation, elevated concentrations of proinflammatory cytokines—particularly IL-6—can stimulate platelet release from megakaryocytes. This mechanism may explain the inverse relationship between inflammation and MPV.
Interestingly, MPV was independently associated with disease activity in our multivariable regression models. This association remained significant after adjustment for other clinical variables. Although MPV was lower overall in patients with RA compared with healthy controls, its relative increase within the RA cohort appears to be a marker of more severe disease. This paradoxical observation warrants further investigation and may be related to complex platelet turnover dynamics and megakaryocyte activation during chronic inflammation, suggesting that MPV’s association with disease activity involves mechanisms not fully reflected by CRP levels alone.
Correlation analyses with CRP levels showed that both neutrophil count and NLR demonstrated moderate positive correlations with CRP, whereas several parameters exhibited only weak correlations with disease activity. These findings highlight the limited role of routine hematological parameters in assessing RA activity but suggest that, when interpreted alongside clinical indices, they may still provide complementary value in comprehensive disease evaluation.
Limitations
This study has several limitations. Its retrospective and single-center design introduces potential selection bias and the possibility of unmeasured confounding factors. Moreover, the absence of comprehensive longitudinal data on treatment response limits the ability to evaluate changes in hematological parameters over time or their utility in disease monitoring.
Conclusion
In conclusion, NLR and PLR demonstrate moderate utility in assessing RA activity but are not superior to traditional inflammatory markers such as ESR and CRP. MPV may be associated with higher disease activity; however, further studies are required to confirm its clinical relevance. Although hematological parameters can aid in diagnosing RA and distinguishing disease activity, they should not be considered standalone biomarkers.
Supplemental Material
sj-doc-1-tab-10.1177_1759720X261417917 – Supplemental material for Evaluation of hematological parameters as biomarkers of disease activity in drug-naïve patients with rheumatoid arthritis: a retrospective analysis
Supplemental material, sj-doc-1-tab-10.1177_1759720X261417917 for Evaluation of hematological parameters as biomarkers of disease activity in drug-naïve patients with rheumatoid arthritis: a retrospective analysis by Esra Dilsat Imrak, Alev Cetin Duran, Ugur Ergun, Nihan Balta and Betul Koyuncu in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
