Abstract
Background:
Interleukin 8 (IL-8) is a chemokine involved in the pathogenesis of rheumatoid arthritis (RA). However, the clinical significance of its circulating levels remains poorly defined, partly due to technical challenges in measuring IL-8 in serum. Consequently, data on the relationship between serum IL-8 concentrations and specific disease manifestations in RA are limited.
Objective:
In this study, we aimed to investigate the associations between serum IL-8 levels and clinical, serological, and cardiovascular features in patients with RA.
Design:
Cross-sectional study.
Methods:
A total of 216 RA patients were recruited. They underwent comprehensive evaluations, including disease-related characteristics and disease activity indices. Moreover, complete lipid profile, insulin resistance indices, metabolic syndrome criteria, and carotid ultrasound for carotid stiffness, intima-media thickness and carotid plaque detection were assessed. The SCORE2 –Systemic Cardiovascular Risk Estimation– was also calculated. IL-8 serum levels were measured using Simoa (Single Molecule Array) technique. A multivariable linear regression analysis was performed to examine the associations between disease characteristics and IL-8.
Results:
C-reactive protein and IL-6 were significantly associated with higher IL-8 levels after multivariable adjustment. Likewise, disease activity scores were independently and positively correlated with higher serum IL-8 concentrations after adjustment for covariates. In contrast, rheumatoid factor and anti-citrullinated protein antibodies positivity showed no association with IL-8 levels. With respect to cardiovascular features, higher SCORE2 and carotid intima media thickness values were positively related to increased IL-8 concentrations.
Conclusion:
Serum IL-8 levels are independently and positively associated with disease activity scores in patients with RA. These findings suggest that IL-8 may have potential as a biomarker in this population but requires prospective validation and comparison with standard markers. The observed relationship between IL-8 levels and the SCORE2 risk calculator suggests a potential association between IL-8 and cardiovascular disease.
Plain language summary
This study looked at the role of interleukin-8 (IL-8), a molecule involved in inflammation, in people with rheumatoid arthritis (RA). Although IL-8 is known to play a part in RA, its importance in everyday clinical practice has been unclear, partly because it is difficult to measure accurately in the blood. To address this, we measured IL-8 levels in 216 patients with RA and examined how these levels related to various aspects of the disease, including how active it was, blood test results, and risk factors for heart disease. We found that people with more active disease and higher levels of inflammation also had higher amounts of IL-8 in their blood. Additionally, those with a greater estimated risk of cardiovascular disease, as measured by the SCORE2 tool, tended to have higher IL-8 levels as well. However, the presence of certain antibodies commonly seen in RA was not linked to IL-8 concentrations. Overall, these findings suggest that IL-8 could be a useful marker for monitoring disease activity and possibly cardiovascular risk in people living with RA.
Introduction
Interleukin 8 (IL-8) is part of the extensive chemokine superfamily, which encompasses more than 40 different chemokines produced by a diverse array of cell types and acting on numerous target cells through specific receptors. Specifically, IL-8 is classified within the CXCL (C-X-C motif) chemokine subgroup and is also known as CXCL8. Its synthesis occurs in a range of cells, such as monocytes, lymphocytes, granulocytes, fibroblasts, endothelial and epithelial cells, hepatocytes, mesangial cells, and chondrocytes, but it is predominantly released in response to inflammatory stimuli. 1 Platelets also contain IL-8 within their granules, enabling them to rapidly secrete this chemokine during inflammation. 2 As a result, the acute inflammatory signal mediated by IL-8 can be initiated by multiple cell types and tissues, contributing to coordinated host defense responses. 1 The key function of IL-8 is to recruit neutrophils to sites of inflammation and, through its interaction with cell adhesion molecules, facilitate their infiltration into inflamed tissues. 3 Apart from recruiting neutrophils, IL-8 activates them to release granule contents, induces superoxide production, and increases adhesion molecule expression. Additionally, IL-8 regulates neutrophil egress from the bone marrow, contributing to both acute inflammatory responses and homeostatic neutrophil circulation. 4 Consequently, IL-8 has been identified as a potential therapeutic target to prevent neutrophil-mediated organ damage. 5
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disease characterized by persistent synovial inflammation, leading to joint destruction, pain, and functional impairment. 6 It affects approximately 1% of the global population, with a higher prevalence in women. 7 RA is associated with the presence of autoantibodies, particularly rheumatoid factor and anti-citrullinated protein antibodies (ACPAs). In addition to articular involvement, RA often presents with extra-articular manifestations affecting the lungs, skin, eyes, and cardiovascular (CV) system. 8 The development of RA results from a multifaceted interplay of genetic predisposition, epigenetic modifications, and environmental exposures, ultimately leading to the loss of immune self-tolerance and persistent, symmetrical inflammation of the synovial joints. Within this context, the initiation and maintenance of RA are strongly influenced by autocrine and paracrine signaling, as immune and stromal cells generate a complex cascade of proinflammatory cytokines that drive and sustain the inflammatory process. 9 Inflammatory cells are recruited to the synovium by the actions of multiple ILs and chemokines. Chemokines, including IL-8, are involved in enhancing transmigration of leukocytes into the synovial tissue. 5 Then the T and B cells become organized and activated in the presence of these cytokines, and the proliferative and destructive component of synovitis is initiated by the inflammatory environment.
RA is also associated with metabolic syndrome, endothelial cell dysfunction, and an increased frequency of CV disease, including myocardial infarction resulting from accelerated atherosclerosis.10–13 Interestingly, a recent study showed that, in addition to increased levels of platelet-derived IL-6, elevated levels of IL-8 were identified in the secretome of patients with acute myocardial infarction. These levels correlated with clinical markers of myocardial injury. 14 These observations highlight the potential role of IL-8 in endothelial dysfunction and ischemic heart disease.
To our knowledge, no studies have evaluated serum IL-8 levels in large cohorts of patients with RA. In the present study, we measured IL-8 concentrations using the highly sensitive Simoa (Single Molecule Array) technique, which enables reliable detection of low IL-8 levels. Our RA cohort was comprehensively characterized, incorporating both disease-specific features and CV comorbidities. The objective of our work was to examine the association between serum IL-8 levels and disease activity and the presence of extra-articular manifestations. Besides we aim to analyze the relation of IL-8 to the CV comorbidity that frequently accompany RA patients like parameters associated with metabolic syndrome, carotid ultrasound data as a surrogate marker of atherosclerosis and the Systematic Coronary Risk Evaluation 2 (SCORE2) that is a risk prediction model developed by the European Society of Cardiology to estimate the 10-year risk of fatal and non-fatal CV disease, myocardial infarction and stroke, in apparently healthy people. 15
Methods
Study participants
This was a cross-sectional study that included 216 consecutively recruited RA patients, all of whom were 18 years or older and met the 2010 ACR/EULAR (American College of Rheumatology/European League Against Rheumatism) classification criteria. 16 They had been diagnosed by rheumatologists and attended regular follow-up appointments at rheumatology outpatient clinics. Patients were recruited between April 2024 and May 2025. For the purpose of inclusion in the present study, the duration of RA disease was required to be ⩾1 year. Since glucocorticoids are commonly used in the treatment of RA, patients taking prednisone or an equivalent dose of ⩽10 mg/day were included in the study. Exclusions were made for those with previous CV events, cancer, chronic diseases (e.g., hypothyroidism, heart or respiratory diseases, nephrotic syndrome) or active infections. Based on these inclusion and exclusion criteria, a total of 300 subjects with a clinical diagnosis of RA were initially preselected for this study. The patient selection process is detailed in Supplemental Figure 1. From an initial assessment of 300 patients, 50 were excluded due to an unconfirmed RA diagnosis (remaining 250). An additional 20 were excluded for age/consent criteria (remaining 230), 5 for comorbidities or active infection (remaining 225), and 6 for recent therapeutic changes (remaining 219). Finally, 3 were excluded for other reasons, yielding 216 patients included in the final analysis (Supplemental Figure 1). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 17 (Supplemental Checklist).
Data collection
Participants underwent a comprehensive examination, which included a questionnaire on CV risk factors and medication use. The physical examination assessed body mass index (BMI), abdominal circumference, and blood pressure (both systolic and diastolic) under standardized conditions. Information on smoking, diabetes, and hypertension was collected, and specific diagnoses and medication details were verified through medical record reviews. Disease activity in patients with RA was measured using the Disease Activity Score (DAS28) in 28 joints, 18 the Clinical Disease Activity Index (CDAI), 19 and the Simple Disease Activity Index (SDAI). 20 DAS28-erythrocyte sedimentation rate (ESR) and DAS28-C-reactive protein (CRP) were classified into distinct categories based on predefined thresholds: remission (<2.6), low (>2.6–3.2), moderate (>3.2–5.1), or high disease activity (>5.1) as previously described. 21 Likewise, the SDAI categories were defined as follows: remission (⩽3.3), low disease activity (>3.3 and ⩽11), moderate disease activity (>11 and ⩽26), and high disease activity (>26). Similarly, the CDAI was categorized into remission (⩽2.8), low disease activity (>2.8 and ⩽10), moderate disease activity (>10 and ⩽22), and high disease activity (>22). These categorizations adhere to established criteria. 22
The presence of metabolic syndrome was determined using the National Cholesterol Education Program (NCEP/ATPIII) criteria. 23 According to the NCEP ATP III definition, metabolic syndrome is diagnosed when three or more of the following five criteria are met: waist circumference exceeding 102 cm in men or 88 cm in women, blood pressure greater than 130/85 mmHg, fasting triglyceride levels above 150 mg/dL, fasting high-density lipoprotein (HDL) cholesterol levels lower than 40 mg/dL in men or 50 mg/dL in women, and fasting blood glucose levels exceeding 100 mg/dL. 23 The Systematic Coronary Risk Estimation-2 (SCORE2) CV risk tool was calculated as previously described using age, gender, smoking status, systolic blood pressure, and non-HDL-cholesterol. 15 SCORE2 estimates an individual’s 10-year risk of fatal and non-fatal CV disease events in individuals aged 40–69 years. For healthy people aged ⩾70 years, the SCORE2-OP (older persons) algorithm estimates 5-year and 10-year fatal and nonfatal CV disease events.
Laboratory assessments
Cholesterol, triglycerides, and HDL-cholesterol were measured using the enzymatic colorimetric assay (Roche Farma, Madrid, Spain) in serum. Lipoproteins were assessed using a quantitative immunoturbidimetric assay (Roche Farma, Madrid, Spain) in serum. Cholesterol ranged from 0.08 to 20.7 mmol/L (intra-assay coefficient of variation of 0.3%); triglycerides ranged from 4 to 1.000 mg/dL (intra-assay coefficient of variation of 1.8%); and HDL-cholesterol ranged from 3 to 120 mg/dL (intra-assay coefficient of variation of 0.9%). The atherogenic index was calculated using the total cholesterol: HDL-cholesterol ratio according to the Castelli formula. Low-density lipoprotein (LDL)-cholesterol was calculated using the Friedewald formula. The ESR was determined using the Westergren method. High-sensitivity CRP levels were measured using a high-sensitivity immunoassay.
The homeostatic model assessment (HOMA) method was performed to determine insulin resistance (IR). Briefly, the HOMA model enabled an estimate of insulin sensitivity (%S) and β-cell function (%B) from fasting plasma insulin, C peptide, and glucose concentrations. In this study, we used HOMA2, the updated computer HOMA model. 24 This model can be used to assess insulin sensitivity and β-cell function from paired fasting plasma glucose and specific insulin, or C peptide, concentrations across a range of 1–2200 pmol/L for insulin and 1–25 mmol/L for glucose. C peptide better estimates β-cell function since it is a marker of secretion, and insulin data is preferable when calculating %S since HOMA-%S is derived from glucose disposal as a function of insulin concentration. In our study, IR and %S were calculated using insulin serum levels. Otherwise, %B was calculated using C-peptide serum levels. The computer model provided a value for insulin sensitivity expressed as HOMA2-%S (in which 100% is normal). HOMA2-IR (insulin resistance index) is simply the reciprocal of %S. Insulin (Architect Abbott, 2000I, Madrid, Spain) and C peptide (Immulite 2000, Siemens, Munich, Germany) were determined by chemiluminescent immunometric assays.
IL-8 and IL-6 were measured from serum samples through the Simoa technique using the Cytokine 4-Plex C Advantage PLUS Reagent KitTM (no. 105066, Quanterix, Billerica, MA, USA) following the manufacturer’s instructions. Sample processing and analysis were done using an HD-X analyzer (software version 4.16.2307.14001; Quanterix). All blood samples were collected from fasting patients using standardized serum tubes. Samples were processed within 2 h of collection by centrifugation at 1500 × g for 15 minutes and stored at −80°C until analysis. The assay’s lower limit of quantification for this kit is 0.05 pg/mL, and the upper limit of quantification is 1000 pg/mL. No patients had an IL-8 value below the lower limit of quantification (0.05 pg/mL). Intra-assay and inter-assay coefficients of variation (CVs) were maintained below 7%. Internal controls were incorporated on each plate, and batch effects were not detected.
Carotid ultrasound assessment
A carotid ultrasound was performed to assess the thickness of the carotid intima-media wall (cIMT) in the common carotid artery. The goal was to identify any localized plaques in the extracranial carotid arteries, located outside the skull (extracranial carotid tree). The measurements were carried out using the EsaoteMylab 70 ultrasound system from Genova, Italy. This system is equipped with a 7- to 12 -MHz linear transducer and employs the Quality Intima Media Thickness in real-time automated software-guided radiofrequency technique developed by Esaote in Maastricht, Holland. The assessment process followed the guidelines established by the Mannheim Consensus, 25 which establishes criteria for identifying plaques within the accessible extracranial carotid arteries. These arteries include the common carotid artery, the bulb, and the internal carotid artery. Plaque criteria were established as the presence of a localized bulge within the arterial lumen, with a measurement of cIMT exceeding >1.5 mm. Additionally, the bulge needed to be at least 50% larger than the adjacent cIMT or result in an arterial lumen reduction of >0.5 mm. 25
Carotid arterial stiffness was assessed using high-resolution ultrasound imaging with an Esaote® system equipped with radiofrequency-based echo-tracking technology. The Esaote echo-tracking software automatically calculated key stiffness parameters, including pulse wave velocity (PWV), augmentation index (AIx), distensibility coefficient, compliance, and the β-stiffness index, based on simultaneous recordings of arterial diameters and brachial blood pressure. All measurements were synchronized with the electrocardiogram to ensure accurate timing of systolic and diastolic phases. This standardized protocol allowed for the reliable and reproducible assessment of carotid stiffness, providing valuable insights into early vascular alterations associated with CV risk. 26
Statistical analysis
Demographic and clinical characteristics in patients with RA were described as means (standard deviation) or percentages for categorical variables. For non-normally distributed continuous variables, data were expressed as median and interquartile range (IQR). Missing data were addressed by carefully reviewing all collected variables for completeness prior to analysis. Cases with missing key clinical or laboratory data were excluded from specific analyses to avoid introducing bias. The extent of missing data was minimal, and details of missing data are transparently reported. Prior to study initiation, we powered the analysis to detect a correlation of r = 0.20 between DAS28‑ESR and IL‑8 using Fisher’s z transformation for a two‑sided α = 0.05 and 80% power. This corresponds to an approximate required sample size of n ≈ 193. To provide a margin for model adjustment and minor attrition, we planned to enroll at least 200 participants. This prospective calculation ensured adequate sensitivity to detect a moderate association considered clinically meaningful. The relationship between disease characteristics and IL-8 serum levels was analyzed using multivariable linear regression analysis. Potential confounders were identified using a change-in-estimate approach. Specifically, variables were considered confounders and retained in the multivariable model if their inclusion resulted in a change of 10% or more in the beta coefficient of the main exposure variable. This criterion was applied iteratively, adjusting the model to control for confounding while maintaining parsimony. The initially selected potential confounders included age, sex, BMI, smoking and diabetes. Log-transformed variables were used for correlation and regression analyses to address non-normal distributions. Normality of the transformed variables was assessed using appropriate statistical tests for normality, such as the Shapiro-Wilk test, complemented by graphical methods including Q–Q plots and histograms to ensure the adequacy of the transformations. We evaluated linear regression assumptions via residuals-versus-fitted (homoscedasticity, mean-zero) and Q–Q plots (normality). Linearity was checked with partial residuals/splines; independence with Durbin–Watson; and influence via studentized residuals, leverage, and Cook’s distance. Multicollinearity was assessed using the variance inflation factor (VIF) (<5). Results were robust across sensitivity checks. All analyses were performed at a 5% two-sided significance level using Stata software, version 17/BE (StataCorp, College Station, TX, USA). p-values <0.05 were considered statistically significant. Figure generation was performed using Python. Julius AI (Julius AI Inc., San Francisco, CA, USA; https://julius.ai) was used solely as a coding assistant for plot scripting; no individual-level patient data were uploaded to any third-party server, and all analyses were executed locally.
Results
Demographics and disease-related data
This study included a total of 216 patients diagnosed with RA. IL-8 values were 10.1 ± 5.8 pg/mL. Serum IL-8 exhibited marked right-skewness and substantial deviation from normality on the Q–Q plot. Logarithmic transformation (log10 IL-8) yielded a more symmetric distribution, with improved alignment to the theoretical normal curve on both histogram and Q–Q plot, rendering the log scale suitable for parametric statistical modeling (Supplemental Figure 2). Demographic- and disease-related characteristics of the participants are shown in Table 1. The study population had a mean age of 59 ± 9 years, with 83% of the participants being women. BMI was 29 ± 5 kg/m2. The prevalence of traditional CV risk factors was generally high in the study population. Specifically, 23% were current smokers at the time of enrollment, 13% had diabetes mellitus, and 41% were diagnosed with hypertension. Additionally, 56% of participants met the diagnostic criteria for metabolic syndrome, and 35% and 10% were, respectively, on statin and aspirin therapy. The values from the SCORE2 CV risk calculator were a median of 4.2 (IQR 2.4–6.2), with 57% of RA patients categorized as low risk, 35% as intermediate risk, and 8% as high risk. Regarding subclinical atherosclerosis markers, PWV was 7.6 ± 1.7 m/s and augmentation index was 7.7 (3.6–11.8)%. Additionally, the mean intima-media thickness was 721 ± 141 microns, and 54% of patients presented with carotid plaque. Besides, information on the full lipid profile and insulin resistance indices is shown in Table 1.
Demographics, cardiovascular risk factors, and disease-related data in RA patients.

Data represent means ± SD or median (IQR) when data were not normally distributed.
ACPA, anti-citrullinated protein antibodies; BMI, body mass index; CDAI, Clinical Disease Activity Index; cIMT, carotid intima media thickness; CRP, C reactive protein; CV, cardiovascular; DAS28, Disease Activity Score in 28 joints; DMARD, disease-modifying antirheumatic drug; ESR, erythrocyte sedimentation rate; HDL, high-density lipoprotein; HOMA, homeostatic model assessment; IL, interleukin; LDL, low-density lipoprotein; NSAID, nonsteroidal anti-inflammatory drugs; RA, rheumatoid arthritis; SCORE2, Systematic COronary Risk Evaluation 2; SDAI, Simple Disease Activity Index; TNF, tumor necrosis factor.
The median duration of the disease was 13 ± 9 years. At the time of the study, the mean values of hs-CRP and ESR were 2.3 mg/L (IQR 0.2–5.0) and 18 ± 14 mm/1 h, respectively. Rheumatoid factor was positive in 84% of patients, and 74% were positive for ACPA. The disease activity, as measured by DAS28-ESR, was 3.2 ± 1.3. The DAS28-CRP had a value of 2.8 ± 1.2, and SDAI and CDAI were 8 (IQR 5–16) and 7 (IQR 3–14), respectively. Thirty-six percent of the patients were treated with prednisone, and 88% were receiving at least one conventional disease-modifying anti-rheumatic drug, with methotrexate being the most widely used (69%). Nineteen percent of the patients were receiving anti-tumor necrosis factor therapies. The frequency of use of other treatments and historical disease-related data are provided in Table 1.
Disease features and their association with IL-8 levels
The relationship between demographic and disease-related characteristics and IL-8 levels is presented in Table 2. Age, sex, and anthropometric measurements were not associated with IL-8 concentrations. In contrast, among CV risk factors, patients who smoked or had diabetes or used statins exhibited significantly higher IL-8 levels compared to those without these conditions. A similar trend was observed for hypertension, although this did not reach statistical significance (Table 2).
Relation of disease characteristics to IL-8 serum levels.
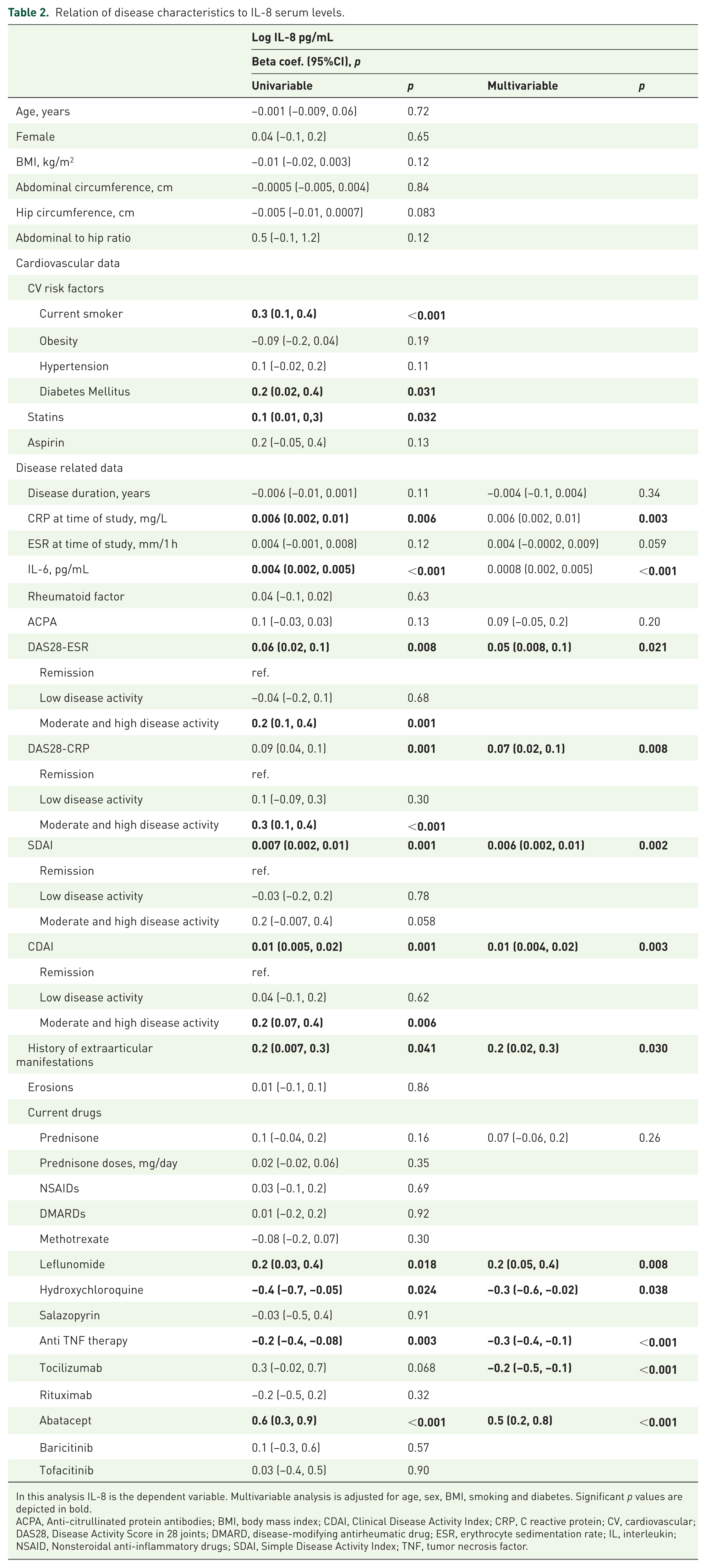
In this analysis IL-8 is the dependent variable. Multivariable analysis is adjusted for age, sex, BMI, smoking and diabetes. Significant p values are depicted in bold.
ACPA, Anti-citrullinated protein antibodies; BMI, body mass index; CDAI, Clinical Disease Activity Index; CRP, C reactive protein; CV, cardiovascular; DAS28, Disease Activity Score in 28 joints; DMARD, disease-modifying antirheumatic drug; ESR, erythrocyte sedimentation rate; IL, interleukin; NSAID, Nonsteroidal anti-inflammatory drugs; SDAI, Simple Disease Activity Index; TNF, tumor necrosis factor.
Regarding disease characteristics, acute phase reactants CRP and IL-6, but not ESR, were significantly associated with IL-8 levels after multivariable adjustment. Similarly, all disease activity scores consistently correlated with higher IL-8 levels, whether analyzed as continuous or categorical variables (Figure 1). Remarkably, compared correlations showed that the log-IL-8/CDAI correlation was not significantly different from ESR/CDAI (p = 0.389), but was significantly stronger than CRP–CDAI (p = 0.010) (Supplemental Figure 3). In contrast, positivity for rheumatoid factor or ACPA was not related to IL-8 levels. Finally, among treatments, patients under anti-TNF therapy, hydroxychloroquine or tocilizumab exhibited lower IL-8 levels, whereas patients receiving abatacept or leflunomide had higher levels (Table 2).

Association between disease activity and IL-8 levels. Left panel: scatterplot of DAS28-ESR versus log IL‑8. Right panel: distribution of log IL‑8 across DAS28-ESR categories (remission, low activity, moderate, and high activity) using boxplots with overlaid individual observations. Boxes indicate medians and interquartile ranges; whiskers denote variability outside the upper and lower quartiles; points represent individual subjects.
Relationship between cardiovascular comorbidity data of patients with RA and serum IL-8 levels
The associations between CV-related disease characteristics and IL-8 levels are presented in Table 3. The presence of metabolic syndrome, lipid profile, and insulin resistance indices showed no association with circulating IL-8 levels. Notably, subclinical atherosclerosis, as assessed by cIMT, was significantly associated with elevated serum values of IL-8. However, the SCORE2 CV risk calculator showed a positive and significant association with serum IL-8 levels, both when analyzed as a continuous variable and when categorized into risk groups. This analysis was not adjusted for covariates, as SCORE2 is derived from multiple variables, making further adjustment inappropriate due to potential collinearity (Table 3).
Relation of cardiovascular disease features to IL-8 serum levels.

In this analysis, IL-8 serum values are the dependent variable. Regression analyses for metabolic syndrome and SCORE2 were not adjusted because these variables are derived from composite calculations. Multivariable analysis is adjusted for body mass index, smoking, hypertension, and diabetes. Significant p values are depicted in bold.
cIMT, carotid intima media thickness; HDL, high-density lipoprotein; HOMA, homeostatic model assessment; LDL, low-density lipoprotein; SCORE2, Systematic COronary Risk Evaluation 2.
Discussion
Most studies investigating IL-8 in RA have focused primarily on experimental and molecular aspects, particularly within synovial tissues. However, circulating IL-8 levels have been less systematically explored, and their associations with clinical features remain insufficiently characterized. Therefore, the present study aims to address this gap by assessing serum IL-8 levels in patients with RA. Based on our findings, IL-8 could have a role as a biomarker of disease activity in this population. Additionally, IL-8 was independently associated with acute phase reactants, including CRP and IL-6. Moreover, IL-8 levels also correlated with SCORE2, which is clinically relevant for calculating overall CV disease, suggesting a potential link of IL-8 with CV disease in RA.
Numerous studies have highlighted the potential role of IL-8 in the pathogenesis of RA. In this regard, IL-8 protein levels have been described to be increased in clinical involved joints compared with clinically uninvolved joints, which was confirmed by immunohistochemistry and in situ hybridization in RA patients. 27 Moreover, IL-8 activates polymorphonuclears and subsequently releases neutrophil-derived chemotactic factor acting as chemotactic factors for monocytes and T cells in RA. 27 IL-8 has also been reported to be produced by cells located within the sublining layer that is in direct contact with the cartilage, thereby suggesting its role in the expression of collagenases and consequent cartilage damage. 28 In this respect, IL-8 has been identified as an autocrine regulator of RANKL-induced osteoclastogenesis, by promoting the expression of RANKL by osteoblastic stromal cells. 29
We found an independent association between disease activity and circulating IL-8 levels in our study. This relationship remained consistent across various disease activity scores. IL-8 also demonstrated a significant positive correlation with both CRP and IL-6 levels. We feel that, considering the established molecular role of IL-8 in synovial inflammation, the observed association between disease activity and serum IL-8 levels may be biologically plausible. To our knowledge, this relationship has not been examined in previous research. In this regard, the large sample size of our cohort enabled robust multivariable adjustment, supporting the reliability of our findings regarding the link between disease activity and IL-8 concentrations.
Our analysis did not reveal any relationship between the presence of rheumatoid factor or ACPA and IL-8 concentrations. This finding suggests that these autoantibodies, which are well-established markers of disease prognosis in RA, do not appear to influence IL-8 expression. Therefore, IL-8 may reflect disease activity and inflammatory burden through mechanisms distinct from those driving autoantibody production. This distinction is clinically relevant, as it highlights the potential utility of IL-8 measurement in both seropositive and seronegative patients. Consequently, IL-8 could serve as a potential biomarker for assessing disease activity in RA, independent of serological status.
In our study, patients treated with anti-TNF therapies had significantly lower IL-8 levels after multivariable adjustment. This association likely reflects the effectiveness of anti-TNF agents in controlling inflammation and reducing disease activity in RA, given the close link between IL-8 and inflammatory processes. In other words, it probably represents a direct suppression of the TNF-α–dependent proinflammatory cytokine cascade, including IL-8. Although our cross-sectional design does not allow for causal inference, it is plausible that the reduction in IL-8 among these patients is a consequence of better disease control achieved through anti-TNF therapy. In contrast, patients receiving abatacept showed significantly higher levels of IL-8. The mechanism underlying this observation remains unclear. Abatacept works by inhibiting T-cell costimulation, primarily affecting adaptive immune responses, whereas IL-8 production is largely determined by innate immune pathways and proinflammatory cytokines such as TNF-α and IL-6. Therefore, abatacept may have a less direct effect on innate immune IL-8 production. It is possible that abatacept does not sufficiently suppress IL-8 synthesis or that its mechanism of action does not directly affect the pathways responsible for IL-8 production. Alternatively, patients treated with abatacept may represent a subgroup with more refractory or complex disease, which could correlate with persistently elevated IL-8 levels despite therapy. However, this interpretation should be made with caution, as only 11 patients in our cohort were receiving abatacept, limiting the robustness of this finding. For this reason, these findings should be regarded as exploratory, serving as preliminary signals for future research rather than evidence of causality.
Since the inflammatory nature of atherosclerosis was established, evidence in the scientific literature unequivocally supports the role of IL-8 in initiating and maintaining the inflammatory microenvironment within the affected vascular wall. 30 In the EPIC-Norfolk population study, baseline IL-8 concentrations were measured in 785 apparently healthy individuals who subsequently developed fatal or nonfatal coronary artery disease during follow-up, along with 1570 matched controls. 31 Baseline IL-8 concentrations were higher in cases than in matched controls. The risk of future coronary artery disease increased with increasing quartiles of IL-8, and among individuals in the highest IL-8 quartile the odds ratio for future coronary artery disease was still significant after adjustment for traditional risk factors (OR, 1.58; 95% CI, 1.19–2.09; p = 0.002) and after additional adjustment for CRP and white cell count. 31 Besides, in vitro studies and animal models have confirmed the involvement of IL-8 in atherosclerosis. For example, IL-8 levels are elevated in coronary atherectomy specimens compared to control tissues, and IL-8 is a key mediator of angiogenesis within atherosclerotic plaques. 32 Through its pro-angiogenic effects, IL-8 may contribute to both plaque development and progression. 32 Similarly, IL-8 acts as a chemotactic factor, recruiting inflammatory cells to atherosclerotic lesions and contributing to the inflammatory microenvironment of the vascular wall. 30 In our study, we did not find a significant association between IL-8 levels and individual CV risk factors, including insulin resistance or dyslipidemia. However, we did observe a significant relationship between IL-8 concentrations and both cIMT and the SCORE2 risk calculator, which provides an integrated assessment of CV risk by combining multiple traditional risk factors. We believe the relation of SCORE2 to IL-8 is because this risk calculator combines multiple traditional CV risk factors, many of which are influenced by systemic inflammation. This suggests that IL-8 might potentially serve as a marker of overall CV risk burden rather than being linked to any single risk factor in isolation. Accordingly, while IL-8 showed a consistent positive association with SCORE2, its correlations with imaging surrogates were less consistent. We believe, this distinction likely reflects SCORE2’s focus on modifiable systemic risk and inflammation versus imaging’s detection of structural vascular changes. These findings may suggest IL-8 may primarily capture systemic inflammatory burden rather than advanced morphological alterations. Our findings reinforce the concept that systemic inflammation, as indicated by elevated IL-8 levels, plays a central role in the pathogenesis of CV disease in patients with RA.
Anti-IL-8 monoclonal antibody-based therapies are being investigated in several diseases. For example, HuMax-IL8 (now known as BMS-986253), a novel fully human monoclonal antibody that inhibits IL-8, has been tested in solid tumors in phase I trials. 33 Reparixin and ladarixin, which are inhibitors of the CXCR1 and CXCR2 receptors, which block IL-8–mediated signaling, have also been evaluated in pilot studies for cancer and transplant rejection.34,35 Although these therapies have not yet been evaluated in RA, our findings suggest that targeting IL-8 could be beneficial in this population 36 ; however, this hypothesis-generating proposal requires validation through future clinical studies.
Limitations
The measurement of IL-8 in our study was performed using the highly sensitive Simoa platform, which offers superior precision and reliability compared to conventional assays. This advanced technology enables the detection of very low serum IL-8 concentrations, ensuring high specificity and reproducibility, and closely reflecting biological activity. However, we recognize certain limitations, including the cross-sectional nature of our research, which limits causal inference, and the absence of a healthy control group. Our findings warrant validation in prospective longitudinal studies and randomized intervention trials to establish temporal relationships and clinical utility. Nevertheless, the primary aim of our study was to explore the associations between serum IL-8 and clinical features within the RA population, rather than to compare levels with those of healthy individuals. Furthermore, our study was not designed to determine whether IL-8 is a better predictor of disease activity than established markers such as ESR, CRP, or DAS28. Addressing that question would require a different study with a specific design aimed at comparing the predictive performance of these biomarkers. Therefore, we cannot draw conclusions on the relative utility of IL-8 versus other inflammatory or clinical markers for assessing RA activity. We also recognize that the clinical utility of measuring IL-8 is currently limited by its high cost and technical complexity in laboratory settings. However, it is anticipated that ongoing advancements in ultrasensitive detection technologies will reduce these barriers, facilitating broader implementation, including integration into routine clinical practice in the future. Nevertheless, IL-8 measurement is not currently widely accessible in routine clinical practice, as it requires specialized assays not routinely available in standard laboratory settings. Also, some treatment-related associations should be interpreted with caution due to the small patient numbers involved. Finally, our analysis demonstrates multiple associations between disease features and serum IL-8 levels without formal multiplicity adjustment. Consequently, p-values from these exploratory comparisons should be interpreted cautiously.
Conclusion
In summary, our findings indicate that IL-8 is linked to disease activity in RA. These results suggest that serum IL-8 could represent a potential biomarker for assessing disease activity in these patients, although further studies are needed to confirm this association. While confirmation in prospective studies is warranted, our findings also suggest a potential association between IL-8 and CV disease in patients with RA, as reflected by the observed relationship between IL-8 levels and the SCORE2 risk calculator. Future studies should validate these findings prospectively, compare IL-8 with established markers such as CRP and ESR, and evaluate its performance across different RA subpopulations.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261436835 – Supplemental material for Serum interleukin-8 levels relate to disease activity in patients with rheumatoid arthritis
Supplemental material, sj-docx-1-tab-10.1177_1759720X261436835 for Serum interleukin-8 levels relate to disease activity in patients with rheumatoid arthritis by Diamela León-Cabrera, Juan C. Quevedo-Abeledo, Cristina Almeida-Santiago, Enrique García-Barrera, Luisa M. Villar, Patricia Quiroga-Colina, Javier Gonzalo Ocejo-Vinyals, Alberto Javier-Ormazábal, Miguel Á. González-Gay and Iván Ferraz-Amaro in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X261436835 – Supplemental material for Serum interleukin-8 levels relate to disease activity in patients with rheumatoid arthritis
Supplemental material, sj-docx-2-tab-10.1177_1759720X261436835 for Serum interleukin-8 levels relate to disease activity in patients with rheumatoid arthritis by Diamela León-Cabrera, Juan C. Quevedo-Abeledo, Cristina Almeida-Santiago, Enrique García-Barrera, Luisa M. Villar, Patricia Quiroga-Colina, Javier Gonzalo Ocejo-Vinyals, Alberto Javier-Ormazábal, Miguel Á. González-Gay and Iván Ferraz-Amaro in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
