Abstract
Objectives:
To evaluate, in a multicentric Italian cohort of axial spondyloarthritis (axSpA) patients on Secukinumab (SEC) followed for 24 months: (1) the long-term effectiveness and safety of SEC; (2) the drug retention rate and low disease activity (LDA) measured as Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) < 4/Ankylosing Spondylitis Disease Activity Score (ASDAS) < 2.1 and very low disease activity (VLDA) measured as BASDAI < 2/ASDAS < 1.3; (3) any differences in outcomes according to line of biological treatment (naïve/non-naïve), gender (male/female), subtype of axSpA [radiographic axSpA (r-axSpA)/non-radiographic axSpA (nr-axSpA)].
Methods:
Consecutive axSpA patients treated with SEC were evaluated prospectively. Disease characteristics, previous/ongoing treatments, comorbidities, and follow-up duration were collected. Disease activity/functional/clinimetric scores and biochemical-values were recorded at baseline (T0), 6 (T6), 12 (T12), and 24 (T24) months. Effectiveness was evaluated over-time with descriptive statistics; multivariate Cox and logistic regression models were used to evaluate predictors of drug discontinuation and LDA at T6. Infections and adverse events were recorded.
Results:
A total 249 patients (47.8% male; median age 51) were enrolled; 40.9% had HLA-B27; 53.8% had r-axSpA, and 46.2% nr-axSpA. SEC was prescribed in 28.9% naïve and in 71.1% non-naïve patients. SEC effectiveness was shown as an improvement in several outcomes, such as ASDAS [T0 = 3.5 (2.9–4.4) versus T24 = 1.9 (1.2–2.4); p = 0.02] and BASDAI [T0 = 6.5 (5.0–7.5) versus T24 = 2.8 (1.8–4.0); p = 0.03]. At T24, naïve patients showed better physical functioning and lower disease activity than non-naïve. After 24 months of treatment, 90.7% of naïve and 75.3% of non-naïve patients achieved LDA (BASDAI < 4). Treatment was discontinued in 24.5% patients, mainly due to primary/secondary loss of effectiveness, and in 6.8% due to adverse events. Retention rate at T24 was 75% in the whole population, with some difference depending on gender (p = 0.002).
Conclusion:
In a real-life clinical setting, SEC proved to be safe and effective in axSpA, mainly in naïve-patients, with a notable drug retention rate. No differences were observed between r-axSpA and nr-axSpA.
Keywords
Introduction
Axial spondyloarthritis (axSpA) mainly affects the spine and the sacroiliac joints (SIJs) causing inflammatory back pain, morning stiffness, disability, and impaired quality of life; has an early onset at young age; and can be further subdivided into non-radiographic (nr-axSpA) and radiographic axSpA (r-axSpA), the latter also known as ankylosing spondylitis (AS).1,2 With the development of new effective treatment strategies, the need to identify axSpA patients in an earlier stage of disease has increased.3,4 The Assessment of SpondyloArthritis International Society (ASAS) has established classification criteria to identify patients with early-stage axSpA. 3 If not diagnosed early and treated appropriately, the disease often causes substantial functional impairment and a decreased quality of life. 5 Until the 2000s, axSpA patients were treated with non-steroidal anti-inflammatory drugs (NSAIDs), commonly used as the first-line treatment, often with unsatisfactory results, for the purpose to disease control and radiographic progression.5,6 The European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) guidelines recommended to treat active axSpA patients with high disease activity despite NSAIDs with biological disease-modifying antirheumatic drugs (bDMARDs).4,7 The advent of tumor necrosis factor inhibitors (TNFi) changed the natural history of axSpA over the last two decades, by significantly improving quality of life and reducing damage progression,8,9 as reported by randomized, placebo-controlled trials (RCTs).10–15 However, around 40% of axSpA patients fail to respond to the first TNFi, due to loss of efficacy, adverse events, or tolerability issues.16,17 Therefore, the need to find an alternative treatment led to the development of new pharmacological agents able to block other pivotal cytokines involved in the pathogenesis of axSpA, including the interleukin-23 (IL-23)/IL-17 axis theory.18,19 The fully human IgG1 monoclonal antibody targeting IL-17A (secukinumab) has provided significant and sustained improvement in the signs and symptoms of AS, as evidenced in several multicenter phase III RCTs (MEASURE 1,20,21 MEASURE 2,22,23 MEASURE 3, 24 MEASURE 4, 25 MEASURE 2-J, 26 and MEASURE 5). 27 Recently, PREVENT – the first phase III study evaluating secukinumab (SEC) in 555 nr-axSpA patients – showed a significant and sustained improvement in the axial signs and symptoms by week 16, an improvement that was sustained through week 52. 28 Until now, few registries and limited prospective studies have presented real-life data on the treatment of axSpA with SEC.29–31 Therefore, real-life data on the wide spectrum of patients affected by the disease is needed, to evaluate the effectiveness and safety of this biologic drug, while considering multiple features (lines of biological treatment (LoBT), gender, axSpA subtype).
The aim of our prospective observational study was to (1) evaluate the long-term effectiveness of SEC in the management of axSpA manifestations in a real-life clinical setting during a 24-month follow-up; (2) identify the differences in the outcome measures of effectiveness according to LoBT (naïve/non-naïve), gender (male/female), and axSpA subtype (r-axSpA/nr-axSpA); (3) analyze very low disease activity (VLDA)/low disease activity (LDA) states [Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) < 2/ < 4, Ankylosing Spondylitis Disease Activity Score (ASDAS) < 1.3/ < 2.1]; (4) identify predictors of low disease activity and treatment discontinuation at T6; (5) describe any comorbidities; (6) report any adverse events or infections; and (7) evaluate drug survival at 24 months.
Materials and methods
Patients
The present longitudinal study included consecutive axSpA patients who received SEC from September 2018 to March 2021 in 12 Italian Rheumatology centers. The study was supported by the Italian Society of Rheumatology (SIR) ‘Spondyloarthritis and Psoriatic Arthritis study group A. Spadaro’. Patients started treatment with SEC according to the EULAR and ACR guidelines.4,7
A detailed description of the enrolment and screening assessment of patients eligible to start SEC treatment had been previously published. 32 Inclusion criteria were: age ⩾ 18 years; a diagnosis of axSpA for >6 months, in accordance with the ASAS criteria; 2 the indication to start SEC treatment for a moderate or severe disease. If a peripheral involvement was also present, the concomitant use of conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) and low-dose corticosteroids (daily dose of ⩽10 mg of prednisone or equivalent) was allowed. The discontinuation or reduction in csDMARD dosage was permitted during the study period, if the patient’s clinical situation was favorable. We excluded axSpA patients who showed any of the following: a history of malignancy (over the last 5 years), systemic infections, congestive heart failure and demyelinating diseases.
Screening and assessments
Before being enrolled and starting treatment, patients underwent a series of screening tests, as suggested by the European guidelines.4,7 Screening included chest X-ray, laboratory tests (screening for HIV and hepatitis B and C viruses), the Mantoux TB skin test or Quantiferon TB Gold test, and a pregnancy test for all women of childbearing age. Patients presenting latent tuberculosis underwent a complete 6-month prophylaxis with isoniazid (300 mg/day). Patients were evaluated at baseline (T0) and after 6 (T6), 12 (T12), and 24 (T24) months of SEC treatment. Relevant demographics and clinical, biochemical, metrological, and imaging data were collected. These included the LoBT, which was considered as a dichotomous variable (SEC as a first biologic/as a second or more biologic) and axSpA subtype, which was considered a categorical variable (r-axSpA/nr-axSpA). Clinical variables – such as peripheral arthritis, psoriasis, dactylitis, enthesitis, uveitis, inflammatory bowel disease (IBD), and smoking status – were considered as dichotomous (yes/no) on patient enrolment. Baseline pelvic X-rays and magnetic resonance imaging (MRI) images were evaluated on site. For the analyses, axSpA patients were subdivided in subgroups: (1) naïve to any biologic drug (naïve) and subjects who had failed TNFi (non-naïve); (2) r-axSpA and nr-axSpA; (3) male and female. The presence of comorbidities and concomitant therapies were investigated (yes/no) during a face-to-face interview at one of the scheduled assessment visits, and by reviewing the patients’ medical records. Information on previous therapies with bDMARDs (and in particular the LoTB used) and concomitant csDMARDs (methotrexate, leflunomide, sulfasalazine, hydroxychloroquine, and cyclosporin) or corticosteroid therapy or NSAIDs were recorded at baseline and throughout the study.
Effectiveness measures and outcomes
Relevant patient reported outcomes (PROs), 33 such as the Visual Analogue Scale of pain (VAS-pain) and global health (VAS-gh), Health Assessment Questionnaires modified for spondyloarthritis (HAQ-S), Bath Ankylosing Spondylitis Functional Index (BASFI) and BASDAI were collected. The clinical evaluation, made by an experienced rheumatologist (the same assessor at each time point), included the Bath Ankylosing Spondylitis Metrology Index (BASMI), the Leeds Enthesitis Index (LEI), and the ASDAS. 34 The following LDA and VLDA measures were calculated at T6, T12, and T24: BASDAI-LDA defined as <4, BASDAI-VLDA defined as <2, ASDAS-LDA defined as <2, ASDAS-VLDA defined as <1.3.
These inactive and LDA measures were also calculated after the subdivision of all population in subgroups according to LoTB (naïve/non-naïve patients), gender (male/female), and axSpA subtype (r-axSpA/nr-axSpA). Biochemical acute phase reactants [erythrocyte sedimentation rate (ESR)] and the C-reactive protein (CRP) value were determined and analyzed. Our normal lab ranges were as follows: ESR 0–28 mm/h male, 2–38 mm/h female; CRP 0–6 mg/L. Metrological indexes such as height, weight, and body mass index (BMI) were also evaluated.
Administration of SEC
All patients were treated with SEC, administered subcutaneously at a dosage of 150 mg weekly for the five first injections and every 4 weeks thereafter, in accordance with the manufacturer’s instructions. 20 In subjects affected by active and severe psoriasis – according to the treating physician – a dosage up to 300 mg per administration could be employed. Patients were taught to perform self-injections. The drug’s safety was evaluated by assessing adverse events and through standard laboratory testing, and the patients’ vital signs were evaluated and recorded throughout the study, and for a 60-day period following the last treatment. Drug survival was calculated as the number of days in which the patient remained on therapy. The start date was the day the first dose was taken, and the stop date was the day the treatment was interrupted. Temporary interruptions (e.g. due to infections or surgery ⩽3 months before) were acceptable according to our study protocol.
Statistical analysis
Data were expressed as frequencies and percentages for categorical variables, and as mean and standard deviations or medians and interquartile range for continuous variables. Patients’ characteristics were compared between subgroups [LoBT (naïve/non-naïve), gender (male/female), axSpA subtype (rx-axSpA/nr-axSpA)], using the Chi-square test or the Fisher’s exact test for categorical variables and the t-test or the Wilcoxon rank test for continuous variables, based on data distribution. Effectiveness data on axial symptoms and signs were compared between T0 and T24 with the Chi-square test or the Wilcoxon rank test, as appropriate.
The time-to-event analysis was performed according to the Kaplan–Meier method. Survival curves were compared – by log-rank test – between: (1) LoBT (naïve/non-naïve); (2) gender (male/female); (3) axSpA subtype (r-axSpA/nr-axSpA). A logistic regression analysis was carried out to identify predictors of LDA (BASDAI < 4/ASDAS < 2.1) response at T6, and a Cox regression model was created to identify predictors of time-to-SEC discontinuation. In both cases, baseline independent factors which, based on literature data,30,32 could be expected to have an effect on short-term inactive disease/LDA achievement or on drug discontinuation, were considered. Results were expressed as odds ratio (OR) for logistic regression and hazard ratio (HR) 95% for Cox regression, together with their 95% confidence interval (CI). All statistical analyses were carried out with the SPSS 13.0 software (SSPS Inc., IL, USA). Two-tailed p-values lower than 0.05 were considered statistically significant.
Confidentiality of information/ethical aspects and reporting guidelines
Patients’ written consent was obtained, according to the Declaration of Helsinki, when they were first entered into the database for the treatment. The Ethics Committee’s approval was obtained from all participating centers (approval no. 23943), as well as the written informed consent for the anonymous use of personal data from every patient, in compliance with Italian Legislative Decree 196/2003. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement guidelines for reporting observational studies were followed. 35
Results
Patients’ features
Two-hundred forty-nine axSpA patients [47.8% men; median age 51 (41–59)] were enrolled; median disease duration was 9 (4–14) years, and mean treatment duration was 18.5 (13.0–34.5) months. SEC was prescribed as first-line biologic treatment in 72 (28.9%; naïve) patients and as second (or more) line biological treatment in 177 (71.1%; non-naïve) patients; 102 (40.9%) patients had HLA-B27; 134 (53.8%) were r-axSpA; and 115 (46.2%) nr-axSpA. The patients’ clinical and laboratory baseline characteristics, such as concomitant treatments, are summarized in Table 1.
Baseline characteristics of 249 axSpA patients treated with secukinumab (naïve versus non-naïve) in the period September 2018 to March 2021.

ASDAS, Ankylosing Spondylitis Disease Activity Score; axSpA, axial spondyloarthritis; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; BMI, body mass index; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; ESR, erythrocyte sedimentation rate; gh, global health; HAQ-S, Health Assessment Questionnaire modified for spondyloarthritis; HLA-B27, human leukocyte antigen (HLA) B27; IBD, inflammatory bowel disease; LEI, Leeds Enthesitis Index; MRI, magnetic resonance imaging; N/A, not applicable; naïve, naïve to anti-TNF-alpha inhibitors; non-naïve, TNF-alpha failure inhibitors; nr-axSpA, non-radiographic axial spondyloarthritis; NS, not statistical significant; NSAIDs, non-steroidal inflammatory drugs; r-axSpA, radiographic axial spondyloarthritis; SIJ, sacroiliac joint; SpA, spondyloarthritis; VAS, Visual Analogue Scale; X-rays; conventional radiography.
Data are expressed as median [interquartile range (IQR)] or number [percentage (%)] unless otherwise specified; range of possible values are indicated in round brackets.
p < 0.05 [ANOVA (Kruskal–Wallis) at T0].
At T0, non-naïve (versus naïve) patients were older and more frequently HLA-B27 positive, had a longer duration of axial symptoms, a more frequent peripheral involvement, a worse functional status (HAQs and BASMI), higher ESR and CRP values, a higher intake of SEC 300 mg/injection (Table 1). Few clinical and anthropometric features varied between males and females: at T0, males were more frequently HLA-B27 positive and presented a shorter disease duration, an earlier age of onset of axial pain, a higher prevalence of radiographic sacroiliitis, a higher smoking status, while females had a higher prevalence of peripheral arthritis, enthesitis and psoriasis, higher ESR values, were mostly non-naïve, and were receiving more frequently SEC 300 mg/injection (Supplementary Table 1). Few clinical and anthropometric features varied between r-axSpA and nr-axSpA patients: at T0, r-axSpA were mostly males and frequently HLA-B27 positive, presented a longer disease, while nr-axSpA had a higher prevalence of peripheral arthritis, dactylitis, enthesitis and psoriasis, and a higher intake of cDMARDs and glucocorticoids (Supplementary Table 2).
Therapeutic effectiveness
Of all 249 axSpA patients, 229 (91.9%) were evaluated at T6, 199 (79.9%) at T12, and 139 (55.8%) at T24. The whole population achieved a significant decrease in all indexes (Supplementary Table 3).
At T24, we observed better physical functioning and lower inflammatory activity in naïve versus non-naïve subjects (Table 2). Similarly, at T24, we observed better physical functioning and lower inflammatory activity in males versus females. ASDAS scores remained higher in females than males (Supplementary Table 4). At T24, we observed worse clinical measures (BASMI) and physical functioning (HAQs), and higher inflammatory activity (ESR) in r-axSpA versus nr-axSpA subjects (Supplementary Table 5).
Clinical, functional, disease activity, and serological parameters of naïve (n = 72) and non-naïve (n = 177) axSpA patients during the 24-month follow-up.
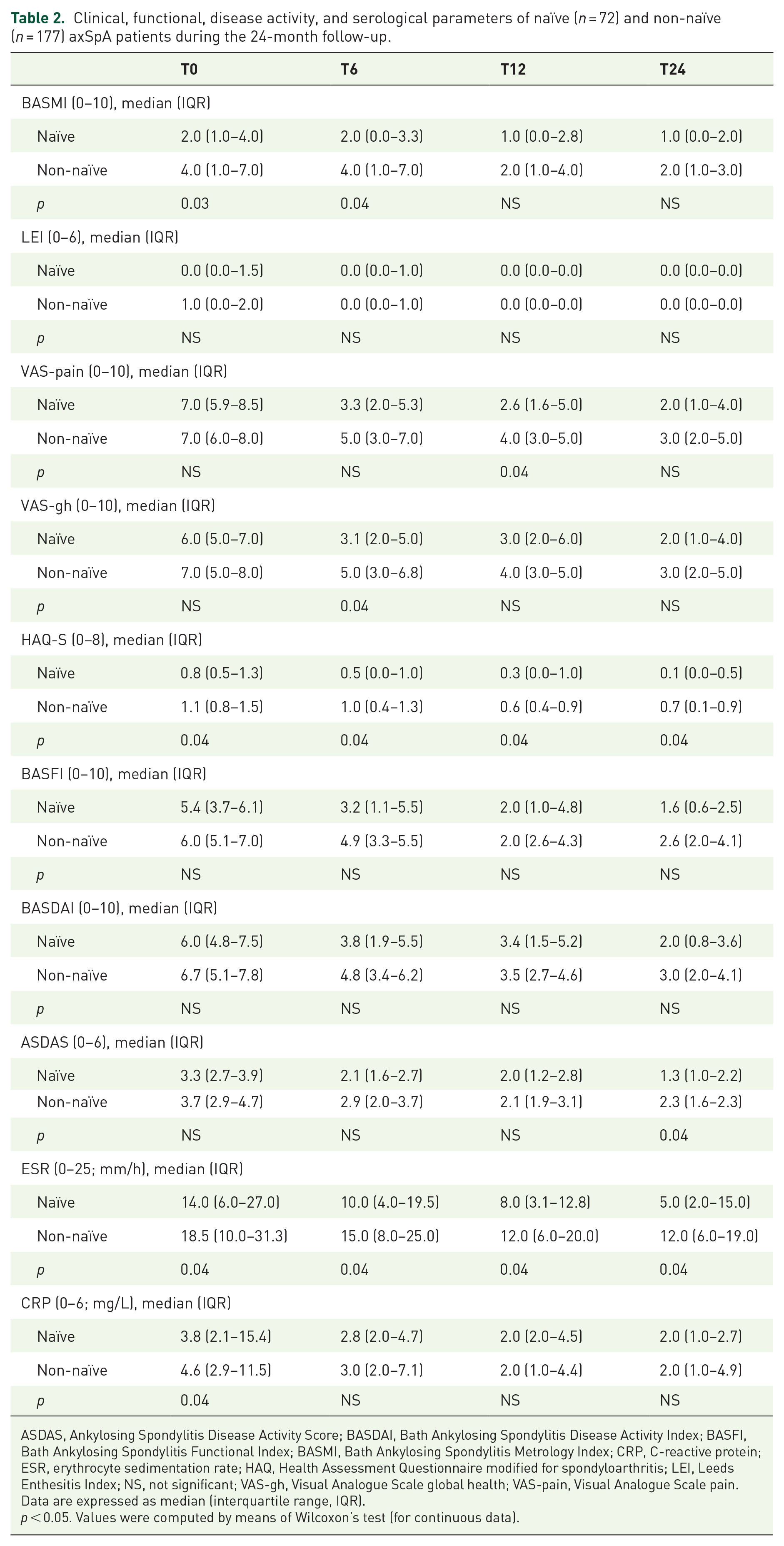
ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HAQ, Health Assessment Questionnaire modified for spondyloarthritis; LEI, Leeds Enthesitis Index; NS, not significant; VAS-gh, Visual Analogue Scale global health; VAS-pain, Visual Analogue Scale pain.
Data are expressed as median (interquartile range, IQR).
p < 0.05. Values were computed by means of Wilcoxon’s test (for continuous data).
At T24, naïve patients achieved VLDA/LDA–BASDAI states [Figure 1(a)–(d)] and VLDA/LDA–ASDAS states [Figure 2(a)–(d)] in a higher proportion than non-naïve. We also evaluated the proportion of achievements of VLDA/LDA according to gender and axSpA. As shown in Figure 1(b)–(e), males achieved VLDA/LDA–BASDAI states in a higher proportion than females and similarly for VLDA/LDA–ASDAS states [Figure 2(b)–(e)]. At T24, more r-axSpA patients than nr-axSpA achieved LDA–BASDAI [Figure 1(c)] and VLDA/LDA–ASDAS states [Figure 2(c)–(f)]. No differences were observed in VLDA–BASDAI achievement in relation to the axSpA subtype [Figure 1(f)].

BASDAI < 4 (percentage, %) of overall population and after their subdivision in two groups, according to lines of (a) biological treatment (LoBT; naïve/non-naïve), (b) gender (male/female), and (c) axSpA subtype (r-axSpA/nr-axSpA). BASDAI < 2 (percentage, %) of overall population and after their subdivision in two groups, according to lines of (d) biological treatment (LoBT; naïve/non-naïve), (e) gender (male/female), and (f) axSpA subtype (r-axSpA/nr-axSpA).

ASDAS < 2.1 (percentage, %) of overall population and after their subdivision in two groups, according to lines of (a) biological treatment (LoBT) (naïve/non-naïve), (b) gender (male/female), and (c) axSpA subtype (r-axSpA/nr-axSpA). ASDAS < 1.3 (percentage, %) of overall population and after their subdivision in two groups, according to lines of (d) biological treatment (LoBT) (naïve/non-naïve), (e) gender (male/female), and (f) axSpA subtype (r-axSpA/nr-axSpA).
The proportion of patients on csDMARDs was higher at T0 (14.5%, n = 36) than at T6 (13.1%, n = 30), at T12 (12.6%, n = 25) and at T24 (11.5%, n = 16), as was the proportion of patients treated with glucocorticoids, who were 14.5% (n = 36) at T0, 6.9% (n = 16) at T6, 6.0% (n = 12) at T12, and 3.6% (n = 5) at T24. A high reduction in NSAIDs intake was observed from T0 (59.4%, n = 148) to T6 (36.2%, n = 83), T12 (29.2%, n = 58), and T24 (25.2%, n = 35).
Drug survival
The retention rate at T24 was good (75%) in the whole population [Figure 3(a)–(c)]. The Kaplan–Meier curves did not show any differences between naïve and non-naïve (log-rank 0.435; p = 0.51) and between r-axSpA and nr-axSpA (log-rank 1.152; p = 0.283), whereas a significant difference was found between males and females (log-rank 9.319; p = 0.002).
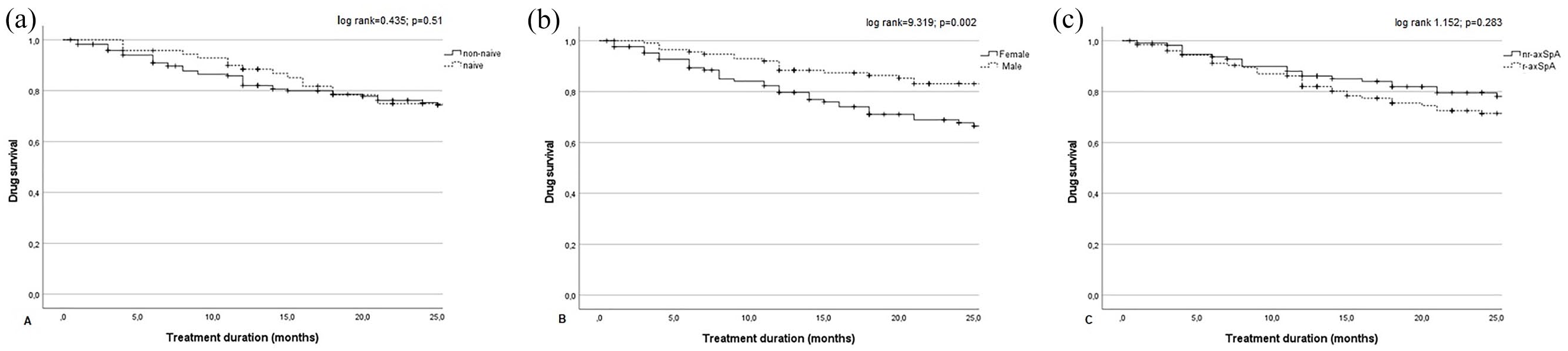
Drug survival in the overall population and after their subdivision in two groups, according to lines of (a) biological treatment (LoBT; naïve/non-naïve), (b) gender (male/female), and (c) axSpA subtype (r-axSpA/nr-axSpA).
Predictors of effectiveness
A multivariable regression analysis was performed to identify any baseline predictors of LDA achievement (according to BASDAI < 4 and ASAS < 2.1) at T6. As shown in Table 3, male gender was associated with a higher chance of BASDAI < 4 achievement at T6, while a higher CRP at T0, an intake of NSAIDs at T0 and the first LoTB were associated with a higher chance of ASDAS < 2.1 achievement. A Cox regression analysis was performed to identify predictors of time-to-SEC discontinuation. The variable that was independently associated with a higher chance of SEC discontinuation was the female gender (Table 3).
Multivariable logistic regression model with LDA (BASDAI < 4/ASDAS < 2.1) at T6 as outcome and Cox regression model with time-to-SEC-withdrawal as outcome.

ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; Bio-naïve, patients naïve to anti-TNF-alpha inhibitors; Coeff, coefficient; CI, confidence interval; CRP, C-reactive protein; HLA-B27, human leukocyte antigen B27; HR, hazard ratio; LDA, low disease activity; MRI, magnetic resonance imaging; NSAIDs, non-steroidal anti-inflammatory drugs; OR, odds ratio; +, positive; SEC, Secukinumab; SIJ, sacroiliac joint; T0, 0 months; T6, 6 months.
p < 0.05. Significant p-values have been highlighted in bold.
Comorbidities
The most frequently observed comorbidities were: metabolic syndrome (MetS; 10.8%, n = 27), hypertension (30.5%, n = 76), ischemic heart disease (5.2%, n = 13), dyslipidemia (24.1%, n = 60), hyperuricemia (7.2%, n = 18), type II diabetes (5.2%, n = 13), gastritis, gastric ulcer or dyspeptic disorders (9.6%, n = 24), liver disease (e.g. steatosis; 6.8%, n = 17), pneumopathies (4.8%, n = 12), thyroid disorder (14.5%, n = 36), osteoporosis (12.9%, n = 32), kidney failure (0.8%, n = 2), depression (8.8%, n = 22), fibromyalgia (19.3%, n = 48), neurological disorders (such as neuropathy; 6.8%, n = 17), positive Mantoux RB skin test or Quantiferon TB Gold test (6.8%, n = 17) without active tuberculous disease, previous hepatitis B (8.0%, n = 20), previous hepatitis C (0.8%, n = 2), previous eradicated cancer (4.4%, n = 11). The frequency of these comorbidities were described in both naïve and non-naïve patients as well as in males/females and r-axSpA/nr-axSpA subjects (Supplementary Tables 6–8). A higher prevalence of dyslipidemia, osteoporosis, and fibromyalgia was found in the non-naïve subjects (Supplementary Table 6). Males more frequently had ischemic cardiovascular diseases, MetS, hyperuricemia, and liver steatosis, while females had a higher prevalence of thyroid disorders, osteoporosis, depression, and fibromyalgia (Supplementary Table 7). A higher prevalence of hypertension, cardiovascular diseases, dyslipidemia, liver diseases, and gastric disorders was found in r-axSpA patients, while fibromyalgia was more prevalent in nr-axSpA (Supplementary Table 8).
Safety and discontinuation
Reasons for discontinuation
Sixty-one patients (24.5%) stopped the treatment during the follow-up, mainly because of primary and secondary loss of effectiveness (20 and 24, respectively). Only 17 patients suspended SEC because of adverse events [7 for reactions at the injection site or skin manifestations, 1 for gastro-intestinal complications, 1 for relapsing uveitis, 1 for hypertransaminasemia, 4 for severe recurrent infections, 3 due to the onset of new cancer (1 thyroid neoplasm, 1 breast cancer, 1 Hodgkin lymphoma)]. Liver and renal functions were monitored in all patients during the study period, and only one patient presented abnormal values.
Infections
A low number of episodes of mild infections (42) occurred during the study period (19 respiratory tract infections, 2 dental infections, 7 oral or vaginal candidiasis, 3 herpetic labial infections, 2 herpes zoster, 1 gastroenteritis, 2 skin infections, 6 urinary tract infections); all resolved following oral antimicrobial treatment, without hospitalization or drug discontinuation. Four patients presented severe infections, which caused them to suspend the SEC treatment (two bronchopneumonia, one erysipelas with sepsis from St. Aureus with hospitalization, and one urinary infection with sepsis from E. coli). No difference between gender/LoBT/axSpA subtype in terms of safety was observed (data not shown).
Discussion
In this prospective multicenter study in patients with active axSpA, SEC was effective, safe, and well tolerated. In addition, over 80% patients managed to achieve LDA-BASDAI at 6 months, and majority of them (75%) remained on treatment after 24 months.
At T6, T12, and T24, SEC reduced all the clinical and inflammatory indexes, in addition to PROs, providing significant improvement in disease activity, physical function, and quality of life in axSpA patients who were either naïve to a prior biologic therapy or had shown an inadequate response to TNF inhibition. We also observed a marked improvement in the BASDAI and ASDAS-CRP scores during the first 6 months of treatment. The efficacy outcomes of this study are consistent with previous RCTs.20–28 MEASURE 1 reported an ASAS20/40 response of 80.2%/61.6% in the SEC 150 mg arm at week 156, and sustained improvements were also seen in BASDAI, BASFI, BASMI, and across all other endpoints. 36 Similarly, in MEASURE 2, SEC 150 mg provided sustained improvements in the ASAS20/40 response rates at week 156 (70.1%/60.9%) versus week 52 (74.2%/57.0%). 23 Furthermore, in the PREVENT, SEC 150 mg met both primary endpoints at weeks 16 and 52 in naïve patients with nr-axSpA. 28 Of note, data on SEC retention rate in our study appeared good, in line with RCTs and other registries. In MEASURE 1, the 2-year SEC retention was 78%; 37 in MEASURE 2, the 3-year retention rate was 86% for SEC 150 mg every 4 weeks; 23 and in MEASURE 3, the 1-year retention rate was 87%. 24 Similarly, the retention rate was high in the PREVENT, with 95% of randomized patients completing week 24 and 86.7% completing week 52. 28 These results were slightly higher than the overall 2-year retention rate of 75% achieved in our real-life study, which instead appeared similar to those found in previous real-life observational studied: EuroSpA – a research collaboration network among 13 registries which presented data from over 1860 axSpA patients treated with SEC – reported a 1-year retention rate of 72%, 38 while an Italian study of 39 axSpA subjects reported a 2-year retention rate of 78.2%. 29 Compared with the patients in MEASURE 2 and 3, the patients in our study were older (mean age 51 versus 42 and 43), had a shorter time from diagnosis (4.0 versus 7.0 and 6.0 years), a lower proportion were TNFi-naïve (28.9% versus 61% and 57%) and we included patients with both r-axSpA and nr-axSpA, whereas only patients with r-axSpA were included in MEASURE 2 and 3.23,24 Other baseline axSpA features were similar. These results obtained from our real-life observational study confirm the prompt effectiveness of SEC on axSpA symptoms and systemic inflammation.
We also focused on searching for any differences in clinical response between patients with different LoBT, gender, or axSpA subtype. Regarding LoBT, our data on clinical response were consistent with literature,20–27 since no differences in retention rate were observed between naïve and non-naïve axSpA patients. In fact, the 12- and 24-month SEC retention rate for naïve patients in our study (86% and 76%) was similar to the 12- and 24-month retention rate to first-line TNFi in 842 naïve AS patients (74% and 63%), 39 as well as to other studies on first-line TNFi treatment. 40 However, effectiveness differed significantly between naïve and non-naïve subjects, with naïve axSpA having better response rates and disease state. In fact, for naïve patients, the achievement of 12-month and 24-month LDA in our study was comparable to those of naïve axSpA patients in the EuroSpA study, in which 12-month BASDAI < 4 was achieved by 76% and ASAS20/40 response by 69%/55% of patients, respectively. 36 These findings in our cohort were slightly higher than those of non-naïve axSpA. The efficacy of SEC turned out to be independent of treatment line and previous bDMARDs use, and the drug can therefore be considered effective both as first-line therapy and in multi-failure patients. Data from multinational registries, such as EuroSpA, have also compared the characteristics of patients starting treatment with SEC or TNFi, and the effectiveness of SEC was similar in both naïve and non-naïve axSpA patients. 41 Therefore, SEC can be considered as a drug of choice in patients with one or more previous TNFi failure.42–44 Accordingly, a comparative real-life cohort study of two modes of biological action in axSpA, after prior withdrawal from at least one TNFi, showed that patients treated with SEC experienced outcomes comparable to those of patients treated with an alternative TNFi. 45
Furthermore, no differences in drug retention emerged between r-axSpA and nr-axSpA subjects, while some emerged regarding functional outcomes: r-axSpA patients presented worse clinical measures, physical functioning, and inflammatory activity.
An important observation is the influence of gender on the achievement and maintenance of LDA and VLDA. Males reached a better disease control than females; male sex was also found to be a predictor of LDA achievement at T6. Interestingly, in literature, female gender was associated with poorer bDMARDs response rates and with a lower probability of achieving remission. 46 Similarly, the PREVENT study performed a subgroup analysis, which revealed that male axSpA patients treated with SEC had higher relative responses than female patients with ASAS40 response rates of 51.2% and 31.7%, respectively. 47 As far as time-to-SEC discontinuation is concerned, male sex appeared to be protective against drug discontinuation. This finding is in accordance with other reports concerning a higher discontinuation rate of first- and second-line biologics among female axSpA.48,49 Female axSpA patients more frequently present an entheseal involvement, often compounded by fibromyalgia, which amplifies the perception of pain and fatigue, and therefore negatively impacts the self-reported disease activity. 50 Furthermore, concomitant fibromyalgia may represent a challenge to the therapeutic strategy, since fibromyalgia-associated symptoms can alter the assessment of clinical response to treatments over time. This aspect is very relevant, since in our population a considerable prevalence of fibromyalgia was found (19.3%), mainly in females (n = 42; 32.3%) versus males (n = 6; 5.0%).
Overall, treatment with SEC was well tolerated among our axSpA cohort (only 17 cases leading to drug withdrawal for adverse events). The safety profile was consistent with the established safety profile across the approved indications.51,52 One of the most commonly reported infections – not usually leading to IL-17 inhibitors interruption – was mucocutaneous candidiasis,32,38 which was observed in only seven patients, preceded by more frequent respiratory tract infections. Other infections were mild-to-moderate and did not lead to treatment discontinuation. New-onset IBD was not identified for up to 2 years of treatment, while only one patient discontinued SEC for relapsing uveitis: these findings appeared consistent with previously reported data on SEC in patients with AS.24–26 Seventeen (6.8%) patients presented positive Mantoux RB skin test or Quantiferon TB Gold test at baseline. None of these patients developed an active tuberculous disease during the course of treatment. No difference was observed between LoBT/gender/axSpA subtype in terms of safety.
Conversely, some differences in comorbidities distribution were observed between the subgroups, without them affecting the continuity of treatment with SEC. A higher prevalence of hypertension, cardiovascular diseases, dyslipidemia, liver diseases, and gastric disorders was found in r-axSpA, as well as a higher prevalence of dyslipidemia and osteoporosis in non-naïve subjects (probably due to a longer history of axSpA disease and side effects from multiple treatment lines, in particular steroids and csDMARDs). As expected, males had more frequently ischemic cardiovascular diseases, MetS, hyperuricemia, and liver steatosis, while females had a higher prevalence of thyroid disorders, osteoporosis, depression, and fibromyalgia. These findings were corroborated by previous studies.46–50 Of note, fibromyalgia was more prevalent in nr-axSpA subjects; the possible reason can be attributed to a higher prevalence (58.3%) of the female gender in the nr-axSpA subgroup.
The major strengths of this study are (1) the prospective study design, (2) the large multicenter study population reflecting routine care, (3) the 24-month follow-up, and (4) the inclusion of a wide cohort of nr-axSpA patients.
Some of the limitations of our study include (1) a registry study data quality lower than RCTs, due to missing data on response outcomes across the registries; (2) the absence of imaging follow-up (pelvic and spinal X-rays and MRI), although imaging was often performed to confirm the diagnosis at baseline; (3) the use of the BASDAI cut-offs <2 and <4 (there is no consensus on the ideal cut-off for BASDAI remission in axSpA, although the cut-off of 4 has previously been recognized in literature for the definition of active AS); and (4) the choice to schedule evaluations after 6 months the starting treatment considering the unavailability of complete data at 3 months.
In conclusion, in this clinical setting, SEC was safe and effective in axSpA, regardless of the biologic treatment line. As suggested by the significant drug retention rate, SEC was able to maintain its effectiveness over a considerably long treatment period. Male gender appears to correlate with LDA and inactive disease achievement and drug persistence, whereas the subtype of axSpA (r-axSpA or nr-axSpA) does not appear to impact the drug-discontinuation rate. The safety of SEC allows for its use in patients with comorbidities. This study demonstrated that SEC can be a viable option to treat the entire spectrum of axSpA, that is, from early to late stage or from nr-axSpA to r-axSpA. Further head-to-head studies are warranted to better understand the differences on treatment effectiveness of TNFi and IL-17 pathway inhibitors, and to potentially identify specific subsets of patients who may benefit most from switching to IL-17 inhibitors.
Supplemental Material
sj-doc-2-tab-10.1177_1759720X221090310 – Supplemental material for Effectiveness and safety of secukinumab in axial spondyloarthritis: a 24-month prospective, multicenter real-life study
Supplemental material, sj-doc-2-tab-10.1177_1759720X221090310 for Effectiveness and safety of secukinumab in axial spondyloarthritis: a 24-month prospective, multicenter real-life study by Roberta Ramonda, Mariagrazia Lorenzin, Maria Sole Chimenti, Salvatore D’Angelo, Antonio Marchesoni, Carlo Salvarani, Ennio Lubrano, Luisa Costa, Ylenia Dal Bosco, Elena Fracassi, Augusta Ortolan, Mario Ferraioli, Antonio Carriero, Elisa Visalli, Riccardo Bixio, Francesca Desiati, Alberto Bergamini, Elisa Pedrollo, Andrea Doria, Rosario Foti and Antonio Carletto in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221090310 – Supplemental material for Effectiveness and safety of secukinumab in axial spondyloarthritis: a 24-month prospective, multicenter real-life study
Supplemental material, sj-docx-1-tab-10.1177_1759720X221090310 for Effectiveness and safety of secukinumab in axial spondyloarthritis: a 24-month prospective, multicenter real-life study by Roberta Ramonda, Mariagrazia Lorenzin, Maria Sole Chimenti, Salvatore D’Angelo, Antonio Marchesoni, Carlo Salvarani, Ennio Lubrano, Luisa Costa, Ylenia Dal Bosco, Elena Fracassi, Augusta Ortolan, Mario Ferraioli, Antonio Carriero, Elisa Visalli, Riccardo Bixio, Francesca Desiati, Alberto Bergamini, Elisa Pedrollo, Andrea Doria, Rosario Foti and Antonio Carletto in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors wish to thank the Italian Society of Rheumatology (SIR) ‘Spondyloarthritis and Psoriatic Arthritis Study Group—A. Spadaro’. The authors would like to thank T&T Attubato S.a.s. for native English check of this article. Coordinators of the Spondyloarthritis and Psoriatic Arthritis SIR Study Group ‘Antonio Spadaro’: Antonio Marchesoni, MD, PhD, Rheumatology, Humanitas San Pio X, Milan, Lombardia, Italy; Alberto Cauli, MD, PhD, Rheumatology Unit, Department of Medical Sciences, AOU and University of Cagliari, Cagliari, Sardegna, Italy; Salvatore d’Angelo, MD, PhD, Rheumatology Institute of Lucania (IReL), Rheumatology Department of Lucania, San Carlo Hospital of Potenza and Madonna delle Grazie Hospital of Matera, Potenza Local Health System, Potenza, Basilicata, Italy; Ennio Lubrano, MD, PhD, Academic Rheumatology Unit, Dipartimento di Medicina e Scienze per la Salute ‘Vincenzo Tiberio’, University of Molise, Campobasso, Molise, Italy; Roberta Ramonda, MD, PhD, Rheumatology Unit, Department of Medicine–DIMED, University of Padova, Padova, Veneto, Italy; Carlo Salvarani, MD, PhD, Rheumatology Unit, Department of Internal Medicine, Azienda USL-IRCCS, Istituto di Ricovero e Cura a Carattere Scientifico, Reggio Emilia, Italy; University of Modena and Reggio Emilia, Modena, Emilia Romagna, Italy. Contributing authors of the Spondyloarthritis and Psoriatic Arthritis SIR Study Group ‘Antonio Spadaro’. Alberto Cauli, MD, PhD, Rheumatology Unit, Department of Medical Sciences, AOU and University of Cagliari, Cagliari, Sardegna, Italy; Angelo Semeraro, MD, Rheumatology Unit, Martina Franca-ASL Taranto, Puglia, Italy; Leonardo Santo, MD, Rheumatology Unit, ASL BT Andria – DSS4 Barletta, Italy, Barletta-Andria-Trani, Puglia, Italy; Emanuela Praino, MD, Rheumatology Unit, ASL BT Andria – DSS4 Barletta, Italy, Barletta-Andria-Trani, Puglia, Italy; Giorgio Amato, MD, Rheumatology Unit, A.O.U. Policlinico S. Marco, Catania, Sicilia, Italy; Nicolò Girolimetto, MD, Rheumatology Unit, Department of Internal Medicine, Azienda USL-IRCCS, Istituto di Ricovero e Cura a Carattere Scientifico, Reggio Emilia, Emilia Romagna, Italy.
Author contribution(s)
Spondyloarthritis and Psoriatic Arthritis SIR Study Group “Antonio Spadaro” Project administration
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RR has received honoraria and speaker fees from Novartis, AbbVie, Pfizer, MSD, and Janssen. AM has received honoraria and speaker fees from AbbVie, Pfizer, MSD, UCB, Novartis, Janssen, and Eli-Lilly. LS received speaker fees from Jansen, Novartis, Pfizer, UCB, MSD, and Sanofi.
The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This support was funded by Novartis Farma.
ORCID iDs
Data availability statement
De-identified data are available upon reasonable request, by contacting the corresponding author by email.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
