Abstract
Introduction:
Chronic inflammatory diseases (CIDs), including rheumatic diseases and other inflammatory conditions, often affect women of reproductive age. Tumor necrosis factor inhibitors (TNFi) are widely used to treat CID, but there is limited information on outcomes of TNFi-exposed pregnancies. We evaluated pregnancy outcomes from 1392 prospectively reported pregnancies exposed to certolizumab pegol (CZP), a PEGylated, Fc-free TNFi with no to minimal placental transfer.
Methods:
CZP-exposed pregnancies in patients with CID from the UCB Pharmacovigilance global safety database were reviewed from the start of CZP clinical development (July 2001) to 1 November 2020. To limit bias, the analysis focused on prospectively reported cases with known pregnancy outcomes.
Results:
In total, 1392 prospective pregnancies with maternal CZP exposure and known pregnancy outcomes (n = 1425) were reported; 1021 had at least first-trimester CZP exposure. Live birth was reported in 1259/1425 (88.4%) of all prospective outcomes. There were 150/1425 (10.5%) pregnancy losses before 20 weeks (miscarriage/induced abortion), 11/1425 (0.8%) stillbirths, and 5/1392 (0.4%) ectopic pregnancies. Congenital malformations were present in 30/1259 (2.4%) live-born infants, of which 26 (2.1%) were considered major according to the Metropolitan Atlanta Congenital Defects Program criteria. There was no pattern of congenital malformations.
Discussion and conclusion:
No signal for adverse pregnancy outcomes or congenital malformations was observed in CZP-exposed pregnancies. Although the limitations of data collected through this methodology (including underreporting, missing information, and absence of a comparator group) should be considered, these data provide reassurance for women with CID who require CZP treatment during pregnancy, and their treating physicians.

Graphical Summary
Introduction
Chronic inflammatory diseases (CIDs), including rheumatoid arthritis (RA), axial spondyloarthritis (axSpA), psoriatic arthritis (PsA), Crohn’s disease (CD), and psoriasis (PSO), often affect women of reproductive age. Many of these diseases manifest in early adulthood, meaning disease activity often overlaps with peak reproductive years for women.1,2 There is evidence that uncontrolled disease activity in CID during pregnancy can increase the rate of adverse pregnancy outcomes such as miscarriage, preterm delivery, and low birth weight; therefore, these pregnancies may be considered higher risk when left untreated.3–12 Careful management of women with CID who are pregnant, potentially pregnant, or planning pregnancy at initiation of a pharmacological treatment is therefore crucial, including ensuring patients reach a stable condition with low inflammatory activity prior to pregnancy. Moreover, because many pregnancies are unplanned, reproductive health and potential for pregnancy should be discussed with women of childbearing age with CID early in the course of disease.13,14
Some medications commonly used to treat rheumatic disease, including corticosteroids, nonsteroidal anti-inflammatory drugs (NSAIDs), methotrexate, and opioids, have been associated with adverse pregnancy outcomes,15–19 resulting in a need to reduce their intake during pregnancy. In line with current treatment guidelines,14,20–22 tumor necrosis factor inhibitors (TNFi) are widely used to treat CIDs, and currently available data suggest their use is compatible with the first trimester of pregnancy, when organogenesis occurs, and for some TNFi, the second and third trimesters.12,20 Evidence is sufficient to indicate that certolizumab pegol (CZP) can be continued for the entire duration of pregnancy in women with rheumatic disease, though some guidelines suggest stopping other TNFi, including etanercept, adalimumab, and infliximab in the weeks or months prior to delivery to decrease fetal exposure.14,20,23 Given this changing treatment landscape for CID in pregnancy, it is important health care providers have access to all available information to enable individual decision making in women of childbearing age, and ensure the benefits and risks of biologic therapies are appropriately understood.
Currently, most evidence on pregnancy outcomes following exposure to biologics comes from observational and cross-sectional studies. 24 However, prospective/retrospective registries, real-world evidence, and pharmacovigilance databases may provide additional evidence in biologic-treated women during pregnancy.
CZP is a PEGylated, Fc-free TNFi established for the treatment of several inflammatory diseases, including RA, axSpA, PsA, CD, and PSO.25,26 Unlike other TNFi, CZP lacks an Fc fragment and thus does not bind the neonatal Fc receptor, which mediates maternal–fetal transfer of antibodies from the first trimester onward.27,28 Clinical pharmacokinetic studies showed that CZP had no to minimal levels of placental transfer during the third trimester;29,30 continued CZP use is therefore considered to be compatible with pregnancy in women with a CID if clinically needed.25,26 Furthermore, when prescribed postpartum, it has been shown to have minimal transfer from plasma to breast milk. 31
Data on pregnancies exposed to CZP from UCB’s Pharmacovigilance global safety database, using cutoff dates in 2014 and 2017, have been previously reported, with 528 prospective pregnancies with known outcomes reported up to 2017.32,33 These reports showed no increase in adverse outcomes in CZP-exposed prospective pregnancies, compared with the general population. Here, we report data up to November 2020 from 1392 prospective pregnancies with known outcomes and CZP exposure from the UCB Pharmacovigilance global safety database, including 1021 with first-trimester CZP exposure. Data collected include rates of adverse maternal/fetal outcomes, congenital malformations, as well as preterm delivery and birth weight. Pregnancy outcomes across different indications for CZP treatment are broken down for further review. In addition, confounding factors potentially contributing to adverse pregnancy outcomes in severe CID patients treated with CZP are evaluated.
Methods
Data source and patient populations
The methodology used to collect pregnancy data has been previously reported. 33 Details of pregnancies with CZP exposure were obtained from the UCB Pharmacovigilance global safety database from the start of CZP clinical development (earliest report 12 July 2001) up to a cutoff date of 1 November 2020. Data were obtained from spontaneous reports (information received directly from the patient or health care provider), or from interventional and non-interventional studies (including non-pregnancy registry studies). Organization of Teratology Information Specialists (OTIS) case reports were excluded, since they were collected as part of an ongoing prospective pregnancy registry (NCT01797224).
Pregnancy outcomes were collected for prospective and retrospective pregnancies with CZP exposure. A prospective pregnancy is one in which CZP exposure was reported to UCB Pharma prior to knowledge of the pregnancy outcome or prior to detection of a congenital malformation on prenatal examination. A retrospective pregnancy is one in which CZP exposure was reported to UCB Pharma after the outcome of the pregnancy was known or after detection of a congenital malformation on prenatal test. To limit potential reporting bias associated with retrospective cases, here we focus on outcomes from prospectively reported pregnancies with maternal CZP exposure up to the cutoff date, including those previously published.32,33
Pregnancy outcomes
Pregnancy outcomes were descriptively reported and included live birth, ectopic pregnancy, pregnancy loss before 20 weeks of gestation due to miscarriage (non-induced embryonic/fetal death before 20 weeks’ gestation) or induced abortion (medically indicated or elective), stillbirth (fetal death occurring at or after 20 weeks’ gestation), and congenital malformations. For live births, we analyzed the following outcomes: congenital malformations, preterm delivery (delivery before 37 weeks’ gestation), preterm delivery according to reported maternal corticosteroid use, and low birth weight (<2500 g).34,35 Where both birth weight and gestational age at delivery were recorded, the percentage of live-born infants with low birth weight who were small-for-gestational age (SGA; birth weight <10th centile 36 ) was calculated.
Pregnancies with unknown outcomes included ongoing pregnancies and those with no pregnancy outcome reported; these pregnancies were not included in the analysis.
Timing of CZP exposure during pregnancy was recorded by trimester, defined based on the first day of the last menstrual period. The first trimester was defined as the period up to 12 weeks and 6 days’ gestation, the second as 13–28 weeks and 6 days’ gestation, and the third as any time at or after 29 weeks’ gestation. If no information on timing of CZP exposure during pregnancy was available, this was reported as ‘unknown’ in the safety database. Also collected in the database were maternal indication(s) for CZP treatment, maternal age at the start of pregnancy, maternal comorbidities (gestational diabetes, hypertensive disease of pregnancy, infections during pregnancy, and postpartum), country of origin, and concomitant medications.
Cases of congenital malformations were reviewed by an external clinical teratologist and classified as major or minor using the Metropolitan Atlanta Congenital Defects Program (MACDP) 37 and the European Surveillance of Congenital Anomalies (EUROCAT) criteria. 38
Analysis of variables associated with pregnancy outcomes
A univariate logistic regression analysis was used to identify baseline variables associated with pregnancy loss at <20 weeks’ gestation, preterm birth, and low birth weight. The following baseline variables (yes/no unless otherwise stated) were included: maternal age (⩽35 or >35 years); CZP indication: RA, PsA, axSpA, CD, and PSO; gestational diabetes; hypertensive disease of pregnancy (including hypertension, preeclampsia, and eclampsia); maternal infections; and any use of the following drugs during pregnancy: systemic corticosteroids excluding topical and inhaled preparations, NSAIDs, opioid analgesics, and methotrexate/leflunomide. Dexamethasone use, recorded in three cases, was not excluded from the model because the indication for its prescription was not reported. Each variable also included an ‘unknown’ category to account for missing information not reported in the safety database. All of these variables, if present in at least 10% of the CZP-exposed population, were entered into a multivariable stepwise regression model. The multivariable model used a forward selection criterion of p < 0.05 and a backward elimination criterion of p ⩾ 0.1. Given the heterogeneity and sparsity of the information reported, it was not possible to include dose and time of exposure of concomitant medications in either the univariate or multivariable analysis.
Outcomes from the univariate and multivariable analyses are reported as odds ratios (ORs) with 95% confidence intervals (CIs).
A checklist for the reporting of studies conducted using observational routinely collected health data statement for pharmacoepidemiology (RECORD-PE) is provided in the supplementary material (Supplementary Table 1). 39
Patient and public involvement
Neither patients nor other members of the public were involved in the design, conduct, reporting, or dissemination of this research.
Results
Patient disposition and baseline characteristics
A total of 5681 reports of pregnancies (including both prospective and retrospective cases) with CZP exposure were identified in the UCB Pharmacovigilance global safety database from the start of CZP clinical development up to the cutoff date of 1 November 2020, from all reporting sources, as described above. Of these, 5576/5681 (98.2%) and 105/5681 (1.8%) had maternal and paternal CZP exposure, respectively (Figure 1).

Reports of pregnancies exposed to CZP identified in the UCB Pharmacovigilance global safety database.
Of all 4234 reported prospective pregnancies, 1392 (32.9%) had known outcomes (n = 1425 pregnancy outcomes; see Figure 1 and Table 1). Of these pregnancies, the most common indication for CZP treatment was RA (n = 643), followed by CD (n = 293), axSpA (n = 215), PsA (n = 113), PSO (n = 61), and other (n = 52); diagnosis data were missing in 60 patients; some patients had multiple indications.
Demographics and baseline characteristics of prospective pregnancies with maternal CZP exposure.
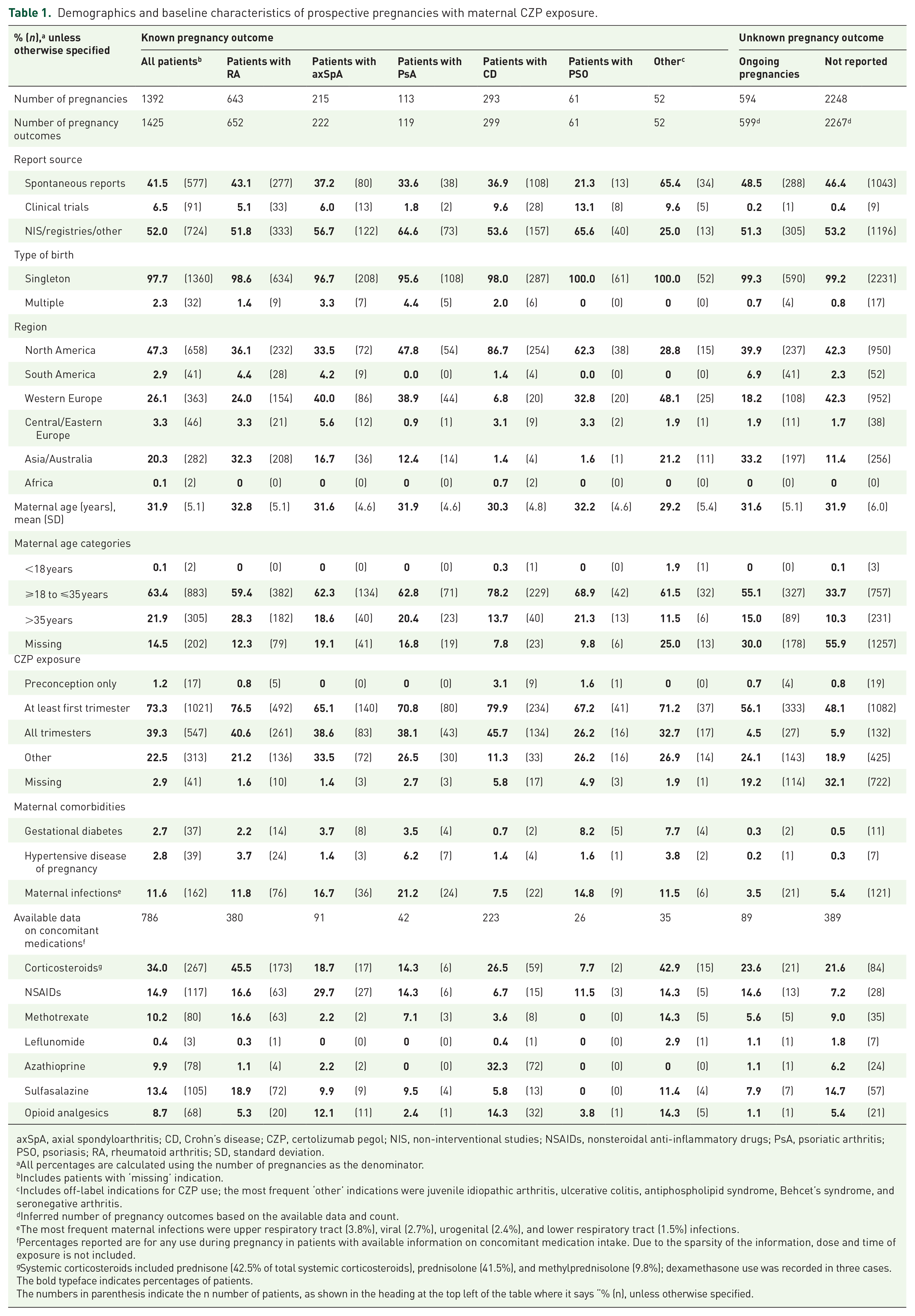
axSpA, axial spondyloarthritis; CD, Crohn’s disease; CZP, certolizumab pegol; NIS, non-interventional studies; NSAIDs, nonsteroidal anti-inflammatory drugs; PsA, psoriatic arthritis; PSO, psoriasis; RA, rheumatoid arthritis; SD, standard deviation.
All percentages are calculated using the number of pregnancies as the denominator.
Includes patients with ‘missing’ indication.
Includes off-label indications for CZP use; the most frequent ‘other’ indications were juvenile idiopathic arthritis, ulcerative colitis, antiphospholipid syndrome, Behcet’s syndrome, and seronegative arthritis.
Inferred number of pregnancy outcomes based on the available data and count.
The most frequent maternal infections were upper respiratory tract (3.8%), viral (2.7%), urogenital (2.4%), and lower respiratory tract (1.5%) infections.
Percentages reported are for any use during pregnancy in patients with available information on concomitant medication intake. Due to the sparsity of the information, dose and time of exposure is not included.
Systemic corticosteroids included prednisone (42.5% of total systemic corticosteroids), prednisolone (41.5%), and methylprednisolone (9.8%); dexamethasone use was recorded in three cases.The bold typeface indicates percentages of patients. The numbers in parenthesis indicate the n number of patients, as shown in the heading at the top left of the table where it says “% (n), unless otherwise specified.
Prospective pregnancies with known outcomes
Maternal demographics
In total, 577/1392 (41.5%) records of prospective pregnancies with maternal CZP exposure were obtained from spontaneous reports and 91/1392 (6.5%) from clinical trials. The remaining 724/1392 (52.0%) came from non-interventional studies, including non-pregnancy registry data or other sources. Most reports originated from patients based in North America (658/1392; 47.3%) or Western Europe (363/1392; 26.1%) (Table 1). The mean maternal age was 31.9 years, with most patients (883/1392; 63.4%) aged ⩾18 to ⩽35 years. The mean maternal age across indications was comparable, ranging from 29.2 to 32.8 years. Most prospective pregnancies (1021/1392; 73.3%) were exposed to CZP at least during the first trimester of the pregnancy; 547/1392 (39.3%) were exposed to CZP during all trimesters. Where information was available, maternal comorbidities and concomitant medication intake, including corticosteroids, NSAIDs, methotrexate, leflunomide, azathioprine, sulfasalazine, and opioid analgesics, are reported (Table 1). Baseline characteristics of pregnancies with unknown outcomes were comparable to pregnancies with known outcomes (Table 1).
Pregnancy outcomes
The 1392 pregnancies with 1425 known pregnancy outcomes resulted in 1259/1425 (88.4%) live births (Figure 2). Across all indications, live birth was the most common pregnancy outcome.

Pregnancy outcomes of prospectively reported pregnancies with maternal CZP exposure.
There were 150/1425 (10.5%) pregnancy losses before 20 weeks’ gestation (Figure 2), including 111/1425 (7.8%) miscarriages and 39/1425 (2.7%) elective or medically indicated abortions. There were 11/1425 (0.8%) stillbirths in total: five occurred in patients with RA and two each in patients with axSpA, PSO, or CD. Ectopic pregnancies were reported in 5/1392 (0.4%) cases: three occurred in patients with RA and one each in women with PSO or CD. In total, 124/1259 (9.8%) live births were preterm and 101/1259 (8.0%) of infants had low birth weight (Table 2). In the overall cohort, quantitative birth weight information plus exact gestational age was only available in 116 live-born infants, precluding estimation of the percentage of SGA infants for the overall cohort.
Adverse outcomes in prospectively reported pregnancies with known pregnancy outcome and maternal CZP exposure.

axSpA, axial spondyloarthritis; CD, Crohn’s disease; CZP, certolizumab pegol; PsA, psoriatic arthritis; PSO, psoriasis; RA, rheumatoid arthritis.
Percentages of ectopic pregnancies are reported using the total number of pregnancies as the denominator.
<2500 g. Both gestational age and birth weight were recorded in 82/101 infants with low birth weight; of these, 54/82 (65.9%) were small-for-gestational age.The numbers in parenthesis indicate the n number of patients, as shown in the heading at the top left of the table where it says “% (n), unless otherwise specified
Congenital malformations
Congenital malformations were reported in 35/1425 known pregnancy outcomes (2.5%), of which 30/1259 presented in live-born infants (2.4%) (Table 2). Of all cases with congenital malformations, 6/35 (17.1%) did not have known first-trimester CZP exposure (unknown exposure in three cases). A total of 26 congenital malformations that presented in 22 live-born infants were classified as major according to the MACDP criteria, representing a prevalence of 2.1% (26/1259) in live-born infants. According to EUROCAT criteria, 17 congenital malformations in 16 live-born infants were major (1.4% (17/1259) of all live births). All five congenital malformations occurring in pregnancies ending in either miscarriage or elective abortion were major according to MACDP criteria (Table 3); according to EUROCAT criteria, only two were major (classification was ‘unknown’ in one case).
Congenital malformations in prospectively reported pregnancies with maternal CZP exposure..

axSpA, axial spondyloarthritis; CD, Crohn’s disease; CZP, certolizumab pegol; EUROCAT, European Surveillance of Congenital Anomalies; MACDP, Metropolitan Atlanta Congenital Defects Program; NOS, not otherwise specified; PsA, psoriatic arthritis; PSO, psoriasis; RA, rheumatoid arthritis; VACTERL, vertebral defects, anal atresia, cardiac defects, tracheo-esophageal fistula, renal anomalies, and limb abnormalities.
Reported as part of VACTERL syndrome.
Factors associated with outcomes in prospective CZP-exposed pregnancies
Results of the initial univariate analyses, performed to evaluate associations between baseline variables and pregnancy outcomes (see ‘Methods’ section), are presented in Supplementary Figure 1.
In the multivariable stepwise regression model, reported corticosteroid use (i.e. any use during pregnancy) was identified as associated with increased odds of preterm birth [OR (95% CI): 2.1 (1.3–3.4), p < 0.005] and low birth weight [OR (95% CI): 1.7 (1.0–2.9), p < 0.05] but decreased odds of pregnancy loss at <20 weeks [OR (95% CI): 0.5 (0.3–0.9), p < 0.05] (Figure 3). Reported NSAID use (i.e. any use during pregnancy) was associated with increased odds of pregnancy loss at <20 weeks [OR (95% CI): 2.2 (1.2–4.0), p < 0.05], as was methotrexate/leflunomide use [OR (95% CI): 3.2 (1.7–6.2), p < 0.0005] (Figure 3). The analysis also identified an association between a diagnosis of CD and pregnancy loss at <20 weeks [OR (95% CI): 2.5 (1.5–4.1), p = 0.0005], and between RA and low birth weight [OR (95% CI): 1.9 (1.1–3.3), p < 0.05] (Figure 3). Finally, maternal infections were associated with increased odds of preterm birth [OR (95% CI): 1.9 (1.1–3.5), p < 0.05] (Figure 3).

Potential confounders identified as significantly associated with pregnancy outcomes from multivariable stepwise regression analysis.a
Retrospective pregnancies with known outcomes
Out of 1362 outcomes from 1342 retrospectively reported maternal exposure pregnancies, 1259 had known outcomes, including 1014 (80.5%) live births, 199 (15.8%) miscarriages, 11 (0.9%) medically indicated abortions, 9 (0.7%) elective abortions, 13 (1.0%) stillbirths, and 13 (1.0%) ectopic pregnancies; 71/1014 (7.0%) live births were reported preterm and 43/1014 (4.2%) infants had low birth weight. Congenital malformations were reported in 38 cases out of 1259 (3.0%) outcomes, of which 28 cases out of 1014 (2.8%) outcomes presented in live-born infants.
Discussion
This analysis reports outcomes from the largest pregnancy safety database dedicated to CZP use in patients with CID. Outcomes are reported for 1392 prospective pregnancies with maternal CZP exposure, including 1021 with first-trimester exposure. Over 85% of all prospectively reported pregnancies with CZP exposure and known outcomes resulted in live birth, with no observed increase in adverse outcomes.
In the general population, rates of miscarriage range from 12% to 25%,40,41 abortion 1% to 20.4%,42,43 major congenital malformations 2% to 3%,44,45 preterm birth 12%, 46 and low birth weight 8.2%. 46 Rates of adverse pregnancy outcomes reported in this analysis are in keeping with those from the general population,47–51 those from previous registry studies evaluating biologics in pregnancy in women with CID,52,53 and also with previous publications from the UCB Pharmacovigilance global safety database reporting prospective pregnancy outcomes using cutoff dates in 2014 (339 pregnancies) 32 and 2017 (528 pregnancies). 33
In particular, the rates of major congenital malformations in prospective pregnancies from this database were comparable to those reported in the general population,44,45 although caution is required in assessing data collected through different methodologies. Nonetheless, no pattern of congenital malformations was identified, and there was no discernible trend between maternal CZP indication and major congenital malformations. According to MACDP criteria, 37 26 major congenital malformations occurred in 1259 live-born infants (2.1%), while by EUROCAT criteria, 38 17 were major (1.4%), highlighting that methodological approaches for classifying abnormalities can impact their reported prevalence. In retrospectively reported maternal exposure pregnancies, rates of congenital malformations (2.8% in live births) and other adverse pregnancy outcomes were of similar magnitude, despite the reporting bias inherently associated with retrospective cases.
Whether in utero exposure to TNFi is associated with increased risk of neonatal infections has been investigated in several studies that used heterogeneous methodological approaches and population cohorts. Recent studies on large cohorts of TNFi-exposed pregnancies from Nordic population databases indicated significantly increased odds of neonatal infections during the first year of life compared with matched standard of care-exposed pregnancies,54,55 while other studies in pregnant women with inflamatory bowel disease (IBD)52,56 or RA 57 found no increased risk of neonatal infection after biologic exposure. Of note, these studies recruited mostly pregnancies exposed to etanercept, infliximab, or adalimumab. While the CZP pharmacovigilance database captures neonatal infection information, insufficient reporting makes it difficult to assess the potential impact of CZP on neonatal infection. CZP has no to minimal placental transfer during the third trimester so might be expected to have a minimal impact on infants, but this cannot be confirmed by the current analysis.
In patients with CID, adverse pregnancy outcomes have been associated with disease activity, increased comorbidity rates, and increased use of medications such as corticosteroids. 15 Although details on maternal disease activity were unavailable, information on concomitant medication and comorbidities was available from the database. Stepwise regression analysis identified maternal infection in pregnancy as associated with preterm birth, a link that is well-established in the literature.58,59
Current treatment guidelines recommend adoption of strategies to minimize or limit corticosteroid intake during pregnancy.14,15,20,60 This analysis identified an association between maternal corticosteroid use and preterm birth and low birth weight. While these findings are in keeping with previously published data,15,60 dose and timing of exposure was only recorded in a small number of cases, and therefore these could not be included in our analysis. In addition, CID activity is not routinely recorded in this registry, so the use of corticosteroids could not be adjusted for this important predictor or preterm birth. Given this, limited conclusions on the impact of corticosteroids on CZP-exposed pregnancy outcomes can be drawn from our data.
Similarly, the current analysis identified an association between NSAID use and pregnancy loss at <20 weeks. There are conflicting results about NSAID use in pregnancy and miscarriage.20,61,62 A caveat of these analyses is that, as with corticosteroid data, there were limited data available on the dose, duration, or timing of medications, any of which may have an impact on outcomes. For instance, it is possible that NSAIDs were used to manage pain associated with pregnancy loss at <20 weeks; thus, the association could be biased by indication. 63
Although this analysis focused on prospective data only, it is constrained by the methodology associated with the spontaneous reporting system and the collection of pharmacovigilance data from a number of heterogeneous sources. Since information is collected through a passive reporting system, it has several inherent limitations, including underreporting, incomplete reporting of information, and missing pregnancy outcome data. Efforts to collect data and increase the number of cases with known pregnancy outcomes will therefore be an important focus for the future. Furthermore, limited information was reported in the database on disease activity during pregnancy or other items that may be of relevance, such as family history, dose and time of exposure to concomitant medication, and definitions used to diagnose comorbidities. The absence of a comparator group (pregnancies in women with CID and no CZP exposure) is a further limitation of these analyses. However, these limitations are mitigated by the large number of outcomes available, which can increase confidence in the conclusions drawn.
To the best of our knowledge, this is one of the largest analyses of pregnancy outcomes with TNFi exposure from a pharmacovigilance database, with outcomes reported from 1392 prospective CZP-exposed pregnancies. Where pregnancy outcomes were known, we found no evidence of increased risk of adverse outcomes or patterns of congenital malformations in pregnancies with maternal CZP exposure compared with the general population. This information is important for women with CID who may have higher risk pregnancies, and will be informative to clinicians and patients in assessing the benefits and risks of their therapeutic decisions before and during pregnancy. Future data from the ongoing prospective OTIS registry study, which will report outcomes from CZP-exposed and non-exposed pregnancies, in addition to continued collection of data in the UCB Pharmacovigilance global safety database, will further add to these findings.
Supplemental Material
sj-doc-2-tab-10.1177_1759720X221087650 – Supplemental material for Pharmacovigilance pregnancy data in a large population of patients with chronic inflammatory disease exposed to certolizumab pegol
Supplemental material, sj-doc-2-tab-10.1177_1759720X221087650 for Pharmacovigilance pregnancy data in a large population of patients with chronic inflammatory disease exposed to certolizumab pegol by Megan Clowse, Rebecca Fischer-Betz, Catherine Nelson-Piercy, Angela E. Scheuerle, Brigitte Stephan, Marla Dubinsky, Thomas Kumke, Rachna Kasliwal, Bernard Lauwerys and Frauke Förger in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221087650 – Supplemental material for Pharmacovigilance pregnancy data in a large population of patients with chronic inflammatory disease exposed to certolizumab pegol
Supplemental material, sj-docx-1-tab-10.1177_1759720X221087650 for Pharmacovigilance pregnancy data in a large population of patients with chronic inflammatory disease exposed to certolizumab pegol by Megan Clowse, Rebecca Fischer-Betz, Catherine Nelson-Piercy, Angela E. Scheuerle, Brigitte Stephan, Marla Dubinsky, Thomas Kumke, Rachna Kasliwal, Bernard Lauwerys and Frauke Förger in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors thank the patients and caregivers, the investigators, and their teams who took part in this study. The authors also acknowledge Simone E. Auteri, MSc, EMS, PhD, UCB Pharma, for publication coordination; Maureen Cooney, Tamara Klapka, Laura Shaughnessy, Christine Taeter, and Marie Teil, UCB Pharma, for contributions to analysis and interpretation of the data and critical review; and Jessica Patel, PhD, and Julia Stevens, PhD, from Costello Medical, UK, for medical writing and editorial assistance based on the authors’ input and direction. This study was funded by UCB Pharma.
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.C.: Consultancy fees from UCB Pharma; grant/research support from GSK, Janssen, and Pfizer. R.F.-B.: Consultancy fees from AbbVie, BMS, Celgene, Chugai, Eli Lilly, Janssen, Novartis, Pfizer, Sanofi, and UCB Pharma. C.N.-P.: Consultancy fees from Alliance Pharma and UCB Pharma; speakers bureau for Alexion, Janssen, Sanofi, and UCB Pharma. A.E.S.: Consultancy fees for Antiretroviral Pregnancy Registry, Harmony Biosciences, IQVIA, PPD, Sanofi-Genzyme, Syneos, UCB Pharma, and ViiV. B.S.: Lecture fees from AbbVie and UCB Pharma. M.D.: Consultancy fees from AbbVie, Arena, BMS, Celgene, Eli Lilly, Gilead, Janssen, Pfizer, Takeda, and UCB Pharma. T.K.: Employee and shareholder of UCB Pharma. R.K.: Employee and shareholder of UCB Pharma; shareholder of Biogen. B.L.: Employee and shareholder of UCB Pharma. F.F.: Grant/research support from UCB Pharma; speakers bureau for Mepha, Roche, and UCB Pharma.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by UCB Pharma. Support for third-party writing assistance for this article, provided by Jessica Patel, PhD, and Julia Stevens, PhD, from Costello Medical, UK, was funded by UCB Pharma in accordance with Good Publication Practice (GPP3) guidelines (![]() ).
).
Informed consent
This article reports anonymized case data from a pharmacovigilance safety database. In accordance with Article 9(2)(i) of the General Data Protection Regulation (GDPR), 64 which recognizes that processing of health data without informed consent is necessary to fulfill pharmacovigilance legislation, informed consent was not required. Routine pharmacovigilance procedures and follow-up are also exempt from data privacy in the United States according to the Health Insurance Portability and Accountability Act of 1996 (HIPAA), Public Law 104-191. 65
Data sharing
Data from non-interventional studies are outside of UCB’s data sharing policy.
Supplemental material
Supplemental material for this article (including the video abstract) is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
