Abstract
Dipeptidyl peptidase-4 inhibitors are oral antihyperglycemic medications often used as adjuncts to other antidiabetic agents to treat type 2 diabetes mellitus. Henoch–Schönlein purpura, also known as ImmunoglobulinA (IgA) vasculitis, is a form of IgA-mediated leukocytoclastic vasculitis, which is rarely reported in adults. Herein, we report the case of a 52-year-old male with type 2 diabetes mellitus who presented with petechiae, ecchymoses, and palpable purpura below the knees bilaterally after starting sitagliptin, a dipeptidyl peptidase-4 inhibitor. A punch biopsy for direct immunofluorescence showed granular Immunoglobulin A, M, and complement 3 in vessel walls, suggestive of Henoch–Schönlein purpura. Sitagliptin was discontinued, and the patient was treated with celecoxib, colchicine, and cefadroxil for wound infection. Three months after initial presentation, brown hyperpigmentation was appreciated, suggestive of post-inflammatory changes and resolution of the lesions. However, urinalysis revealed new 0.3 g/L protein. Overall, this case highlights a new potential association of dipeptidyl peptidase-4 inhibitors with Henoch–Schönlein purpura, which may have systemic consequences.
Introduction
An estimated 828 million adults are affected by diabetes worldwide, with type 2 diabetes mellitus (T2DM) representing 98% of global diabetes diagnoses. 1 Dipeptidyl peptidase-4 (DPP-4) inhibitors are oral antihyperglycemic medications often used as adjuncts to other antidiabetic agents such as metformin. 2 Although adverse cutaneous reactions to DPP-4 inhibitors have been reported, leukocytoclastic vasculitis (LCV) remains an exceptionally rare complication.3,4 LCV, a cutaneous small vessel vasculitis, is mediated by immune-complex deposition in small vessel walls and activation of the complement system. 5 Henoch–Schönlein purpura (HSP), also known as IgA vasculitis, is a form of IgA-mediated LCV whereby only 3.4 to 14.3 cases per million occur in adults. 6 Various triggers have been proposed, including upper respiratory tract infections, medications, vaccinations, and malignancies; however, the etiology of HSP remains elusive. 7 Herein, we present the rare case of a patient who developed HSP after beginning sitagliptin.
Case report
A 52-year-old male with T2DM presented with a 2-week history of a purpuric rash on the lower limbs bilaterally, which acutely worsened over the past 72 h. The patient denied pruritus but endorsed edema, burning, and numbness in the toes. Associated symptoms included fatigue, but no arthralgias or abdominal issues. He also reported a severe headache during the initial eruption. Past medical history included T2DM, hypertension, dyslipidemia, and possible sleep apnea as described by the patient. Medications included sitagliptin, which was newly initiated 3 weeks prior to eruption onset, as well as telmisartan, rosuvastatin, and metformin. Metformin monotherapy was previously well tolerated. The patient reported seasonal allergies and alcohol consumption (3 alcoholic drinks per day) but denied drug allergies, smoking, and recreational drug use.
Physical examination revealed obesity and several petechiae, ecchymoses, and palpable purpura below the knees to the distal toes bilaterally, many with central vesicles. There was no torso or upper extremity involvement. Soles of the feet were spared (Figure 1).
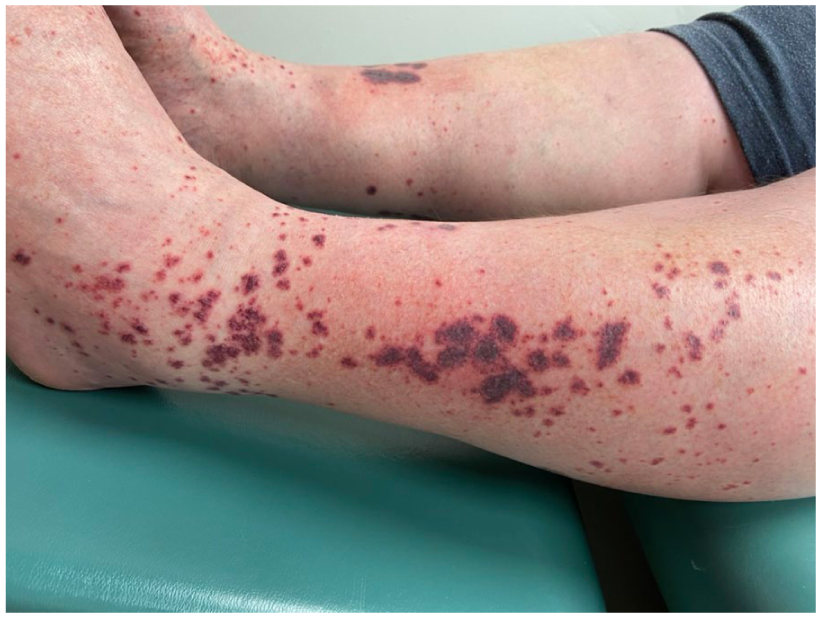
Initial presentation of petechiae, ecchymoses, and palpable purpura below the knees bilaterally, some with central vesicles.
Laboratory investigations revealed mild leukocytosis (11.5 × 109/L, normal: 3.5 × 109/L–10.5 × 109/L) and elevated erythrocyte sedimentation rate (29 mm/h, normal: 0–10 mm/h). Hemoglobin, platelets, and C-reactive protein were within normal limits. Anti-nuclear antibody, anti-nuclear cytoplasmic antibody, and extractable nuclear antibodies were negative. Serum complement C3 and C4 levels were increased at 2.43 g/L (normal: 0.9–1.80 g/L) and 0.49 g/L (normal: 0.10–0.40 g/L), respectively. Urea and creatinine were within normal ranges. Liver function tests and liver enzymes were normal except for protein (elevated at 80 g/L, normal: 63–75 g/L) and gamma glutamyl transferase (elevated at 178 U/L, normal: 11–105 U/L). Urinalysis showed no protein, erythrocytes, or casts at presentation. A punch biopsy for direct immunofluorescence showed granular IgA, IgM, and C3 in vessel walls, suggestive of HSP.
Based on these findings, a diagnosis of a sitagliptin-induced HSP was made. Conservative management was first pursued per patient wishes with compression socks and betamethasone valerate 0.1% cream twice daily as needed. Sitagliptin was discontinued.
At 1-month follow-up, the patient’s purpura remained with new punched-out ulcers at the site of prior vesicles. He was thus more eager for resolution and was prescribed celecoxib 100 mg po twice daily, colchicine 0.6 mg po twice daily, and a 10-day course of cefadroxil for wound infection. Wound care was ordered: hydrocolloid dressings for ulcerated areas and compression therapy with 2-layer Coban. At the 2-month follow-up, burning symptoms completely resolved. Upon examination, multiple non-blanching erythematous violaceous macules across the lower legs were appreciated, some of which contained greatly improved crusted centers (Figure 2). The patient forgot to perform his urinalysis. Wound care was continued for another 2 weeks along with his current regimen of celecoxib and colchicine. At the 3-month follow-up, brown hyperpigmentation and multiple non-blanching red-purple patches were appreciated across the lower legs, suggestive of post-inflammatory changes, scarring, and resolution of the lesions (Figure 3). His colchicine was decreased to daily with the plan to taper off one medication at a time every 2–4 weeks. Urinalysis revealed new 0.3 g/L protein, and the patient was referred to nephrology.

Non-blanching erythematous violaceous macules with improved crusted centers at 2-month follow-up.

Brown hyperpigmentation and non-blanching red-purple patches suggestive of post-inflammatory changes and scarring at 3-month follow-up.
Discussion
DPP-4 inhibitors are frequently used to manage T2DM, often in combination with metformin. 8 While metformin increases glucagon-like peptide-1 (GLP-1) synthesis, DPP-4 inhibitors prevent GLP-1 degradation. 8 This synergistic effect makes metformin-DPP-4 inhibitor combination therapy an attractive option for managing T2DM, as seen in our patient, who was started on sitagliptin in addition to pre-existing metformin. However, several adverse cutaneous reactions have been reported after initiating DPP-4 inhibitor therapy, including bullous pemphigoid and Stevens–Johnson syndrome. 4
To our knowledge, this is the first case of sitagliptin-induced HSP and the third case of sitagliptin-induced LCV documented in the literature. A study that searched the French pharmacovigilance and World Health Organization’s global individual case safety reports databases reported three cases of antidiabetic-induced IgA vasculitis, which occurred after vildagliptin, repaglinide, and benfluorex. 9 Uçan et al. 10 reported a 46-year-old male who developed LCV 15 days after sitagliptin initiation, resolving within 10 days after discontinuation. Sargin et al. 3 reported LCV in a 70-year-old female beginning sitagliptin/metformin therapy, which resolved within 1 week of drug discontinuation and prednisolone initiation. 3 Two additional cases of DPP-4 inhibitor-induced LCV have been reported, one after vildagliptin and another after alogliptin.8,11
The consequences of HSP on health outcomes can be profound. Despite discontinuation of sitagliptin and appropriate interventions, our patient’s cutaneous lesions required wound care, and achieved resolution 3 months after initial presentation. This may have been due to the delay in seeking Dermatology care and thus the delay in diagnosis and cessation of sitagliptin (total drug exposure was 5 weeks). Furthermore, our patient developed systemic involvement as evidenced by proteinuria, likely related to HSP-associated IgA disease. Although patients with IgA vasculitis usually develop renal manifestations within a few weeks after initial presentation, it is imperative to note that this can be delayed for several months. 12 Therefore, more frequent monitoring, which may include urinalysis and blood pressure monitoring, should be continued if clinical or biologic abnormalities persist. 12
Overall, this case emphasizes the criticality of awareness of HSP as a rare but serious adverse effect associated with DPP-4 inhibitors. Unexplained rashes in diabetic patients should be evaluated promptly to initiate appropriate management.
Footnotes
Acknowledgements
None.
Consent to participate
Informed written consent was obtained from the patient for the publication of this case.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
