Abstract
Aims:
Rheumatoid arthritis (RA) is associated with cardiovascular disease (CVD), but the influence of CVD risk factors on RA outcomes is limited. We examined if CVD risk factors alone are associated with RA disease activity and disability.
Methods:
We performed a cross-sectional analysis of participants in the Ontario Best Practices Research Initiative, RA registry. Patients were categorized into mutually exclusive CVD categories: (1) No established CVD and no CVD risk factors; (2) CVD risk factors only including ⩾1 of hypertension, dyslipidemia, diabetes, or smoking; or (3) history of established CVD event. Multivariable regression analyses examined the effect of CVD status on Disease Activity Score 28 (DAS28-ESR), Clinical Disease Activity Index (CDAI), and Health Assessment Questionnaire Disability Index (HAQ-DI) scores at baseline.
Results:
Of 2033 patients, 50% had at least 1 CVD risk factor, even in the absence of established CVD. The presence of ⩾1 CVD risk factor was independently associated with higher CDAI [β coefficient 1.59, 95% confidence interval (CI) 0.29–2.90, p = 0.02], DAS28-ESR (β coefficient 0.20, 95% CI 0.06–0.34, p = 0.01) and HAQ-DI scores (β coefficient 0.15, 95% CI 0.08–0.22, p < 0.0001). The total number of CVD risk factors displayed a dose response, as >1 CVD risk factor was associated with higher disease activity and disability, compared with having one or no CVD risk factors.
Conclusion:
CVD risk factors alone, or in combination, are associated with higher disease activity and disability in RA. This emphasizes the importance of risk factor recognition and management, not only to prevent CVD, but also to improve potential RA outcomes.
Introduction
Cardiovascular disease (CVD) is associated with rheumatoid arthritis (RA). Systemic inflammation is a key mediator in the pathogenesis of CVD, and there have been notable correlations between cumulative inflammatory burden and an increased risk of CVD events in RA.1–3 Reducing disease activity and inflammation – with the use of conventional synthetic disease modifying antirheumatic drugs (csDMARD) and biologic DMARDs (bDMARDs) – has been a promising strategy to reduce endpoints such as myocardial infarction and even CVD-related death.4,5
Despite highly effective therapies, RA remains a disease of multimorbidity. Traditional CVD risk factors, including hypertension, diabetes, and dyslipidemia, are highly prevalent in RA. 6 These CVD risk factors are more likely to be underdiagnosed and often do not reach recommended treatment targets. 7 This gap in care is important because comorbidities reduce the success of achieving therapeutic goals in RA.8–10 Multiple comorbidities may also complicate the choice of treatment, decrease medication adherence, and cause drug interactions from polypharmacy. 8
Studies have focused on the association of total number of comorbidities or non-CVD comorbidities (e.g., depression, respiratory disease) on patient-reported outcomes.8,10 However, whether CVD risk factors specifically, in the absence of established CVD, impact RA disease activity or disability negatively has not been well studied. If associated, this information could facilitate greater attention to improve CVD risk factor control, not only to prevent CVD endpoints, but also to potentially modify poor prognostic outcomes in RA.
The objective of this study was to examine the association between CVD risk factors on RA outcomes. We hypothesized that the presence of CVD risk factors, even in the absence of a history of established CVD events, would be independently associated with higher disease activity and disability.
Methods
Data source and participants
We conducted a cross-sectional study of patients enrolled in the Ontario Best Practices Research Initiative (OBRI) Rheumatoid Arthritis Registry. This is a multicenter, prospective registry across Ontario, Canada, collecting data from both rheumatologists and patients with RA. It incorporates rheumatologist assessments from approximately one-third of rheumatologists in the province of Ontario. Patients are eligible to be enrolled if they are ⩾16 years of age at the time of diagnosis, ⩾18 years of age at enrolment, have a rheumatologist confirmed RA diagnosis, and have at least one swollen joint. Treatment (standard of care) is left to the discretion of the rheumatologist, aiming for remission or low disease activity.
For this study, we included the first visit (baseline) for subjects enrolled in OBRI from January 2008 to January 2017, with complete data including comorbidity profile, medications, and RA disease outcome measures.
Ethics approval and consent
Ethics approval was obtained from the University Health Network (UHN) (REB# is 07-0729 AE) for investigators working at UHN and for the OBRI data management center. We also obtained approval from each of the individual research ethics boards of all institutions before recruitment. Participants provided verbal and written informed consent for both the OBRI study participation and the reporting and publication of the aggregated de-identified data.
Clinical assessments
The following data were obtained: patient global assessment (PtGA), physician global assessment (PhGA), 28-joint tender count (TCJ-28), 28-joint swollen count (SJC-28), 28-joint disease activity score (DAS28-ESR), clinical disease activity index (CDAI), erythrocyte sedimentation rate (ESR) level (mm/h), C-reactive protein (CRP) level (mg/l), health assessment questionnaire disability index (HAQ-DI), HAQ pain score (or VAS pain score), rheumatoid factor (RF), anti-cyclic citrullinated peptide (anti-CCP), presence of joint erosions verified by radiographs, comorbidity profile recorded by physicians (including CVD events and individual CVD risk factors), and current medication use such as nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids, csDMARD, and bDMARDs. Non-rheumatic medication, including statin use, was obtained through patient self-report.
Exposures
Patients were divided into mutually exclusive groups by CVD status as reported by physicians: (1) no history of established CVD and no CVD risk factors; (2) CVD-risk factors only (yes/no), including ⩾1 of hypertension, dyslipidemia, diabetes or current smoking; and (3) established CVD (with or without documented traditional CVD risk factors), including coronary artery disease (including prior myocardial infarction and coronary interventions), transient ischemic attack (TIA)/stroke, or peripheral arterial disease When there was uncertainty, patient-reported CVD status along with their medication profile was reviewed to arrive at consensus.
Outcomes
Our co-primary outcomes of interest were disease activity and disability. We evaluated both the DAS28-ESR and CDAI as continuous disease activity measures. The HAQ-DI was used to measure disability. Our secondary outcome examined the influence of CVD status on categorical disease activity: DAS28-ESR and CDAI remission (DAS28-ESR < 2.6, or CDAI ⩽ 2.8), or low disease activity (LDA, 2.6 ⩽ DAS28-ESR < 3.2, or 2.8 < CDAI < 10).
Covariates
We considered the following explanatory variables: age, sex, body mass index (BMI), education, RA duration, CRP, and non-CVD comorbidities (depression, respiratory disease, gastrointestinal or liver disease, malignancy, and osteoarthritis). Treatment variables were also considered: use of statin, csDMARDs, bDMARDs, oral corticosteroids, and nonsteroidal anti-inflammatory drugs (NSAIDs).
Statistical analysis
Data were described using means and standard deviation (SD) for continuous variables or counts and proportions for categorical variables. Comparison of variables by CVD status was made by the analysis of variance (ANOVA) or Kruskal–Wallis test (continuous variables) or the chi-square or Fisher’s exact test (categorical variables) as appropriate.
We performed separate unadjusted and adjusted linear or logistic regression analyses to determine the independent effect of CVD status on disease outcomes. A priori, we included all clinically important confounders felt to potentially modify the association between CVD status and outcome (see below). We also analyzed for any possible dose effect of CVD risk factors on outcomes by categorizing them to none, 1 or >1 CVD risk factor.
We conducted a sensitivity analysis by separating our exposure of interest to individual CVD risk factors (hypertension, dyslipidemia, diabetes or current smoking) to determine their association with continuous measures of disease activity and disability outcome.
Missing data for covariates were imputed with the Markov Chain Monte Carlo methods. A total of 20 datasets were imputed and results were combined using Rubin’s rules. 11 A total of 625 subjects needed data imputation. All statistical analyses were performed using SAS 9.4 (SAS Institute Inc. ; Cary, NC, USA).
Results
The analytic sample included 2033 subjects. Almost half of the subjects (917, 45%) had no established CVD and no CVD risk factors, 1006 (50%) had CVD-risk factors only, and 110 (5.3%) had established CVD, with a prior history of myocardial infarction/coronary artery disease (4.7%), followed by TIA/stroke (0.5%), and peripheral arterial disease (0.1%). Not all patients with established CVD had documented CVD risk factors. In descending order of frequency, CVD risk factors in the risk-factor-only group included hypertension (66%), smoking (32.4%), dyslipidemia (31.3%), and diabetes (14.5%) (Table 1). The majority (68.3%) had only one risk factor, which was most commonly a diagnosis of hypertension (49%).
Characteristics of the OBRI cohort (n = 2033) according to CVD status.
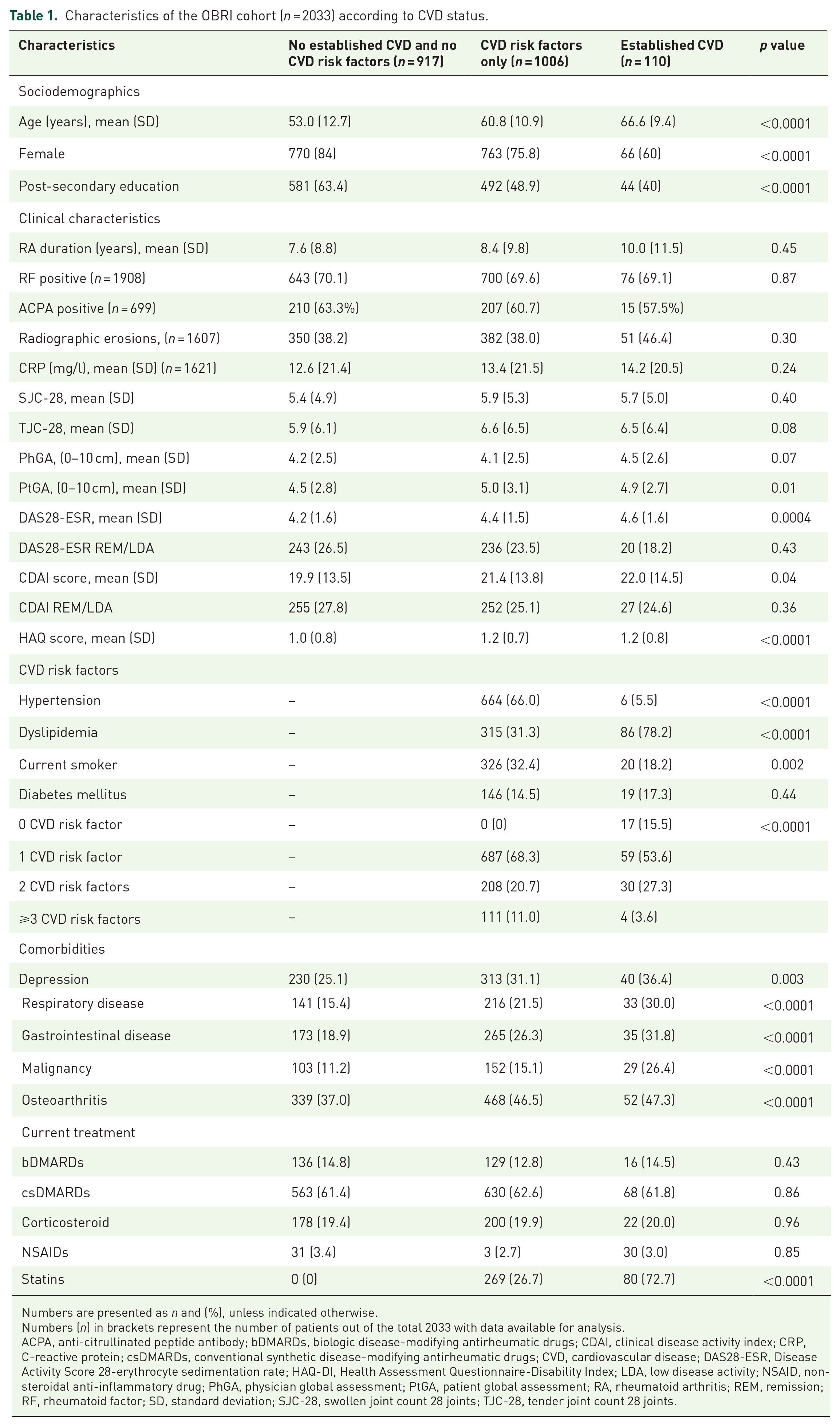
Numbers are presented as n and (%), unless indicated otherwise.
Numbers (n) in brackets represent the number of patients out of the total 2033 with data available for analysis.
ACPA, anti-citrullinated peptide antibody; bDMARDs, biologic disease-modifying antirheumatic drugs; CDAI, clinical disease activity index; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; CVD, cardiovascular disease; DAS28-ESR, Disease Activity Score 28-erythrocyte sedimentation rate; HAQ-DI, Health Assessment Questionnaire-Disability Index; LDA, low disease activity; NSAID, non-steroidal anti-inflammatory drug; PhGA, physician global assessment; PtGA, patient global assessment; RA, rheumatoid arthritis; REM, remission; RF, rheumatoid factor; SD, standard deviation; SJC-28, swollen joint count 28 joints; TJC-28, tender joint count 28 joints.
Patient characteristics were significantly different among the groups stratified by CVD status. Patients with established CVD or CVD risk factors were older, male, had less post-secondary education, and lower household income (Table 1). The established CVD and CVD risk factor only groups had a higher prevalence of non-cardiac comorbidities. The frequency or types of current RA therapies did not differ according to baseline CVD status. As expected, statin use was higher for the established CVD and CVD risk factor only groups (Table 1).
In univariable analyses, disease activity and disability scores were significantly higher in the established CVD and CVD risk-factor-only groups, compared with the group with no established CVD and no CVD risk factors.
After multivariable adjustment for clinically important confounders, the association between CVD risk factors and higher disease activity and disability persisted. The presence of at least one CVD risk factor was associated independently with higher baseline CDAI (β coefficient 1.59, 95% CI 0.29–2.90, p = 0.02), DAS28-ESR (β coefficient 0.20, 95% CI 0.06–0.34, p = 0.01) and HAQ-DI scores (β coefficient 0.15, 95% CI 0.08–0.22, p < 0.0001) (Table 2).
Association between CVD status and RA disease activity (CDAI or DAS28-ESR) and disability (HAQ-DI).
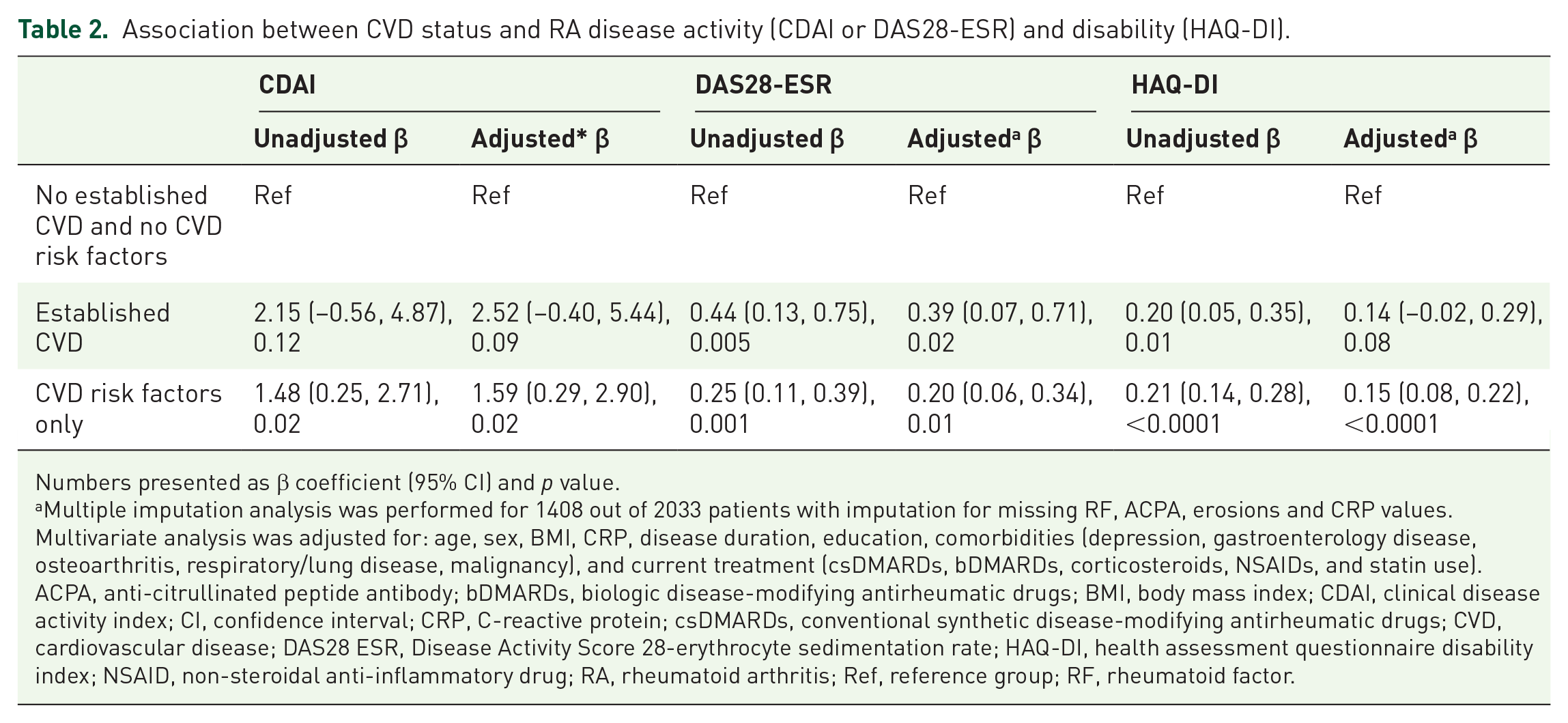
Numbers presented as β coefficient (95% CI) and p value.
Multiple imputation analysis was performed for 1408 out of 2033 patients with imputation for missing RF, ACPA, erosions and CRP values. Multivariate analysis was adjusted for: age, sex, BMI, CRP, disease duration, education, comorbidities (depression, gastroenterology disease, osteoarthritis, respiratory/lung disease, malignancy), and current treatment (csDMARDs, bDMARDs, corticosteroids, NSAIDs, and statin use).
ACPA, anti-citrullinated peptide antibody; bDMARDs, biologic disease-modifying antirheumatic drugs; BMI, body mass index; CDAI, clinical disease activity index; CI, confidence interval; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; CVD, cardiovascular disease; DAS28 ESR, Disease Activity Score 28-erythrocyte sedimentation rate; HAQ-DI, health assessment questionnaire disability index; NSAID, non-steroidal anti-inflammatory drug; RA, rheumatoid arthritis; Ref, reference group; RF, rheumatoid factor.
The number of CVD risk factors also demonstrated a dose-response relationship. Having one CVD risk factor was associated with a 1.61 increase in CDAI (95% CI 0.28–2.95, p = 0.02), whereas having >1 CVD risk factor was associated with a 2.48 increase in baseline CDAI score (95%CI 0.38–4.58, p = 0.02). Similarly, the magnitude of association was significantly larger for >1 CVD risk factor and DAS28-ESR and HAQ-DI scores (Table 3).
Association between the number of CVD risk factors and RA disease activity (CDAI or DAS28-ESR) and disability (HAQ-DI).

Multiple imputation analysis was performed for 1408 out of 2033 patients with imputation for missing RF, ACPA, erosions and CRP values. Multivariate analysis was adjusted for: age, sex, BMI, CRP, disease duration, education, comorbidities (depression, gastroenterology disease, osteoarthritis, respiratory/lung disease, malignancy) and current treatment (csDMARDs, bDMARDs, corticosteroids, NSAIDs and statin use).
ACPA, anti-citrullinated peptide antibody; bDMARDs, biologic disease-modifying antirheumatic drugs; BMI, body mass index; CDAI, clinical disease activity index; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; CVD, cardiovascular disease; DAS28-ESR, Disease Activity Score 28-erythrocyte sedimentation rate; HAQ-DI, health assessment questionnaire disability index; NSAID, non-steroidal anti-inflammatory drug; RA, rheumatoid arthritis; Ref, reference group; RF, rheumatoid factor.
The influence of CVD status on disease activity state (remission or LDA) was explored as a secondary outcome. A history of established CVD or CVD risk factors alone (without established CVD) was associated with lower odds of DAS28-ESR remission or LDA but was not associated with CDAI-defined remission or LDA (Table 4).
Association between CVD risk factor status and disease activity state (remission or low disease activity).

Numbers presented as OR (corresponding 95% CI) and p value.
Multiple imputation analysis was performed for 1408 out of 2033 patients with imputation for missing RF, ACPA, erosions, and CRP values. Multivariate analysis was adjusted for: age, sex, BMI, CRP, disease duration, education, comorbidities (depression, gastroenterology disease, osteoarthritis, respiratory/lung disease, malignancy), and current treatment (csDMARDs, bDMARDs, corticosteroids, NSAIDs, and statin use).
ACPA, anti-citrullinated peptide antibody; bDMARDs, biologic disease-modifying antirheumatic drugs; BMI, body mass index; CDAI, clinical disease activity index; CI, confidence interval; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; CVD, cardiovascular disease; DAS28 ESR, Disease Activity Score 28-erythrocyte sedimentation rate; HAQ-DI, health assessment questionnaire disability index; LDA, low disease activity; NSAID, non-steroidal anti-inflammatory drug; OR, odds ratio; Ref, reference group; RF, rheumatoid factor.
We also explored the association between individual CVD risk factors and outcomes. Current smoking was associated with higher CDAI (β coefficient 2.08, 95% CI 0.54–3.61, p = 0.01), higher DAS28-ESR (β coefficient 0.20, 95% CI 0.03–0.37, p = 0.02), and higher HAQ-DI scores (β coefficient 0.13, 95% CI 0.05–0.21, p = 0.002, Table 5). Dyslipidemia was associated with higher disease activity (CDAI β coefficient 5.69, 95% CI 2.06–9.32, p = 0.002) and DAS28-ESR, (β coefficient 0.47, 95% CI 0.07–0.88, p = 0.02) but not with disability (β coefficient 0.13, 95% CI −0.06–0.32, p = 0.19). Diabetes was associated with higher DAS28-ESR (β coefficient 0.3, 95% CI 0.05–0.55, p = 0.02) and HAQ-DI (β coefficient 0.16, 95% CI 0.04–0.28, p = 0.01), while the most common comorbidity of hypertension was not associated with either disease activity or disability.
Association between individual CVD risk factors and RA disease activity (CDAI or DAS28-ESR) and disability (HAQ-DI).

Numbers presented as β coefficient (95% CI), and p value.
Multiple imputation analysis was performed for 1408 out of 2033 patients with imputation for missing RF, ACPA, erosions and CRP values. Multivariate analysis was adjusted for: age, sex, BMI, CRP, disease duration, education, comorbidities (depression, gastroenterology disease, osteoarthritis, respiratory/lung disease, malignancy) and current treatment (csDMARDs, bDMARDs, corticosteroids, NSAIDs and statin use).
ACPA, anti-citrullinated peptide antibody; bDMARDs, biologic disease-modifying antirheumatic drugs; BMI, body mass index; CDAI, clinical disease activity index; CI, confidence interval; CRP, C-reactive protein; csDMARDs, conventional synthetic disease-modifying antirheumatic drugs; CVD, cardiovascular disease; DAS28-ESR, Disease Activity Score 28-erythrocyte sedimentation rate; HAQ-DI, health assessment questionnaire disability index; NSAID, non-steroidal anti-inflammatory drug; RA, rheumatoid arthritis; Ref, reference group; RF, rheumatoid factor.
Discussion
In this large, real-world RA cohort, we evaluated the relationship between CVD comorbidity and clinically relevant RA outcomes. The presence of CVD risk factors alone, even in the absence of established CVD events, was associated with higher DAS28-ESR, CDAI, and HAQ-DI scores. Our study contributes to new knowledge as we also observed a significant dose–response relationship between the number of CVD risk factors and disease activity and disability in RA.
Overall, the absolute increases in disease outcomes did not meet standard definitions for minimum clinically important differences in DAS28-ESR, CDAI, or HAQ-DI. 12 However, these small numerical increases, particularly for CDAI scores, could result in re-classification of a patient from remission to LDA and thus have the potential to alter treat-to-target approaches in routine clinical care.
Comorbid illnesses have been shown to impact other core components used to calculate composite indices. Diabetes and obesity have been associated with higher joint counts, and correlate with worse PtGA. 9 Depression, fibromyalgia, and osteoarthritis have been shown to skew RA response measures by affecting self-reported joint counts and pain scores. 10 However, we found an independent association between CVD risk factors and RA outcomes even after adjusting for non-CVD comorbidities that may have impacted outcomes. In a recent study, high PtGA score prevented achievement of remission in patients, and this was influenced primarily by self-perceived impact of pain and function. 13 We did not explore the association of each CVD risk factor and individual DAS28-ESR or CDAI components to determine what may be driving this association. Baseline pain, joint counts, and CRP level were similar among groups but PtGA was marginally higher in the CVD risk factor only group so differential patient perceptions by CVD status may be playing a role.
It has been suggested that treatment bias may explain the higher disease activity observed in the CVD risk factor only group. Ageing and comorbidity may cause “channeling” bias due to fear of adverse events or multiple drug interactions, limiting the aggressive use of DMARDs. 14 However, our results did not support this theory. Despite a greater number of non-CVD comorbidities, patients with established CVD and CVD risk factors were treated equally with corticosteroids, csDMARDs, and bDMARDs compared with the younger, no CVD/no CVD risk factor group. Of course, we were not able to assess if adherence may have differed by comorbidity profile. Future research is needed to understand how medication prescribing, adherence, and sustainability may vary by CVD status.
We also explored the contribution of individual CVD risk factors to disease outcomes. Current smoking was strongly and consistently associated with all disease outcomes measured. This aligns with evidence that smoking adversely affects RA disease severity, and supports the notion that smoking cessation can be a modifiable risk factor for disease activity and treatment response.15–17 It should be noted that we did not have information on pack-years of smoking to determine whether there was a dose response effect, or whether duration of smoking cessation was associated with poorer clinical outcomes.
The effect of other traditional CVD risk factors was inconsistent across disease outcomes. Prior studies have found an association between higher CDAI, DAS28-ESR, and the presence of diabetes. 9 It has been suggested that insulin resistance is correlated directly with higher swollen joint counts and CRP levels via shared metabolic pathways.18–20 In our study, diabetes was associated with a small increase in DAS28-ESR but not with CDAI or HAQ-DI. This difference may be explained partially by the inflammatory marker measurement essential to calculate the DAS28, but not other composite scores. Dyslipidemia, even when adjusted for treatment with statins, was also an independent risk factor for worse disease activity. This is in contrast to findings that statins may exert potent anti-inflammatory effects to improve synovial inflammation and patient-reported pain and stiffness, possibly through inhibition of key cytokines interleukin 1 and interleukin 6. 21 It is plausible that dyslipidemia in our cohort was associated with some unmeasured variables, for example patient adherence to medications, that impacted disease activity or treatment non-response. This observed relationship may also reflect detection bias, whereby patients with more severe or active RA are also more likely to receive testing and treatment for dyslipidemia. Our findings need to be replicated in other cohorts and additional research is needed to explore whether a particular combination, or severity, of CVD risk factors, differentially affects RA disease activity or disability.
Our results were novel in showing the additive effect of traditional CVD risk factors on disease outcome in RA. A similar dose–response relationship between greater CVD risk factors and higher disease activity has been observed in psoriatic arthritis (PsA). In a recent study, PsA patients with >3 CVD risk factors had the highest adjusted disease activity compared with those with fewer or no CVD risk factors. 22 Cardiometabolic conditions are over-represented in PsA and the prevalence of CVD risk factors was 73% in this cohort, compared with 50% in our study. While the different populations and disease outcomes studied limit our ability to make direct comparisons, these findings underscore the influence that burden of CVD comorbidity may exert on inflammatory arthritis course and is worthy of investigation in other RA and PsA cohorts.
Strengths of our study should be noted. We focused on CVD comorbidities in a large, heterogeneous sample of RA patients. The inclusion of “real-world” patients enhances generalizability to cohorts with similar patient characteristics and practice patterns. In addition, we adjusted analyses for a range of clinically important and demographic variables that are known to impact disease activity and disability.
The cross-sectional nature of the study limits any causal inference between CVD risk factors and RA outcome. We did not have all possible CVD risk factors available to study, such as a family history of CVD, nor did we have information on important lifestyle habits (e.g., alcohol, diet, exercise) that may have impacted our outcomes. Those who had an extensive past smoking history were not classified as smokers, potentially reducing the strength of the association. There may have been misclassification of CVD risk factors as our definitions were not contingent on anthropometric or laboratory values. The sequence of CVD risk factor development and degree of control (e.g., severity of diabetes, dyslipidemia, or hypertension) was also unknown, so we cannot ascertain how this may have affected RA disease outcomes. Last, we had to contend with missing data for some variables that were imputed into multivariable models. However, we would expect these to be “missing at random” and not different among the exposure groups, and thus not lead to biased estimates.
In summary, we identified the presence of CVD risk factors to be associated with higher disease activity and disability. This knowledge can help practicing rheumatologists identify at-risk patients who may benefit from closer follow up or tailored therapeutic strategies, according to their CVD status. This could facilitate prevention of CVD events by heightened attention to CVD risk factor control, and also help improve RA treatment targets. Further longitudinal studies are needed to confirm our findings and to evaluate specific barriers and facilitators of RA response measures in the setting of CVD comorbidity.
Footnotes
Acknowledgements
We would like to thank all of the OBRI participants and OBRI investigators for contributing their data. OBRI investigators include: Ahluwalia, V., Ahmad, Z., Akhavan, P., Albert, L., Alderdice, C., Aubrey, M., Bajaj, S., Bensen, B., Bombardier, C., Bookman, A., Campbell, D., Carette, S., Carmona, R., Chow, A., Ciaschini, P., Cividino, A., Cohen, D., Dixit, S., Haaland, D., Hanna, B., Haroon, N., Hochman, J., Jaroszynska, A., Johnson, V., Joshi, S., Kagal, A., Karasik, A., Karsh, J., Keystone, E., Khalidi, N., Kuriya, B., Larche, M., Lau, A., LeRiche, N., Leung, F., Mahendira, D., Matsos, M., McDonald-Blumer, H., Mittoo, S., Mody, A., Montgomery, A., Mulgund, M., Norris, E., Ng, T., Papneja, E., Pavlova, V., Perlin, L., Pope, J, Purvis, J., Rohekar, G., Rohekar, S., Rubin, L., Samadi, N., Shaikh, S., Shickh, A., Shupak, R., Smith, D., Soucy, E., Stein, J., Thompson, A., Thorne, C., and Wilkinson, S.
Author contributions
The corresponding author affirms that all authors (KC, MM, CB, BK) contributed to the planning, conduct, and reporting of the work described in the article.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: OBRI was funded by peer reviewed grants from CIHR (Canadian Institutes for Health Research), Ontario Ministry of Health and Long-Term Care (MOHLTC), Canadian Arthritis Network (CAN) and unrestricted grants from: Abbvie, Amgen, Celgene, Hospira, Janssen, Lilly, Merck, Novartis, Pfizer, Roche, Sanofi, and UCB.
