Abstract
These authors contributed equally to this work.
Introduction
Myelin proteolipid protein (PLP) is the most abundant protein in central nervous system (CNS) myelin (Eng et al., 1968; Norton and Poduslo, 1973). The tetraspan protein plays a major role in the structure and function of CNS myelin (Klugmann et al., 1997; Baumann and Pham-Dinh, 2001) and is important for communication between oligodendrocytes (myelin-producing cells in the CNS) and axons (Gruenenfelder et al., 2011). Additionally, PLP serves a neuroprotective role. It is required for the transport of certain proteins into the myelin compartment believed to be important for axo-glial metabolism and long-term support of axons by oligodendrocytes (Werner et al., 2007).
PLP1 gene expression is regulated in a spatiotemporal manner (Wight and Dobretsova, 2004), with very high levels occurring in oligodendrocytes. It is estimated that during the active myelination period, 10% of the mRNA in oligodendrocytes is derived from PLP1 (Baumann and Pham-Dinh, 2001). Other select cells in the CNS and periphery express the gene, albeit to much lower levels, except for olfactory ensheathing cells (OECs) where levels are also high (Griffiths et al., 1995; Dickinson et al., 1997). PLP1-related disorders include Pelizaeus-Merzbacher disease and spastic paraplegia type 2 (SPG2), which arise from alterations in PLP1 gene dosage, missense mutations, or indels (Inoue, 2005; Garbern, 2007; Hobson and Garbern, 2012). Expression of the gene must be tightly regulated since increased copy number (Sistermans et al., 1998; Inoue et al., 1999; Mimault et al., 1999; Hodes et al., 2000; Wolf et al., 2005; Clark et al., 2013) or deletion (Raskind et al., 1991; Inoue et al., 2002; Matsufuji et al., 2013) of PLP1 both cause problems. It is known from transgenic mouse studies, which utilize the mPlp1 promoter to drive expression of the lacZ reporter, that inclusion of mPlp1 intron 1 is essential for attaining good levels of expression in brain, concurrent with the active myelination period (Li et al., 2002). The intron is relatively large (8.1 kb for mPlp1; Wight and Dobretsova, 1997), comprising half of the gene. Several regions within mPlp1 intron 1 possess enhancer-like activity. One of these was identified by deletion-transfection analysis and named antisilencer/enhancer (ASE; 83 bp) based on its properties in the N20.1 and Oli-neu oligodendroglial cell lines (Dobretsova and Wight, 1999; Dobretsova et al., 2000; Meng et al., 2005; Pereira et al., 2011). Two other regions in mPlp1 intron 1 were identified as putative enhancers based on their ability to elicit expression of a lacZ transgene containing a basal heterologous (hsp) promoter (Tuason et al., 2008). The wmN1 region (1171 bp) conferred high levels of transgene expression in myelinating oligodendrocytes and OECs, while the wmN2 region (313 bp) led to high-level expression in Schwann cells and their progenitors, DRG satellite cells, and OECs and weak expression in oligodendrocyte lineage cells (Tuason et al., 2008). However, it is unclear whether wmN1 or wmN2 functions in association with the native (mPlp1) promoter.
Not much is known about the mechanisms that govern expression of hPLP1. PLP1 promoter and coding sequences are highly conserved between human and mouse (Macklin et al., 1987). Even intron 1 is moderately conserved (60% identity) between these species. The first intron is a little larger in human, comprising 8,579 bp. Both human and mouse contain minor (supplementary) exons in PLP1 intron 1 that results in novel splice variants due to exon inclusion (Bongarzone et al., 1999; Li et al., 2009; Sarret et al., 2010). Although the sequences for these minor exons are fairly conserved between human and mouse, they are not utilized in both species due to lack of one or more splice sites. There are two supplementary exons in hPLP1 intron 1 (exon AB and exon C; Sarret et al., 2010), which are positionally distinct from those in mPlp1 intron 1 (exon 1.1 and exon 1.2; Bongarzone et al., 1999; Li et al., 2009). Due to an internal splice donor and acceptor site in exon AB, two additional splice variants may be formed (hPLP A and hPLP A′) which are translated into a protein having additional nine amino acids at the N-terminus compared to the classic product (Sarret et al., 2010). However, transcripts which possess either exon AB in its entirety or exon C are predicted to initiate translation from an internal ATG site (at the end of exon 4), yielding a protein that corresponds to the last 72 amino acids of hPLP (residues 205–276). Exon AB is located just upstream of the sequence orthologous to the wmN1 region, while exon C is fully contained within the wmN1 region (Figure 1).
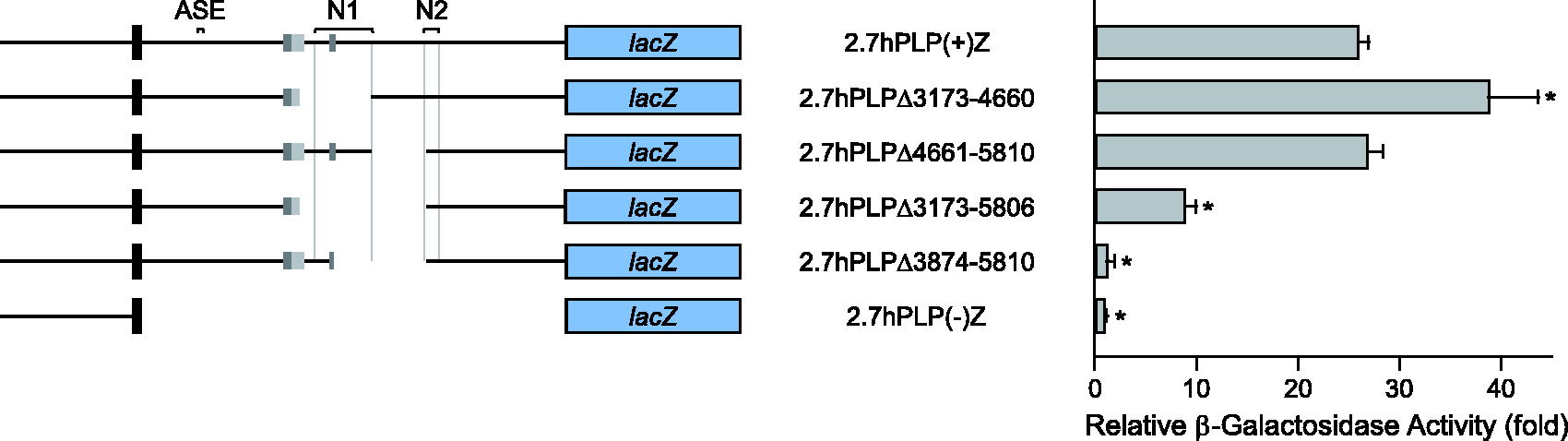
The first intron of the hPLP1 gene is required for maximal expression of hPLP1-lacZ constructs in Oli-neu cells. The structure of the hPLP1 gene is diagramed at the top with the seven major exons depicted by black boxes. 5′-flanking DNA and introns are indicated by black lines. Alternatively spliced exons AB and C are indicated by gray boxes, which get incorporated separately in splice variants. Due to an internal splice donor and acceptor site in exon AB, additional splice variants may be formed which incorporate sequences only from the dark gray region; the light gray portion indicates the region specific to splice variants that contain all of exon AB. Regions orthologous to mouse ASE, wmN1 (N1) and wmN2 (N2) enhancers are indicated as well. Transfection results with various hPLP1-lacZ constructs are shown below. The structure of the hPLP1-lacZ constructs is illustrated toward the left. These constructs utilize hPLP1 sequences to drive lacZ reporter gene expression and contain hPLP1 sequences from the proximal 6.2 or 2.7 kb of 5′-flanking to the first 38 bp of exon 2. All of hPLP1 intron 1 is missing from 6.2hPLP(−)Z and 2.7hPLP(−)Z; the minus sign indicates absence of the intron while the plus sign indicates its presence. (The bent gray line denotes the juncture between regions in 2.7hPLP(−)Z.) Oli-neu cells were transiently cotransfected with equimolar amounts of hPLP1-lacZ constructs along with a set amount of RSVL to correct for differences in transfection efficiency. Transfection results represent the fold mean ± SD of β-gal activity (n = 8) relative to that obtained for 2.7hPLP(−)Z, which was arbitrarily set at 1 in every experiment. *Significant difference (p < .001) between the indicated pair of constructs by one-way ANOVA with Tukey’s post hoc analysis.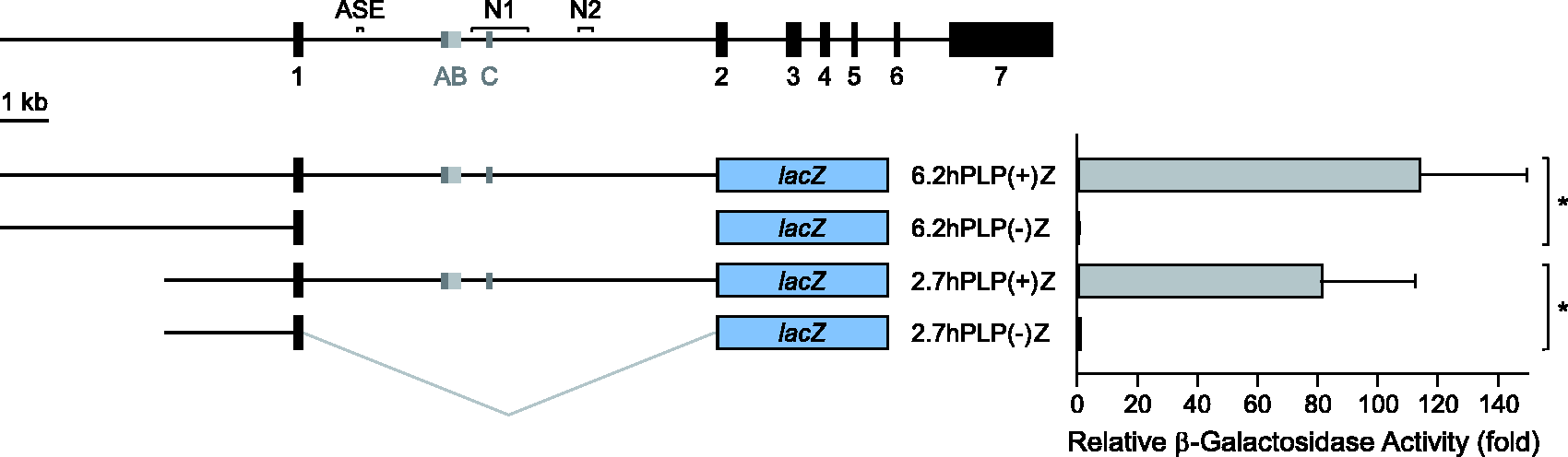
To test whether the first intron is important for the regulation of hPLP1 expression, and if so, whether sequences orthologous to the ASE, wmN1 and wmN2 regions are involved, hPLP1-lacZ constructs were generated that contain various portions of hPLP1 intron 1 DNA. The constructs were transfected in Oli-neu cells in order to identify important regulatory elements, as well as to determine if the supplementary exons from hPLP1 are incorporated in splice variants.
Material and Methods
Cell Culture
The Oli-neu cell line was generated by infecting mouse primary oligodendrocytes (and their precursors) with a replication-defective retrovirus that expresses the t-neu oncogene (Jung et al., 1995). Oli-neu cells were grown at 37℃ in SATO medium devoid of mitogens (ODM) according to the modifications by He et al. (2007), which was supplemented with 1% horse serum (Invitrogen, Carlsbad, CA), and maintained in an atmosphere of 10% CO2.
Construction of hPLP1-lacZ Plasmids
Constructs that utilize hPLP1 genomic sequences to drive lacZ reporter gene expression were generated by a multistep process. The source of hPLP1 DNA came from a fosmid in bacterial clone W122-8877L5 obtained from the Wellcome Trust Sanger Institute (Hixton, Cambridge, UK). Bacteria were grown in 2XYT growth media containing 25 µg/ml chloramphenicol for 14 hr at 37℃, and fosmid DNA was subsequently isolated using the FosmidMAX DNA Purification Kit (Epicentre Biotechnologies, Madison, WI) according to the manufacturer’s specifications. Fosmid DNA was partially digested with BamHI, and fragments were randomly inserted in the pGEM-11Zf(+) vector (Promega, Madison, WI) by shotgun cloning. Plasmids were screened for insertion of a single BamHI fragment, whose orientation was determined by restriction enzyme mapping. Plasmid hPLP6.2F contains hPLP1 genomic DNA that spans the proximal 6.2 kb of 5′-flanking DNA to the first half of exon 1, inserted in an orientation consistent with the vector numbering system (forward orientation; F). Plasmid hPLP9.5F contains a 9.5 kb fragment of hPLP1 genomic DNA that extends from the BamHI site in exon 1 to a BamHI site which overlaps the intron 2/exon 3 border, inserted in the forward direction. The plasmids were then modified to generate subclones used in the construction of hPLP1-lacZ fusion genes (see Supplementary Figure 1 for overview [available at http://asn.sagepub.com/supplemental]).
To convert the ApaI site in hPLP1 exon 2 to an AgeI site, a SacI fragment was first deleted from hPLP9.5F to generate the hPLP9.5FΔSacI plasmid. This essentially removes the 5′-half of hPLP1 sequences from hPLP9.5F, including an ApaI site in intron 1. Subsequently, hPLP9.5FΔSacI was digested with ApaI and resealed with DNA ligase to remove hPLP1 sequences downstream of the ApaI site in exon 2. The resulting plasmid (hPLP9.5FΔSacI/ΔApaI) was then digested with ApaI and HindIII and ligated to an ApaI-HindIII adapter (sense: 5′-TT
The 2.7hPLP(+)Z construct, which is similar to 6.2hPLP(+)Z but contains only 2.7 kb of hPLP1 5′-flanking DNA, was generated by a multistep process. First, the 6.2hPLPF plasmid was digested with XhoI and KpnI to remove some of the upstream hPLP1 5′-flanking DNA and then ligated to an XhoI-KpnI adapter (sense: 5′-TCGAGGGCCCAGATTCTCTGAGGTAC-3′, antisense: 5′-CTCAGAGAATCTGGGCCC-3′) which reconstitutes the native ApaI site located immediately upstream of the KpnI site. The resulting plasmid (hPLP6.2ΔXhoI-KpnI + ApaI adapter) was digested with XhoI and BamHI, and the hPLP1-containing fragment inserted between the XhoI and BamHI sites of BA8.7 to generate 2.7hPLP(+). Lastly, 2.7hPLP(+) was digested with XhoI and AgeI, and the XhoI-AgeI fragment containing hPLP1 sequences were isolated and inserted into pGAL5 between the XhoI and AgeI sites to produce 2.7hPLP(+)Z.
To generate plasmids similar to 2.7hPLP(+)Z and 6.2hPLP(+)Z, but missing all of hPLP1 intron 1 DNA, 2.7hPLP(+) and 6.2hPLP(+) were digested with BstXI and AgeI to remove the intron and were ligated to an adapter which essentially fills in the gap with cDNA sequence (sense: 5′-ATGGGCTTGTTAGAGTGCTGTGCAAGATGTCTGGTAGGGGCCTTA-3′; antisense: 5′-GGCCATTCCGGGGATGGTCTGTAGAACGTGTCGTGAGATTGTTCGGGTACAGA-3′). The resulting plasmids, 2.7hPLP(−) and 6.2hPLP(−), were digested with XhoI and AgeI, and the hPLP1-containing fragment was cloned into pGAL5 between the XhoI and AgeI sites to yield 2.7hPLP(−)Z and 6.2hPLP(−)Z, respectively. Partial deletion constructs lacking a portion of hPLP1 intron 1 were constructed either directly from 2.7hPLP(+)Z or via a shuttle vector. Deletions were generated through the use of restriction enzymes, and a SalI site was added at the deletion junction site by means of SalI linkers (5′-CGGTCGACCG-3′) or custom-designed adapters (Integrated DNA Technologies) unless noted, otherwise. Klenow enzyme was used to fill in or digest back-staggered ends produced by restriction enzyme digestion, prior to ligation with linkers. Partial deletion constructs were named according to the missing hPLP1 intron 1 sequence, based on numbering the intron from positions 1 to 8579.
The following hPLP1-lacZ partial deletion constructs were generated directly from 2.7hPLP(+)Z through use of the enzymes and adapters or linkers indicated in parentheses: 2.7hPLPΔ212–5810 (PmlI and SbfI; digest back with Klenow enzyme; SalI linker); 2.7hPLPΔ5811–6221 (SbfI and SphI; SbfI-SphI adapter: sense 5′-CGGTCGACCGCATG-3′ and antisense 5′-CGGTCGACCGTGCA-3′); 2.7hPLPΔ6228–7569 (SphI and BstAPI; SphI-BstBI adapter: sense 5′-CGGTCGACCGTCT-3′ and antisense 5′-CGGTCGACCGCATG-3′); 2.7hPLPΔ7573–8167 (BstAPI and XmaI; BstAPI-XmaI adapter: sense 5′-CGGTCGACCG-3′ and antisense 5′-CCGGCGGTCGACCGAGA-3′).
Other hPLP1-lacZ partial deletion constructs were generated via intermediary shuttle vectors whose modifications were ultimately incorporated in the background of 2.7hPLP(+)Z. Only the initial shuttle vectors containing the modifications are specified. The 2.7hPLPΔ3874–5810 plasmid was generated by digesting the BA8.7 shuttle vector with PstI followed by removal of the staggered ends with Klenow enzyme and ligation to the SalI linker. The 2.7hPLPΔ1685–3168 plasmid was generated by digesting the BA8.7ΔSphI shuttle vector with PpuMI and XbaI followed by ligation with the PpuMI-XbaI adapter which contains a SalI site (sense: 5′-GTCCGGTCGACCG-3′; antisense: 5′-CTAGCGGTCGACCG-3′).
Some hPLP1-lacZ partial deletion constructs were generated using a “mix-and-match” approach. 2.7hPLPΔ5811–8167 was generated by digestion of 2.7hPLPΔ5811–6221 and 2.7hPLPΔ7573–8167 with SalI and HindIII, followed by ligation of the appropriate fragments. 2.7hPLPΔ212–3168 was generated by digesting 2.7hPLPΔ212–5810 and 2.7hPLPΔ1685–3168 with XhoI and SalI, followed by treatment with Antarctic phosphatase (New England BioLabs, Ipswich, MA) and ligation of the appropriate fragments.
Additionally, the 2.7hPLP(+)Z construct was modified to generate two plasmids, each having a unique BstBI site. A BstBI site was introduced at the XbaI site just upstream of the wmN1 region through digestion of the BA8.7ΔSphI, a shuttle vector with XbaI, followed by dephosphorylation with Antarctic phosphatase, and ligation to a 5′-phosphorylated BstBI adapter (sense and antisense: 5′-CTAGTTCGAA-3′) to eventually yield 2.7hPLP(+)Z/XbaI → BstBI. Likewise, the EcoRI site at the 3′ end of the wmN1 region was converted to a BstBI site through digestion of the BA8.7ΔSfiI-PmlI shuttle vector (BA8.7 lacking an SfiI-PmlI fragment) with EcoRI, followed by fill in of the staggered ends with Klenow enzyme, and ligation to a double BstBI linker (5′-TTCGAATTCGAA-3′) to ultimately produce 2.7hPLP(+)Z/EcoRI → BstBI. Subsequently, 2.7hPLP(+)Z/XbaI → BstBI and 2.7hPLP(+)Z/EcoRI → BstBI were digested with HindIII and BstBI, and the appropriate fragments were ligated to form 2.7hPLPΔ3173–4660 (2.7hPLPΔwmN1). Furthermore, the 2.7hPLPΔ3173–5806 construct was generated by digestion of 2.7hPLPΔ3173–4660 with BstBI and SbfI, followed by ligation to a BstI-SbfI adapter containing an internal SalI site (sense: 5′-CGGTCGACCGTGCA-3′; antisense: 5′-CGGTCGAC-3′).
Lastly, the EcoRI site at the 3′ end of the wmN1 region was converted to a SalI site by digesting a shuttle vector (BA8.7ΔSfiI-PmlI/XbaI → BstBI) with EcoRI, which was followed by S1 nuclease treatment to remove the staggered ends and ligation to a SalI linker. A fragment containing this modification was eventually linked to the SalI site introduced at the SbfI site to generate 2.7hPLPΔ4661–5810.
The sequence for both strands of hPLP1 intron 1 DNA was determined by the dideoxynucleotide chain termination method, using many of the plasmids described above. The DNA sequence of hPLP1 intron 1 in 2.7hPLP(+)Z and 6.2hPLP(+)Z consists of 8,579 bp and was deposited in GenBank under the accession number KP313870.
Transfection Analysis
Oli-neu cells were seeded at a density of 3.5 × 105 cells per 35-mm well (six-well plates) in 2 ml of growth medium, 16 to 18 hr prior to transfection. Cells were transfected with equimolar amounts of hPLP1-lacZ constructs and a fixed quantity (0.35 µg/well) of the RSV-luciferase reporter, RSVL (de Wet et al., 1987). Empty vector (pBluescript SK+; Stratagene) was added as necessary to keep the total amount of DNA constant. Two types of transfection reagents were used. For transfection with the FuGENE 6 Transfection Reagent (Roche Diagnostics, Indianapolis, IN; Figures 1 and 2), a combination of 4.5 μl FuGENE 6 and 3 µg total DNA were added to each well. In other experiments (Figures 3–5), the X-tremeGENE HP Transfection Reagent (Roche Diagnostics) was used at a 5:2 ratio with total DNA (7.5 μl X-tremeGENE HP and 3 µg DNA per well; or 5.183 μl X-tremeGENE HP and 2.47 µg DNA per well, simply by reducing the amount of empty vector added). The culture medium was replaced the following day (∼16 hr post DNA addition) only for those cells treated with X-tremeGENE HP. Each test (hPLP1-lacZ) construct was transfected in duplicate, per experiment. Cell lysates were prepared in 190 μl of Reporter Lysis Buffer (Promega) according to manufacturer’s instructions, 48 hr post-DNA addition.
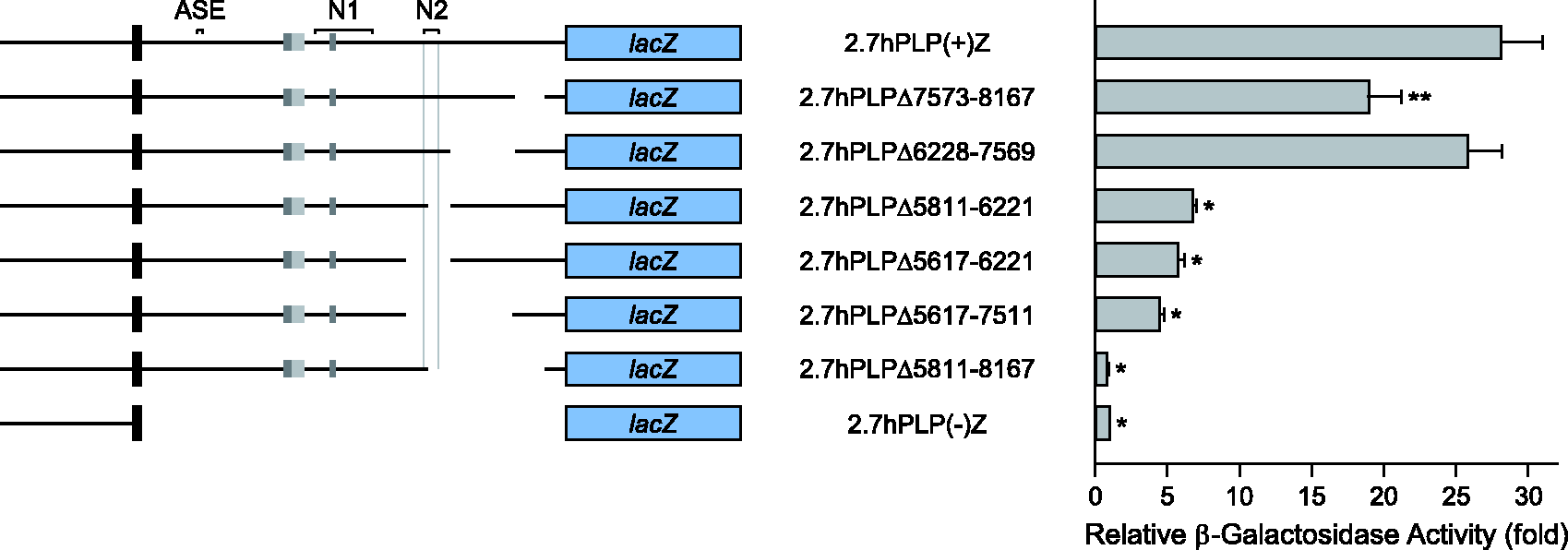
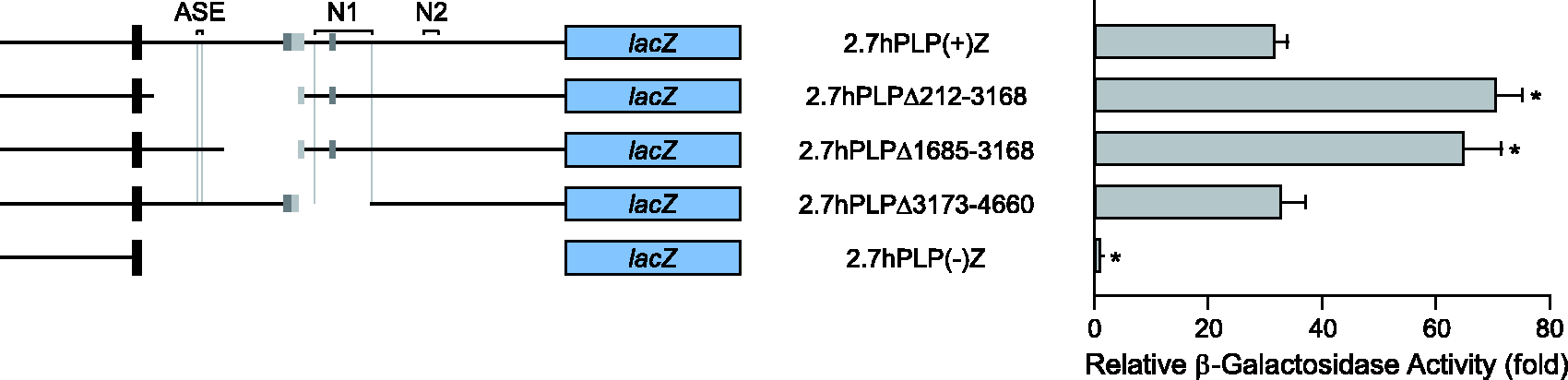
The first intron of hPLP1 intron 1 contains positive regulatory element(s) that are active in Oli-neu cells. Partial deletion constructs were named according to the omitted sequence in hPLP1 intron 1. For instance, 2.7hPLPΔ212-5810 lacks hPLP1 intron 1 positions 212–5810, based on numbering the entire intron from 1 to 8579. The constructs were transfected in Oli-neu and subsequently analyzed for relative levels of expression (see Figure 1 for more details). Transfection results represent the fold mean ± SD of β-gal activity (n ≥ 7) relative to that obtained for 2.7hPLP(−)Z, which was arbitrarily set at 1 in every experiment. *Significant difference (p < .001) between 2.7hPLP(+)Z and other constructs by one-way ANOVA with Tukey’s post hoc analysis. The wmN2 enhancer is active in Oli-neu cells, as well as a positive regulatory element located further downstream in hPLP1 intron 1. Transfection analysis was performed with various hPLP1-lacZ partial deletion constructs, which are missing part of hPLP1 intron 1 primary downstream of position 5810 (see Figure 1 for further details). Results represent the fold mean ± SD of β-gal activity (n = 8) relative to that obtained for 2.7hPLP(−)Z, which was arbitrarily set at 1 in every experiment. *Significant difference (p < .001) between 2.7hPLP(+)Z and other constructs by one-way ANOVA with Tukey’s post hoc analysis; **(p = .002). The human sequence, orthologous to mouse ASE, is nonfunctional. Transfection analysis was performed in Oli-neu cells with the indicated hPLP1-lacZ constructs whose structure is diagramed on the left side (see Figure 1 for further details). Transfection results are plotted on the right side and represent the fold mean ± SD of β-gal activity (n = 6) relative to that obtained for 2.7hPLP(−)Z, which was arbitrarily set at 1 in every experiment. *Significant difference (p < .001) between 2.7hPLP(+)Z and other constructs by one-way ANOVA with Tukey’s post hoc analysis. The human sequence, orthologous to mouse wmN1, exhibits functional redundancy with another element located directly downstream in hPLP1 intron 1. Transfection analysis was performed in Oli-neu cells with the indicated hPLP1-lacZ constructs whose structure is diagramed on the left side (see Figure 1 for further details). Transfection results are plotted on the right side and represent the fold mean ± SD of β-gal activity (n = 6) relative to that obtained for 2.7hPLP(−)Z, which was arbitrarily set at 1 in every experiment. *Significant difference (p < .001) between 2.7hPLP(+)Z and other constructs by one-way ANOVA with Tukey’s post hoc analysis. Only when the wmN1 region is deleted in conjunction with the immediate downstream sequence (positions 4661-5806) is a decrease in β-gal activity observed (2.7hPLPΔ3173-5806).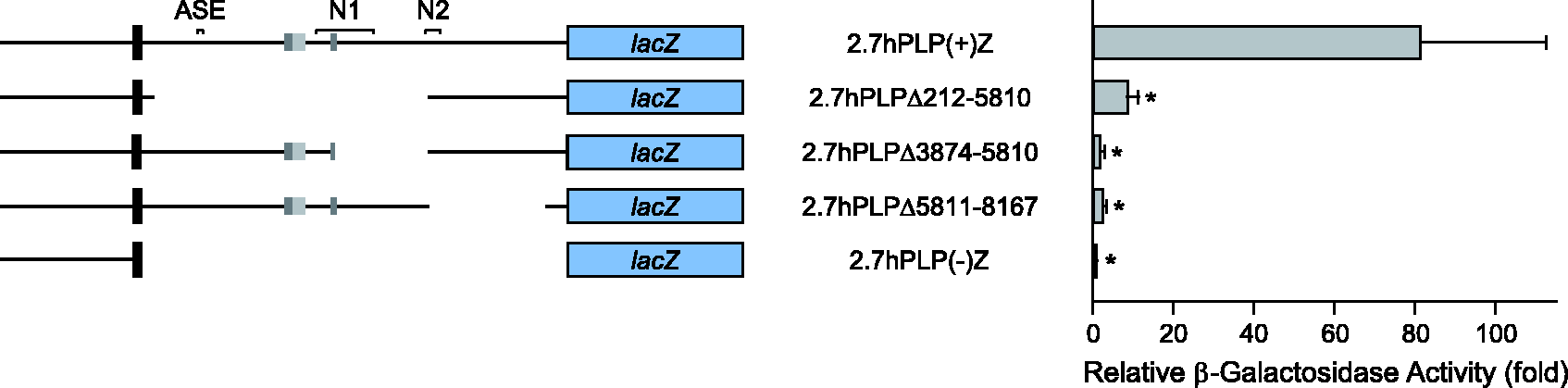
Aliquots of lysate (10 μl each) were evaluated for reporter gene expression in triplicate using the Galacto-Light Plus Kit (Tropix, Bedford, MA) and Luciferase Assay System (Promega) for determination of β-galactosidase (β-gal) and luciferase activities, respectively, as previously described (Dobretsova and Wight, 1999). Luminescence was measured as relative light units (RLU) with an AutoLumat LB 953 luminometer (EG&G Berthold, Gaithersburg, MD). Background levels of luminescence were determined from cells transfected solely with pBluescript SK+, and subtracted, accordingly. Differences in transfection efficiency were abrogated by reporting the amount of β-gal activity, for a set amount (2 × 106 RLU) of luciferase activity. Results are presented as the normalized mean ± SD of β-gal activity relative to that obtained for 2.7hPLP(−)Z transfected cells, which arbitrarily was set at 1 (fold) in every experiment. Results are the compilation of three or more independent experiments among similar conditions (i.e., same transfection reagent). Data were analyzed by one-way ANOVA, with Tukey’s post-hoc analysis, using SigmaPlot 12.5 (Systat Software, Inc., San Jose, CA). p < .05 were considered statistically significant.
Reverse Transcription-Polymerase Chain Reaction
Total RNA was extracted from Oli-neu cells which had been transiently transfected with equimolar amounts of either 2.7hPLP(+)Z, 2.7hPLPΔ3874–5810, or 2.7hPLP(−)Z as described above, except for lack of the RSVL plasmid. RNA was extracted using the Trizol reagent (Invitrogen) according to the manufacturer’s specifications. First strand cDNA synthesis was performed with the iScript cDNA synthesis Kit (Bio-Rad, Hercules, CA) per the supplier’s instructions using 1.0 µg of total RNA in a final reaction volume of 20 μl. Conditions for RT were as follows: 5 min at 25℃, 30 min at 42℃, and then 5 min at 25℃. The ensuing cDNA reactions were aliquoted and stored at −70℃ until use as template DNA, for PCR.
PCR was carried out in a final reaction volume of 20 µl containing the SsoFast Probes Supermix (Bio-Rad, Hercules, CA) and 1 µl of the RT reaction according to the manufacturer’s specifications. Sense (forward) primers that detect hPLP A-, hPLP AB-, and hPLP C-related splice variants, respectively, were 5′-AAAGAAGACCAAGTGAGGACAG-3′ (A-For), 5′-AGGAGGAAATGGTGGGAGA-3′ (AB-For), and 5′-ACTACTGCAGGCCCTGAT-3′ (C-For). A common antisense (reverse) primer, 5′-AATATCGCGGCTCAGTTCG-3′ (TrpS-Rev), from the lacZ expression cassette was used in all reactions. Initially, PCR reactions were heated at 95℃ for 1.5 min to denature the template, followed by 38 cycles of amplification (15 s at 95℃, 1 min at 60℃). The TaqMan® Gene Expression Assay for β-actin (Mm00607939_s1; Life Technologies, Grand Island, NY) was used as an internal control. PCR conditions were the same except for only 32 cycles of amplification. PCR products were resolved by electrophoresis on a 3% agarose gel.
Results
Inclusion of PLP1 Intron 1 in hPLP1-lacZ Constructs Is Required for Maximal Expression in Oli-neu Cells
Previously, we have shown that Plp1 intron 1 is required to attain maximal expression of mPlp1-lacZ constructs in Oli-neu cells (Pereira et al., 2011) and transgenic mice (Li et al., 2002). However, not much is known about the mechanisms that specifically control hPLP1 expression. To test whether the first intron of the human gene is likewise important, two pairs of hPLP1-lacZ constructs were generated and subsequently analyzed in Oli-neu cells. Each construct contains hPLP1 sequences (promoter, exon 1, and the initial 38 bp exon 2) which were used to drive expression of a lacZ reporter cassette. The resulting product contains the first 13 amino acids of classic PLP coupled to β-gal; the classic PLP isoform contains 277 residues counting the initiator methionine. The constructs were named based on their amounts of hPLP1 5′-flanking DNA (2.7 or 6.2 kb), and whether they possess (+) or lack (−) hPLP1 intron 1 DNA in its entirety (Figure 1). 2.7hPLP(+)Z and 2.7hPLP(−)Z are analogous to mPlp1-lacZ constructs used in previous studies, which contain 2.4 kb of 5′-flanking mPlp1 DNA (Li et al., 2002; Pereira et al., 2011). Transfection analysis demonstrated that the presence of hPLP1 intron 1 is required for maximal expression of hPLP1-lacZ constructs in Oli-neu cells (Figure 1). Levels of β-gal activity in 2.7hPLP(+)Z and 6.2hPLP(+)Z transfected cells were 81- and 114-fold higher, respectively, than 2.7hPLP(−)Z (arbitrarily set at 1), whereas the level for 6.2hPLP(−)Z (0.53-fold) was slightly reduced relative to 2.7hPLP(−)Z. Together, these data indicate that hPLP1 intron 1 must contain at least one positive regulatory element, operative in Oli-neu cells.
hPLP1 Intron 1 Contains Multiple Regulatory Elements
To map the location of positive regulatory element(s) in hPLP1 intron 1, various segments of the intron were excised from 2.7hPLP(+)Z, and the ensuing levels of expression from the partial deletion constructs were assessed in Oli-neu cells. Partial deletion constructs were named according to the omitted sequence. For instance, 2.7hPLPΔ212–5810 is missing intron 1 positions 212 to 5810 based on numbering the entire intron from 1 to 8579. Initially, three constructs were tested whose deletions collectively span most of the intron (from positions 212 to 8167). Although all constructs exhibited elevated levels of β-gal activity relative to 2.7hPLP(−)Z, the levels from the partial deletion constructs were markedly reduced in comparison to 2.7hPLP(+)Z, which was 81-fold higher than 2.7hPLP(−)Z (Figure 2). In comparison, the levels of β-gal activity for 2.7hPLPΔ212–5810 (ninefold), 2.7hPLPΔ3874–5810 (twofold), and 2.7hPLPΔ5811–8167 (twofold) were closer to 2.7hPLP(−)Z (onefold), suggesting that one or more positive regulatory elements exist between hPLP1 intron 1 positions 212 and 8167 (Figure 2). Additionally, it is possible that a negative regulatory element lies between positions 212 and 3874 since the level of β-gal activity was lower for 2.7hPLPΔ3874–5810 than 2.7hPLPΔ212–5810. Alternatively, the presence of exon AB in 2.7hPLPΔ3874–5810, which is missing from 2.7hPLPΔ212–5810, may help to explain the lower amount of β-gal activity attained with 2.7hPLPΔ3874–5810. Products derived from splice variants containing all of exon AB (or exon C) must use an internal ATG site (codon 91 of lacZ; Kalnins et al., 1983) from which to initiate translation, instead of the classic start site near the end of hPLP1 exon 1, due to the introduction of multiple (premature) stop codons. Because the N-terminal region of β-gal is required for enzymatic activity (Mohler and Blau, 1996), exon AB-containing splice variants from 2.7hPLPΔ3874–5810 are not expected to contribute to the overall amount of β-gal activity produced.
Altogether, the results in Figure 2 further support the notion that hPLP1 intron 1 contains at least one positive regulatory element which is necessary for maximal expression in Oli-neu cells. It is possible that a single element, which straddles position 5810, was disrupted in the partial deletion constructs. Alternatively, the intron may contain multiple positive elements situated upstream and downstream of position 5810. To discriminate between these possibilities, additional hPLP1-lacZ constructs (which lack hPLP1 intron 1 sequences primarily downstream of position 5810) were assessed in Oli-neu cells. Initially three constructs were tested that, collectively, are missing intron 1 positions 5811 to 8167. In this set of experiments, the level of β-gal activity for 2.7hPLP(+)Z was 28-fold, whereas 2.7hPLPΔ5811–8167 was 0.8-fold, relative to 2.7hPLP(−)Z (Figure 3). The decrease in activity with 2.7hPLPΔ5811–8167 largely is due to a loss of positions 5811to 6221 (2.7hPLPΔ5811–6221 at sevenfold), followed by a modest drop with elimination of positions 7573 to 8167 (2.7hPLPΔ7573–8167 at 19-fold) (Figure 3). However, removal of the intervening sequence (positions 6228–7569) had no effect (compare 2.7hPLPΔ6228–7569 at 26-fold with 2.7hPLP(+)Z at 28-fold). Because 2.7hPLPΔ5811–6221 still retains the proximal third (102 bp) of the wmN2 region, additional constructs were analyzed to test whether removal of this entire region resulted in an additional drop in β-gal activity. However, at best, there was only a slight decrease in activity upon complete removal of the wmN2 region (compare 2.7hPLPΔ5617–6221 at sixfold and 2.7hPLPΔ5617–7511 at fourfold with 2.7hPLPΔ5811–6221 at sevenfold; Figure 3). Altogether, these data suggest that an important regulatory element does not straddle position 5810, and that the absence of multiple positive regulatory elements (situated between positions 5811–6221 and 7573–8167), cumulatively, is responsible for the low level of activity observed with 2.7hPLPΔ5811 to 8167.
To map the location of regulatory elements upstream of hPLP1 intron 1 position 5810, additional partial deletion constructs were evaluated in Oli-neu cells. Similar to the preceding experiments, results are reported as the fold amount of β-gal activity relative to 2.7hPLP(−)Z. Sequences orthologous to the ASE and wmN1 region were investigated initially. As shown in Figure 4, both 2.7hPLPΔ212 to 3168 (71-fold) and 2.7hPLPΔ1685–3168 (65-fold) demonstrated high levels of activity which surpassed that for 2.7hPLP(+)Z (32-fold). These results are consistent with the removal of a negative regulatory element(s) situated between positions 1685 to 3168. Additionally these results seem to indicate that, unlike the mouse ASE, the orthologous sequence in human is not operative in Oli-neu cells. Likewise, deletion of positions 3173 to 4660, which encompasses sequence orthologous to the wmN1 region, did not cause a decrease in β-gal activity (compare 2.7hPLPΔ3173–4660 at 33-fold with 2.7hPLP(+)Z at 32-fold in Figure 4).
Because removal of positions 3874 to 5810 from 2.7hPLP(+)Z abrogated any beneficial effect from hPLP1 intron 1 (Figure 2), and loss of the wmN1 region did not decrease β-gal activity (see 2.7hPLPΔ3173–4660 in Figure 4), deletion of positions 4661 to 5810 was investigated next. As shown in Figure 5, 2.7hPLPΔ4661–5810 and 2.7hPLP(+)Z were 27- and 26-fold higher, respectively, than 2.7hPLP(−)Z, suggesting that loss of hPLP1 intron 1 positions 4661 to 5810 had no effect in Oli-neu cells. However, removal of positions 3173 to 4660 actually resulted in higher levels of β-gal activity (2.7hPLPΔ3173–4660 at 39-fold), while deletion of positions 3874 to 5810 caused levels to plummet (2.7hPLPΔ3874–5810 at onefold) on par with 2.7hPLP(−)Z. Therefore, deletion of a larger segment, which spans both sectors, was evaluated. β-gal activity for 2.7hPLPΔ3173–5806 was only ninefold higher than 2.7hPLP(−)Z (Figure 5), suggesting that loss of regulatory element(s) located between positions 3173 to 4660 may be compensated by regulatory element(s) contained within positions 4661 to 5810, and vice versa. Taken together, these results indicate that multiple positive regulatory elements reside between intron 1 position 3874 and 5810, which are active in Oli-neu cells, and are functionally redundant.
Supplementary Exons Within hPLP1 Intron 1 are Utilized to Generate Splice Variants in Oli-neu Cells
Reverse Transcription-Polymerase Chain Reaction (RT-PCR) analysis was performed to test whether the apparent conundrum in the transfection results between 2.7hPLPΔ3173–5806 (ninefold) and 2.7hPLPΔ3874–5810 (onefold) in Figure 5 may be due to alternative splicing. As discussed earlier, splice isoforms that contain all of exon AB or exon C are enzymatically inactive, while isoforms that contain only a portion of exon AB (A or A′ sectors) produce β-gal activity similar to that of the classic fusion protein (which lacks any hPLP1 intron 1-derived sequence). As expected, RNA isolated from Oli-neu cells transfected with 2.7hPLP(−)Z did not yield any RT-PCR products containing either exon AB or exon C (Figure 6). However, cells transfected with 2.7hPLP(+)Z express splice variants containing either exon AB (including merely the A portion) or exon C (Figure 6). Exon AB (or just the A portion) was incorporated in splice variants derived from 2.7hPLPΔ3874–5810, but not exon C (Figure 6); the distal (3′) third of exon C is missing from 2.7hPLPΔ3874–5810. Altogether, these results demonstrate that the supplementary exons in hPLP1 intron 1 are utilized in Oli-neu cells and, as a consequence, can affect the overall output of β-gal activity from hPLP1-lacZ constructs.
The supplementary exons in hPLP1 intron 1 are utilized in Oli-neu cells transiently transfected with hPLP1-lacZ constructs. Oli-neu cells were transfected with equimolar amounts of the indicated constructs and total RNA isolated 48 h post DNA addition. RT-PCR analysis was performed using primer pairs specific for products that contain only the A region of exon AB (hPLP A), exon AB (hPLP AB), or exon C (hPLP C). A β-actin primer set was used as a positive control. Only cells transfected with 2.7hPLP(+)Z produced all three types of splice variants.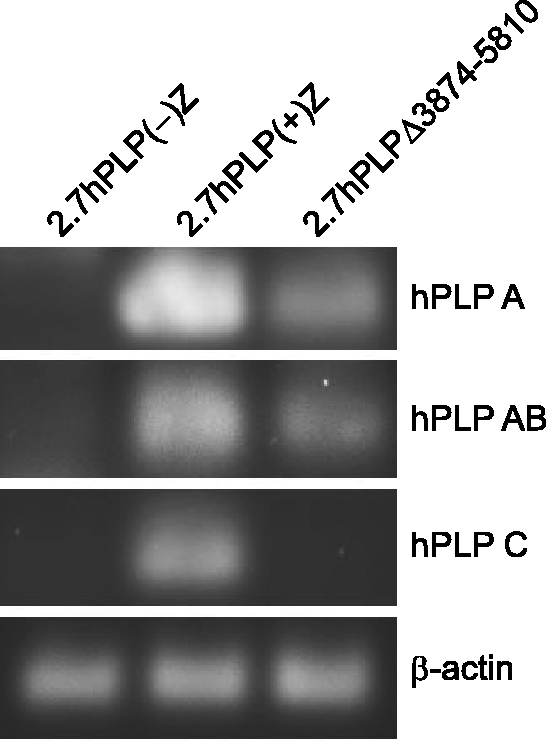
Discussion
Although the PLP1 gene encodes the most abundant protein in CNS myelin, not much was known about the mechanisms that govern expression of the human gene. Previous studies with the mouse gene showed that intron 1 was required to promote good activity of a mPlp1-lacZ transgene in brain, which developmentally mirrored expression of the endogenous mPlp1 gene (Wight et al., 1993; Li et al., 2002). Several purported enhancers have been identified in mPlp1 intron 1. One of these is located just over 1 kb downstream of exon 1 and was named ASE because it exhibited both antisilencer (Dobretsova and Wight, 1999) and enhancer (Dobretsova et al., 2000; Meng et al., 2005) properties in the N20.1 (oligodendroglial) cell line. Enhancer-like features were also apparent in another oligodendroglial cell line, Oli-neu, which appears to be at a slightly more mature stage of differentiation than N20.1 based on the relative abundance and splice isoforms generated for several myelin genes, including mPlp1 (Pereira et al., 2011). Located further downstream in the intron are two evolutionary conserved regions that were capable of augmenting expression of a minimally promoted reporter gene (eGFPlacZ) in transgenic mice. The wmN1 enhancer region (1,171 bp in all) led to transgene expression in myelinating oligodendrocytes and OECs, while the wmN2 enhancer region (313 bp) fostered expression in oligodendrocyte progenitors, oligodendrocytes actively elaborating myelin sheaths, OECs, Schwann cells and their progenitors, and the neural crest and its derivatives (Tuason et al., 2008). However, whether the wmN1 or wmN2 region could function in collaboration with its own promoter—not just a basal promoter (HSV thymidine kinase promoter)—was unknown.
The current study was undertaken to test whether the first intron of hPLP1 is required for hPLP1 gene expression, and if so, whether sequences orthologous to the enhancers identified in mPlp1 intron 1 or yet unidentified regulatory regions are responsible. Unfortunately, there exist only a few oligodendroglial cell lines of human origin (Buntinx et al., 2003), none of which expresses the hPLP1 gene except for a hybrid cell line derived by fusing a 6-thioguanine-resitant mutant of the human rhabdomyosarcoma RD with human oligodendrocytes cultured from a surgical specimen (McLaurin et al., 1995). Therefore, we were constrained to use a mouse cell line (Oli-neu), which expresses mPlp1. Oli-neu cells have been used as a model system to study many aspects of oligodendroglial biology including cell-type-specific gene regulation (Aruga et al., 2007), differentiation (Joubert et al., 2010), and growth factor-induced apoptosis (Schulz et al., 2009), whose results have been corroborated with primary cultures of oligodendrocyte precursor cells or in vivo. Previously we have shown that Oli-neu cells, when grown under the same conditions used here (i.e., differentiation medium), may be moving toward the immature oligodendrocyte stage based on the splice variants expressed for mPlp1 and other myelin-related genes including myelin basic protein (Mbp) and 2′,3′-cyclic nucleotide 3′-phosphodiesterase (Cnp) (Pereira et al., 2011).
Similar to mPlp1, an ApaI fragment of hPLP1 genomic DNA, which spans sequences from the proximal 2.7 kb of 5′-flanking DNA to an internal site in exon 2, was able to drive lacZ reporter gene (2.7hPLP(+)Z) expression to high levels in Oli-neu cells, relative to a related construct (2.7hPLP(−)Z) which totally lacks hPLP1 intron 1 (Figure 1). Thus, the first intron is required to achieve substantial levels of expression. Inclusion of an additional 3.5 kb of 5′-flanking in the 6.2hPLP(+)Z construct led to a further (although modest) increase in β-gal activity, suggesting that a positive regulatory element may reside within this upstream segment. Previously, an evolutionarily conserved region in mPlp1 termed 4250-Opo (positions −4088 to −2170 relative to the major transcription site identified by Macklin et al. (1987)) was shown to direct expression of a minimally promoted eGFPlacZ reporter gene in discrete neuronal populations including spinal cord gray matter, hippocampus, striatum, and numerous ventral neuronal nuclei in brain but not in glia, in the same study from which the wmN1 and wmN2 enhancers were identified (Tuason et al., 2008). The orthologous 4250-Opo region is fully present in hPLP1-lacZ constructs containing 6.2 kb of hPLP1 5′-flanking DNA, but largely absent from those having only 2.7 kb of 5′-flanking DNA. Yet if the 3.5 kb portion of 5′-flanking hPLP1 DNA unique to the 6.2hPLP-related constructs contains a positive regulatory element, one would have expected the level of β-gal activity in 6.2hPLP(−)Z transfected cells to be higher than 2.7hPLP(−)Z, which was not the case (Figure 1). Thus, if a regulatory element lies within this distal portion of 5′-flanking hPLP1 DNA, its influence is minimal in Oli-neu cells in comparison to those from the first intron.
Of the three purported enhancers from mPlp1 intron 1, only the human sequence orthologous to wmN2 unequivocally demonstrated activity in Oli-neu cells (Figure 3). There is 85% identity between the wmN2 sequences in mouse and human (313 and 314 bp, respectively). The wmN2 activity from hPLP1 appears to emanate from the latter two thirds of the region since there was not a significant difference in the levels of β-gal activity between 2.7hPLPΔ5811–6221 and 2.7hPLPΔ5617–6221 (Figure 3). Locate downstream of wmN2 is a novel regulatory region (intron positions 7573–8167) that acts to modestly increase hPLP1-lacZ activity in Oli-neu cells. Loss of this element, along with wmN2, presumably explains the very low level of β-gal activity observed with 2.7hPLPΔ5811–8167, which essentially was the same as 2.7hPLP(−)Z (Figure 3).
While the ASE sequence from mouse (mPlp1 intron 1 positions 1093–1177) displays substantial enhancer activity in Oli-neu cells (Pereira et al., 2011), the orthologous region in hPLP1 does not, as evidenced by the high level of β-gal activity observed with 2.7hPLPΔ212–3168. Moreover a segment (positions 1685–3168), directly downstream of orthologous ASE region, may possibly contain a negative regulatory element; levels of β-gal activity for 2.7hPLPΔ212–3168 and 2.7hPLPΔ1685–3168 were more than twice that of 2.7hPLP(+)Z (Figure 4). Thus, the hPLP1 sequence orthologous to the ASE is not likewise operative in Oli-neu cells. This is consistent with results from an earlier study which showed that the human sequence was incapable of functionally substituting for the mouse ASE in mPlp1-lacZ constructs tested in N20.1 cells (Meng et al., 2005).
Initially, the sequence orthologous to the wmN1 region in hPLP1 was deemed not to function as an enhancer since its loss, along with the adjoining 331 bp of 5′-sequence, did not cause a decrease in β-gal activity. In fact, the level of β-gal activity was somewhat higher for 2.7hPLPΔ3173–4660 than 2.7hPLP(+)Z (Figure 5). Because removal of intron 1 positions 3874 to 5810 (distal 705 bp of wmN1 and adjoining 1,232 bp, downstream) resulted in such a dramatic decrease β-gal activity, the sequence immediately downstream of wmN1 was targeted for deletion. Surprisingly, 2.7hPLPΔ4661–5810 and 2.7hPLP(+)Z generated similar levels of β-gal activity in Oli-neu cells. However, combined loss of the wmN1 region, and the immediate downstream sequence in 2.7hPLPΔ3173–5806 did result in a decrease in β-gal activity relative to 2.7hPLP(+)Z. These data suggest that the orthologous wmN1 region functions as a positive regulatory element; however, its loss can be functionally compensated by the presence of another element located immediately downstream. Only when both elements are eliminated does their significance become apparent.
Because the higher level of β-gal activity for 2.7hPLPΔ3173–4660 over 2.7hPLP(+)Z in Figure 5 might be due to changes in the pools of splice variants, RT-PCR analysis was performed (Figure 6). (Incorporation of exon AB or exon C in splice variants leads to the addition of multiple [in-frame] stop codons downstream of the classic translation start site in PLP1 exon 1. The longest open reading frame [ORF] with splice variants containing exon AB or exon C begins at codon 91 of lacZ. Since the N-terminal region of β-gal is required for enzymatic activity (Mohler and Blau, 1996), these splice products would not contribute to the overall amount of β-gal activity.) Oli-neu cells transfected with 2.7hPLP(+)Z generated splice variants containing either exon AB (or just the A portion) or exon C, unlike cells transfected with 2.7hPLP(−)Z (Figure 6). Similarly, 2.7hPLPΔ3874–5810 transfected cells also expressed splice variants containing all or just a part of exon AB (A portion), but not hPLP C-related splice variants as exon C is missing from this construct. Thus, the supplementary exons in hPLP1 intron 1 are utilized even in Oli-neu cells, indicating that the splice factors needed for their incorporation are conserved between human and mouse. Because 2.7hPLPΔ3173–4660 and 2.7hPLPΔ3173–5806 only express isoforms having β-gal activity, this would explain why their levels were higher than 2.7hPLP(+)Z or 2.7hPLPΔ3874–5810, respectively (Figure 5); 2.7hPLP(+)Z and 2.7hPLPΔ3874–5810 are capable of generating isoforms devoid of enzymatic activity as indicated by the detection of exon C- or exon AB-containing transcripts (Figure 6).
In conclusion, transfection analysis in Oli-neu cells indicates the importance of hPLP1 intron 1 in regulating hPLP1 expression. The intron contains multiple positive regulatory elements that are required for maximal expression of hPLP1-lacZ constructs. The strongest of these in Oli-neu cells appears to be the wmN2 enhancer. There is 85% identity between the wmN2 sequences from mouse and human (313 and 314 bp, respectively). Activity of the human wmN2 sequence appears to emanate from the latter two thirds since 2.7hPLPΔ5811–6221, which contains the first 102 bp of wmN2, showed similar levels of activity as 2.7hPLPΔ5617–6221, which lacks wmN2 altogether (Figure 4). While the levels of conservation between wmN1 and ASE sequences in human and mouse are quite similar (72% and 71% identity, respectively), only the wmN1 enhancer from hPLP1 appears to be active in Oli-neu cells. Even so, removal of the wmN1 region did not cause β-gal activity levels to fall (2.7hPLPΔ3173–4660 in Figure 5). Only in concert with the loss of additional sequences (hPLP1 intron 1 positions 4661–5810) was a decrease observed. Thus, there appears to be functional redundancy of the wmN1 enhancer with another element located directly downstream. It is not altogether surprising that activity of wmN1 is modest in comparison to wmN2 in Oli-neu cells, due to the developmental status (oligodendrocyte precursor/immature oligodendrocyte) of the cell line. Moreover, transfection results with the wmN1-deleted construct (2.7hPLPΔ3173–4660) are confounded by alterations in splice variants, due to the partial or complete lack of exons AB and C, respectively. Similarly, absence of the proximal two thirds of exon AB in 2.7hPLPΔ1685–3168 may help to explain why it showed higher levels of β-gal activity than 2.7hPLP(+)Z (Figure 4). Thus, our initial interpretation of a negative element mapping to hPLP1 intron 1 positions 1685 to 3168 may be incorrect if 2.7hPLP(+)Z produces a substantial amount of the splice variant containing exon AB in its entirety. Nevertheless, the current transfection analysis demonstrates for the first time that the wmN1 and wmN2 enhancers are capable of functioning in collaboration with their native promoter—not just a basal heterologous promoter—to augment hPLP1 expression. In addition to these enhancers, novel positive regulatory elements reside between hPLP1 intron 1 positions 7573 to 8167 and 4661 to 5810 (exhibits functional redundancy with wmN1), which are operative in Oli-neu cells. Thus, hPLP1 expression is controlled via multiple positive regulatory elements in intron 1. It is possible that the relative activity mediated through these regulatory modules changes during primary myelination, myelin maintenance, and remyelination.
Summary
This study demonstrates that human PLP1 intron 1 contains several positive transcriptional regulatory elements as well as a couple of human-specific alternatively spliced exons that are utilized in an oligodendroglial cell line (Oli-neu) of mouse origin.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the National Multiple Sclerosis Society (RG 2705) and National Institutes of Health (R01 NS037821), and a gift from the Rampy MS Research Foundation.
Author Contributions
S. H. constructed most of the Plp1-lacZ plasmids used in the study, with assistance from H. H. H. H., N. T. K., and L. A. J. conducted the transfection analyses, while N. T. K. performed the RT-PCR experiments. P. A. W. conceived and directed the study. H. H. and P. A. W. wrote the manuscript, with significant input from N. T. K. All authors participated in editing the final manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
