Abstract
Background:
P-loop and αC-helix compressing (PACC) mutations are a distinct structural subset of atypical epidermal growth factor receptor (EGFR) alterations in non-small-cell lung cancer (NSCLC), yet no standard first-line strategy has been established.
Objectives:
This study aimed to evaluate first-line treatment outcomes in patients with PACC-mutant NSCLC.
Design:
A real-world retrospective study.
Methods:
This retrospective cohort study enrolled 100 patients with PACC-mutant NSCLC who were diagnosed between February 2015 and April 2025. The efficacy of different first-line treatment strategies in this population was evaluated.
Results:
Among the 100 patients, compared with chemotherapy, treatment with tyrosine kinase inhibitors (TKIs) significantly prolonged progression-free survival (PFS; 14.2 vs 5.2 months, hazard ratio (HR) = 0.348, p = 0.005). The inverse probability of treatment weighting (IPTW)-weighted Cox analysis produced results consistent with the primary analysis (IPTW-adjusted HR = 0.487, p = 0.039). Among patients treated with TKIs, both second-generation (13.9 vs 13.1 months, HR = 0.338, p = 0.015) and third-generation TKIs (16.8 vs 13.9 months, HR = 0.239, p = 0.011) appeared to be associated with longer PFS compared with first-generation TKIs; however, given the relatively small subgroup sizes, these comparisons should be interpreted with caution. In patients with brain metastases, third-generation TKIs were associated with numerically longer PFS (28.0 vs 11.2 months, HR = 0.324, p = 0.132) and significantly longer overall survival (not reached vs 29.0 months, HR = 0.097, p = 0.030) compared with second-generation TKIs. In patients with a single PACC mutation, second-generation TKIs significantly prolonged PFS compared with non-second-generation TKIs (18.0 vs 11.0 months, HR = 0.347, p = 0.032). In addition, subgroup analyses indicated that, relative to the E709X subset, patients with S768I mutations had significantly prolonged PFS under TKI treatment (24.6 vs 10.6 months, HR = 0.263, p = 0.027).
Conclusion:
TKIs may represent a promising first-line option for advanced NSCLC with PACC mutations. The choice of specific TKIs may depend on genomic characteristics and brain metastasis status.
Plain language summary
Some people with advanced lung cancer have rare changes in a gene called EGFR. One group of these changes is known as PACC mutations. For people with this type of mutation, it is not yet clear which first treatment works best. This makes treatment decisions more challenging.
The research team reviewed the medical records of 100 people with this rare EGFR mutation. They compared the effectiveness of different first-line treatment options to better understand which treatments may be more suitable for these patients.
Overall, people who received targeted treatment tended to live longer without their cancer getting worse compared with those who received chemotherapy. In certain groups of patients—such as those whose cancer had spread to the brain or who had specific mutation patterns—some newer targeted medicines appeared to work better.
These results suggest that targeted treatment may be a promising first option for people with this rare type of lung cancer. However, further research is needed to better determine which treatment is most appropriate for different patients.
Introduction
Alterations in the epidermal growth factor receptor (EGFR) gene occur in approximately 45%–50% of Asian patients with lung adenocarcinoma. 1 Among these, deletions affecting amino acid residues 747–750 in exon 19 and the L858R point mutation in exon 21 represent the most prevalent EGFR mutation subtypes. 2 With the rapid development and widespread adoption of next-generation sequencing (NGS) technologies, an expanding spectrum of EGFR alterations has been identified. To date, at least 200 distinct EGFR mutation types have been reported in non-small-cell lung cancer (NSCLC). 3 Although tyrosine kinase inhibitors (TKIs) directed against mutant EGFR have substantially improved clinical outcomes in EGFR-positive NSCLC, considerable heterogeneity in therapeutic efficacy persists among different EGFR mutation subtypes, as well as among patients with the same mutation treated with different classes of TKIs.4–8
In the setting of marked therapeutic heterogeneity among EGFR-mutant NSCLC, P-loop and αC-helix compressing (PACC) mutations have emerged as a distinct subgroup of atypical EGFR alterations. These mutations are characterized by compression of the phosphate-binding loop and/or the αC-helix within the EGFR kinase domain, resulting in conformational changes in the ATP-binding pocket. 9 Although PACC mutations account for approximately 12.5% of all EGFR alterations, 10 their clinical significance remains underexplored. Structural and functional analyses by Robichaux et al. 9 have demonstrated that PACC mutations exhibit differential sensitivity to various classes of EGFR TKIs, highlighting their potential contribution to the observed heterogeneity in treatment responses. However, despite their relatively high prevalence and distinct biological features, studies addressing optimal first-line therapeutic strategies for NSCLC patients harboring PACC mutations remain extremely limited, and no widely accepted standard first-line treatment regimen has been established to date.
Therefore, in the present study, we retrospectively explored potential prognostic determinants in NSCLC patients harboring PACC mutations, with particular focus on PACC mutation subtypes, the presence or absence of brain metastases, and distinct treatment modalities.
Methods
Study design and patients
This retrospective cohort study screened 2000 patients with EGFR-mutant NSCLC and ultimately identified 100 patients with lung adenocarcinoma harboring PACC mutations who were diagnosed at Zhejiang Cancer Hospital between February 2015 and April 2025. The classification of PACC mutations followed published expert consensus and established EGFR mutation classification frameworks, with detailed mapping provided in Table S1. The inclusion criteria were as follows: (1) histologically or cytologically confirmed primary advanced lung adenocarcinoma; (2) presence of a PACC mutation detected by multiplex polymerase chain reaction or NGS, including compound PACC mutations with or without co-occurring classical sensitizing EGFR mutations; (3) receipt of at least two cycles of first-line treatment; (4) availability of at least one measurable target lesion; and (5) availability of survival follow-up data, regardless of event occurrence. The exclusion criteria included the following: (1) presence of non-PACC EGFR mutations such as exon 19 deletion, L858R, exon 20 insertion, or L861Q; (2) squamous cell carcinoma, small-cell lung cancer, or mixed histological subtypes; (3) incomplete medical records; and (4) patients with potentially curative treatment options, including radical surgery or radiotherapy. The reporting of this study conforms to the STROBE guidelines for cohort studies. 11 This study was approved by the Ethics Committee of Zhejiang Cancer Hospital (approval number: IRB-2025-1164), and all procedures were conducted in accordance with the principles of the Declaration of Helsinki.
Outcome assessment
Radiologic assessments were performed during systemic treatment according to routine clinical practice, typically at 6- to 8-week intervals. Brain MRI was routinely performed at baseline. During follow-up, repeat brain MRI examinations were conducted according to routine clinical practice, either at regular intervals or when neurological symptoms were suspected. Tumor response was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1. 12 The objective response rate (ORR) was defined as the proportion of patients who achieved a complete response (CR) or partial response (PR). The disease control rate (DCR) was defined as the proportion of patients who achieved CR, PR, or stable disease. Progression-free survival (PFS) was defined as the interval from the initiation of first-line systemic treatment to documented disease progression, death from any cause, or the date of last follow-up, whichever occurred first. Overall survival (OS) was defined as the time from the initiation of first-line systemic treatment to death from any cause or the date of last follow-up. The data cutoff for this study was August 22, 2025.
Statistical analysis
Survival curves were estimated using the Kaplan–Meier method and compared by the log-rank test. Categorical variables were analyzed using the Chi-square test, and continuous variables were compared using Student’s t test or the Mann–Whitney U test, as appropriate. To address potential confounding in the comparison between first-line EGFR-TKI and chemotherapy, inverse probability of treatment weighting (IPTW) based on propensity scores was performed. Propensity scores were estimated using multivariable logistic regression, including prespecified baseline covariates (age, sex, smoking status, Eastern Cooperative Oncology Group (ECOG) performance status, disease stage, liver metastasis, brain metastasis, and treatment era). Weights were calculated to estimate the average treatment effect and truncated at the 1st and 99th percentiles to reduce the influence of extreme weights. Covariate balance before and after weighting was assessed using standardized mean differences (SMD). Variables with residual imbalance (SMD >0.1) after weighting were additionally adjusted in sensitivity analyses. Weighted Kaplan–Meier estimators and weighted Cox proportional hazards models with robust variance estimators were applied to estimate treatment effects in the IPTW-adjusted cohort. For other comparative and subgroup analyses, conventional multivariable Cox regression models were used. Patients without documented progression or death were censored at the date of last follow-up. The proportional hazards assumption was evaluated using Schoenfeld residuals. All tests were two-sided, with p < 0.05 considered statistically significant. Statistical analyses were performed using R (version 4.4.1; R Foundation for Statistical Computing, Vienna, Austria).
Results
Clinicopathological characteristics
A total of 100 patients with lung adenocarcinoma who received either first-line EGFR-TKIs (n = 88) or platinum-based chemotherapy (n = 12) were retrospectively analyzed. Among patients treated with EGFR-TKIs, first-generation TKIs included icotinib (n = 10) and gefitinib (n = 1); second-generation TKIs included afatinib (n = 52), sutetinib (n = 3), and mehwatinib (n = 1); and third-generation TKIs included Osimertinib (n = 6), furmonertinib (n = 9), almonertinib (n = 5), and lazertinib (n = 1). Baseline clinicopathological characteristics, including age, sex, smoking status, clinical stage, brain metastases, liver metastases, and ECOG performance status, were compared between the treatment groups. The detailed clinical and demographic characteristics are summarized in Table 1.
Baseline characteristics.
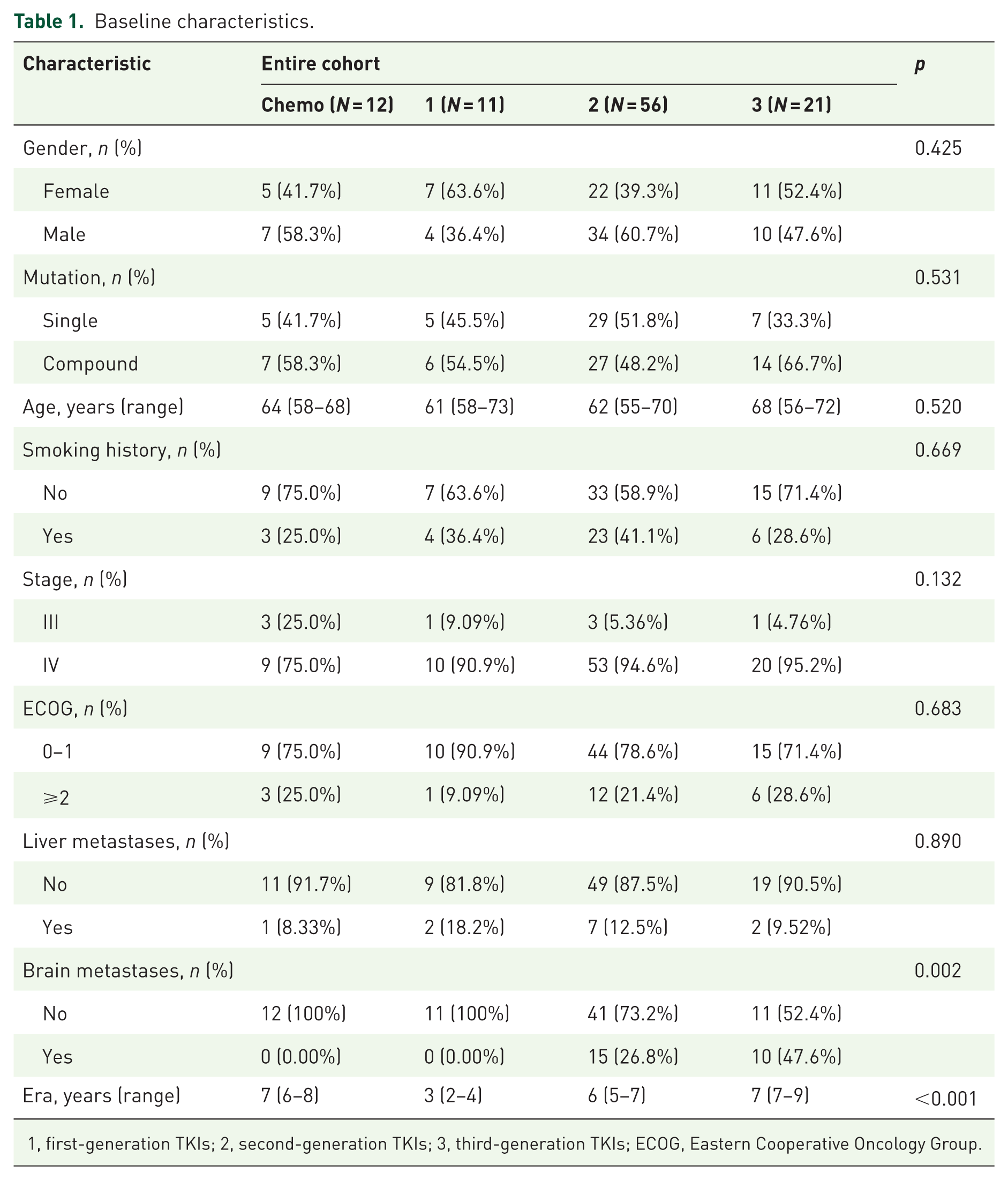
1, first-generation TKIs; 2, second-generation TKIs; 3, third-generation TKIs; ECOG, Eastern Cooperative Oncology Group.
Distribution of PACC mutation subtypes
The spectrum of PACC mutations in the 100 enrolled patients is summarized in Table 2. Among these patients, the most frequent mutation was G719X, followed by E709X and S768I, which were identified in 68 (68%), 12 (12%), and 11 (11%) patients, respectively. The remaining nine patients (9%) harbored other rare PACC mutations.
Distribution of PACC mutations identified in 100 patients from our center.
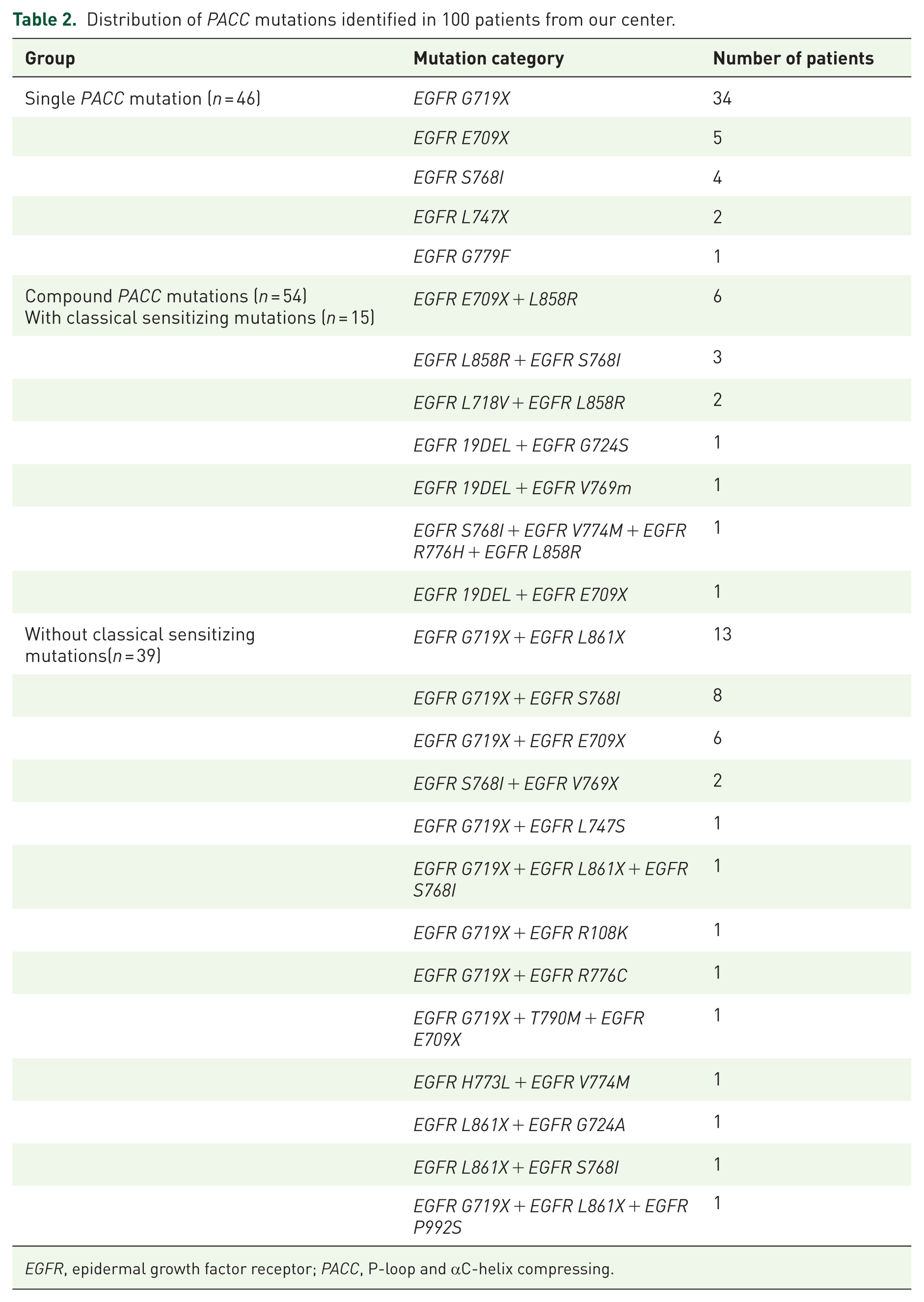
EGFR, epidermal growth factor receptor; PACC, P-loop and αC-helix compressing.
In total, 46 patients carried a single PACC mutation, whereas 54 patients harbored compound PACC mutations. Among those with compound mutations, 15 patients (27.8%) also carried a classical sensitizing EGFR mutation. In addition, 22 patients (40.7%) carried at least 2 distinct PACC mutations simultaneously, including 1 patient harboring 3 different PACC mutations. One patient additionally carried an EGFR L861Q mutation, and another harbored an EGFR T790M mutation.
Among the 46 patients with a single PACC mutation and the 54 patients with compound PACC mutations, G719X remained the most predominant alteration, accounting for 73.9% (34/46) and 63.0% (34/54), respectively.
Response to different first-line therapies
Among the 100 patients with PACC mutations, 12 received first-line platinum-based chemotherapy, and 88 received first-line EGFR-TKIs. One patient treated with TKIs lacked a response evaluation. The ORR in the chemotherapy and TKIs groups was 25.0% and 55.7%, respectively (p = 0.066), while the DCR was 83.3% and 95.5%, respectively (p = 0.154; Figure 1). Among patients who received TKIs, 11, 56, and 21 patients were treated with first-, second-, and third-generation TKIs, respectively. The ORR among patients treated with first-, second-, and third-generation TKIs was 36.4%, 55.4%, and 66.7%, respectively (p = 0.394), and the corresponding DCR was 90.9%, 94.6%, and 100.0% (p = 0.283). Further comparisons between patients treated with first-generation TKIs and those treated with non-first-generation TKIs demonstrated ORR of 36.4% and 58.4%, respectively (p = 0.322), and DCR of 90.9% and 96.1%, respectively (p = 0.392).

Treatment timeline for 100 NSCLC patients included in the study. The dashed vertical line separates the first-line treatment phase (left) from the later-line treatment phase (right). Bar length represents the duration of disease control. On the left, different colors indicate efficacy outcomes for first-line therapy. On the right, bar colors denote treatment groups (purple for chemotherapy, pink for first-generation TKIs, light blue for second-generation TKIs, and green for third-generation TKIs).
Among the 88 patients treated with first-line TKIs, 11 carried E709X mutations, 61 carried G719X mutations, 9 carried S768I mutations, and 7 harbored other rare PACC subtypes. The ORR for these subgroups were 45.5%, 55.7%, 55.6%, and 71.4%, respectively (p = 0.804), and the corresponding DCR were 90.9%, 95.1%, 100.0%, and 100.0% (p = 0.004). Within the most common subtype, G719X, the ORR for patients treated with first-, second-, and third-generation TKIs were 37.5%, 53.8%, and 71.4%, respectively (p = 0.388), and the corresponding DCR were 87.5%, 94.9%, and 100.0% (p = 0.050).
Survival outcomes with different first-line treatments
PFS and OS in the overall cohort
The impact of different first-line treatment strategies on PFS and OS was evaluated in the overall PACC-mutant population. Both multivariable Cox regression analysis and Kaplan–Meier curves demonstrated that patients treated with first-line TKIs had a significantly longer median PFS (mPFS) than those treated with platinum-based chemotherapy (14.2 vs 5.2 months, HR = 0.348, p = 0.005; Figure 2(a) and Table S2), indicating that targeted therapy significantly delayed disease progression compared with chemotherapy. By contrast, no significant difference in median OS (mOS) was observed between the two groups (65.9 vs 49.6 months, HR = 1.616, p = 0.385; Figure 2(b)). IPTW-adjusted mPFS (7.2 vs 14.2 months; IPTW-adjusted HR = 0.487, p = 0.039; Figure S1(A)) and mOS (65.9 vs 56.2 months; IPTW-adjusted HR = 1.622, p = 0.327; Figure S1(B)) were consistent with the unadjusted analysis. The balance of baseline covariates before and after IPTW weighting is illustrated by the SMD plot (Figure S2). In the sensitivity analysis incorporating additional covariates with SMD >0.1, the results remained consistent with the primary analysis (Table S3).
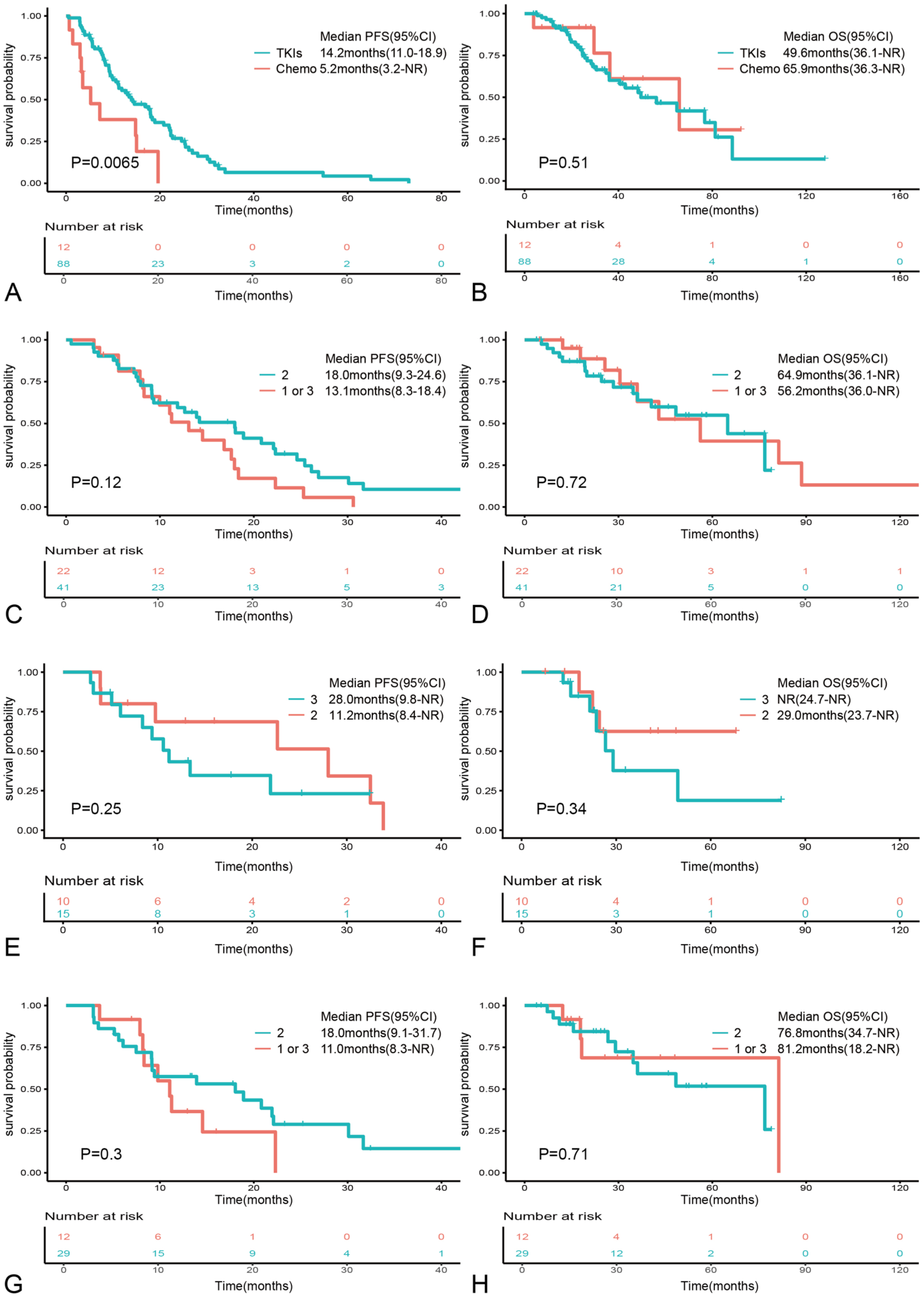
Kaplan–Meier curves for PFS and OS. (a, b) PFS and OS of the different TKIs in the overall population. (c, d) PFS and OS of the different TKIs in the non-brain metastasis cohort. (e, f) PFS and OS of the different TKIs in the brain metastasis cohort. (g, h) PFS and OS of different TKIs in a single PACC mutation cohort.
Kaplan–Meier analyses further revealed no statistically significant differences in mPFS (13.1 vs 13.9 vs 16.8 months, p = 0.64; Figure S3(A)) or mOS (68.7 vs 49.6 vs not reached, p = 0.64; Figure S3(B)) among patients treated with first-, second-, and third-generation TKIs, respectively. However, multivariable Cox analyses suggested a trend toward delayed disease progression with second-generation TKIs (HR = 0.338, p = 0.015) and third-generation TKIs (HR = 0.239, p = 0.011) compared with first-generation TKIs. Given the relatively small and uneven subgroup sizes, these comparisons should be interpreted with caution and considered exploratory.
Survival outcomes according to brain metastasis status
We next investigated the prognostic impact of different treatment strategies in patients stratified by the presence of brain metastases. In patients without brain metastases, those treated with second-generation TKIs experienced numerically longer PFS (18.0 vs 13.1 months, p = 0.12; Figure 2(c)) and OS (64.9 vs 56.2 months, p = 0.72; Figure 2(d)) compared with those treated with first- or third-generation TKIs; however, these differences were not statistically significant (p > 0.05). By contrast, among patients with brain metastases, Kaplan–Meier curves and multivariable Cox regression analysis showed that third-generation TKIs were associated with numerically longer PFS (28.0 vs 11.2 months, HR = 0.324, p = 0.132; Figure 2(e)) and significantly longer OS (not reached vs 29.0 months, HR = 0.097, p = 0.030; Figure 2(f)) compared with second-generation TKIs. To account for potential confounding from local therapy, we further compared concomitant local treatments during first-line therapy. No significant differences were observed between groups (Table S4).
Survival outcomes according to PACC mutation subtypes
We further analyzed treatment efficacy according to specific PACC mutation patterns. Among patients with a single PACC mutation, the use of second-generation TKIs was associated with a longer PFS compared with non-second-generation TKIs (18.0 vs 11.0 months, p = 0.30; Figure 2(g)), although this difference was not statistically significant. No significant difference was observed in mOS (76.8 vs 81.2 months, p = 0.71; Figure 2(h)). Multivariable Cox regression analysis, however, identified second-generation TKIs as an independent protective factor for PFS in this subgroup (HR = 0.347, p = 0.032), while no significant association was found with OS (p > 0.05). In patients with compound PACC mutations, neither Kaplan–Meier analysis nor multivariable Cox regression demonstrated a significant difference in PFS (12.6 vs 17.9 months, HR = 1.737, p = 0.185; Figure S3(C)) or OS (49.6 vs 56.2 months, HR = 2.227, p = 0.166; Figure S3(D)) between those treated with second-generation TKIs and those treated with non-second-generation TKIs.
Among patients harboring the most common subtype, G719X, no significant differences in PFS (14.5 vs 18.0 months, p = 0.53; Figure S3(E)) or OS (81.2 vs 64.9 months, p = 0.44; Figure S3(F)) were observed between patients treated with first- or third-generation TKIs and those treated with second-generation TKIs. Finally, when comparing different PACC mutation subtypes, both Kaplan–Meier and multivariable Cox analyses indicated that patients with G719X mutations tended to have longer PFS (17.6 vs 10.6 months, HR = 0.585, p = 0.197; Figure S3(G)) and OS (76.8 vs 29.8 months, HR = 0.488, p = 0.112; Figure S3(H)) than those with E709X mutations. Notably, patients with S768I mutations exhibited a significantly prolonged PFS (24.6 vs 10.6 months, HR = 0.263, p = 0.027; Figure S3(G)) compared with the E709X subgroup, whereas no statistically significant difference in OS was observed (40.5 vs 29.8 months, HR = 0.593, p = 0.467; Figure S3(H)). These findings suggest that treatment benefit may vary among distinct PACC subtypes in NSCLC.
First-line TKI therapy in PACC-mutant NSCLC
We systematically summarized ongoing and previously reported studies evaluating first-line TKIs therapy in PACC-mutant NSCLC7,9,10,13–28 (Table 3). Based on the available literature, current evidence remains limited, largely derived from small cohorts, with heterogeneous treatment outcomes across different genomic subtypes and therapeutic strategies.
Summary of studies on first-line TKIs therapy in PACC-mutant NSCLC.

NR, not reached or not reported; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; TTF, time to treatment failure.
Discussion
In this retrospective study, we evaluated the differential efficacy of various first-line treatment strategies and the potential determinants of treatment outcomes in patients with advanced NSCLC harboring PACC mutations. Our findings underline the clinical relevance of distinct patient characteristics and PACC mutation subtypes in guiding therapeutic decision-making for this specific population.
In recent years, the therapeutic landscape for advanced NSCLC harboring classical EGFR-sensitive mutations has evolved substantially, with continued expansion of first-line and post-resistance treatment strategies.4–8 The FLAURA2 trial demonstrated that the combination of EGFR TKIs with chemotherapy can extend mOS to approximately 4 years in this population. 29 In addition, the HARMONi-A 30 and OptiTROP-Lung04 studies 31 have provided alternative treatment options for patients with EGFR TKIs resistance who lack identifiable targetable resistance mechanisms upon re-biopsy, thereby broadening the therapeutic paradigm beyond platinum-based chemotherapy. However, clinical evidence regarding the efficacy of TKIs in patients with rare EGFR-mutant NSCLC remains limited, particularly in those harboring PACC mutations. Using a preclinical model, Robichaux et al. 9 predicted that PACC mutations would be more sensitive to second-generation TKIs than to other EGFR inhibitors. Nevertheless, in real-world clinical practice, the optimal selection of TKIs and the patient subgroups most likely to benefit from specific agents in the context of PACC mutations have remained unclear.
Our results showed that, in patients with advanced NSCLC harboring PACC mutations, platinum-based chemotherapy did not demonstrate superior survival benefit compared with TKIs, consistent with the findings reported by Miura et al. 7 Previous studies have suggested that differences in the tumor microenvironment among patients with atypical EGFR mutations may influence their response to immunotherapy. 32 However, as only one patient in our cohort received platinum-based chemoimmunotherapy, the impact of immunotherapy on outcomes in patients with PACC mutations could not be meaningfully evaluated. Furthermore, our findings suggest that second- and third-generation TKIs may be associated with numerically improved outcomes compared with first-generation TKIs, consistent with the results reported by Gu et al. 28 Similarly, Watanabe et al. 14 reported limited efficacy of gefitinib in patients harboring the G719X subtype. By contrast, Leventakos et al. 13 observed clinical benefit with erlotinib in a subset of patients with the S768I mutation. Given that current evidence regarding the efficacy of first-generation TKIs in PACC-mutant NSCLC is largely derived from small cohorts, further validation in larger prospective studies is warranted.
In the subgroup of patients with brain metastases, compared with second-generation TKIs, third-generation TKIs were associated with numerically longer PFS and significantly improved OS, with no significant differences in the use of local therapies between the two groups. By contrast, among patients without brain metastases, second-generation TKIs were associated with numerically longer PFS and OS compared with other TKI generations. These observations may be partly attributable to the higher intracranial penetration of third-generation TKIs. 33 Collectively, our findings suggest that third-generation TKIs may be a reasonable first-line consideration in patients with concomitant brain metastases.
We also evaluated the impact of PACC mutation burden (single vs compound) on treatment outcomes. The results indicated that in patients harboring compound PACC mutations, those treated with second-generation TKIs had shorter PFS and OS compared with patients treated with first- or third-generation TKIs. By contrast, our results indicated that among patients with a single PACC mutation, second-generation TKIs might be associated with improved treatment outcomes. These findings are in line with the results reported by Atlagh et al., 34 who observed that osimertinib may be associated with longer PFS compared with other TKIs in patients with compound PACC mutations.
Finally, we further examined the association between different PACC mutation subtypes and treatment outcomes. Our analyses revealed that, compared with the E709X subtype, the S768I mutation may serve as an independent predictive factor for favorable outcomes in PACC-mutant patients receiving TKIs therapy. Furthermore, in our cohort, subgroup analyses of the most prevalent PACC mutation, G719X, revealed no significant differences in PFS or OS between patients treated with first- or third-generation TKIs and those receiving second-generation TKIs. These findings suggest that the efficacy of third-generation TKIs may vary across specific PACC mutation subtypes. Although previous analyses from the LUX-Lung 2, 3, and 6 trials have demonstrated that both G719X and S768I mutations are sensitive to second-generation TKIs, 35 multiple studies have also suggested that third-generation TKIs may also be associated with clinical benefit in patients harboring PACC mutations. These results warrant further validation in large-scale prospective clinical studies. Importantly, comparisons across different EGFR-TKIs generations in this study should be considered exploratory and hypothesis-generating rather than confirmatory, given the limited sample size and potential residual confounding inherent in retrospective analyses.
Several limitations of this study should be acknowledged. First, this was a single-center retrospective analysis with a relatively limited sample size, and selection bias cannot be completely excluded. Second, owing to the limited sample size, we were unable to further compare the efficacy of different TKI generations across specific PACC mutation subtypes. Third, given the extended study period and the evolution of detection technologies over time, inconsistencies in testing methodologies may have increased the risk of false-negative results for certain gene alterations. Fourth, only one patient in our cohort received platinum-based chemoimmunotherapy, precluding any meaningful statistical evaluation of immunotherapy in PACC-mutant NSCLC. Fifth, the lack of tumor-derived RNA samples precluded further mechanistic investigations into the biological basis underlying the prognostic differences observed among PACC subtypes. Sixth, the majority of patients in this cohort were treated with second-generation EGFR-TKIs, which may limit the generalizability of our findings in the current era dominated by third-generation TKIs. Therefore, the clinical significance and heterogeneity of responses among PACC subtypes in the context of third-generation EGFR-TKI-based treatment require further exploration in future studies.
Conclusion
In conclusion, our study suggests that, for patients with advanced NSCLC harboring PACC mutations, TKI-based therapy may represent a promising first-line treatment strategy. In patients with concomitant brain metastases, third-generation TKIs were associated with improved survival outcomes. However, given the relatively small and uneven subgroup sizes and the retrospective nature of this study, comparisons across different EGFR-TKI generations should be interpreted with caution and considered exploratory and hypothesis-generating. Further prospective studies with larger sample sizes are warranted to validate these findings and to determine the optimal first-line therapeutic strategy for patients with PACC-mutant NSCLC.
Supplemental Material
sj-docx-2-tam-10.1177_17588359261442625 – Supplemental material for A real-world study on the comparative effectiveness of first-line treatment regimens in advanced NSCLC patients with PACC mutations
Supplemental material, sj-docx-2-tam-10.1177_17588359261442625 for A real-world study on the comparative effectiveness of first-line treatment regimens in advanced NSCLC patients with PACC mutations by Dujiang Liu, Xinyue Li, Kailai Yin, Yi Lu, Na Han and Yanjun Xu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359261442625 – Supplemental material for A real-world study on the comparative effectiveness of first-line treatment regimens in advanced NSCLC patients with PACC mutations
Supplemental material, sj-docx-3-tam-10.1177_17588359261442625 for A real-world study on the comparative effectiveness of first-line treatment regimens in advanced NSCLC patients with PACC mutations by Dujiang Liu, Xinyue Li, Kailai Yin, Yi Lu, Na Han and Yanjun Xu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-1-tam-10.1177_17588359261442625 – Supplemental material for A real-world study on the comparative effectiveness of first-line treatment regimens in advanced NSCLC patients with PACC mutations
Supplemental material, sj-pdf-1-tam-10.1177_17588359261442625 for A real-world study on the comparative effectiveness of first-line treatment regimens in advanced NSCLC patients with PACC mutations by Dujiang Liu, Xinyue Li, Kailai Yin, Yi Lu, Na Han and Yanjun Xu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
