Abstract
Background:
While targeted therapy has become the standard treatment for certain non-small-cell lung cancer (NSCLC) patients with gene mutation positivity, there remains a lack of enough reports of the efficacy of mesenchymal–epithelial transition (MET) alterations in the real world.
Objectives:
We aimed to explore the efficacy and toxicity of targeted therapy in NSCLC patients with different types of MET alterations and hope to provide more clinical medication guidance.
Design:
Designed different subgroups to compare the efficacy and safety of targeted therapy in NSCLC patients with MET alterations.
Methods:
We conducted analyses on the efficacy and safety of mesenchymal–epithelial transition factor-tyrosine kinase inhibitor (MET-TKI) therapy in NSCLC patients with MET alterations. Tumor response was evaluated based on the Response Evaluation Criteria in Solid Tumors version 1.1 criteria, and both progression-free survival (PFS) and overall survival were determined using the Kaplan–Meier method.
Results:
Our study encompassed 116 NSCLC patients with MET alterations, including MET ex14 skipping mutation (n = 50), MET primary amplification (amp) (n = 25), and secondary amp (n = 41). Among treated patients, 34 achieved a partial response, while 52 exhibited stable disease. The overall response rate for the entire cohort was 29.31%, with a disease control rate of 74.14%. A significant difference was observed in the median PFS among patients with MET ex14 skipping mutation, MET primary amplification (amp), and secondary amp (10.4 versus 6.6 versus 4.5 months, p = 0.002). In all, 69 patients experienced drug-related adverse effects, with the most common being peripheral edema (35.34%), nausea and vomiting (21.55%), and fatigue (10.34%). In total, 29 patients (25%) encountered drug-related adverse reactions of grade 3 or higher.
Conclusion:
MET-TKI therapy works better for MET ex14 skipping mutation than other types of MET gene alteration. In the two MET amplified groups, the secondary amp was less effective. This study may provide more research support for the treatment of these patients.
Introduction
Lung cancer ranks as the most prevalent and lethal form of cancer in China, presenting in two main categories – small-cell lung cancer (SCLC) and non-SCLC (NSCLC), the latter representing 80–85% of cases. The median survival for those with advanced lung cancer is a mere 10 months, 1 and the overall 5-year survival rate stands at a disheartening 15%. 2 Despite significant strides in lung cancer diagnosis and treatment, the current therapeutic approaches still fall short of achieving optimal outcomes.
In the past decade, amid the swift progress of precision medicine, numerous carcinogenic driver genes have been identified in NSCLC, such as Epidermal Growth Factor Receptor (EGFR), Kirsten rat sarcoma viral oncogene (KRAS), ROS proto-oncology 1 ( ROS1), mesenchymal–epithelial transition (MET), and others. These oncogenic driver genes are assuming an increasingly crucial role in guiding treatment decisions and determining therapeutic outcomes. Multiple studies have consistently demonstrated that MET alterations, encompassing MET exon 14 skipping mutation, MET amp, MET overexpression, and MET fusion, manifest in various roles within NSCLC. These alterations can function as either primary driver genes or serve as bypass mechanisms leading to secondary drug resistance. MET exon 14 skipping mutation, occurring in 2–3% of NSCLC (including adenocarcinoma, squamous cell carcinoma, and more commonly sarcomatoids), is notably associated with a poor prognosis.3,4 MET amp can be categorized into primary and secondary types, with the latter often arising after prolonged exposure to EGFR-TKIs.
In contrast to NSCLC patients with various types of oncogenic driver genes, those with MET gene alterations are frequently associated with an older age group, in particular individuals carrying the MET exon 14 skipping mutation are often aged over 70 years.5,6 Consequently, there is a heightened need for attention to the patient’s ability to tolerate drug-related adverse effects. Therefore, in comparison to traditional chemotherapy, targeted therapy, characterized by its more precise effects, emerges as a promising treatment avenue for NSCLC patients with MET alterations. Currently, series MET tyrosine kinase inhibitors (MET-TKIs), including crizotinib, capmatinib, savolitinib, tepotinib, and so on, have been developed to address these alterations. In addition, various novel drugs are undergoing clinical trials, though challenges persist in terms of efficacy and the identification of reliable biomarkers. Notably, there is still a lack of enough reports of clinical medication in the real world and the efficacy of MET-TKIs remains to be definitively established.
This study aims to evaluate the efficacy and safety of MET-TKIs for NSCLC patients with MET alterations, including MET exon 14 skipping mutation and MET amp, offering increased research support for treatment alternatives in NSCLC patients with MET gene alterations.
Methods
Patient inclusion criteria
This study included a cohort of 116 NSCLC patients with MET alterations who underwent treatment at Zhejiang Cancer Hospital between February 2015 and November 2023. The patient classification was determined through next-generation sequencing (NGS) or fluorescence in situ hybridization (FISH), where MET/CEN7 ⩾ 1.8 or gene copy number ⩾ 4 indicated MET amp. The patient groups were categorized into MET exon 14 skipping mutation, MET primary amp, and MET secondary amp. For patients without MET amp who received initial treatment and experienced resistance to drugs, all of them should receive the detection of MET secondary amp by NGS or FISH. Before using the MET-TKIs, the condition of the MET secondary amp should be ensured. The study followed the principles of the Declaration of Helsinki, and individual consent for this retrospective analysis was waived. The research protocol received approval from the Zhejiang Cancer Hospital Committee and other relevant institutions.
Treatment and response assessment
Clinical response was evaluated through a computed tomography (CT) scan based on Response Evaluation Criteria in Solid Tumors version 1.1, which includes categories such as complete response (CR), partial response (PR), disease stabilization (SD), and disease progression (PD). Progression-free survival (PFS) was characterized as the duration from the initiation of first-line targeted therapy to the date of PD diagnosis. Overall survival (OS) was defined as the duration from the commencement of targeted therapy to either death or the final follow-up. The objective response rate (ORR) is calculated as the combined total of CR and PR, while the disease control rate is the sum of CR, PR, and SD.
Statistical analysis
The last follow-up date was on 30 November 2023, with a complete follow-up rate of 100%. All patients underwent evaluations for PFS and OS. Kaplan–Meier survival analysis was employed to assess patient survival, and the log-rank test was used to compare survival among different prognostic factors. Univariate and multivariable analyses were conducted using Cox regression models. Statistical significance for all tests was set at a two-sided p-value of <0.05. The statistical analyses were carried out using SPSS 26.0 (IBM Corporation, Armonk, NY, USA) and GraphPad Prism 10.0 (GraphPad Software Inc., San Diego, CA, USA).
Results
Patient characteristics
The patient cohort was stratified into three groups: MET exon 14 skipping mutation (n = 50), MET primary amp (n = 25), and MET secondary amp (n = 41). All patients presented with stage IIIB, IIIC, or IV tumors, and their Eastern Cooperative Oncology Group Performance Status (ECOG PS) was within the range of 0–1. Histological examination indicated that the adenocarcinoma subtype was predominant, constituting 82.75% of the total tumors. Of the 25 patients with primary amplification, 16 (64.0%) patients had only MET amplification, 1 (4.0%) patient had KRAS mutation, and 8 (32.0%) patients had EGFR mutation. And in 41 patients with MET secondary amplification, 26 (63.4%) had a previous EGFR mutation, 4 (9.8%) had an Anaplastic lymphoma kinase (ALK) mutation, and 11 (26.8%) had no driver gene mutation. Before receiving targeted therapy, 98 patients (84.48%) had developed distant metastases. A summary of the baseline characteristics of the patients is presented in Table 1.
Patients characteristics.

ECOG PS, Eastern Cooperative Oncology Group Performance status.
Treatment response and survival analysis
In our study, 96 patients were assessable for response evaluation. None of the patients achieved CR, while 34 patients demonstrated PR, 52 patients had SD, and 10 patients exhibited PD. Among the 34 patients with PR, 19 were patients with MET exon 14 skipping mutation, 7 were patients with MET primary amp, and 8 were patients with MET secondary amp. In the evaluabled-cases, the ORR for the MET exon 14 skipping mutation group (47.5%) was higher than the other two groups, followed by the MET primary amp group (31.82%), and the MET secondary amp group had the lowest ORR (23.53%). Further details are illustrated in Figure 1.

The Falls map shows the response for all NSCLC patients with MET alterations. (a) The best response of MET-TKIs for patients with (a) MET exon 14 skipping mutation, (b) MET primary amp, and (c) MET secondary amp.
There is a noticeable difference in median PFS (mPFS) among MET exon 14 skipping mutation, primary amp, and secondary amp groups (10.4 versus 6.6 versus 4.5 months, p = 0.002). The efficacy of MET exon 14 skipping mutation shows no difference between first-line and posterior-line therapy (10.4 versus 7.1 months, p = 0.615), and similar results are observed in patients with MET primary amp (6.2 versus 6.6 months, p = 0.577). The median OS rates for MET exon 14 skipping mutation, primary amp, and secondary amp are 14.3, 10.1, and 9.4 months (p = 0.247), respectively, as illustrated in Figure 2.
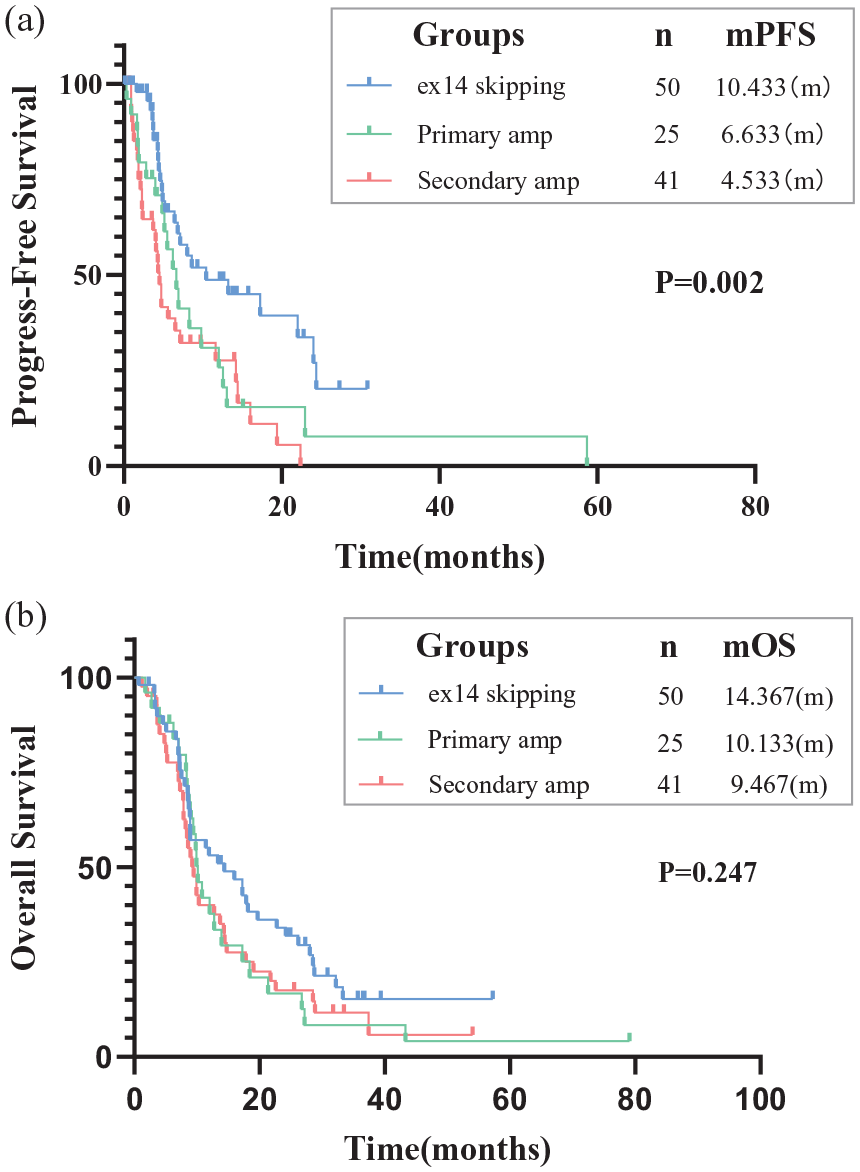
Kaplan–Meier estimates of PFS and OS. (a) PFS of MET ex14 skipping mutation, MET primary amp, and MET secondary amp groups (10.433 versus 6.633 versus 4.533 months, p = 0.002). (b) OS of MET ex14 skipping mutation, MET primary amp, and MET secondary amp groups (14.367 versus 10.133 versus 9.467 months, p = 0.247).
In addition, we evaluated the extent of amp in patients with MET primary amp. Out of the 25 patients, 14 could be assessed for the extent of amplification, with 3 of them classified as high amp (defined as MET/CEN7 ⩾5 or Gene copy number ⩾10) and 11 as low amp (defined as MET/CEN7 ⩾1.8, <5 and Gene copy number ⩾4, <10). Comparing the efficacy of these two groups, we found that the mPFS of the high amp group was significantly longer than that of the low amp group (6.6 versus 23.0 months, p = 0.019). No difference was observed in the mPFS between the MET secondary high (n = 10) and low (n = 19) amp groups (1.8 versus 4.7 months, p = 0.123). Further details are presented in the Appendix.
Multivariate analysis of the clinical features and prognosis
In the multivariate analysis of mPFS, we assessed the following criteria: gender, age, smoking history, Eastern Cooperative Oncology Group Performance Status (ECOG PS) score, stage, distant metastasis, surgery history, previous radiotherapy, and type of MET gene alteration (MET exon 14 skipping mutation/primary amp/secondary amp). Except for the type of MET gene alteration [95% confidence interval (CI): 1.076–1.459, p = 0.004], none of the other factors significantly impacted mPFS (p > 0.05). Detailed information is provided in Figure 3.

Forest plot of potential factors affecting progression-free survival in NSCLC patients with MET alterations, using a Cox proportional hazards model.
Toxicity evaluation
Out of the 116 patients, 69 (59.48%) experienced drug-related adverse effects. Common adverse events included peripheral edema (35.34%), nausea and vomiting (21.55%), fatigue (10.34%), erythema (9.48%), blurred vision (2.58%), elevated creatinine (5.17%), hypoalbuminemia (5.17%), and so on. Within the entire cohort, the incidence of drug-related adverse reactions at grade 3 and higher was 25%. Specific details of drug-related adverse reactions are outlined in Table 2.
Toxicities evaluation for MET-TKIs in NSCLC patients.

MET-TKI, mesenchymal–epithelial transition factor-tyrosine kinase inhibitor; NSCLC, non-small-cell lung cancer.
Discussion
Our study represents the largest sample exploration of MET-TKI therapy in NSCLC patients with various subtypes of MET alterations. It systematically investigates the efficacy of MET-TKIs in these patients while analyzing the drug-related toxicity of MET-TKIs. Significant variations in therapeutic effects were observed among the three MET-altered subtypes, demonstrating promising efficacy in MET exon 14 skipping mutation and MET primary amp. Importantly, the administration of MET-TKIs was well tolerated in these patients.
In recent decades, chemotherapy has been a prevalent treatment for malignancies and is extensively employed in the therapeutic management of NSCLC. However, the effectiveness of chemotherapy is generally constrained in advanced or metastatic NSCLC, particularly in patients with MET exon 14 skipping mutations. 7 In addition, immunotherapy has demonstrated limited efficacy in individuals with MET alterations.8 –11 Consequently, there is a compelling need to explore novel treatment modalities with enhanced efficacy for these patients. As precision medicine continues to advance, targeted therapy has assumed an increasingly pivotal role in the treatment of lung cancer. Notably, in other oncogene-driven lung cancers, such as those involving EGFR alterations, targeted therapy has demonstrated favorable outcomes.
Previous studies on the use of MET-TKIs for NSCLC patients with MET genetic changes have predominantly been retrospective with small sample sizes. Despite variations in efficacy, the overall results have demonstrated positive therapeutic effects. Generally, MET alteration is often considered a marker of poor prognosis, particularly in patients with MET amp. In the study conducted by Wolf et al., 12 the mPFS for patients with MET exon 14 alteration treated with capmatinib ranged from 5.4 to 12.4 months, whereas for MET amp patients, it was only 2.7–4.2 months. Similar findings have been reported in other preceding studies on MET-TKIs. In these studies, the mPFS for patients with MET exon 14 skipping mutation ranged from 5.4 to 12 months, while for those with MET amp, it was only 1.8–6.7 months.12 –23 The findings in our study align with these results. Notably, within all patients with MET amp, those with secondary amp exhibited poorer efficacy compared to the primary amp. In the study conducted by Yang et al., 13 patients with secondary MET amp were treated with MET-TKIs yet the overall mPFS was only 4 months. This may be attributed to the continuous activation of EGFR-independent pathways downstream in cases of MET secondary amp, providing a bypass pathway in the presence of an EGFR inhibitor.24,25 Several studies investigating the treatment outcomes of MET-TKIs have been succinctly summarized in Table 3.
Response for all the patients.
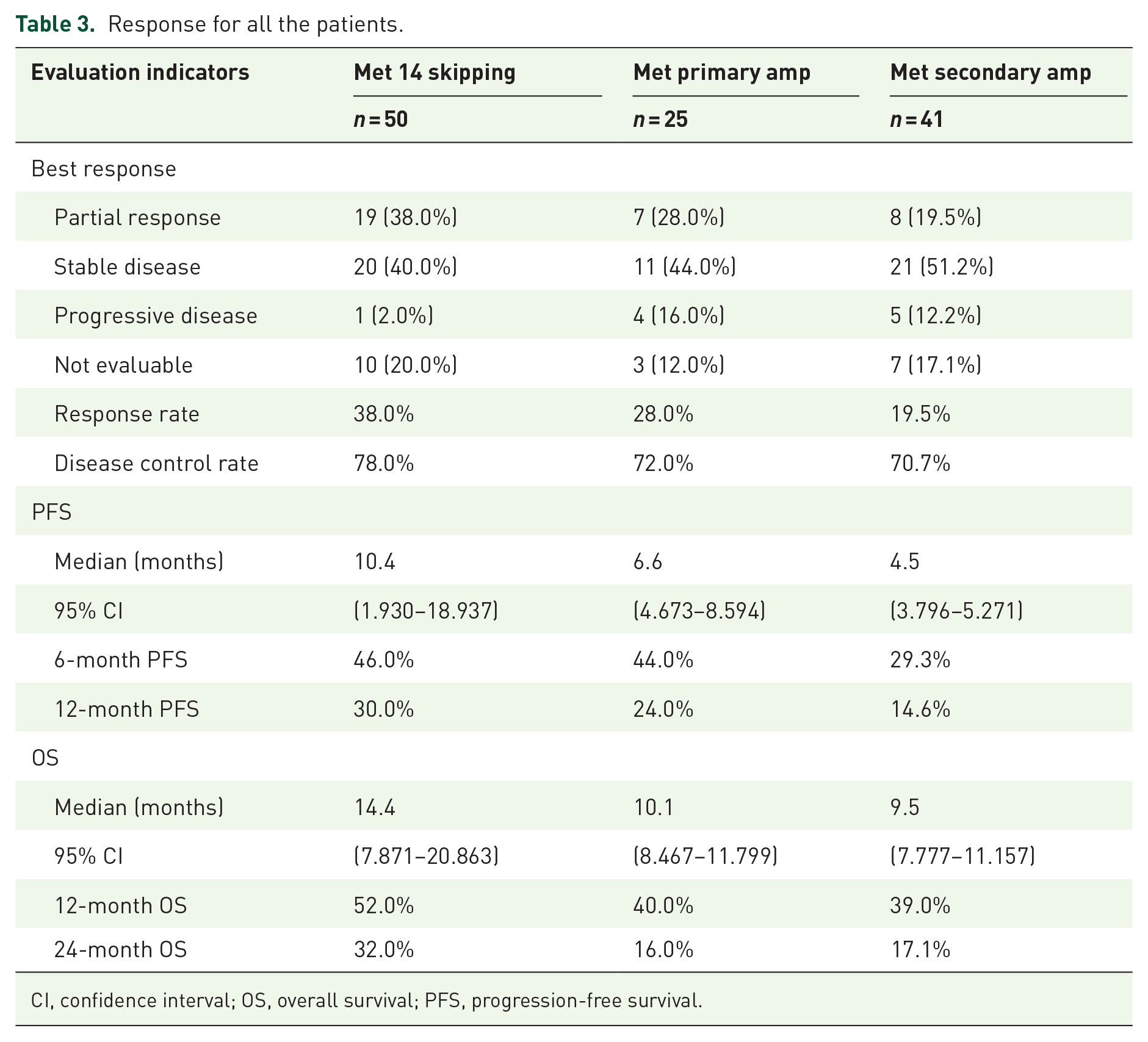
CI, confidence interval; OS, overall survival; PFS, progression-free survival.
Furthermore, earlier studies have indicated a potential correlation between the efficacy of targeted therapy and the degree of MET amp. In the study by Wolf et al., 12 the objective response rate (ORR) of carbatinib ranged from 7% to 12% in patients with MET amplified gene copies less than 10. However, in patients with a MET amp gene copy number of 10 or higher, the ORR reached 29–40%. Similar results were observed in the study by Camidge et al., 14 where they categorized the extent of MET amp and found that the mPFS of the high MET amp group was notably longer than that of the low MET amp group (6.7 versus 1.8 months). Our study yielded analogous results, with the mPFS of the high amp group significantly exceeding that in the low amp group (6.6 versus 23.0 months, p = 0.019). This suggests that the degree of MET amp may serve as an indicator of the predicted efficacy of targeted therapy, although further validation through prospective studies is necessary.
Special consideration should be given to the occurrence of drug-related adverse effects when administering MET-TKIs. Common adverse reactions encompass peripheral edema, nausea and vomiting, erythema, and fatigue. In our study, peripheral edema emerged as the most prevalent, accounting for 35.34%, followed by nausea and vomiting at 21.55%. Notably, 29 patients (25%) experienced drug-related adverse reactions of grade 3 or higher while no previously undocumented drug-related adverse effects were observed.
This study has some limitations, mainly due to its retrospective nature, and some data were not fully recorded. For example, only 14/25 patients in the primary amplification group and only 29/41 in the secondary amplification group were mentioned to the extent of the amplification. The incidence of adverse reactions is likely to be underestimated due to patient recall or medical records. In addition, since the confirmation of the amplification threshold is still controversial, the threshold we determined may include patients with are polyploid but not truly amplified, which needs more perfect exploration and definition to perfect.
Conclusion
Statistically significant differences were observed among NSCLC patients with different subtypes of MET alterations. Patients with MET exon 14 skipping mutations and primary amp demonstrated promising efficacy while limited effectiveness was observed in patients with secondary amp. Importantly, no previously unknown drug-related adverse effects occurred during the treatment period. The findings from this study may offer valuable research support for refining treatment options for these patients.
Footnotes
Appendix
Basic information of NSCLC patients with secondary MET amplification.
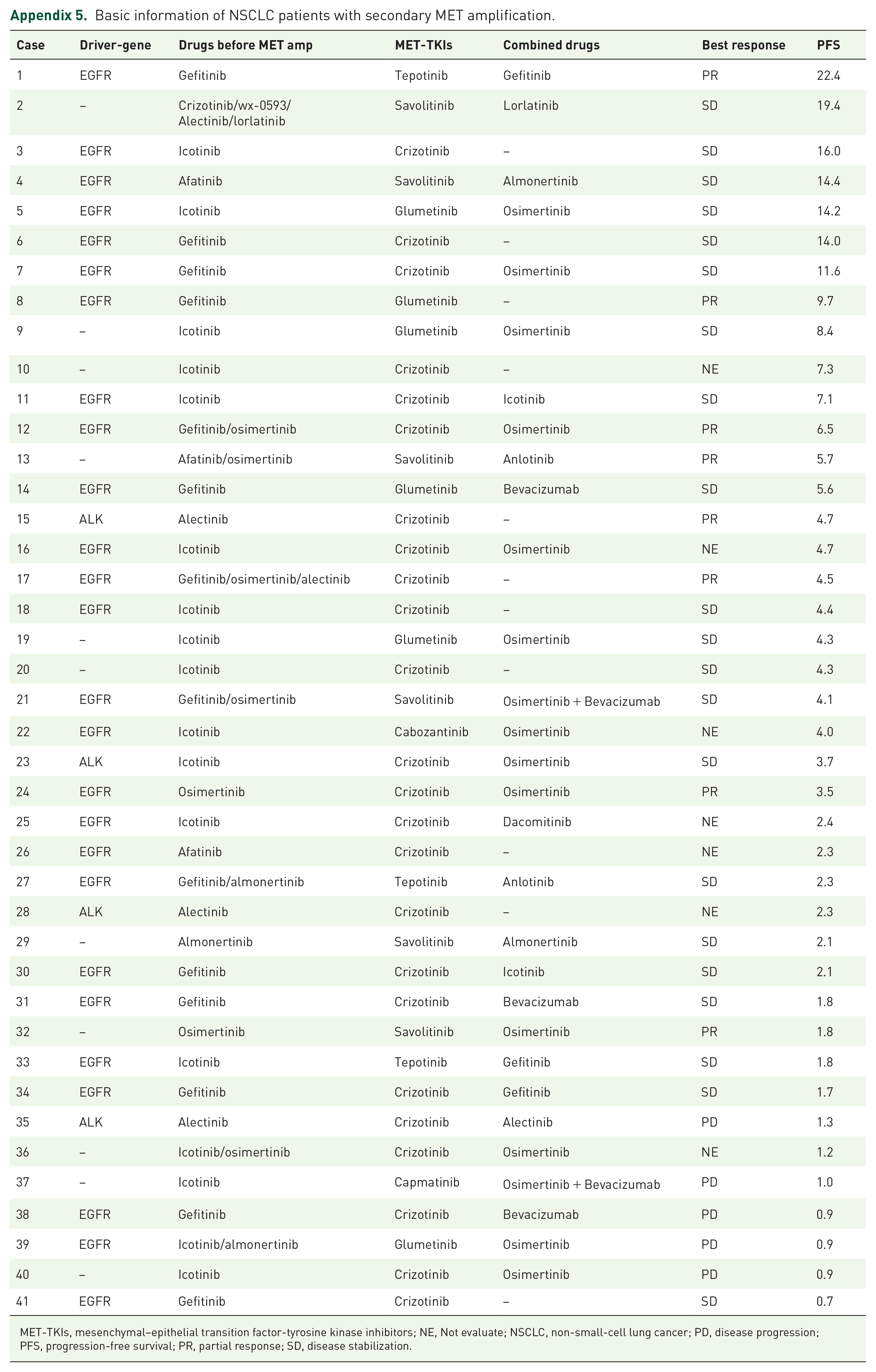
| Case | Driver-gene | Drugs before MET amp | MET-TKIs | Combined drugs | Best response | PFS |
|---|---|---|---|---|---|---|
| 1 | EGFR | Gefitinib | Tepotinib | Gefitinib | PR | 22.4 |
| 2 | – | Crizotinib/wx-0593/ Alectinib/lorlatinib |
Savolitinib | Lorlatinib | SD | 19.4 |
| 3 | EGFR | Icotinib | Crizotinib | – | SD | 16.0 |
| 4 | EGFR | Afatinib | Savolitinib | Almonertinib | SD | 14.4 |
| 5 | EGFR | Icotinib | Glumetinib | Osimertinib | SD | 14.2 |
| 6 | EGFR | Gefitinib | Crizotinib | – | SD | 14.0 |
| 7 | EGFR | Gefitinib | Crizotinib | Osimertinib | SD | 11.6 |
| 8 | EGFR | Gefitinib | Glumetinib | – | PR | 9.7 |
| 9 | – | Icotinib | Glumetinib | Osimertinib | SD | 8.4 |
| 10 | – | Icotinib | Crizotinib | – | NE | 7.3 |
| 11 | EGFR | Icotinib | Crizotinib | Icotinib | SD | 7.1 |
| 12 | EGFR | Gefitinib/osimertinib | Crizotinib | Osimertinib | PR | 6.5 |
| 13 | – | Afatinib/osimertinib | Savolitinib | Anlotinib | PR | 5.7 |
| 14 | EGFR | Gefitinib | Glumetinib | Bevacizumab | SD | 5.6 |
| 15 | ALK | Alectinib | Crizotinib | – | PR | 4.7 |
| 16 | EGFR | Icotinib | Crizotinib | Osimertinib | NE | 4.7 |
| 17 | EGFR | Gefitinib/osimertinib/alectinib | Crizotinib | – | PR | 4.5 |
| 18 | EGFR | Icotinib | Crizotinib | – | SD | 4.4 |
| 19 | – | Icotinib | Glumetinib | Osimertinib | SD | 4.3 |
| 20 | – | Icotinib | Crizotinib | – | SD | 4.3 |
| 21 | EGFR | Gefitinib/osimertinib | Savolitinib | Osimertinib + Bevacizumab | SD | 4.1 |
| 22 | EGFR | Icotinib | Cabozantinib | Osimertinib | NE | 4.0 |
| 23 | ALK | Icotinib | Crizotinib | Osimertinib | SD | 3.7 |
| 24 | EGFR | Osimertinib | Crizotinib | Osimertinib | PR | 3.5 |
| 25 | EGFR | Icotinib | Crizotinib | Dacomitinib | NE | 2.4 |
| 26 | EGFR | Afatinib | Crizotinib | – | NE | 2.3 |
| 27 | EGFR | Gefitinib/almonertinib | Tepotinib | Anlotinib | SD | 2.3 |
| 28 | ALK | Alectinib | Crizotinib | – | NE | 2.3 |
| 29 | – | Almonertinib | Savolitinib | Almonertinib | SD | 2.1 |
| 30 | EGFR | Gefitinib | Crizotinib | Icotinib | SD | 2.1 |
| 31 | EGFR | Gefitinib | Crizotinib | Bevacizumab | SD | 1.8 |
| 32 | – | Osimertinib | Savolitinib | Osimertinib | PR | 1.8 |
| 33 | EGFR | Icotinib | Tepotinib | Gefitinib | SD | 1.8 |
| 34 | EGFR | Gefitinib | Crizotinib | Gefitinib | SD | 1.7 |
| 35 | ALK | Alectinib | Crizotinib | Alectinib | PD | 1.3 |
| 36 | – | Icotinib/osimertinib | Crizotinib | Osimertinib | NE | 1.2 |
| 37 | – | Icotinib | Capmatinib | Osimertinib + Bevacizumab | PD | 1.0 |
| 38 | EGFR | Gefitinib | Crizotinib | Bevacizumab | PD | 0.9 |
| 39 | EGFR | Icotinib/almonertinib | Glumetinib | Osimertinib | PD | 0.9 |
| 40 | – | Icotinib | Crizotinib | Osimertinib | PD | 0.9 |
| 41 | EGFR | Gefitinib | Crizotinib | – | SD | 0.7 |
MET-TKIs, mesenchymal–epithelial transition factor-tyrosine kinase inhibitors; NE, Not evaluate; NSCLC, non-small-cell lung cancer; PD, disease progression; PFS, progression-free survival; PR, partial response; SD, disease stabilization.
Acknowledgements
The authors would like to thank all patients and their families for their cooperation and participation. In addition, we are thankful to all research staff and co-investigators involved in this research.
