Abstract
Background:
Leptomeningeal metastasis (LM) is a serious complication of advanced non-small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) mutation. There are no guidelines or consensus on the optimal treatment regimens and strategies for LM yet.
Objectives:
To analyze the influence of different treatment methods on the survival of patients and explore the factors influencing the prognosis of patients with LM. We profiled the cerebrospinal fluid (CSF) immune microenvironment in patients with LM using genetic and lymphocyte subset analyses.
Design:
We identified 209 patients with advanced NSCLC who developed LM without extracranial progression following third-generation EGFR-tyrosine kinase inhibitor (EGFR-TKIs) therapy. Their medical records were reviewed to extract relevant data.
Methods:
Survival curves were plotted and prognostic factors were analyzed using R software and SPSS statistical software, respectively. Statistical analysis was performed on the results of CSF genetic testing and lymphocyte subset profiling.
Results:
The median overall survival (mOS) time of patients who received high-dose EGFR-TKI monotherapy or in combination with other treatments was longer than that of patients with standard-dose EGFR-TKI combined with other treatments (19.7 vs 9.0 months), patients who received EGFR-TKI combined with anti-angiogenic drugs was longer than that of patients who did not combine anti-angiogenic drugs (19.6 vs 12.0 months). Analysis of CSF genomic profiles from 124 patients with LM identified a greater frequency of EGFR L858R (64.5%) versus exon 19 deletions (35.5%). We also observed a significant enrichment of B-cell subsets in the CSF of patients with LM compared to those without, and the B-cell count gradually decreased during the treatment process.
Conclusion:
For patients developing LM after third-generation EGFR-TKI resistance, high-dose EGFR-TKIs or EGFR-TKIs combined with anti-angiogenic drugs represent viable therapeutic strategies, and B lymphocytes may serve as a promising immunotherapy target for LM.
Background
Leptomeningeal metastasis (LM) represents a devastating complication in non-small cell lung cancer (NSCLC) patients, resulting in extremely unfavorable clinical outcomes.1,2 The incidence of LM has been steadily increasing, possibly due to prolonged patient survival resulting from effective systemic treatments, which consequently elevates the cumulative risk of central nervous system (CNS) progression. This trend is particularly notable in lung cancer patients with epidermal growth factor receptor (EGFR) mutations, with reported incidence rates as high as 9%–16% in studies of Chinese populations.1,3–5 Patients with NSCLC-LM often experience severe clinical symptoms and face an extremely poor prognosis. Prior to the widespread use of third-generation EGFR-tyrosine kinase inhibitor (EGFR-TKIs) such as osimertinib, the median overall survival (mOS) following an LM diagnosis was only 3–6 months.1,6 Osimertinib, with its superior blood–brain barrier penetration, has significantly improved treatment outcomes, extending mOS to approximately 10–16 months and providing more effective control of CNS disease.1,4,7–11 Notwithstanding these improved outcomes, the incidence of LM remains high at around 10%, 12 and the issue of drug resistance remains unavoidable.
As the optimal treatment regimen and strategy for NSCLC-LM remains incomplete and is still in the exploratory stage, current management primarily depends on the histology and molecular subtype of NSCLC, the progression of systemic disease, neurological function, and prognosis.13,14 Previous studies have not reached a unanimous conclusion on whether whole brain radiotherapy (WBRT) can improve the survival of EGFR-NSCLC patients with LM,1,3,5,15–19 further studies are needed. The efficacy of conventional chemotherapy for LM is generally limited due to the blood–brain barrier. 20 This is corroborated by a phase II clinical trial, which reported a clinical benefit rate of only 15.8% (3/19) and a mOS of 43 days in patients with LM originating from lung or breast cancer. 21 Intrathecal chemotherapy (ITC) effectively bypasses the blood–brain barrier, achieving high drug concentrations in the cerebrospinal fluid (CSF) at low systemic doses. Its efficacy in NSCLC-related LM, with a mOS of 3–9 months, has been demonstrated in clinical trials and pooled analysis.22–24 Despite various intracranial treatment regimens being proposed, there is still no consensus on the optimal drug, dosage, frequency, or duration. The combination of third-generation TKIs and bevacizumab is also a common medication combination in clinical practice. A phase II clinical study 25 showed that the median progression-free survival (PFS) and mOS of 14 NSCLC patients with LM and EGFR-sensitive mutations treated with osimertinib combined with bevacizumab were 9.3 and 12.6 months, respectively. Whether this combined treatment is effective for LM after resistance to third-generation EGFR-TKIs remains to be verified. In the BLOOM trial, 41 patients with LM who had progressed on first- or second-generation EGFR-TKIs were given osimertinib at a double dose of 160 mg once daily, achieving an intracranial objective response rate of 62.0% and mOS of 11.0 months. 11 Further studies are needed to determine whether high-dose TKIs are effective for NSCLC-LM patients after third-generation EGFR-TKI treatment failure.
Immunotherapy continues to demonstrate limited efficacy in patients with LM. A European study of 19 individuals with NSCLC and LM who were treated with PD-1 inhibitors reported a mOS of only 3.7 months. 26 Similarly, a separate analysis of 13 patients with LM from various primary tumor types who received pembrolizumab showed a mOS of 4.9 months. 27 Given the limitations of systemic immunotherapy, novel delivery strategies are under investigation. Among these, intrathecal immunotherapy has emerged as a promising avenue. Although still preliminary, its therapeutic potential warrants further clinical trials validation (NCT06901817, NCT06132698, NCT06462222). Furthermore, existing study has conducted single-cell RNA sequencing based on CSF, revealing the immune cell landscape of LM and providing potential therapeutic targets for overcoming LM. 28 However, the current understanding of the CSF immune microenvironment in patients with LM remains limited, requiring further multidisciplinary efforts and advanced technologies to unravel its complexities and guide precision therapy.
So far, there is no clear consensus on the treatment regimens for NSCLC patients after LM, especially for NSCLC patients who develop LM after resistance to the third-generation EGFR-TKIs. Therefore, this study retrospectively analyzed the real-world effects of different treatment regimens on prognosis in this patient population and assessed prognostic factors associated with LM. Furthermore, we analyzed the differences in lymphocyte subsets in the CSF between patients with LM and those without LM, and compared the changes in EGFR gene mutations before and after LM.
Methods
Research subjects and inclusion criteria
The study subjects were patients with advanced NSCLC who developed LM after resistance to treatment with third-generation EGFR-TKIs targeted therapy between January 2020 and December 2024 at Shandong Cancer Hospital. The case inclusion criteria were as follows: (1) patients with histopathological confirmed NSCLC; (2) patients had an EGFR gene mutation and treated with third-generation EGFR-TKIs; (3) patients with confirmed CSF cytology or/and cranial magnetic resonance imaging (MRI) diagnosis of LM; (4) no extracranial progression when LM was diagnosed after resistance to the third-generation EGFR-TKIs. This retrospective study was approved by the Ethics Committee of Shandong Cancer Hospital (approval No. SDTHEC202506005) and was conducted in accordance with the principles of the Declaration of Helsinki. Informed consent was waived by our Institutional Review Board because of the retrospective nature of our study.
The medical records of these enrolled patients were reviewed. Data extracted from the records included demographic information, histological type, EGFR mutation status, clinical manifestations at the time of LM diagnosis, and treatment regimens after LM diagnosis, etc.
Evaluation of intracranial response and treatment toxicity
Overall survival (OSLM) in this study was defined as the time from diagnosis of LM to death or last follow-up (March 11, 2025). Intracranial progression-free survival (iPFSLM) was defined as the time from the date of LM diagnosis to the first documented progression of intracranial lesions or death due to intracranial progression.
The Response Assessment in Neuro-Oncology criteria for LM integrate neurological exam, CSF analysis, and neuroaxis MRI for treatment response assessment.29,30 Nonetheless, these criteria face several challenges. 13 Neurological evaluation may be confounded by other conditions, such as concurrent brain metastasis progression or treatment toxicity.29,30 CSF cytology has low sensitivity, and a negative finding does not rule out disease. Radiographic assessment is particularly difficult as many LM lesions are nonmeasurable, and about 20%–30% of patients show normal MRI results. 24 Given these practical limitations and in line with the methodologies of previous relevant clinical and retrospective studies,24,31,32 this study adopted the assessment criteria for intracranial LM efficacy from those prior works. This evaluation criteria consists of five layers as follows: Complete response (CR) is defined as almost normal neurological examination and Karnofsky performance status (KPS) score ⩾90, obvious response (OR) as significant neurological improvement and KPS score ⩾70 or increase of ⩾30 from baseline, partial response (PR) as partial neurological improvement and a KPS score of 50–70 or an increase of 10–20 from baseline, stable disease (SD) as no visible neurological improvement and KPS score increased ⩽10 from baseline, progressive disease (PD) as worsening neurological symptoms/signs and KPS score decreased compared to baseline. Clinical response was defined as the continuous presence of CR, OR, or PR within an interval of at least 1 week; CR, OR, and PR were considered effective, SD and PD were considered ineffective. 24 Clinical evaluation was performed once per week from the beginning of treatment to 4 weeks after the whole treatment cycle.
Adverse events (AEs) were assessed based on the Common Terminology Criteria for Adverse Events (CTCAE, version 5.0). Toxicity was categorized into five grades: mild (grade 1), moderate (grade 2), severe (grade 3), life-threatening (grade 4), and fatal (grade 5, death associated with AEs).
Survival analysis
To elucidate the optimal management for LM, this study evaluates a spectrum of treatment strategies. We conducted comparative analyses of (1) dose intensity, to compare patients receiving high-dose versus standard-dose EGFR-TKIs; (2) systemic combination therapy, to compare regimens containing EGFR-TKIs with versus without anti-angiogenic agents; and (3) local interventions, to compare outcomes with versus without ITC or WBRT.
Statistical analysis
Kaplan–Meier survival curves were plotted using the R software (version 4.4.3), and differences in survival rates between subgroups were compared using the log-rank test. Correlation bar graphs of CSF lymphocyte subgroups were plotted using GraphPad Prism 8.0. Cox proportional hazards models were used to assess the effect of clinical and tumor characteristics on survival, and hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated. Multivariate Cox regression analyses were performed for variables with a p value <0.1 in univariate analyses. p Value less than 0.05 was considered statistically significant. All data analyses were performed using IBM SPSS 25.0 statistical software.
We used the STROBE reporting guideline 33 to draft this manuscript, and the STROBE reporting checklist when editing, included in Supplemental Material.
Results
Patient characteristics
A total of 209 patients were included in the study according to the inclusion criteria, and their characteristics are shown in Table 1. The population consisted of 86 males and 123 females, and the median age of the patients was 57 (range: 30–80). Almost all patients (98.6%) were diagnosed with adenocarcinoma. Analysis of EGFR mutation status showed that 143 patients (68.4%) harbored the exon 21 L858R mutation, while 54 patients (25.8%) had the exon 19 deletion mutation. In total, 142 patients (67.9%) had brain metastases in the past or at the time of diagnosis of LM.
Baseline characteristics of enrolled patients.

Others: Patient received non-EGFR-TKI therapies (e.g., immunotherapy, chemotherapy).
EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor; ITC, intrathecal chemotherapy; KPS, Karnofsky performance status; LM, leptomeningeal metastasis; MRI, magnetic resonance imaging; NSCLC, non-small-cell lung cancer; WBRT, whole brain radiotherapy.
Clinical presentation and treatment of patients with LM
The median interval from diagnosis of lung cancer to the development of LM was 21.7 months (range: 1.2–149.2; Table 1). At the time of LM diagnosis, 139 patients (66.5%) had good physical activity (KPS ⩾80), 23 (11.0%) were asymptomatic, and 128 (61.2%) showed intracranial pressure ⩾280 mmH2O on lumbar puncture manometry. Among enrolled patients, LM was diagnosed by cranial MRI alone in 100 (47.8%), by CSF cytology alone in 62 (29.7%), and by both methods in 47 (22.5%). After the diagnosis of LM, 79 (37.8%) patients received standard-dose EGFR-TKIs combined with other treatments, 113 (54.1%) patients received high-dose EGFR-TKIs monotherapy or combined with other treatments, 17 (8.1%) patients received treatments other than EGFR-TKIs. After LM, 43.1% of the patients received WBRT and 72.7% of the patients received ITC.
Treatment response assessment
All patients underwent clinical assessments after treatment, and the results of the intracranial assessments of treatment response are summarized in Table 2. In the assessment of intracranial treatment response, 21 patients (10.0%) achieved CR, 44 (21.1%) showed OR, 55 (26.3%) had PR, 36 (17.2%) were classified as SD, and 53 (25.4%) experienced PD. The efficiency of intracranial treatment was 57.4%.
Clinical efficacy in the study population.

Toxicity and safety
Treatment-related toxicities and AEs were systematically evaluated across three therapeutic modalities (Table 3). Among the three cohorts, myelosuppression was the emerged as the predominant AE, with incidence rates of 22.8%, 16.8%, 29.4%, respectively. The whole population demonstrated a 5.7% (12/209) incidence of grade 3–4 myelosuppression. Hepatic dysfunction (8.6%) and electrolyte imbalance (6.7%) are also relatively common AEs that occur during the treatment of LM. Notably, no grade 5 treatment-related AEs were reported in any study cohort.
Toxicities and adverse events.

Cohort 1, the patient received standard-dose EGFR-TKI combined with other treatments; Cohort 2, the patient received high-dose EGFR-TKI monotherapy or combined with other treatments; Cohort 3, the patient received treatments other than EGFR-TKIs.
EGFR-TKI, epidermal growth factor receptor-tyrosine kinase inhibitor.
Survival and prognostic factors
We conducted univariate and multivariate Cox regression analyses to determine the prognostic factors related to iPFS and OS in patients with LM (Figures 1 and 2). Variables with p < 0.1 in univariate analysis were included in multivariate analysis. The results showed that patients age ⩾60 had poorer iPFS ((p = 0.009, HR: 1.72, 95% CI: 1.15–2.57) and OS (p = 0.004, HR: 1.79, 95% CI: 1.21–2.65). The KPS score (⩾80), receiving high-dose EGFR-TKIs monotherapy or in combination with other treatments, and ITC were associated with a better clinical prognosis.
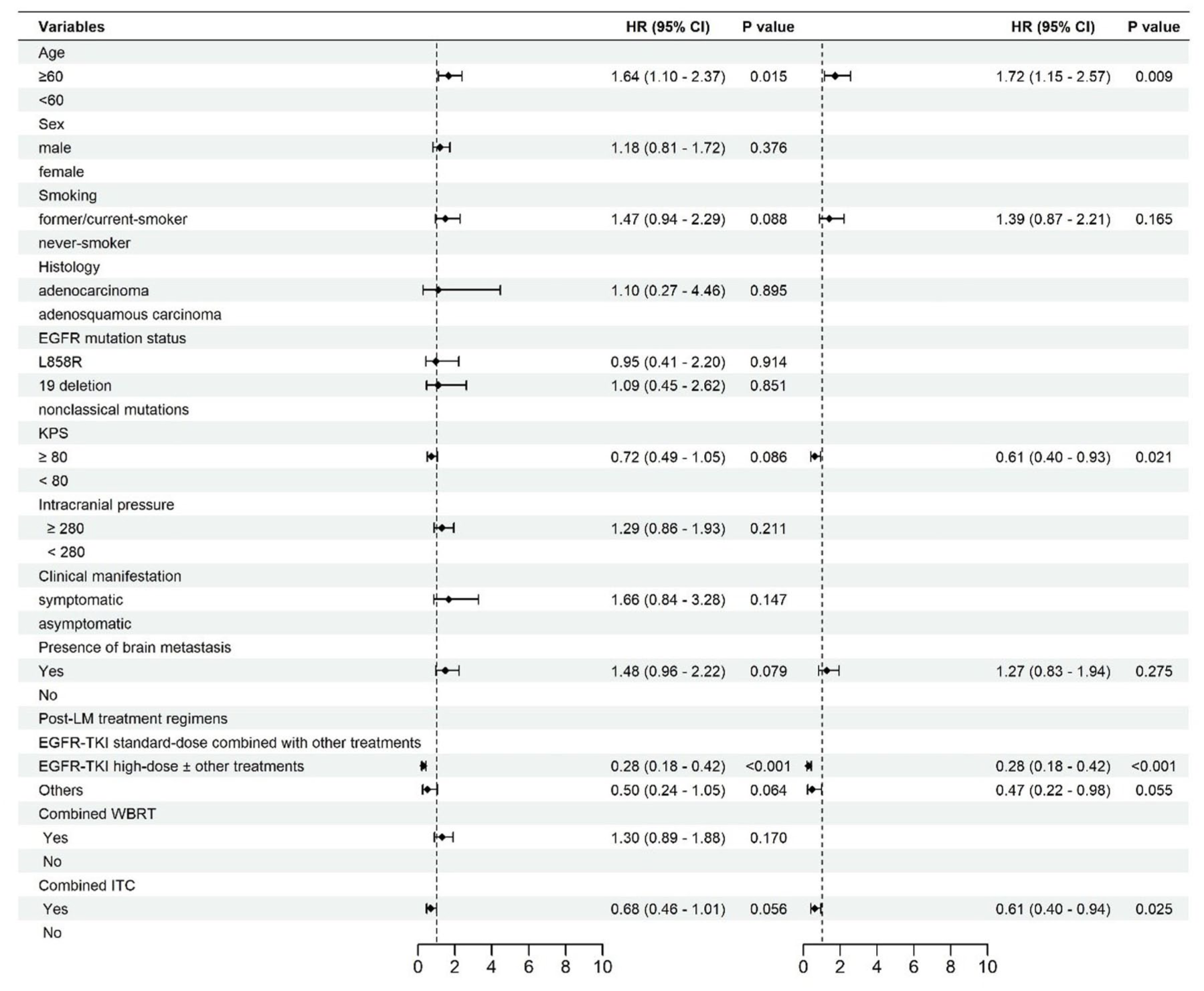
Forest plot showing Cox regression univariate and multivariate analysis of iPFS.

Forest plot showing Cox regression univariate and multivariate analysis of OS.
The final follow-up of this study was March 11, 2025. The median iPFSLM was 9.3 months (95% CI: 7.8–12.2; Figure 3(a)), median OSLM in the total population was 14.1 months (95% CI: 12.0–19.7; Figure 3(b)). In our study, the survival period of 113 patients who received high-dose EGFR-TKI monotherapy or in combination with other treatments was longer than that of patients with standard-dose EGFR-TKI combined with other treatments (19.7 vs 9.0 months, p < 0.0001, Figure 4(e)), and significant statistical results were also observed in the iPFS analysis (15.0 vs 6.2 months, p < 0.0001, Figure 4(a)). The OS of 58 patients who received EGFR-TKI combined with anti-angiogenic drugs was longer than that of patients who did not combine anti-angiogenic drugs (19.6 vs 12.0 months, p = 0.017, Figure 4(f)). Similarly, patients treated with EGFR-TKI combined with anti-angiogenic drugs have a longer iPFS (11.9 vs 8.0 months, p = 0.036, Figure 4(b)). The effect of ITC on prolonging iPFS (10.9 vs 7.8 months, p = 0.055, Figure 4(c)) and OS (15.0 vs 10.8 months, p = 0.062, Figure 4(g)) in patients is not obvious enough, and studies with larger sample sizes may be needed for further verification. In our cohort, patients received WBRT had no statistically significant effect on the prolongation of iPFS (8.2 vs 11.6 months, p = 0.17, Figure 4(d)) and OS (12.0 vs 15.2 months, p = 0.18, Figure 4(h)).

Kaplan–Meier analysis estimated iPFS and OS in all enrolled populations. (a) iPFS after the diagnosis of LM. (b) OS after the diagnosis of LM.

Kaplan–Meier analysis showing iPFS and OS curves by different treatment method: (a and e) with high-dose EGFR-TKI versus with standard-dose EGFR-TKI, (b and f) EGFR-TKI combined with anti-angiogenic agents versus did not combine with anti-angiogenic agents, (c and g) with ITC versus without ITC, (d and h) with WBRT versus without WBRT.
The EGFR gene mutation status before and after LM
Although prior evidence has implicated specific co-mutations in LM pathogenesis, the question of which EGFR subtypes confer the highest risk remains unresolved. From a total of 209 LM patients, we collected 124 cases who had both initial genetic testing results after lung cancer diagnosis and genetic testing results of CSF obtained after the development of LM (Table 4). Statistical analysis shows that 64.5% of patients with LM carry the EGFR exon 21 L858R mutation, TP53, PIK3CA, and CDK4/6 are common single drug resistance mutations after LM. Additionally, 35.5% of patients had EGFR exon 19 deletion mutations, and 40.9% of patients did not acquire new resistance mutations after LM. Based on our research results, we speculate that patients carrying the EGFR exon 21 L858R mutation may be more prone to LM, and TP53, PIK3CA, and CDK4/6 may play important roles in the occurrence of LM.
Comparison of genetic test results before and after LM.

CSF, cerebrospinal fluid; LM, leptomeningeal metastasis.
CSF biochemistry and lymphocyte subset analysis
To characterize the immune landscape of LM, we collected CSF from 24 patients with LM and 20 patients without LM for lymphocyte subsets comparison. The results showed that the level of B cells in the CSF of patients with LM was significantly higher than in those without LM (p = 0.046, Figure 5(a)). Moreover, flow cytometry analysis of CSF in a patient with LM demonstrated a declining trend in B-cell levels during treatment (Figure 5(e)).

Comparative analysis of lymphocyte subsets in CSF between patients with LM and those without LM. (a) B cells, (b) CD3+ T cells, (c) CD4+ T cells, (d) CD8+ T cells, (e) the flow cytometry results of a LM patient who underwent three CSF lymphocyte subset detections during the treatment period.
Given this immunological insight into the LM microenvironment, there is growing interest in modulating the CSF immune response for therapy. As such, intrathecal treatment with immune drugs represents a current research hotspot. Some clinical trials are exploring the potential of intrathecal PD-1 immune checkpoint inhibitors (whether used alone or in combination with other drugs) for the treatment of LM (Table 5).
Ongoing clinical trials on LM from solid tumors including NSCLC.

Source: From https://ClinicalTrials.gov, last update June 20, 2025.
LM, leptomeningeal metastasis; NSCLC, non-small-cell lung cancer.
Discussion
Despite the establishment of third-generation EGFR-TKIs as a first-line standard for advanced EGFR-mutated NSCLC based on improved efficacy and tolerability, the emergence of LM remains a unique and challenging pattern of disease progression, which carries a poor prognosis. In this study, we retrospectively analyzed 209 patients who developed LM with stable extracranial disease following failure of third-generation EGFR-TKI therapy. The entire cohort showed a mOS of 14.1 months and a median iPFS of 9.3 months, which was almost longer than that of previous studies.1,3,18,34 This outcome is likely attributable to the relatively high baseline KPS score (KPS ⩾80) of most enrolled patients, combined with recent advancements in the diagnosis and management of LM. As our analysis results show, the KPS score (⩾80), receiving high-dose EGFR-TKIs monotherapy or in combination with other treatments, and ITC were associated with a better clinical prognosis.
The incidence of LM has risen steadily, with a significantly higher rate observed in EGFR-mutant NSCLC patients compared to their wild-type counterparts. 35 Among the CSF gene test results of 124 patients, 64.5% of patients harbored EGFR exon 21 L858R mutations, and 35.5% of patients carried the EGFR exon 19 deletion mutations, suggesting a potential association between EGFR exon 21 L858R mutations and elevated LM risk. The same findings have also been made in other studies, verifying our discovery.1,6 Moreover, the results of CSF gene testing after LM revealed that EGFR L858R mutations often coexisting with TP53, CDK4/6, and PIK3CA resistance mutations simultaneously. These findings suggest that alterations in these drug resistance genes may be involved in the pathogenesis of NSCLC-LM through the cell cycle and DNA damage response pathways. Our observations align with the previously reported findings by Fan et al., 36 further supporting this mechanistic hypothesis. Based on our findings, we support that comprehensive CSF genotyping should be considered in the diagnostic workup of suspected LM. This approach facilitates risk stratification and informs personalized combination therapy, supporting more proactive management for EGFR-positive patients, especially the L858R subgroup.
Third-generation EGFR-TKIs have become an important treatment option for the management of LM in individuals with EGFR mutations, owing to their superior CSF permeability and intracranial activity. 37 In cases where patients receiving standard-dose third-generation EGFR-TKI therapy achieve sustained extracranial control without developing acquired resistance mutations, the occurrence of LM may be attributed to “pharmacokinetic failure”—that is, insufficient drug concentration in the CNS. In such scenarios, dose escalation of third-generation EGFR-TKIs may serve as an effective approach for treating LM.38,39 Considering the characteristics of the enrolled population in this study, along with the absence of new resistance mutations in CSF genetic testing in some patients after LM occurrence, it suggests that treatment failure may be attributed to pharmacokinetic rather than pharmacodynamic factors. This perspective is further supported by our analysis, which shows that patients receiving high-dose EGFR-TKI monotherapy or combination therapy with other agents had better survival outcomes compared to those receiving standard-dose EGFR-TKI combined with other treatments. Furthermore, we found that patients receiving EGFR-TKIs combined with antiangiogenic agents demonstrated significantly longer iPFS and OS than those without combination therapy, suggesting this regimen warrants clinical consideration for LM management. Our findings may provide a strategic framework for patients with LM. We suggest that clinicians consider high-dose EGFR-TKI escalation when pharmacokinetic failure is suspected, and that combining EGFR-TKIs with anti-angiogenic therapy could be prioritized as a potential approach to improve survival outcomes.
ITC is another pivotal treatment for LM, enabling direct drug delivery into the CSF compartment. This method allows for high local drug concentrations with low systemic exposure, thus maximizing cytotoxic efficacy while minimizing systemic toxicity. In contrast to a phase II trial 24 which showed promising activity of intrathecal pemetrexed (mOS was 9.0 months), our analysis found no significant improvement in iPFS or OS with ITC. Larger, dedicated studies are needed to determine the efficacy of ITC in this population.
Although WBRT plays a role in alleviating neurological symptoms, there is a lack of evidence to support its survival benefits, and its efficacy remains controversial.1,3,5,15–19 The research results of this study indicate that WBRT does not improve the iPFS and OS of patients with LM. Given that tumor cells in the CSF of patients with LM exhibit dynamic circulation and dissemination throughout all CSF compartments, 40 the entire craniospinal axis should theoretically be considered the optimal target volume for radiotherapy. However, due to its obvious bone marrow toxicity and the lack of evidence for its survival benefits, craniospinal irradiation is rarely recommended in clinical practice.41,42 Generally, when LM is treated with WBRT, irradiation is typically limited to the intracranial CSF compartments of the CNS.17,40,43 This limited target volume may explain its suboptimal therapeutic efficacy.
In recent years, the unique immunological landscape of LM has been increasingly depicted, it provides unprecedented opportunities for immunotherapy in the management of LM. Studies analyzing CSF have uncovered a profoundly immunosuppressive microenvironment in LM, marked by elevated levels of M2-polarized macrophages, enhanced fatty acid metabolism, and an accumulation of partially exhausted CD8+ T cells and Treg cells.44,45 Further insights from single-cell RNA sequencing have identified a distinct macrophage subtype within the CSF, which is linked to osimertinib resistance and LM progression, suggesting is a promising new therapeutic target. 28 Based on these mechanistic findings, ongoing clinical trials are evaluating whether intrathecal immunotherapy can modulate the CSF immunosuppressive microenvironment to enhance survival outcomes in patients with LM. In this study, we performed preliminary characterization of the CSF immune microenvironment and found that B cell levels in the CSF of patients with LM were significantly elevated compared to those without LM, and gradually decreased following treatment. Given evidence that certain subsets of tumor-associated B lymphocytes in solid tumors can exhibit immunosuppressive effects and promote LM development,46,47 our findings suggest these CSF-infiltrating B cells may actively participate in LM pathogenesis through immunosuppressive mechanisms. This discovery indicates that targeting B cells could be a promising new approach for the treatment of LM.
Our research also has several limitations. First, our research only includes one cohort from a single institution, which may inevitably lead to sample bias. Second, our investigation into the molecular resistance mechanisms and genomic landscape of LM remains exploratory due to the limited cohort size. These findings require further validation in larger cohorts and prospective studies.
Conclusion
Our research indicates that high-dose EGFR-TKIs or combined with anti-angiogenic drugs are feasible therapeutic strategies for NSCLC patients who develop LM after resistance to third-generation EGFR-TKIs. Furthermore, we found that the presence of an EGFR exon 21 L858R mutation was correlated with a higher risk of LM. Additionally, patients with LM showed elevated levels of B cells in their CSF. These results not only provide clinical guidance for LM management but also suggest CSF B cells as a promising immunotherapeutic target, warranting further investigation into their functional subsets and interplay with the tumor microenvironment.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251403396 – Supplemental material for Treatment options for EGFR mutation-positive NSCLC patients with leptomeningeal metastases after third-generation TKI resistance: a retrospective real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251403396 for Treatment options for EGFR mutation-positive NSCLC patients with leptomeningeal metastases after third-generation TKI resistance: a retrospective real-world study by Wenhua Yun, Ailing Liu, Junxu Wen, Kaikai Zhao and Xiangjiao Meng in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
