Abstract
The emergence of targeted therapies, such as epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase inhibitors, has marked a new chapter in the management of non-small-cell lung cancer (NSCLC). However, monotherapy with targeted agents often falls short in achieving optimal outcomes. The addition of radiotherapy (RT) has been shown to enhance therapeutic efficacy in various clinical settings. The LAURA trial (consolidation osimertinib) and POLESTAR trial (consolidation aumolertinib) suggest that targeted consolidation therapy following chemoradiotherapy is a promising approach for Stage III unresectable EGFR-mutant NSCLC. The integration of radiation with targeted therapies, while promising, is complex and requires ongoing research and multidisciplinary efforts to optimize patient outcomes. This review examines the role of radiotherapy in treating EGFR-mutant NSCLC across various stages, highlighting the significance of minimal residual disease and circulating tumor DNA, and identifies key challenges for advancing the field.
Introduction
Lung cancer is the most common and deadliest malignancy worldwide. Non-small-cell lung cancer (NSCLC) is the predominant subtype, accounting for approximately 85% of lung cancer cases, with more than half of these patients harboring key driver gene mutations. 1
Targeted therapies, particularly epidermal growth factor receptor (EGFR) and anaplastic lymphoma kinase (ALK) tyrosine kinase inhibitors (TKIs), have reshaped the treatment landscape for advanced NSCLC, demonstrating robust first-line efficacy. In recent years, the success of the ADAURA trial (adjuvant osimertinib) 2 and the ALINA trial (adjuvant alectinib) 3 has also established the role of targeted therapy in the adjuvant setting post-surgery. Nevertheless, for the majority of patients with locally advanced and advanced NSCLC, disease progression and acquired resistance to targeted therapies remain inevitable, necessitating the exploration and optimization of combination treatment strategies. 4
In the management of NSCLC, about 70% of patients require radiotherapy (RT). 5 In recent years, radiotherapy techniques have rapidly advanced. 6 However, subgroup analyses from the PACIFIC trial, which evaluated consolidation durvalumab following concurrent chemoradiotherapy (cCRT), as well as emerging real-world data (RWD), indicate that the addition of immunotherapy to radiotherapy does not improve survival outcomes in patients with driver gene-positive NSCLC. 7 Meanwhile, the exploration of combining radiotherapy with targeted therapy is being actively pursued. The incorporation of radiotherapy has improved the prognosis for patients with EGFR-mutant NSCLC. 8 At the 2024 American Society of Clinical Oncology (ASCO) Annual Meeting, the phase III LAURA 9 trial, which evaluated consolidation osimertinib following definitive chemoradiotherapy (CRT), demonstrated a significant improvement in progression-free survival (PFS) in patients with unresectable stage III EGFR-mutant NSCLC. The results of the LAURA trial have led to changes in clinical guidelines and reshaped the post-chemoradiotherapy treatment paradigm for unresectable stage III NSCLC. However, many issues remain unresolved, such as selecting the appropriate patients for the combination of radiotherapy and targeted therapy in locally advanced disease and determining the optimal duration of targeted consolidation therapy following combined targeted and radiotherapy in advanced patients. Drug holidays and adaptive therapy based on ctDNA-minimal residual disease (MRD) detection may help to address these questions, further emphasizing personalized treatment for lung cancer. 10
This review comprehensively explores the role of radiotherapy in the treatment of EGFR-mutant NSCLC, as summarized in Figure 1, examines the unresolved challenges in this field, and discusses future directions.
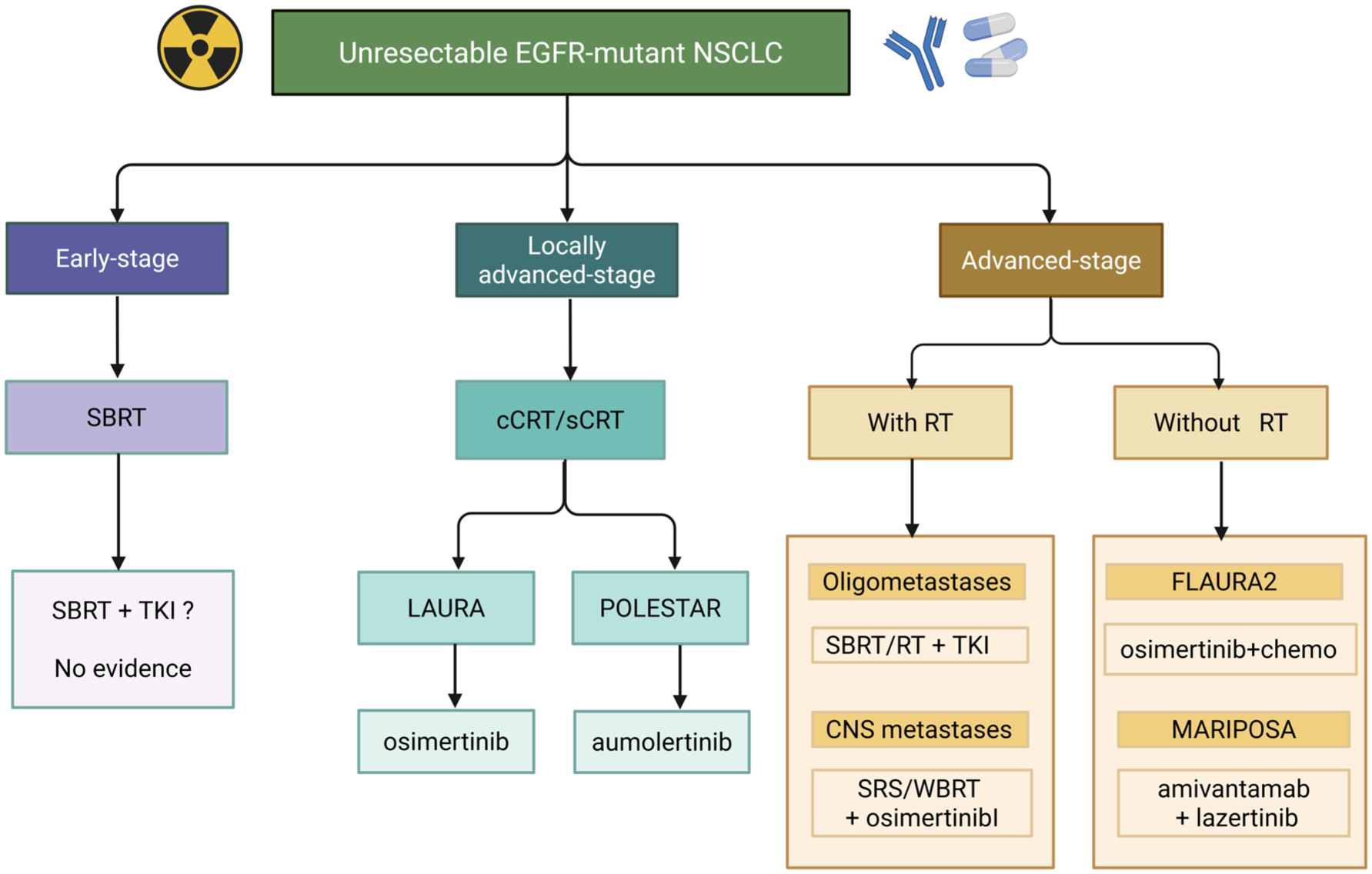
A proposed clinical treatment algorithm for integrating radiotherapy in unresectable EGFR-mutant non-small-cell lung cancer.
EGFR-targeted therapy in advanced NSCLC
Targeted therapies have fundamentally reshaped the treatment landscape for NSCLC harboring sensitizing EGFR mutations. These alterations are present in approximately 40%–50% of Asian patients and 15%–20% of patients worldwide with lung adenocarcinoma. The most prevalent variants are EGFR exon 19 deletions (≈60%) and exon 21 L858R substitutions (≈35%), both of which result in ligand-independent activation of the EGFR signaling pathway.11–13 The landmark IPASS trial established the clinical paradigm of genotype-directed therapy in advanced NSCLC. Subsequently, first-generation EGFR TKIs, including gefitinib and erlotinib, demonstrated superior efficacy over chemotherapy in molecularly selected populations, leading to their regulatory approval.14,15 Second-generation TKIs such as afatinib further improved outcomes in patients with activating EGFR mutations.16,17 In the OPTIMAL trial, erlotinib significantly prolonged PFS compared with chemotherapy (median PFS (mPFS), 13.1 vs 4.6 months), a benefit independently confirmed in the EURTAC study.18,19 Osimertinib, a third-generation EGFR-TKI with activity against the EGFR T790M resistance mutation, was initially approved for T790M-positive disease after progression on earlier TKIs. 20 The phase III FLAURA trial subsequently established osimertinib as the preferred first-line therapy, demonstrating superior PFS (18.9 vs 10.2 months) and overall survival (OS) compared with first-generation TKIs.21,22 More recently, upfront treatment intensification strategies have further improved clinical outcomes in phase III trials.23,24
Contemporary clinical guidelines now incorporate osimertinib plus platinum-based doublet chemotherapy and the chemotherapy-free combination of amivantamab plus lazertinib as recommended first-line options for patients with EGFR exon 19 deletions or exon 21 L858R-positive advanced NSCLC, particularly in higher-risk clinical settings. 25 In the phase III FLAURA2 trial, 26 the addition of platinum-pemetrexed chemotherapy to first-line osimertinib significantly reduced the risk of central nervous system (CNS) progression or death and, in the final analysis, improved OS compared with osimertinib monotherapy (median OS, 47.5 vs 37.6 months; hazard ratio (HR), 0.77; 95% confidence interval (CI), 0.61–0.96; p = 0.02). In the MARIPOSA trial, 27 the mPFS was 23.7 months for the amivantamab plus lazertinib group versus 16.6 months for the osimertinib group (HR = 0.70; 95% CI: 0.58–0.85; p < 0.001). A secondary analysis further demonstrated consistent benefit across poor-prognosis subgroups, including those with TP53 co-mutations, baseline circulating tumor DNA (ctDNA) positivity, lack of early ctDNA clearance, or liver metastases, supporting this regimen as an important therapeutic option for selected high-risk patients. 28
Radiation therapy
Advances in radiotherapy technology and optimization
Modern radiotherapy techniques have markedly improved dose conformity and reduced irradiation of normal tissues, thereby decreasing treatment-related toxicities. A secondary analysis of the phase III NRG Oncology–RTOG 0617 trial demonstrated that intensity-modulated radiotherapy (IMRT), compared with three-dimensional conformal radiotherapy, significantly reduced cardiac dose exposure (heart V40: 16.5% vs 20.5%; p < 0.001), resulting in fewer grade ⩾3 cardiovascular adverse events and improved OS in patients with unresectable stage III NSCLC.
Current clinical practice incorporates image-guided radiotherapy, adaptive planning, and respiratory gating to further limit exposure to non-malignant tissues. FDG-PET-guided target delineation combined with endobronchial ultrasound-guided nodal staging enables selective nodal irradiation. Ongoing trials, including RTOG 1308 and RAPID-RT, are evaluating proton therapy and cardiac avoidance strategies to further optimize the therapeutic ratio.29–32
Role of radiotherapy across the disease spectrum of NSCLC
Radiotherapy plays a central role across the disease spectrum of NSCLC. For medically inoperable, node-negative stage I–II disease, definitive stereotactic body radiotherapy (SBRT) is the standard of care and is typically delivered to a biologically effective dose ⩾100 Gy, achieving long-term local control rates of 90%–95%.33–37
In unresectable stage III NSCLC, treatment has evolved from radiotherapy alone to sequential chemoradiotherapy and ultimately to cCRT, which was established as the standard approach by the RTOG 9410 trial. Until recently, definitive cCRT to 60 Gy in 30 fractions represented standard management, with distant progression remaining the predominant mode of failure. 48 The PACIFIC trial, evaluating consolidation durvalumab—an anti-PD-L1 immune checkpoint inhibitor—after cCRT, significantly improved survival outcomes in the overall population.38–42
However, subgroup and post hoc analyses indicated no clear PFS or OS benefit in patients with EGFR-mutated tumors, underscoring persistent unmet needs in this molecular subgroup.7,42
Radiotherapy also remains essential for symptom palliation in advanced disease. Common regimens include single-fraction 8 Gy or hypofractionated schedules such as 20 Gy in five fractions for painful bone metastases, while stereotactic approaches (e.g., 24 Gy in two fractions) provide superior pain control for spinal lesions. 53 Palliative thoracic irradiation is typically delivered as 20 Gy in 5 fractions or 30 Gy in 10 fractions, with selected use of moderately hypofractionated regimens.43,44
Rationale for combining RT and targeted therapy
The biological rationale for combining EGFR-TKIs with radiotherapy is grounded in the principle of biological cooperation, whereby simultaneous targeting of complementary survival pathways amplifies tumor cell kill and enhances treatment efficacy.45,46 EGFR blockade suppresses downstream PI3K/AKT and RAS/RAF/MEK/ERK signaling cascades, thereby impairing DNA damage repair, promoting apoptosis, and inhibiting cell proliferation. As a result, tumor cells harboring accumulated DNA damage become more susceptible to radiation-induced cytotoxicity.47–49 Beyond pathway inhibition, EGFR-TKIs enhance radiosensitivity through multiple interrelated mechanisms, including redistribution of the cell cycle from the relatively radioresistant S phase toward the more radiosensitive G0/G1 and G2/M phases, attenuation of radiation-induced EGFR activation and autophosphorylation, and downregulation of key DNA repair proteins such as RAD51, leading to delayed repair of radiation-induced DNA double-strand breaks. EGFR-TKIs further potentiate radiation effects by increasing apoptotic signaling, reducing cancer stem cell populations, modulating radiosensitivity-associated gene pathways (e.g., CXCL1 and Egr-1), and improving tumor reoxygenation through anti-angiogenic effects. 46 Importantly, EGFR-mutant NSCLC cell lines, including those harboring resistance-associated mutations, exhibit intrinsically heightened radiosensitivity compared with EGFR wild-type counterparts, providing additional biological justification for combination strategies. Collectively, these mechanisms are supported by robust preclinical evidence demonstrating synergistic tumor growth inhibition and significant reductions in tumor volume in EGFR- and ALK-driven xenograft models, underscoring the therapeutic potential of integrating EGFR-targeted therapy with radiotherapy in oncogene-driven NSCLC.50–53
Radiotherapy in the era of targeted therapy
The development of radiotherapy techniques has progressed rapidly; however, local treatment alone is insufficient to control the risk of distant metastasis. Targeted therapy has demonstrated promising efficacy in the first-line treatment of advanced driver gene-positive NSCLC. Nevertheless, targeted therapy primarily inhibits tumor growth rather than achieving complete tumor eradication. Therefore, integrating targeted therapy with a local treatment modality is of critical importance. For patients with inoperable driver gene-positive NSCLC, the combination of radiotherapy and targeted therapy may be an effective therapeutic strategy. At present, numerous studies are investigating the combination of radiotherapy and targeted therapy in locally advanced and advanced NSCLC, focusing primarily on EGFR-positive NSCLC. Figure 2 illustrates the timeline of the development of this combined treatment approach.

Timeline of combined radiotherapy and targeted therapy.
Locally advanced NSCLC: EGFR-TKI-based strategies combined with thoracic radiotherapy
EGFR-TKI combined with chemoradiotherapy and as post-chemoradiotherapy consolidation
Several phase I–II clinical trials have investigated EGFR-TKI combined with cCRT (Table 1).54–61
Clinical trials of EGFR-TKI combined with chemoradiotherapy or post-chemoradiotherapy consolidation.

CI, confidence interval; cCRT, concurrent chemoradiotherapy; CNS PFS, central nervous system progression-free survival; DFS, disease-free survival; EFGR, epidermal growth factor receptor; GTV, gross tumor volume; HR, hazard ratio; IMRT, intensity-modulated radiotherapy; mPFS, median PFS; MTD, maximum tolerated dose; NSCLC, non-small-cell lung cancer; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PTV, planned target volume; RT, radiotherapy; TKI, tyrosine kinase inhibitor; TTP, time to progression; V20%, total lung volume minus GTV exceeding 20 Gy.
In the first six studies, all patients had stage III disease, with 14–60 patients enrolled per study. Gefitinib or erlotinib was administered, and the radiotherapy dose was ⩾63 Gy. Treatment strategies included the following: (1) EGFR-TKI plus cCRT; (2) induction platinum-based chemotherapy followed by EGFR-TKI plus cCRT; and (3) EGFR-TKI plus cCRT followed by consolidation chemotherapy. Komaki et al. reported a single-arm phase II trial enrolling 48 patients with unresectable stage III NSCLC. Patients received concurrent IMRT (63 Gy in 35 fractions), weekly paclitaxel and carboplatin, and daily erlotinib, followed by two cycles of consolidation chemotherapy. The overall response rate (ORR) was 85%, the median time to progression was 14 months, and the median OS was 36.5 months, with a 5-year survival rate of 36%. Grade ⩾3 toxicities occurred in 25% of patients, with no treatment-related deaths. However, distant relapse, mainly brain metastases (BM), remained common, indicating insufficient systemic control. 56 CALGB 30106 compared cCRT plus gefitinib and cCRT plus erlotinib, followed by chemotherapy. A total of 63 patients with stage III NSCLC were enrolled, including 43% with squamous cell carcinoma and 33% with adenocarcinoma. Patients were stratified into low-risk (performance status (PS) 0–1 or weight loss <5% within 3 months) and high-risk (PS 2 or weight loss ⩾5%) groups. Low-risk patients (n = 39) received cCRT plus gefitinib, whereas high-risk patients received radiotherapy plus gefitinib followed by maintenance gefitinib until disease progression.
The results showed that the PFS and OS were longer in the high-risk group than in the low-risk group. 60 In another study, 60 EGFR wild-type patients with stage III or oligometastatic NSCLC received concurrent erlotinib with radiotherapy and chemotherapy after a limited response to induction chemotherapy. Grade ⩾3 pneumonitis and esophagitis occurred in 8% and 2% of patients, respectively. The ORR was 53.3%, and the median OS was 23.3 months, indicating acceptable safety.54,62
Overall, EGFR-TKI combined with CCRT achieved mOS of 10.2–36.5 months, mPFS of 4.7–14.0 months, and ORRs of 21%–85%, compared with mOS of 13.4–26.8 months with CCRT alone.63–68 Across different strategies, outcomes varied substantially, and toxicity profiles were heterogeneous. Therefore, although combined EGFR-TKI and CCRT may improve outcomes, conclusions remain limited by small sample sizes and heterogeneity among studies.
Building on insights from earlier trials and advances in trial design, multiple pivotal studies have recently investigated targeted consolidation strategies in unresectable stage III NSCLC. The phase III LAURA trial has fundamentally reshaped the treatment landscape for unresectable stage III EGFR-mutated NSCLC by establishing consolidation osimertinib as a highly effective strategy following definitive CRT. LAURA demonstrated an unprecedented improvement in PFS, with a mPFS of 39.1 months for osimertinib compared with 5.6 months for placebo (HR 0.16; 95% CI, 0.10–0.24), representing one of the largest PFS gains observed in this disease setting. Importantly, this benefit was driven largely by a marked reduction in distant and CNS progression, as reflected by a CNS PFS HR of 0.17, highlighting the potent intracranial activity of third-generation EGFR TKIs. Despite these compelling disease control outcomes, OS data from LAURA remain immature. At approximately 31% OS maturity, a favorable but statistically non-significant OS trend was observed with osimertinib (median OS 58.8 vs 54.1 months; HR 0.67), likely confounded by substantial crossover, with 78% of patients in the placebo arm receiving subsequent osimertinib. Consequently, LAURA has not yet resolved the critical question of whether immediate initiation of EGFR TKI consolidation after CRT confers a survival advantage over delayed treatment at disease progression. Several important limitations warrant consideration. Disease progression in LAURA was predominantly locoregional, raising uncertainty regarding the optimal sequencing of systemic versus local therapies. Moreover, the unexpectedly short PFS in the placebo arm—substantially inferior to that observed in the PACIFIC trial and real-world cohorts—has raised concerns about patient selection and staging rigor. Notably, only approximately half of the patients underwent PET imaging before CRT, and repeat staging before randomization was not mandated, potentially allowing occult metastatic disease at baseline. Indeed, retrospective central review identified baseline BM in a subset of enrolled patients. Nevertheless, despite these caveats, the magnitude and consistency of PFS benefit, robust CNS protection, and reduction in distant metastases strongly support consolidation osimertinib as a new standard of care for unresectable stage III EGFR-mutated NSCLC. The regulatory approvals by both the FDA and EMA in 2024 underscore the clinical significance of these findings, while ongoing follow-up and future trials will be essential to refine patient selection, treatment sequencing, and the long-term survival impact of this paradigm. 9
POLESTAR is a phase III, randomized, double-blind trial evaluating aumolertinib as consolidation therapy after definitive platinum-based CRT in patients with unresectable stage III EGFR-mutant (Ex19del or L858R) NSCLC. A total of 147 Chinese patients without disease progression after CRT were randomized in a 2:1 ratio to receive aumolertinib (110 mg once daily) or placebo, with stratification by EGFR mutation subtype, disease stage, and CRT modality. Aumolertinib significantly prolonged PFS. mPFS was 30.4 months with aumolertinib and 3.8 months with placebo (HR, 0.20; p < 0.0001). Investigator-assessed PFS results were concordant. PFS benefit was consistent across all predefined subgroups, including those defined by EGFR mutation subtype, disease stage, PS, age, and CRT modality. Objective response rate (57% vs 22%) and disease control rate (96% vs 74%) favored aumolertinib. The risks of CNS progression (HR, 0.33) and distant metastasis or death (HR, 0.21) were also reduced. OS data were immature at the time of analysis. Aumolertinib demonstrated a favorable safety profile. No treatment-related deaths, interstitial lung disease (ILD), or grade ⩾3 radiation pneumonitis (RP) were observed. Grade ⩾3 treatment-related adverse events occurred in fewer than 10% of patients. Cutaneous adverse events and diarrhea were infrequent and predominantly low grade. Compared with the LAURA trial of osimertinib, POLESTAR enrolled an exclusively Chinese population with a higher proportion of patients with L858R mutations, stage IIIB/IIIC disease, ECOG PS 1, and sequential CRT, representing a more clinically challenging population. While cross-trial comparisons are inherently limited, descriptively lower rates of ILD and severe RP were reported with aumolertinib, suggesting a potential pulmonary safety advantage in the post-CRT setting. This difference in ILD rates may be partly due to the higher use of sequential CRT in POLESTAR. Sequential CRT, where chemotherapy precedes radiation, generally results in lower lung toxicity compared to concurrent CRT, which can exacerbate both acute and late lung damage. Therefore, the reduced ILD rates observed in POLESTAR could reflect the lung-sparing effect of sequential CRT. However, further research is needed to determine whether the observed differences in ILD rates are primarily due to the properties of aumolertinib or the CRT regimen. 69
The APPROACH trial is an ongoing multicenter, randomized phase III study designed to evaluate almonertinib induction followed by MRD-guided maintenance therapy in patients with unresectable stage III EGFR-mutant non-squamous NSCLC. A total of 192 patients with ECOG PS 0–1 are planned for enrollment. All patients receive almonertinib 110 mg once daily for 8 weeks, followed by multidisciplinary team-directed definitive local therapy, including surgery or chemoradiotherapy. Patients are subsequently randomized in a 1:2 ratio to either continuous almonertinib maintenance or ctDNA-based MRD-guided intermittent almonertinib, in which treatment initiation, interruption, and re-initiation are determined by serial MRD status. Radiographic assessment and ctDNA monitoring are performed every 12 weeks. The primary endpoints are the objective response rate after induction therapy and 24-month event-free survival (EFS) assessed by an independent review committee. Secondary endpoints include MRD-defined EFS subgroups, OS, major pathological response in surgical patients, safety, and quality of life. The study was initiated in June 2022 and remains ongoing.
The LAURA and POLESTAR trials establish osimertinib and aumolertinib as guideline-endorsed standard consolidation therapies following definitive chemoradiotherapy in patients with unresectable stage III NSCLC harboring common sensitizing EGFR mutations. However, further optimization requires extensive exploration and large-scale clinical studies.
EGFR-TKI combined with thoracic radiotherapy
Five phase II clinical trials in Table 2 analyzed the feasibility and efficacy of EGFR-TKI combined with radiotherapy alone in treating locally advanced NSCLC.60,63,70–72 Martinez et al. 71 studied 23 patients with stage I–III unresectable NSCLC who were not eligible for chemotherapy. All patients were divided into RT plus a placebo or erlotinib group, showing that the erlotinib group had better efficacy, a lower rate of disease progression (16.7% vs 22.2%), and everyday adverse events. The study from Spain in 2016 73 showed that RT combined with erlotinib had a higher complete response rate (41.5% vs 21.4%, p = 0.0446) and a trend toward more prolonged cancer-specific survival (21.4 vs 17 months, p = 0.0645). The median OS, PFS, and ORR seemed lower than the RT alone group, but the difference was insignificant. The incidence of rash in the combination group was significantly increased, but RT-related adverse reactions did not improve. In 2016, Swaminath et al. 70 showed that in advanced/metastatic NSCLC patients with palliative RT who had poor PS, erlotinib combined with palliative RT had little benefit compared with palliative radiotherapy alone. Still, combined therapy significantly improved the quality of life. A phase I study of gefitinib combined with radiotherapy for locally advanced NSCLC enrolled nine patients, two of whom had EGFR mutations, and both of those patients survived for more than 5 years. The above results showed a significant advantage in combining TKI and RT, with median OS (17.0–28.5 months) comparable to simple concurrent chemoradiation (13.4–26.8 months). 74 The mPFS ranged from 11.0 to 13.4 months and ORR from 53.0% to 83.3%; the targeted therapy group had no significant increase in toxicities. In summary, for unresectable stage III NSCLC, the treatment of TKI combined with RT had encouraging results and needs further exploration.
Clinical trials of EGFR-TKI combined with radiation therapy.
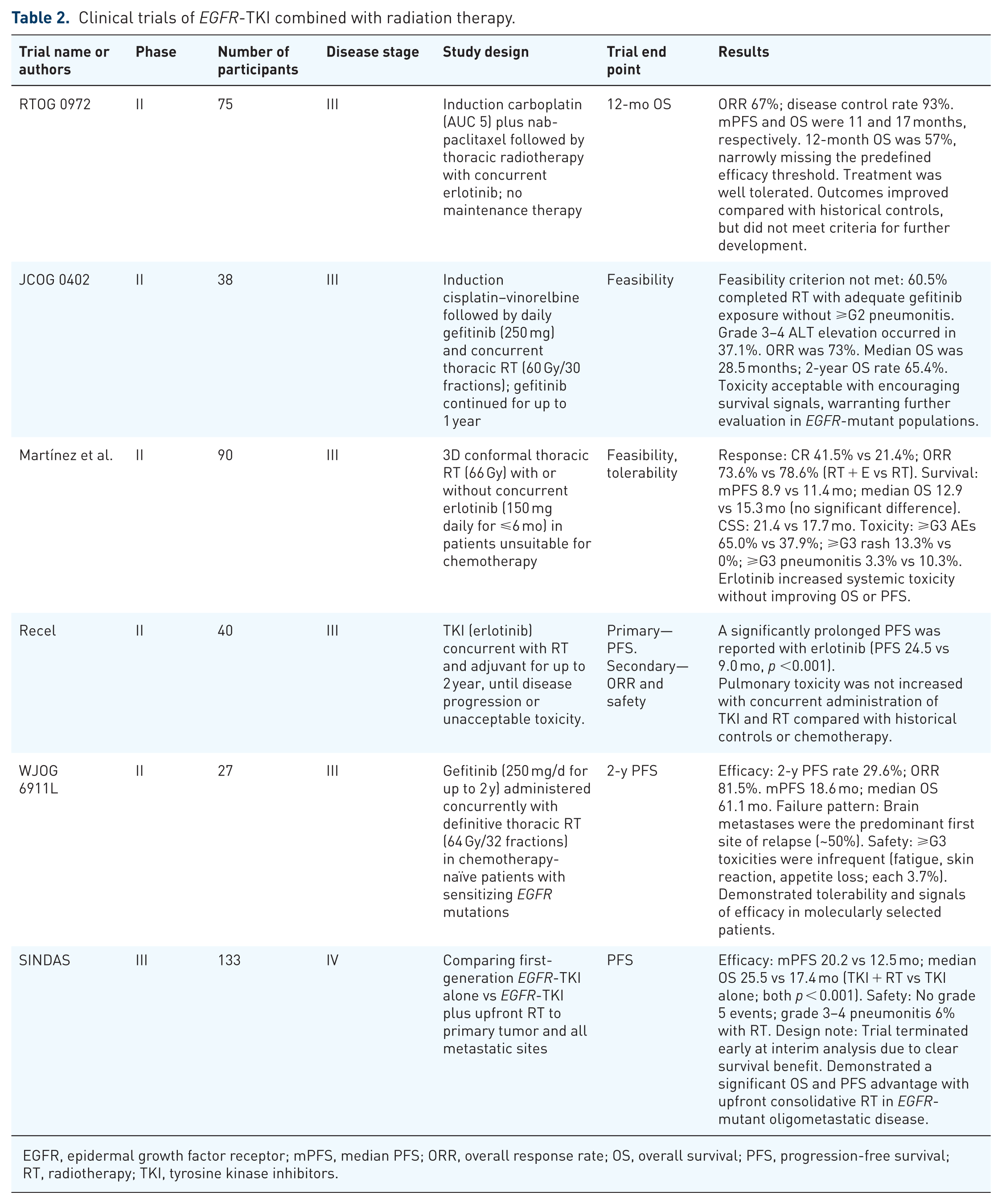
EGFR, epidermal growth factor receptor; mPFS, median PFS; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; RT, radiotherapy; TKI, tyrosine kinase inhibitors.
Two recent phase II studies have reported encouraging clinical outcomes. The WJOG 6911L phase II trial evaluated concurrent gefitinib and thoracic radiotherapy (TRT) in unresectable stage III NSCLC harboring sensitizing EGFR mutations. Gefitinib plus definitive radiotherapy achieved a 2-year PFS rate of 29.6%, with a mPFS of 18.6 months, an objective response rate of 81.5%, and a median OS of 61.1 months. Local control was favorable, with few in-field recurrences, suggesting a radiosensitizing effect of EGFR-TKI therapy. However, BM was the most common site of failure. Treatment was generally manageable, though pneumonitis and hepatic toxicity led to discontinuation in some patients. These findings support further exploration of EGFR-TKI-radiotherapy strategies. 75
Another study, the RECEL trial conducted in China, was a multicenter, randomized, open-label phase II trial. This study enrolling 40 patients with unresectable stage III NSCLC with EGFR mutations. The study compared erlotinib plus definitive radiotherapy with standard cCRT. Erlotinib combined with radiotherapy significantly prolonged PFS compared with cCRT (mPFS, 24.5 vs 9.0 months, p < 0.001), while objective response rates were comparable (70.0% vs 61.9%). Although the primary PFS endpoint was met, the results should be interpreted cautiously given the small sample size and phase II design. 75
ADVANCE (ChiCTR2000040590) was a randomized phase III study comparing an EGFR-TKI-based strategy with standard cCRT in patients with unresectable stage III EGFR-mutant non-squamous NSCLC. Patients aged 18–75 years with EGFR exon 19 deletion or L858R mutation were randomized 1:1 to receive osimertinib induction followed by osimertinib combined with definitive RT and maintenance osimertinib or definitive RT with concurrent cisplatin-pemetrexed chemotherapy. The primary endpoint was investigator-assessed PFS. Between March 2021 and March 2024, 43 patients were enrolled (24 in the experimental arm and 19 in the control arm). At a median follow-up of 25.5 months, mPFS was significantly prolonged with osimertinib-RT compared with cCRT (34.0 vs 7.8 months; HR 0.15, 95% CI 0.06–0.24). Median OS was not reached in the experimental arm and was 30.5 months in the control arm, without a statistically significant difference. The incidence of neutropenia and nausea was significantly higher in the cCRT arm, while quality-of-life measures favored the osimertinib-based approach. Among patients who completed RT without disease progression, the survival benefit was further accentuated, with non-reached PFS and a marked OS advantage in the experimental arm. To externally validate these findings, RWD (NCT04304638) from six centers were analyzed. Between 2012 and 2024, 125 patients with unresectable stage III EGFR-mutant NSCLC were included: 31 received RT plus EGFR-TKI, 33 received chemoradiotherapy plus EGFR-TKI, and 61 received cCRT alone. After a median follow-up of 32.7 months, both RT plus EGFR-TKI and chemoradiotherapy plus EGFR-TKI were associated with significantly longer PFS and OS compared with cCRT alone (PFS: not reached vs 36.7 vs 9.8 months; OS: not reached vs not reached vs 48.9 months; all p < 0.001). Notably, no significant differences in PFS or OS were observed between RT plus EGFR-TKI and chemoradiotherapy plus EGFR-TKI, suggesting that the addition of chemotherapy may not confer additional survival benefit in the context of EGFR-TKI-based multimodality treatment. Together, results from the ADVANCE trial and complementary real-world analyses consistently demonstrate that induction EGFR-TKI followed by EGFR-TKI combined with definitive radiotherapy provides substantial and durable disease control with improved tolerability, supporting this strategy as a promising alternative to standard cCRT in unresectable stage III EGFR-mutant NSCLC. 76
The REFRACT study analyzed 440 patients with unresectable stage III EGFR-mutant lung adenocarcinoma treated in a multicenter real-world cohort. Patients were categorized into three treatment groups: CRT alone; radiotherapy combined with EGFR TKIs (RT + TKI), including either EGFR-TKI administered after CRT or EGFR-TKI given concurrently with definitive radiotherapy, with or without chemotherapy; and EGFR-TKI monotherapy without radiotherapy. After inverse probability of treatment weighting, mPFS was 12.4 months with CRT, 26.2 months with RT + TKI, and 16.2 months with TKI alone (p < 0.001). Median OS was 51.0, 67.4, and 49.3 months, respectively. RT + TKI significantly improved PFS (adjusted HR 0.40) and OS (adjusted HR 0.61) compared with CRT, whereas TKI monotherapy did not improve OS. Notably, within the RT + TKI group, OS did not differ according to chemotherapy use, indicating that the survival benefit was driven primarily by the integration of definitive radiotherapy with targeted therapy rather than by chemotherapy. 77
In unresectable stage III EGFR-mutant NSCLC, CCRT has historically represented the standard treatment. The LAURA trial subsequently established osimertinib consolidation following definitive CCRT as a new therapeutic benchmark for this molecularly defined subgroup. Beyond osimertinib, other third-generation EGFR TKIs are being actively explored. The POLESTAR study demonstrated the feasibility of aumolertinib consolidation after CCRT, while the APPROACH study extended this concept by incorporating ctDNA-based MRD assessment to guide post-CCRT aumolertinib therapy. In parallel, the PLATINUM study (NCT05338619) evaluates lazertinib consolidation using a LAURA-like post-CCRT strategy. In contrast to consolidation-only approaches, the phase III ADVANCE trial investigates a chemotherapy-sparing paradigm that integrates aumolertinib across induction, concurrent radiotherapy, and consolidation. By directly comparing this EGFR-TKI-based strategy with conventional CCRT, ADVANCE provides critical evidence supporting the feasibility of omitting chemotherapy in selected patients with unresectable stage III EGFR-mutant NSCLC.
EGFR-targeted or immune consolidation following definitive chemoradiotherapy
Across multiple retrospective cohorts, converging evidence demonstrates that consolidation strategies for unresectable stage II–III EGFR-mutant (EGFRmut) NSCLC diverge substantially from the immunotherapy-based paradigm established in molecularly unselected populations. In a large international, multi-institutional analysis of 136 patients with stage III EGFRmut NSCLC treated with definitive cCRT, consolidation osimertinib was associated with a markedly superior 24-month real-world PFS (rwPFS) compared with durvalumab or observation (86% vs 30% vs 27%, respectively; p < 0.001), whereas no rwPFS difference was observed between durvalumab and observation. OS did not differ significantly, likely reflecting limited follow-up duration. 78 Similarly, in a separate multi-institutional cohort of 37 patients, consolidation durvalumab following CRT did not improve mPFS compared with CRT alone (10.3 vs 6.9 months; p = 0.993). By contrast, CRT combined with induction or consolidation TKIs was associated with a significantly longer mPFS (26.1 months; p = 0.023). Notably, clinically relevant immune-related toxicities were observed following sequential durvalumab and osimertinib. 79 Consistent findings were observed in a large Chinese cohort of 242 patients with unresectable stage II–III lung adenocarcinoma, in which mutation-positive patients receiving consolidation therapy experienced significantly prolonged mPFS (42.97 vs 24.87 months; p = 0.014) and improved OS (not reached vs 24.37 months; p = 0.006) relative to those without consolidation. Within the EGFRmut subgroup, targeted consolidation was associated with longer PFS than immune consolidation (42.87 vs 27.03 months; p = 0.029), without a statistically significant difference in OS. 80 Collectively, these data support EGFR-mutant locally advanced NSCLC as a biologically distinct entity in which EGFR-directed consolidation strategies may provide superior disease control compared with durvalumab-based immunotherapy.
Advanced NSCLC: local radiotherapy in combination with EGFR-TKI
EGFR-TKI combined with local radiotherapy for oligometastatic NSCLC
Incorporating radiotherapy alongside systemic therapy has been shown to improve both PFS and OS in patients with oligometastatic NSCLC. 81 However, whether these benefits extend to EGFR-mutant NSCLC remains uncertain.
Recent data from two phase II clinical trials have provided encouraging evidence in this molecularly defined population. In a prospective, multicenter, randomized phase II trial, patients with EGFR-mutant oligometastatic NSCLC who achieved stable disease or partial response after 3 months of first-line first-generation TKI therapy were randomized to receive SBRT plus TKI or TKI alone. The primary endpoint was PFS. mPFS was significantly longer in the SBRT plus TKI group than in the TKI-only group (17.6 vs 9.0 months; HR 0.52, 95% CI 0.31–0.89; p = 0.016), with a corresponding improvement in median OS (33.6 vs 23.2 months; HR 0.53, 95% CI 0.30–0.95; p = 0.026). Notably, greater PFS benefit was observed when SBRT was delivered to the primary tumor rather than metastatic sites. 82 In a separate single-arm phase II study, consolidative stereotactic radiotherapy (SRT) was evaluated in patients with EGFR-mutant metastatic NSCLC and oligo-residual disease following first-line third-generation EGFR-TKI therapy. Sixty-one patients received SRT while continuing EGFR-TKI treatment, achieving a mPFS of 29.9 months. Treatment-related toxicities were manageable, with pneumonitis and esophagitis being the most common adverse events. A propensity score-matched analysis demonstrated significantly prolonged PFS compared with EGFR-TKI therapy alone, supporting consolidative SRT as a promising strategy in this setting. 83
Building on the signals of benefit observed in earlier phase II studies, the phase III SINDAS and NROG-002 trials provide higher-level evidence supporting the integration of radiotherapy with EGFR-targeted therapy in patients with EGFR-mutant oligometastatic NSCLC. Two additional phase III trials have further confirmed the efficacy of integrating radiotherapy with targeted therapy in patients with oligometastatic NSCLC. The SINDAS study was a phase III randomized trial that evaluated the impact of SBRT on PFS and OS in treatment-naïve patients with EGFR-mutant oligometastatic NSCLC. The results showed that first-line SBRT with TKI therapy significantly prolonged PFS and OS compared with TKI monotherapy (mPFS: 20.2 vs 12.5 months, median OS: 25.5 vs 17.4 months), indicating a survival benefit from the addition of radiotherapy. This study established the clinical value of combining EGFR-targeted therapy with SBRT in EGFR-mutant oligometastatic NSCLC. 84 Another multicenter, randomized phase III trial, Northern Radiation Oncology Group of China (NROG)-002, evaluated TRT combined with EGFR TKIs in patients with EGFR-mutant oligometastatic NSCLC. TRT plus TKI significantly improved PFS and OS compared with TKI alone (mPFS: 17.1 vs 10.6 months; median OS: 34.4 vs 26.2 months). However, improved local control was accompanied by a higher incidence of grade 3–4 treatment-related adverse events, including radiation esophagitis (6.8% vs 0%), RP (5.1% vs 0%), leukopenia (42.4% vs 16.9%), and neutropenia (33.9% vs 11.9%). Overall, TRT combined with TKIs represents an effective strategy to improve survival in EGFR-mutant oligometastatic NSCLC, albeit at the cost of increased toxicity. 85
In 2024, ASCO reported a multicenter, single-arm phase II study evaluating osimertinib combined with consolidative radiotherapy, predominantly SRT, in patients with advanced EGFR-mutant NSCLC. Eligibility criteria did not restrict the number, location, or size of metastatic lesions. The study enrolled 42 patients, with 76% received consolidative radiotherapy after initial osimertinib treatment. With a median follow-up of 35.7 months, the mPFS was 32.3 months, and the median OS was 45.0 months. Building on prior studies in oligometastatic or locally advanced EGFR-mutant NSCLC, this analysis extends the exploration of osimertinib plus radiotherapy to a broader metastatic setting. Although limited by its single-arm design, the observed outcomes suggest that adding stereotactic ablative radiotherapy (SABR) to osimertinib may improve disease control compared with historical osimertinib monotherapy, warranting further prospective validation.86,87
However, these findings should be interpreted in the context of contemporary first-line intensification strategies evaluated in randomized trials. Although cross-trial comparisons must be made cautiously, the phase III FLAURA2 trial demonstrated that osimertinib plus platinum-pemetrexed chemotherapy significantly improved OS compared with osimertinib alone (median OS 47.5 vs 37.6 months), with higher rates of grade ⩾3 adverse events (70% vs 34%). Similarly, the phase III MARIPOSA trial showed improved survival with amivantamab plus lazertinib, with a 3-year OS of 60% versus 51% for osimertinib, accompanied by increased grade ⩾3 toxicity (80% vs 52%). By contrast, the single-arm phase II study of osimertinib plus consolidative radiotherapy reported a median OS of 45 months and a mPFS of 32.3 months, suggesting survival outcomes comparable in magnitude to systemic intensification, potentially with less systemic toxicity. Mechanistically, FLAURA2 and MARIPOSA represent systemic-intensification approaches supported by phase III evidence, whereas osimertinib plus SABR reflects a local-eradication strategy supported by lower-level evidence. Treatment selection should therefore be individualized, pending randomized validation of SABR-based intensification.
Therefore, these data indicate that local therapy plays a critical role in patients with oligometastatic disease and may contribute to delaying the emergence of resistance to EGFR-TKIs. The integration of EGFR-TKI with radiotherapy can enhance disease control and improve OS, with an acceptable toxicity profile. In patients with oligometastatic NSCLC, the SINDAS study demonstrated that SBRT combined with EGFR-TKI therapy significantly improved both PFS and OS. Collectively, these findings support the concept that appropriately timed radiotherapy may help overcome or delay EGFR-TKI resistance. Future studies should further define the optimal timing, sequencing, and modality of radiotherapy in combination with EGFR-targeted therapy.
EGFR-TKI combined with radiotherapy for NSCLC with BM
The treatment landscape for patients with EGFR-mutant NSCLC and BM is evolving, with a particular focus on the role of osimertinib combined with stereotactic radiosurgery (SRS). The ASTRO guidelines recommend SRS for patients with up to four BM and conditionally endorse it for those with 5–10 metastases with good PS. This recommendation sets the stage for exploring the potential benefits of combining osimertinib, a potent third-generation EGFR-TKI with strong CNS penetration, with SRS. 88
The TURBO-NSCLC study provided valuable insights into the efficacy of combining CNS-active TKIs with SRS. This multicenter retrospective analysis included 317 patients with EGFR or ALK mutation-positive NSCLC and BM, treated with either TKI alone or TKI plus SRS. The results showed that while the combination of TKI and SRS did not improve OS, it significantly prolonged the time to CNS progression and improved local CNS control. Notably, patients with BM ⩾1 cm appeared to derive greater benefit from the addition of SRS. 89
A recent pooled analysis presented at the 2024 World Conference on Lung Cancer further explored the combination of osimertinib and SRS. The study aimed to assess the intracranial PFS (icPFS) at 12 months for patients treated with osimertinib plus SRS versus osimertinib alone. The results indicated a trend toward improved icPFS and OS with the combination therapy, although the differences were not statistically significant. Importantly, the incidence of severe adverse events, such as radionecrosis, was low. 88
Despite these findings, the decision to use osimertinib combined with SRS should be individualized, considering factors such as the size of BM, presence of symptoms, and patient preferences. The ASCO-SNO-ASTRO guidelines suggest that for asymptomatic patients with BM receiving osimertinib, local therapy like SRS may be deferred. However, for patients with larger BM (⩾1 cm) or those with neurological symptoms, the addition of SRS may provide better local control without compromising OS.
In conclusion, both the TURBO-NSCLC study and the pooled analysis based on the LUOSICNS and OUTRUN studies have demonstrated that the combination of osimertinib and SRS as first-line treatment for patients with EGFR-sensitive mutation-positive NSCLC and BM provides a certain degree of local control but does not translate into an OS benefit. For patients with larger BM (⩾1 cm) or those presenting with CNS symptoms, the decision to use osimertinib combined with SRS should be made on an individual basis, considering clinical factors and patient preferences. Importantly, the combination therapy does not appear to compromise OS. Future research should focus on identifying patient subgroups most likely to benefit from this combined approach and further elucidating the optimal timing and sequencing of treatments.
Radiation-related pulmonary toxicity in the era of EGFR-TKI therapy
Radiation-related pulmonary toxicity in the context of EGFR-TKI therapy includes both classical RP and the less frequent radiation recall pneumonitis (RRP). 90 As EGFR-TKIs are increasingly used as consolidation therapy after CRT for unresectable stage III NSCLC, concerns have arisen regarding their interaction with prior thoracic irradiation. RRP represents a distinct EGFR-TKI-triggered injury pattern occurring after a period of radiographic quiescence. In a cohort of 160 patients, 7 cases (4.4%) were reported, with TKI initiation within 90 days of radiotherapy markedly increasing the risk (21% vs 2.1%). Episodes typically emerged within 2 weeks of TKI resumption, progressed rapidly, and were strictly confined to the prior radiation field, consistent with a radiation “memory effect.” A systematic review of 23 cases further indicated that RRP is largely pharmacologically driven rather than dose–volume–dependent, with nearly one-quarter presenting as grade ⩾3. 91
Beyond RRP, dose–response data clarify the mechanistic basis of conventional RP. In a prospective cohort of 159 NSCLC patients, a mean lung dose >16.1 Gy and V20 >30% strongly predicted RP. These metrics quantify the volume of normal lung exposed to clinically relevant radiation thresholds; higher exposure promotes epithelial apoptosis, vascular injury, cytokine activation, and fibrotic remodeling. When EGFR-TKIs are introduced under such unfavorable dosimetric conditions, epithelial repair is further impaired, amplifying susceptibility to both RP and RRP. 92
The study by Mu et al. offers the most comprehensive evaluation to date of RP in patients receiving concurrent TRT and different generations of EGFR-TKIs. In 200 matched EGFR-mutant lung adenocarcinoma patients, RP was assessed using both CTCAE clinical grading and volumetric radiographic criteria. Second-generation TKIs demonstrated the highest RP incidence (clinical 48%; radiographic 58%) and the greatest proportion of grade ⩾3 events, whereas first- and third-generation TKIs showed substantially lower and comparable risks (clinical 29% and 28%; radiographic 33% and 36%). These findings are consistent with the broader and irreversible pan-ErbB inhibition characteristic of afatinib, which may impair epithelial repair and potentiate radiation-induced injury. Multivariable analyses showed that gross tumor volume (GTV) was the only independent predictor for both clinical and radiographic RP, while V20 and TKI generation independently predicted radiographic severity. An overlap duration ⩾30 days between TRT and EGFR-TKI therapy significantly increased RP risk, highlighting the detrimental effect of prolonged concurrent exposure. SBRT, reflecting smaller target volumes and reduced lung irradiation, was associated with markedly lower RP rates compared with conventionally fractionated TRT. Mechanistically, larger GTV increases the volume of lung parenchyma exposed to high-dose radiation, amplifying epithelial apoptosis, endothelial damage, cytokine release, and fibro-inflammatory remodeling. Higher V20 and mean lung dose directly quantify the extent of clinically relevant radiation exposure. Second-generation TKIs further exacerbate these processes by suppressing EGFR-mediated epithelial regeneration—an essential component of post-radiation tissue repair—while extended overlap sustains the synergistic injury. Smoking status, although variably associated with RP in prior literature, was not a significant predictor in this study, likely due to the dominant influence of dosimetric and pharmacologic factors and the low prevalence of smokers in EGFR-mutant populations.
Collectively, these findings delineate a spectrum of radiation-related pulmonary toxicities and highlight the importance of minimizing lung dose, reducing TRT–TKI overlap, controlling GTV and V20, and preferentially selecting first- or third-generation TKIs when TRT is required. 93
Effective mitigation of RP during TRT and targeted therapy requires clear differentiation between infectious and non-infectious pneumonia, as the former mandates antimicrobial treatment while the latter—primarily RP or TKI-related ILD—should be managed with systemic corticosteroids, with hospitalization and antifibrotic agents considered for more severe cases. Targeted therapy may be cautiously resumed once symptoms improve to EGFR-TKIs and radiotherapy and reducing irradiated lung volume further decreases toxicity. Multidisciplinary evaluation, early symptom detection, careful steroid tapering, and vigilance for opportunistic infections remain critical to ensure safe and effective treatment. 94
Future directions
Future treatment strategies for EGFR-mutated stage III unresectable NSCLC
The optimal timing and sequencing of targeted therapy in unresectable stage III EGFR-mutant NSCLC remain undefined. Strategies under investigation include targeted consolidation after chemoradiotherapy, concurrent targeted therapy with radiotherapy, and induction targeted therapy before definitive local treatment. Approximately 20% of patients fail to benefit from cCRT and subsequently lose eligibility for consolidation therapy, highlighting the potential value of earlier systemic intervention. Induction therapy may enable tumor downstaging, reduce radiation target volumes, and improve tolerability, and in selected cases, facilitate conversion to resectability. A “sandwich” treatment model incorporating induction, definitive local therapy, and maintenance is therefore of growing interest. Ongoing clinical trials are expected to clarify the optimal induction approach and inform future treatment paradigms.
Timing of radiotherapy intervention for advanced EGFR-mutated NSCLC
In advanced EGFR-mutated NSCLC, integrating radiotherapy during maximal response to targeted therapy may improve local control and prolong PFS. First-generation EGFR TKIs typically achieve a mPFS of 8–10 months, with the most pronounced tumor shrinkage occurring within the first 3 months. Introducing radiotherapy during this early window may allow treatment of MRD, reduce radiation target volumes, and limit radiation-related toxicity. Early local intervention while tumor cells remain drug-sensitive may also delay the emergence of resistance, rather than reserving radiotherapy as salvage at progression. However, with third-generation EGFR-TKIs such as osimertinib extending mPFS to approximately 18–20 months, the optimal timing of radiotherapy—whether early or deferred—remains undefined and warrants prospective evaluation.
Feasibility of chemotherapy de-escalation strategies in stage III EGFR-mutant NSCLC
Although cCRT remains the standard of care for unresectable stage III NSCLC, emerging evidence suggests that chemotherapy may not be indispensable for all patients with EGFR-mutant disease. In carefully selected individuals, radiotherapy combined with EGFR-targeted therapy has demonstrated durable disease control with potentially reduced treatment-related toxicity. This chemotherapy-free approach may be considered for patients who are unable to tolerate platinum-based regimens, such as elderly individuals, those with poor PS, or patients with significant comorbidities, provided that definitive TRT can be safely delivered. Phase II studies, including RECEL, WJOG6911L, and ADVANCE, support the biological and clinical feasibility of this strategy. However, in the absence of phase III randomized evidence, chemotherapy de-escalation should not be routinely adopted and is best pursued within clinical trials or under strict clinical indications. Ongoing randomized studies are expected to further define appropriate patient selection and treatment sequencing in this setting.
The feasibility of biomarker-driven diagnosis and treatment in NSCLC
ctDNA and MRD assessment are emerging as clinically actionable biomarkers across the NSCLC disease spectrum. In resectable disease, ctDNA-based MRD detection may refine postoperative risk stratification beyond conventional TNM staging. In unresectable stage III NSCLC, ctDNA monitoring has important implications for guiding consolidation strategies after cCRT. Approximately 20% of patients may achieve durable disease control with CRT alone. Studies evaluating ctDNA dynamics after CRT and consolidation immune checkpoint inhibition suggest that ctDNA negativity is associated with favorable outcomes irrespective of consolidation therapy, whereas persistent or rising ctDNA predicts inferior PFS. The phase III APPROACH trial is prospectively evaluating MRD-guided consolidation with targeted therapy. In advanced EGFR-driven NSCLC, the SWOG S1403 study demonstrated that early ctDNA clearance strongly predicts progression-free and OS, outperforming radiographic response criteria. Collectively, these data support ctDNA-guided adaptive treatment strategies to personalize therapy and minimize unnecessary toxicity.95–97
Integrated and personalized management of driver gene-positive NSCLC
Advances in medical technology have driven a shift from uniform treatment strategies toward personalized care for driver gene-positive NSCLC (Figure 3). Within a multidisciplinary team framework, optimal management integrates tumor biology with patient-related factors, including comorbidities and socioeconomic context. Increasingly, treatment success is defined by achieving an appropriate balance between efficacy, toxicity, and quality of life rather than maximal treatment intensity. Emerging strategies such as MRD-guided adaptive therapy and treatment de-escalation, including drug holidays, aim to individualize therapy over the disease course. Patient and family engagement in treatment adherence, toxicity management, and follow-up further supports informed clinical decision-making and optimized long-term outcomes.98,99

A holistic approach to the management of oncogene-positive NSCLC.
Conclusion
The integration of radiotherapy with targeted therapy represents an important advance in the management of driver gene-positive NSCLC. Recent evidence, particularly from the LAURA trial, has established targeted consolidation after chemoradiotherapy as a new paradigm for unresectable stage III EGFR-mutant disease. However, key challenges remain, including optimal timing and sequencing of radiotherapy, appropriate patient selection, and the duration of consolidation therapy in the setting of prolonged systemic disease control. Clinically, current data support a selective, multidisciplinary approach rather than uniform treatment escalation, with decisions guided by molecular characteristics, treatment tolerance, and patient-specific factors. Emerging biomarkers, especially ctDNA-based MRD assessment, provide a feasible framework for adaptive treatment strategies, including personalized consolidation and de-escalation. Future prospective, biomarker-driven trials are essential to refine treatment sequencing and define the evolving role of radiotherapy in the era of targeted therapy.
